Abstract
Innovative cell-based therapies, including hepatic tissue engineering following hepatocyte transplantation, are considered as theoretical alternatives to liver transplant or for partial replacement of liver function in patients. However, recent progress in hepatic tissue engineering has been hampered by low initial hepatocyte engraftment and insufficient blood supply in vivo. We developed an intact 3D scaffold of an extracellular matrix (ECM) derived from a decellularized liver lobe, with layer-by-layer (LbL) heparin deposition to avoid thrombosis, which we repopulated with hepatocytes and successfully implanted as a tissue-engineered liver (TEL) into the portal system. The TEL provided sufficient volume for transplantation of cell numbers representing up to 10% of whole-liver equivalents and was perfused by portal vein blood. Treatment of extended hepatectomized rats with a TEL improved liver function and prolonged survival; mean lifespan was extended from 16 to 72 h. At 72 h postoperation, the TEL sustained functional and viable hepatocytes. In conclusion, we propose the TEL as a state-of-the-art substitute for whole-liver transplantation and as a proof of concept for the technology that will eventually allow for the transplantation of a reconstituted liver.
Introduction
Liver transplantation is currently the only curative therapy for end-stage liver diseases. Increasing demand for transplantable livers, however, far outweighs the donated organ supply. Thus, alternative, innovative, cell-based liver-directed therapies using hepatocytes (e.g., hepatic tissue engineering) are currently under investigation. The aim of these alternatives is to support patients with liver failure until either an organ becomes available or the patient's native liver regenerates; these alternatives could even potentially replace the patient's liver function.
Based on bioreactor technology, extracorporeal bioartificial liver (BAL) systems use functional hepatocytes to temporarily sustain a patient awaiting liver transplant. Although BALs improve clinical and metabolic conditions, increased patient survival rates have not yet been proven (5,24). For intracorporeal liver replacement, transplantation of isolated liver cells desirable. Injection of liver cell suspensions into anatomic structures such as the spleen, pancreas, kidney capsule (17), or peritoneal cavity (4) has been performed in different animal models. In particular, intraportal hepatocyte injection has been reported to be successful in animal models of metabolic deficiencies. However, portal hypertension, portal vein thrombosis, and pulmonary embolism remain problematic when larger numbers of cells are transplanted. At the same time, hepatocellular suspensions show poor cell stabilities and, due to the lack of vasculature in the construct, the achievable aggregate size is limited (22). Hepatocytes are attachment dependent; they maintain their liver-specific functions and survival through the extracellular matrix (ECM) composition and their cell-to-cell contacts. The concept of tissue engineering led to the application of artificial three-dimensional, porous, and biodegradable scaffolds that provide a platform for hepatocyte attachment (8,14). Ohashi et al. developed a method to engineer a uniform, continuous sheet of hepatic tissue in a scaffold-independent manner using isolated primary hepatocytes (18). All of the above techniques have several advantages compared to the injection of cell suspensions into solid organs. However, low initial hepatocyte engraftment and the lack of a vascular network at the implantation site are still major limitations that prevent successful engineering of functional and stable liver tissue at ectopic sites (30).
This study introduces a rat model of a portal system-implantable tissue-engineered liver (TEL) that combines isolated primary hepatic cells with a perfusion-decellularized liver matrix. Using this technology, Taylor and colleagues have engineered a bioartificial heart in vitro (19). The recent investigation indicated that a decellularized liver-derived biomatrix may be a favorable alternative to existing scaffolds for tissue engineering in that it is bioresorbable, can be easily manipulated, and supports long-term hepatocellular functions in vitro (11). Decellularized matrices also provide the appropriate three-dimensional organ architecture and ECM components for cell attachment and accommodation; they offer sufficient volume for transplantation of cell numbers that represent up to 10% of whole-liver equivalents. One essential component of a decellularized matrix is collagen. The potential advantages of collagen are its weak antigenicity and high tensile strength; however, the prothrombotic properties of collagen are a major drawback in applications for blood-contacting devices. To make collagen more thromboresistant, we used a positively charged polymer as a “bridge” to capture the negatively charged heparin and to adsorb to the collagen surfaces via an layer-by-layer (LbL) self-assembly technique. After the matrix internal surface anticoagulant modification was accomplished, the TEL could be implanted into the portal system and perfused by portal venous blood with an affluent hepatotrophic factor. To determine whether the TEL could correct a physiologically significant deficiency in hepatic function, we employed the 90% hepatectomy model of acute liver failure (ALF) in rats. Using the TEL in rats with ALF led to an improvement in liver function and life span extension.
