Abstract
The developing neural cell must form a highly organized architecture to properly receive and transmit nerve signals. Neural formation from embryonic stem (ES) cells provides a novel system for studying axonogenesis, which are orchestrated by polarity-regulating molecules. Here the ES-derived motoneurons, identified by HB9 promoter-driven green fluorescent protein (GFP) expression, showed characteristics of motoneuron-specific gene expression. In the majority of motoneurons, one of the bilateral neurites developed into an axon that featured with axonal markers, including Tau 1, vesicle acetylcholine transporter, and synaptophysin. Interestingly, one third of the motoneurons developed bi-axonal processes but no multiple axonal GFP cell was found. The neuronal polarity-regulating proteins, including the phosphorylated AKT and ERK, were compartmentalized into both of the bilateral axonal tips. Importantly, this aberrant axon morphology was still present after the engraftment of GFP+ neurons into the spinal cord, suggesting that even a mature neural environment fails to provide a proper niche to guide normal axon formation. These findings underscore the necessity for evaluating the morphogenesis and functionality of neurons before the clinical trials using ES or somatic stem cells.
Introduction
Projection neurons, such as pyramidal hippocampal neurons and motoneurons, are featured with one long and thin axon and multiple short dendrites. The projection neurons integrate input signals from dendritic spines and then transmit this information as action potentials to target cells through the axon. In primary hippocampal neurons, axon formation is initiated by the specification of one immature neurite and controlled by conserved neuronal polarization-regulating factors, including the partitioning defect 3 (Par3)/Par6/protein kinase C (PKC) complex, activated phosphatidylinositol 3-kinase (PI3K), mitogen activated phosphorylated kinases (MAPK), and glycogen synthase kinase-3β (GSK-3β) (2,3,27). These factors are compartmentalized into the nascent axon and direct cytoskeletal rearrangement, membrane expansion, and growth cone extension. Overexpressing these regulating factors produced the formation of multiple axons (10,14,28). Damaging an existing axon caused a dendrite to become a new axon, suggesting that an intrinsic inhibitory feedback signal is likely present to prevent secondary axonal polarization (6). It is notable that most studies exploring axon specification have been conducted using primary hippocampal neurons, while the molecular mechanisms underlying axon formation in other types of projection neurons remains largely unexplored.
Pluripotent embryonic stem (ES) cells provide an important resource for cell replacement therapy (5,16,20). For instance, integrating ES-derived neurons into host tissue successfully restores motor function, particularly when the engrafted neurons have a high rate of survival (9,11,19). In addition, precise connections of engrafted neurons with targeting neurons is also crucial for preventing undesirable side effects (17,26). Notably, in a recent study, rats developed allodynia-like hypersensitivity following neural stem cell (NSC) engraftments in the spinal cord, a neuropathy that may have stemmed from NSC-secreted paracrine factors causing aberrant axonal sprouting in sensory neurons along with misconnections (12).
Materials and Methods
Maintenance and Differentiation of Embryonic Stem Cells
The HB9 transgenic ES (HB9-GFP ES) cell, a gift from Dr. Thomas Jessell (Columbia University, USA), was derived from HB9-GFP transgenic mice, in which the expression of GFP was only detected in postmitotic motoneurons (21,22). Basal media and supplements for the ES cell culture and differentiation were all obtained from Invitrogen except when noted otherwise. HB9-GFP ES cells were maintained on mitomycin C-treated mouse embryonic fibroblast cells in high-glucose DMEM supplemented with 15% fetal bovine serum, 2 mM glutamine, 0.1 mM nonessential amino acids, 1 mM pyruvate, 0.1 mM 2-mercaptoethanol (Sigma-Aldrich), and 1000 U/ml leukemia inhibitory factor (Chemicon). The neural differentiation methods, including the serum-free embryoid-body-like (SFEB) (24), neurobasal/N2B27 (25), and stroma cell-derived inducing activity (SDIA) methods (15), have been described previously in detail. The day on which ES cells were seeded to differentiate is defined as differentiation day 0. Retinoic acid (0.1 μM, Sigma-Aldrich) was added into the differentiation medium from day 3 to day 5; exogenous sonic hedgehog (Shh; 200 μM, R&D Systems) and purmorphamine (0.2 μM, Tocris) were added and replaced every other day.