Materials and Methods
Experimental Design
Rat cadaveric liver lobes were decellularized by pylic perfusion with detergents to produce acellular, perfusable constructs for the TEL scaffold (n = 80). After anticoagulative pretreatment (n = 60), the decellularized scaffolds were reseeded with hepatic cells (n = 50) and circumfused for 24 h in a bioreactor to complete formation of the TEL. Finally, the TELs (n = 40) were implanted into the portal systems of 90% hepatectomized rats. Normal rats served as a negative control.
Animals
Inbred male Lewis (LEW, RT) rats weighing 180–250 g (8–12 weeks of age) were obtained from SLAC Animal Laboratory (Shanghai, China). The animals were housed under standard animal care conditions and fed with rat chow ad libitum pre- and postoperation. All conditions and handing of animals in this study were conducted with protocols approved by the Sichuan University Ethical Committee on Animal Use and Care.
Perfusion and Decellularization of Rat Liver Lobes
All rat surgeries were performed under ketamine (100 mg/kg body weight, administered IP, Kelun, Chengdu, China) and xylazine (10 mg/kg IP, Kelun) anesthesia. Under deep anesthesia, a laparotomy was performed and the liver was exposed. After systemic heparinization through the inferior vena cava, all of the liver ligaments were dissected. The portal vein (PV) was liberated, and the pyloric vein, hepatic artery (HA), infrahepatic inferior vena cava (IHIVC), and common bile duct were ligated. The right suprarenal vein and the right renal vein were ligated and dissected after the suprahepatic inferior vena cava (SHIVC) was freed. The anterior wall of the abdominal aorta was then punctured, the diaphragmatic muscle was sheared, and the thoracic aorta was ligated (Fig. 1). Afterward, the liver was perfused slowly with cold (4°C) deionized water (20 ml) containing heparin sodium (50 U/ml, Kelun) through a cannula in the PV, and the SHIVC was transected, allowing outflow of the perfusate. Once the liver was blanched, the median lobe, PV, and SHIVC of the rat liver were removed en bloc. The right median lobe was preserved after left median lobe resection. Two prefilled 16-gauge integrated IV catheters (BD Medical, Sandy, UT, USA) inserted into the PV and the SHIVC, respectively, allowed pylic perfusion. Because the perfusate was heparinized, deionized water containing 10 mM adenosine at a portal perfusion pressure of 25 mmHg at 4°C overnight was followed in sequence by 1% SDS (Promega, San Luis Obispo, CA, USA), 0.5% SDS, and 0.25% SDS in deionized water for 4 h each. This was followed by 30 min of deionized water perfusion and 1 h of perfusion with 1% Triton X-100 (Amresco, Solon, OH, USA) in deionized water. We used antibiotic-containing physiological saline (100 U/ml penicillin-G; Gibco, Carlsbad, CA, USA), 100 U/ml streptomycin (Gibco), and amphotericin B (Sigma, St. Louis, MO, USA) to perfuse the liver for 12 h.

Schematic of the rat right median lobe decellularized as a scaffold for TEL. The median lobe (purple and yellow), PV (red), and SHIVC (red) of the rat liver were removed en bloc, and detergents were perfused in from the PV, as shown by the arrows. The right median lobe (purple) was preserved as the scaffold. ML, middle or median lobe (RML, right median lobe; LML, left median lobe); LLL, left lateral lobe; RLL, right lateral lobe; CL, caudate lobe.
Heparin Pretreatment
Before recellularization and implantation, the decellularized livers were treated for heparin immobilization by the LbL self-assembly technique (27). We used the polyelectrolyte polydiallyldimethylammonium chloride (PDADMAC, Mw = 100–200 kDa, positively charged, Sigma), which had charges opposite to those of heparin (Kelong, Chengdu, China). Typical adsorption conditions were used to form multilayers on the internal surfaces of decellularized livers (1 g/L of PDADMAC and 2 g/L heparin in physiological saline) (Fig. 2). After a 10-min perfusion of PDADMAC or heparin for each step, adsorption occurred for 20 min. After each adsorption, the decellularized livers were washed by perfusion with physiological saline for 10 min to eliminate residual polyelectrolytes. The eight-bilayer (PDADMAC/heparin)8 with heparin on top was deposited onto the internal matrix surfaces of the decellularized livers by an LbL process. All heparin-treated or non-heparin-treated decellularized livers were perfused by 1 mg/L heparin and antibiotic-containing physiological saline for 72 h for sterilization.
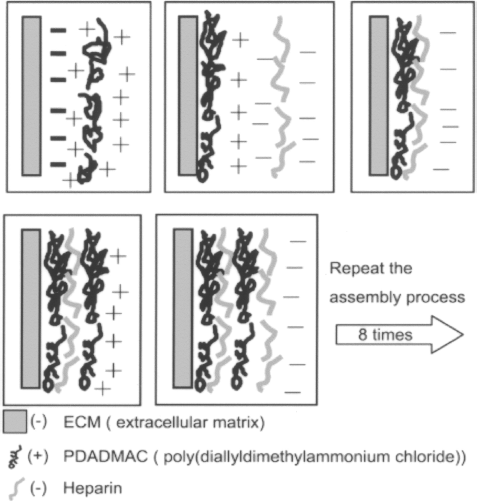
Schematic representation of layer-by-layer self-assembly technique for deposition of heparin.
To evaluate the anticoagulant effect of heparin pretreatment, we implanted both heparin-treated (n = 6) and non-heparin-treated (n = 6) decellularized lobes in vivo to create venous blood perfusion. After the rats were anesthetized and their right kidneys were excised, cuff-method end-to-end anastomosis was performed between the recipient PV branch of the right lateral lobe and donor PV and between the recipient right renal vein and donor SHIVC, located to the left of the decellularized lobes. The wound was closed in layers. The grafts were harvested 3 h postoperation.
Rat Hepatocyte Isolation and Spheroid Formation
Rat hepatocytes were isolated by a modified in situ collagenase perfusion technique, as described previously (7,21). All harvests yielded hepatocytes with viabilities exceeding 90%, as assessed by trypan blue dye exclusion. Freshly isolated hepatocytes were suspended in a culture medium and cultured by a previously described (2) rocking technique for 24 h to form hepatocyte spheroids.
Recellularization of Decellularized Cadaveric Liver Lobes
After 72 h of perfusion with antibiotic-containing physiological saline, we mounted the lobe matrix in a water-jacketed bioreactor vessel to allow closed-circuit perfusion through the PV, and perfused it at 37°C with an oxygenated cell medium for 2 h at a pylic flow rate of 2 ml/min to establish the presence of nutrients and a physiological pH in the ECM seeding bed before cell injection. Approximately 1 × 108 cells with spheroid formations, suspended in 3 ml of culture medium, were introduced into the liver lobe matrix by direct infusion into the PV. After a 4-h static period to allow for attachment, we restarted perfusion at a lower flow rate of 1 ml/min with culture medium only and continued to remove nonadherent cells for the 2-h duration of the culture (Fig. 3). We maintained culture conditions by placing the entire system in a tissue culture incubator. In addition to the 5% CO2 atmosphere of the incubator, we injected humidified carbogen (5% CO2/95% O2) into the medium reservoir.

Schematic of circumfusion recellularized liver bioreactor showing cannulation of PV and SHIVC. Oxygenated medium containing serum and antibiotics entered through the PV and exited through the SHIVC.
TEL Implantation
After the decellularized liver lobes were recellularized for 6 h in bioreactors, the recellularized liver lobes were perfused at 4°C physiological saline for 15 min and harvested as the donor TELs. In recipient rats (Lewis), the anesthesia method, body position, incisal opening, and systemic heparinization were identical to the conditions for the removal of the donor livers. We first performed a modified 90% hepatectomy on recipient rats (13). In the 90% hepatectomy, the left lateral, median, superior right lobe, and both caudate lobes were resected. We then liberated and transected the recipient infrahepatic PV (IHPV) and performed an end-to-end anastomosis of the donor PV and SHIVC to the recipient rat's inferior and superior PV incisions, respectively, using the cuff method. The TEL was connected in series with the residual native liver (Fig. 4). The TEL and residual lobes were harvested after reperfusion for 72 h. Heterotopic transplanted a rat right median lobe without decell/recell using the same surgical technique as the TEL into the recipient's portal system as a transplanted control group (n = 10).
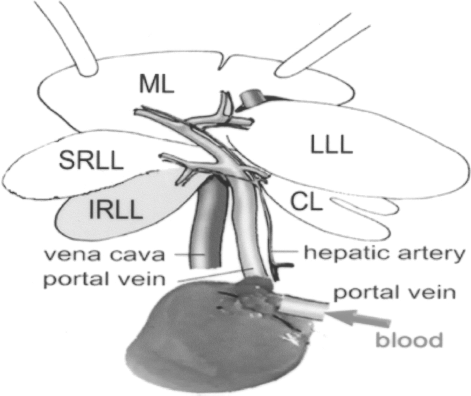
Schematic of the TEL implanted into the 90% hepatectomized rat portal system. Gray: remaining functional liver mass; white: nonfunctional liver mass. Only the IRLL (gray) of the native liver was preserved. The TEL was connected in series with the native liver by the PV. ML, middle or median lobe; LLL, left lateral lobe; RLL, right lateral lobe (SRLL, superior right lateral lobe; IRLL, inferior right lateral lobe); CL, caudate lobe. Red arrow indicates direction of PV blood flow.