Immunohistostaining and Immunocytostaining
Cells were fixed in 4% cold paraformaldehyde and permeabilized with 0.3% Triton X-100. Immunocytochemistry was performed using the following primary antibodies: green fluorescent protein (GFP) (1:200, Aves Labs), MNR2 (1:50, DSHB, Iowa), choline acetyltransferase (ChAT) (1:40, Chemicon), vesicular acetylcholine transporter (VAChT) (1:100, Sigma-Aldrich), microtubule-associated protein 2 (MAP2) (1:100, Convance), Tau 1 (1:100, Chemicon), and Par3 (1:200, Upstate). The antibodies for phosphorylated AKT (pAKT) (1:25), pERKs (1:1000), pJNKs (1:100), and pp38 kinase (1:25) were from Cell Signaling. Cells were washed twice with 0.1% Tween-20 in phosphate-buffered saline (PBS) to remove the first antibody. Appropriate fluorescence-tagged secondary antibodies (all from Jackson Immuno Research) were used for visualization. The 4′,6-diamidino-2-phenylindole (DAPI) was used for nuclear counterstaining. Images of immunostaining were captured using an upright microscope (Nikon ECLIPSE 80I) or confocal microscope (LSM510 Meta, Zeiss).
Flow Cytometry
HB9-GFP+ ES cells and Sox 1+ ES cells (derived from Sox 1-GFP knock-in mouse ES cells; gift from Austin Smith, Cambridge University, UK) (25) on day 6 were fully dissociated with 0.025% trypsin/EDTA and neutralized with ES culture medium. Cells were spun down at 1,000 rpm for 5 min and then resuspended in 500 μl HBSS buffer. Cells were filtered through a 70-μm cell strainer (Becton-Dickinson, Falcon) and analyzed or sorted with flow cytometry (Aria I, nozzle 100 μm, 25 psi, Becton-Dickinson). Cell debris was filtered out by setting thresholds to FSC and SSC parameters.
cDNA Microarray
Total RNA was extracted from the undifferentiated HB9-GFP ES cells, the sorted Sox1+ ES cells, HB9-GFP+ cells, and the mouse cerebellar granule cells on postnatal day 6 using TRIzol C&T (Protech, Taiwan). The quality of RNA was carefully evaluated by the electrophoresis and Bioanalyzer 2100 (Agilent). mRNA (20–50 μg) from each sample was used in each array (Mouse Genome 430 2.0 Array, Affymetrix) by the Affymetrix Gene Expression Service Lab at Academic Sinica, Taiwan (http://ipmb.sinica.edu.tw/affy). The microarray images were scanned and digitized using an Affymetrix scanner (GeneChip scanner 3000). The data were analyzed with Microarray Suite version 5.0 (MAS 5.0) using Affymetrix default analysis settings and global scaling as normalization method. The trimmed mean target intensity of each array was arbitrarily set to 500. The array data were deposited to Gene Expression Omnibus (GEO) repository (Series GSE20899).
Cell Transplantation and Tissue Collection
Eight-week-old mice (C57BL/6; n = 3) weighing 20–25 g were used in this study under permission from the experimental animal committee of Taichung Veterans General Hospital, Taiwan. The mice were anesthetized with 4% isoflurane in induction followed by a maintenance dose (1–2%). HB9-GFP ES cells on day 7 were mechanically pipetted into small cell clumps and concentrated to a density of approximately 1 × 104 cells/μl in neural differentiation medium. The animals received total laminectomy from T8 to T10. Right side microinjections of 2 μl HB9-GFP ES cells into white mater was made at the coordinate of 0.75 mm from the midline and depth of 1.2 mm at the spinal level of T8 and T9 using the infusion pump for 20 min and kept for 5 min after the termination of injection. Left side microinjections of 2 μl HB9-GFP ES cells into the anterior horn were made at the coordinate of 0.5 mm from the midline and depth of 1.2 mm at the same spinal levels and procedures. One animal received 2 μl of PBS injection as a control. One week later, the animals were anesthetized and transcardially perfused with 25 ml of PBS followed by 100 ml of 4% paraformaldehyde. The spinal cord was retrieved and subjected to immunohistochemical staining such as Tau1, GFP, and DAPI. Five consecutive spinal cord sections area (30 μm thickness) of T8 and T9 were obtained for analysis. Of 100 squares with a surface area of 0.01 mm2 each, 20 squares were randomly selected in an ocular grid and used to count the number of GFP+ cells.