Histology and Immunofluorescence
Native livers, decellularized livers, heparin-treated matrixes, recellularized livers, and in vivo TELs were fixed, paraffin embedded, and sectioned following standard protocols. The livers were cut into 5-μm sections and stained with hematoxylin and eosin (H&E), Periodic Acid-Schiff stain (PAS), and Masson trichrome stain, following standard protocols. To stain for heparin content, toluidine blue (0.05 mg/ml) was used on both heparin-treated and non-heparin-treated graft slides. Antigen retrieval was performed on paraffin-embedded tissue for immunohistochemistry (IHC) but not on frozen sections for immunofluorescence (IF). Slides were incubated sequentially for 1 h at room temperature with diluted primary and secondary antibodies. For the IHC test, a primary antibody against albumin (1:50, rabbit polyclonal IgG, A0001, DAKO, Carpinteria, CA, USA) and an HRP-conjugated secondary antibody (Santa Cruz, CA, USA), as well as 3,3-diaminobenzidine (DAB, Pierce, Rockford, IL, USA), were used to produce a brown color. For the IF assay, primary antibodies to collagen I (1:200, rabbit polyclonal IgG, GTX41287, Gene Tex, San Antonio, TX, USA), intracellular adhesion molecule 1 (Icam1) (1:200, mouse monoclonal IgG, sc-53527, Santa Cruz, CA, USA), platelet-endothelial cell adhesion molecule-1 (PECAM-1) (1:400, rabbit polyclonal IgG, BS1574, Bioworld Technology, Minneapolis, MN, USA), and von Willebrand factor (vWF) (1:100, rabbit polyclonal IgG, sc-14014, Santa Cruz) were used. A FITC-conjugated secondary antibody (Santa Cruz) was used to visualize a green color. For apoptotic cell detection, a TUNEL assay (In Situ Cell Death Detection Kit, Roche, Indianapolis, IN, USA) was used according to the manufacturer's instructions. The specimens were counterstained with DAPI (Sigma). Apoptotic nuclei were defined as positive when they displayed a green color; cells without DNA fragmentation displayed only blue nuclear staining. For the cell proliferation assay, a single dose of 5-bromo-2-deoxyuridine (BrdU) (Millipore, Billerica, USA) was injected IP 1 h prior to sacrifice at 50 mg/kg animal weight (5 mg/ml in PBS), and the BrdU In-Situ Detection Kit (BD Pharmingen, San Diego, CA, USA) was used according to the manufacturer's instructions.
Scanning Electron Microscopy (SEM) and Transmission Electron Microscopy (TEM)
We fixed the tissue with 2.5% (v/v) glutaraldehyde in PBS. For SEM, we dehydrated specimens with a series of ethanol solutions of increasing concentrations, beginning with 50% and progressing through 75%, 80%, 95%, and 100% ethanol. We transferred the specimens from 100% ethanol to a HCP-2 Critical-Point Dryer (Hitachi, Tokyo, Japan) and dried them with CO2. We sputter-coated samples with 10 nm AuPd (60%/40% alloy) using a Denton DV-502A high-vacuum system (Denton Vacuum, Moorestown, NJ, USA). We then visualized the samples with a Hitachi S-450 SEM (Tokyo, Japan). For TEM, we postfixed the tissue samples with osmium tetroxide. Samples were cut into ultrathin sections and photographed using a Hitachi H-7500 TEM (Tokyo, Japan).
Determination of mRNA Levels of Key Metabolic Proteins
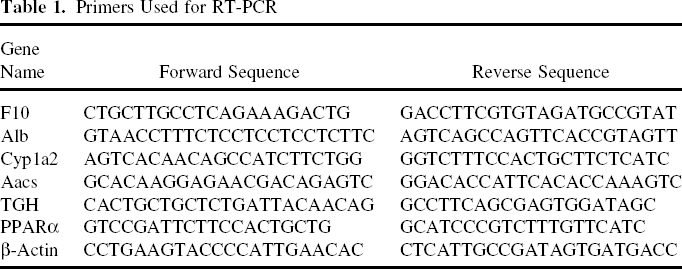
Total RNA was isolated from the TEL (0.2 g) and normal rat liver (0.2 g) using an SV Total RNA Isolation System (Promega). First-strand cDNA was amplified with the gene-specific primers shown in Table 1. Reverse-transcriptase (RT) PCR reactions were performed with approximately 500 ng of total RNA using a TaKaRa SYBR PrimeScript RT-PCR kit (Dalian, China) in a 25-μl reaction volume. The reverse transcription reaction was performed for 15 min at 37°C for reverse transcription. PCR products were separated and visualized on 2% agarose gels, and gel images were scanned using a Bio-Rad Fluor-S MultiImage system (Hercules, CA, USA).
Primers Used for RT-PCR
Quantification of Ammonia
Ammonia concentration was determined using a Fuji Dri-Chem slide (Tokyo, Japan) according to the manufacturer's instructions (23).
Quantification of Relative Cell Mass in TELs
Original livers from 90% hepatectomized rats (n = 5) and TELs (n = 5) were weighed. We used the weight of 90% of a liver, multiplied by 1.1, as the total liver weight. Samples of TELs and original livers were pelleted and aliquoted for DNA analysis. DNA was extracted immediately using a DNA isolation kit (Gentra Systems, Carlsbad, CA, USA). The redissolved purified DNA was characterized, and the DNA concentration was computed using a Gene Quant pro ultraviolet spectrophotometer (Amersham Biosciences, Staffanstorp, Sweden), which produced UV-absorbance measurements.
Statistical Analysis
Results are expressed as means ± SDs. The Kaplan-Meier method was used to calculate survival. Comparisons of survival rates in the different groups were tested by the log rank test. A value of p < 0.05 was considered to be statistically significant.
Results
Decellularization of Rat Cadaveric Livers
Antegrade pylic concentration-gradient SDS perfusion over 12 h yielded a fully decellularized construct with a translucent appearance (Fig. 5A. Histological evaluation revealed no remaining nuclei or cellular material, indicating successful decellularization throughout the entire thickness of the graft (Fig. 5B). Immunofluorescence (Fig. 5C) and SEM (Fig. 5D) showed complete removal of liver cells, while the collagen and fibers were maintained. Pylic perfusion with detergents preserved the underlying ECM and produced an acellular, perfusable vascular architecture and intact lobe geometry.

Perfusion decellularization of partial right lobes of rat livers. (A) Photographs of cadaveric (left) and decellularized (right) right median lobes of rat livers mounted on a perfusion apparatus. The liver lobe became more translucent as cellular material was washed out from the PV to the SHIVC by detergent perfusion. Scale bars: 1 cm. (B) H&E staining of thin sections from cadaveric (left) and decellularized (right) rat liver and Masson trichrome staining for inset (right), showing complete decellularization. Scale bars: 50 μm. (C) Immunofluorescence micrographs of control cadaveric (left) and decellularized (right) rat liver thin sections. No DAPI (blue) staining of intact nuclei was detected in the decellularized liver; the ECM component collagen I was preserved. Scale bars: 50 μm. (D) SEM of cadaveric (left) and decellularized (right) rat liver. The collagen and fibers in the liver lobes were maintained, but cells throughout all tissue layers were removed. H, hepatocytes. Scale bars: 5 μm.
Validation of Anticoagulative Pretreatment
The decellularized liver lobes treated with LbL heparin showed even and positive staining with toluidine blue throughout the entire matrix, whereas decellularized liver lobes without LbL heparin treatment did not stain with toluidine blue (Fig. 6A). These results demonstrate that heparin successfully linked to the entire matrix surface of decellularized lobes. We implanted heparin-treated and nontreated liver scaffolds and perfused them with portal blood to determine whether heparin pretreatment resulted in a significant anticoagulant effect. All rats survived the implantation without infectious or hemorrhagic complications. Histological evaluation revealed that the matrix without heparin treatment formed coagulated blood, but the surface that was heparin treated was thromboresistant (Fig. 6B) after 3 h of perfusion by venous blood in vivo.

Internal surface anticoagulative pretreatment of the decellularized rat liver. (A) The decellularized heparin-treated liver internal surface (left) showed uniform staining with toluidine blue, indicating that heparin linking was maintained throughout the entire ECM. The decellularized non-heparin-treated liver internal surface (right) did not stain with toluidine blue. (B) Corresponding H&E staining of thin sections from heparin-treated (left) or non-heparin-treated (right) decellularized livers, which were implanted and reperfused with blood in vivo for 3 h, shown without or with thrombus. Scale bars: 50 μm.
Construction of the TEL
Hepatocyte Spheroid Formation
After 24 h of rocked culture following hepatocyte isolation, 85% of primary hepatocytes had incorporated into well-formed spheroids greater than 30 μm in diameter. Expression of an important cell adhesive protein, Icam1, remained stable on the surface of hepatocyte spheroids, as seen by immunofluorescence staining (Fig. 7). Spheroid formation created an efficient three-dimensional tissue construct and cell-to-cell adhesion.

Formation of a perfused TEL construct by recellularization of decellularized hepatic ECM. (A) Rat hepatocyte spheroid formation via a rocking technique, viewed by phase microscopy. Rat hepatocytes (1 × 106 cells/ml) were rocked at 0.25 Hz for 24 h to induce spheroid formation. Rat hepatocyte spheroids viewed by phase microscopy (left). Immunofluorescence micrographs of rat hepatocyte spheroids (right) showed that Icam1 (green) was strongly expressed on the surface of hepatocytes during spheroid formation and both normal rat liver (upper inset) and isolated single hepatocytes (lower inset) stainings for control. Cytospin. Scale bars: 50 μm. (B) Photograph of recellularized rat TEL after 2 h of perfusion culture in a liver bioreactor. Scale bar: 2 cm. (C) H&E staining of frozen section from recellularized TEL in vitro show the lattice and multiporous structure formed by the hepatocytes. Scale bars: 50 μm. (D) SEM of recellularized TEL in vitro (left) showing the adhesion of round hepatocytes on the ECM. H, hepatocytes. Scale bar: 5 μm. Immunofluorescence micrographs of recellularized TEL in vitro thin section (right). The distribution of DAPI (blue) staining of nuclei was along the ECM component collagen I. Scale bars: 50 μm.
Recellularization of Decellularized Liver Lobes
We mounted decellularized rat liver lobes in a bioreactor and seeded them with an initial quantity of 1 × 108 cells, freshly formed into hepatocyte spheroids, through pylic injection. By 6 h after cell seeding, liver constructs were uniformly adiaphanous and carneous (Fig. 7B). Upon histological analysis, H&E staining on frozen sections of the recellularized liver lobes revealed lattice-like, multiporous structures formed by the reseeded hepatocytes (Fig. 7C), which accreted along the intrinsic ECM components (Fig. 7D), as shown by SEM and immunofluorescence micrographs. The TELs were successfully constructed when the decellularized liver lobes were recellularized in vitro.
Hepatocellular Masses in TELs and Original Livers
Five TELs and five original livers from rats that would serve as candidates for implantation were weighed, and their DNA was extracted. The concentrations of the extracted DNA were used to represent the cellular mass of TELs and of original livers. The data in Table 2 demonstrate that the cellular masses in TELs were approximately 10% (10.65 ± 0.83%) of whole native liver equivalents.
The Relationship Between the TEL and the Original Liver in Five Recipient Rats

Characteristics of TELs Following Implantation In Vivo
After TELs were implanted into the portal systems of the 90% hepatectomized recipients, TELs were reperfused. The entire portal inflow was perfused through the PV of the TEL graft, sent through the graft, out the SHIVC of the graft, back into the native portal vein, and then through the 10% remaining liver (Fig. 8A). After reperfusion for 72 h by portal venous blood in vivo, histological examination revealed that the hepatocytes maintained the morphologies of native hepatic cells in the inner areas of the scaffolds: pyramidal shapes and large round nuclei were clearly evident. The hepatocyte population was organized into aggregates and formed vessel-like structures, similar to central veins, among the hepatocytes or along the intrinsic basal lamina of vessels inside the scaffolds (Fig. 8B). The morphological transformation and accretion of hepatocytes was illustrated by SEM and compared to those from TEL circumfusion in vitro; the TEM had tight junctions and well-developed bile canaliculi with apical microvilli (Fig. 8C). BrdU-positive staining demonstrated notable hepatocyte proliferation, and the TUNEL assay confirmed minimal numbers of apoptotic cells in both TELs and residual livers (Fig. 8D). The ECM component collagen I was expressed throughout the entire TEL and was accompanied in some areas by cells that were positive for PECAM-1 and vWF (Fig. 8E). A tiny number of PECAM-1 or vWF-positive cells was seen in the absence of reperfusion of endothelial cells and presumably arose from endothelial components derived from PV blood.

Histological analysis of recellularized rat TEL implanted in the 90% hepatectomized rat portal system after 72 h. (A) TEL during reperfusion in vivo. The blue arrow indicates the direction of PV blood flow. Scale barsL 1 cm. (B) H&E staining of thin sections from TEL in vivo after 72 h shows clumps of hepatocytes (left) and blood channel formation among hepatocytes similar to central veins (middle) or along the intrinsic basal lamina of the vascular architecture (right). Scale bars: 50 μm. (C) SEM (left) of TEL reperfusion in vivo after 72 h showing the transformed and accrescent hepatocytes compared to those in TEL circumfusion in vitro. H, hepatocytes. Scale bar: 5 μm. TEM revealed the presence of tight junctions (middle, white arrowhead) and bile canaliculi (right, white arrowhead). Scale bar: 1 μm. (D) Immunohistochemical staining for BrdU-positive cells of the TELs (left) and residual livers (middle) in rats that received 90% hepatectomies at 72 h revealed substantial hepatocyte proliferation. Immunofluorescence staining for TUNEL (green) of TELs in vivo after 72 h (right) displayed low levels of apoptosis. Scale bars: 50 μm. (E) Immunofluorescence micrographs of TEL in vivo thin section for staining the ECM components collagen I (left, green) and endothelial markers PECAM-1 (middle, green) and vWF (right, green). Scale bars: 50 μm.
Use of a TEL in ALF
To determine whether TELs functioned properly and could support hepatic function, we employed a modified 90% hepatectomized model of ALF in rats. A TEL was implanted into the portal system at the time of hepatectomy. At 72 h postoperation, hepatocytes in the TEL expressed several liver-related genes, including coagulation factor X (F10), albumin (Alb), cytochrome P450 (Cyp1a2), acetoacetyl-CoA synthetase (Aacs), triacylglycerol hydrolase (TGH), and a transcription factor (PPARa) involved in lipid oxidation (Fig. 9A). The TEL also showed several characteristics of liver-specific functions, such as albumin synthesis (Fig. 9B) and the presence of glycogen rosettes (Fig. 9C). For untreated rats that underwent 90% hepatectomies, blood ammonia levels increased substantially; however, after treatment with TELs, blood ammonia levels increased more slowly (Fig. 9D). Although TELs did not support long-term survival, they still significantly prolonged the mean life span of rats that underwent 90% hepatectomies, from 16 to 72 h (Fig. 9E). Recipients in the TEL group and liver transplant group both suffered small-for-size syndrome with portal hypertension complicated by gastrointestinal congestion and ascites. These complications were more severe in the TEL group compared to the normal liver graft group.

Functional characteristics of the TEL following implantation into the rat portal system after 72 h. (A) Expression of genes related to liver function and metabolism in TEL (upper) and normal rat liver (lower) as control. F10, coagulation factor X; Alb, albumin; Cyp1a2, cytochrome P450, family 1, subfamily a, polypeptide 2; Aacs, acetoacetyl-CoA synthetase; TGH, triacylglycerol hydrolase; PPARa, peroxisome proliferator activated receptor a. (B) Albumin immunohistochemical staining of TEL (left) and normal rat liver (right) as control. Scale bars: 50 μm. (C) TEM (left) and periodic acid Schiff (PAS) (inset) staining for glycogen in TEL and PAS staining for normal rat liver (right) as control. Glycogens under TEM are black spots (white arrowheads) (N, nucleus; Scale bar: 2 μm) or red spots in PAS staining (Scale bars: 50 μm). (D) Ammonia levels and (E) survival of 90% hepatectomized rat after TEL therapy. TEL group: rats were hepatectomized (90% liver removal) and implanted with TELs (n = 10). Transplanted group: rats were hepatectomized (90% liver removal) and heterotopic transplanted with rat right median lobes (n = 10). 90% PH group: rats underwent 90% hepatectomy (n = 10). Normal group: normal rats serving as controls (n = 5).
Discussion
Recent reports (9,19,26) and our current studies have shown that perfusion with detergents through the PV for liver decellularization is an efficient method to obtain a completely intact liver scaffold with 3D geometry, vasculature, and biocompatibility, in which hepatocytes successfully graft using an organ perfusion system. In this study, seven different decellularization protocols [including different SDS concentrations (0%, 0.1%, 0.25%, 0,5%, 1%) combined with Triton-X 100 (1%) or ammonium hydroxide (0.1%)] have been compared in rat livers for efficiency of complete cell removal and potential for recellularization. The condition in our report was determined to work best. Our liver matrices also supply sufficient volume for the transplantation of cell numbers representing up to 10% of whole-liver equivalents, a cell mass that would be adequate to normalize patients' metabolic situations for many enzyme deficiencies (1). This technology has been successfully used for other organs, such as heart, heart valve, and corneas, and presents great clinical promise for patients in corneal transplantation (3). In addition, Macchiarini et al. (12) reported a decellularized protocol that removed HLA antigens from a donor matrix, leaving only immunologically inert proteins.
Collagen is the main component of the ECM, but it activates the extrinsic coagulation system when it contacts blood. The predominant obstacle to successful implantation of a perfusion-decellularized matrix in vivo is thrombogenicity, which can induce platelet activation. For this reason, a perfusion-decellularized whole-organ matrix that can be implanted in vivo has not been reported until now. To solve this problem, we applied an LbL self-assembly heparin-coating technique to the decellularized matrix. The entire reticular inner surface of the complex matrix was covered by immobilized heparin to create thromboresistance. Both in vitro and in vivo (28) assays indicate that immobilization of heparin on collagen is a very effective way to prevent surface thrombus formation. After the LbL procedure, the adherent primary hepatocytes attached and spread in an accelerated manner on the internal surface, with negatively charged heparin exposed on top. Heparin also enhances the binding of vascular endothelial growth factor (VEGF) to the extracellular matrix, promoting angiogenesis (15). Besides, recently, some studies have shown that heparin forms a hetetodimer and induces a significant increase in serum HGF levels, thus resulting in accelerated hepatocyte proliferation and regeneration (29).
Hepatocyte spheroids formed more efficiently by rocked culture (2) than hanging drops culture (6). Therefore, the rocked culture condition was used for spheroid formation in this study. In preliminary studies, we used single liver cell isolates to recellularize the liver graft by portal vein injection in vitro. But the single liver cells were lost from the liver scaffold much faster than the spheroids. The implanted single liver cells were lost to death more quickly than the spheroids, presumably because of lack of cell–cell contacts. The spheroids integrated into both parenchymal space and vasculature space after recellularization (Fig. 8B). So we perfused the vasculature space for only 2 h to limit spheroid deposition into the vascular space.
A major challenge for bioengineered complex organs is the provision of a functional blood supply. Previous studies preestablished a vasculature bed at ectopic sites and seeded hepatocytes around the network (17,18). However, ectopic sites do not normally offer sufficient hepatotrophic factors, so transplanted hepatocytes are prone to necrosis (30). For these reasons, we chose the PV system as the graft site.
At 72 h after TEL implantation in the PV system, hepatocytes in the TEL showed notable proliferation, colonization, and function. The TEL substituted some functions of a native liver and could help a recipient to survive end-stage liver failure, but did not ultimately support long-term recipient survival. A small-for-size syndrome was observed in both the TEL group and the transplanted group consisting of portal hypertension, gastrointestinal congestion, and ascites. Because the interior of the TEL lacked a true liver architecture, fluid resistance increased when low-pressure blood was infused. The formation of vessel-like channels was observed in the TEL. However, gastrointestinal congestion was still greater in the TEL group compared to the liver transplant group. These observations will be pursued in future studies.
Ongoing studies are also directed towards optimizing reseeding strategies to reduce the internal resistance of the TEL, in order to make portal venous blood perfuse more smoothly, to ameliorate postoperative gastrointestinal congestion, and to support long-term recipient survival. We propose that if the TEL can survive long term in vivo, endothelial cells might penetrate into the cell cluster, grow into a hepatic sinusoid, and reconstitute a hepatic lobule-like structure. However, using the present TEL concept, bile production and drainage are still problematic (24). The foremost limiting issue is the identification of a reliable source of large numbers of highly functional liver cells (5), especially various autologous adult stem cell sources such as oval cells, small hepatoblasts, hematopoietic stem cells, and mesenchymal stem cells. We will search for new sources of hepatocytes for transplantation, in particular focusing on the ex vivo genetic modification of patients' own stem or progenitor cells prior to reimplantation (10,16,20,25).
The current study was limited to rat livers, but we have also successfully applied perfusion decellularization to porcine liver, showing that this technology can be scaled up to livers of human size and complexity. Although further studies in a large-animal model (such as a porcine model) will be needed to confirm potential therapeutic efficacy, the new concept of engineering TELs using a decellularized natural platform with anticoagulant surface modification could be ultimately applied to the creation of a human TEL for future clinical transplantation. In such a clinical application, a TEL would likely be developed from human hepatocytes and a scaffold from xenogeneic animals, such as genetic knock-out and transgenic pigs to minimize complications from vascular rejection.
Footnotes
Acknowledgments
Financial support was received from National Basic Research Program of China (2009CB522401) and National Natural Scientific Foundations of China (30971118 and 30870983). Special thanks to Jinjing Wang, Lijia Cheng, Feng Ye, Lingling Wei, Xianhua Xu, and Song Lei for their expert technical assistance and scientific discussions. The authors are grateful to Zhidan Tu and Mayo Clinic professor Scott Nyberg for their valuable suggestions in the writing of the manuscript.