Statistical Analysis
One-way analysis of variance (ANOVA) was performed to determine significant differences for the distribution of the phosphorylated MAPK-related kinases in the axon of GFP+ cells from two independent experiments. Tukey post hoc test was further used for multiple comparison of the ratio of the polarized distribution. Statistical significance was established at p < 0.01. In addition, the ratios of bi-axon neurons in different fields of tissue sections were also evaluated by a one-way ANOVA test.
Results
HB9-GFP+ Cells Displayed Characteristics of Motoneurons
A serum-free embryoid body-like (SFEB) method was utilized to direct the differentiation of HB9-GFP ES cells into motoneurons. After treatment with RA (0.1 μM) and Shh (200 ng/ml) on day 3 and day 5, respectively (Fig. 1), morphologically neural-like GFP+ cells were consistently detected on day 8 (Fig. 1B). In previous studies, purmorphamine, a small molecule, has been successfully used as an economical alternative to recombinant Shh protein for ventralizing ES-derived neuroepithelial cells (18). In this study, using purmorphamine (0.2 μM) in place of Shh (200 ng/ml) was similarly effective at inducing motoneuron differentiation from the HB9-GFP ES cells (Fig. 1C). Out of the total cell population, 21.4 ± 3.6% of the cells were GFP+ on day 10 according to the FACS analysis (Fig. 1D).

The generation of motoneurons from HB9-GFP ES cells. (A) The induction procedure for obtaining motoneurons from the ES cells. (B) GFP+ cells were derived from ES cells using retinoic acid (0.1 μM) and sonic hedgehog (Shh; 200 μM). (C) Robust motoneurons were also induced using purmorphamine (0.2 μM). (D) The GFP+ cells were sorted and enriched by cell sorting and subjected to the cDNA microarray analysis, comparing their gene expression to that of undifferentiated ES cells, differentiating Sox1+ ES cells, and cerebellar granule cells on postnatal day 6. (E) Distinct to the undifferentiated ES cells, Sox1+ neuroepithelial cells and cerebellar granule cells, the GFP+ cells showed that 793 genes were at least fourfold upregulated. (F–I) The GFP+ cells were further characterized by the expression of MNR2 (F), choline acetyltransferase (ChAT) (G), vesicular acetylcholine transporter (VAChT) (H), and synaptophysin (Syn) (I). Scale bars: 50 μm (B, C), 20 μm (F).
Highly Expressed Genes in HB9-GFP+ Cells, Compared to Those in Undifferentiated ES Cells

Axon Formation in ES-Derived Motoneurons
To examine the morphogenesis of the ES-derived motoneurons, the embryoid bodies were digested with 1% collagenase I on day 8 and the dissociated cells were plated at a low cell density (1 × 104 cells/ml) on 1% Matrigel-coated culture plates. Using time-lapse microscopy to trace cell fate, we found that most GFP+ cells adhered to and migrated on the coated plate, taking less than 12 h to commit axon formation stage in which a distinct, long axon (>80 μm) was readily distinguished (Fig. 2A, arrows, B, C) (11). Because GFP+ cells consistently emerged on day 8, the de novo neuron morphogenesis of motoneurons could be traced from the postmitosis period to the stage of establishment of motoneuron polarization.

Axon organization in ES-derived motoneurons. (A, B) The morphology of the fluorescent HB9-GFP+ cells (A) on day 10 and their counterpart image with a bright field (B). (C) The dissociated uni-axonal GFP+ cells on day 8 were immunoreactive to the axon marker, Tau1. (D, E) Both neurites of the bipolar GFP+ cells (D) were immunoreactive to Tau 1 (E). (F-H) The bi-axons of GFP+ cells were identified using the expression of ChAT (F), VAChT (G), and synaptophysin (Syn) (H). Arrows highlight the neurites in (F), (G), and (H). Scale bar: 30 μm (A, B, D, E); 10 μm (C, F, G, H).
Surprisingly, while all GFP+ cells expressed the classical motoneuron markers, 33.8% (45/133) of the dissociated single GFP+ cells possessed interneuron-like bipolar morphology (Fig. 2A, arrow head, D). In these bipolar GFP+ cells, both neurites expressed axon markers, including Tau1, ChAT, VAChT, and synaptophysin (Fig. 2E-H). In addition, these cells possessed only two axonal processes and no other small neurites were identified (Fig. 2A, B, D), indicating that dendrite specification was disrupted. These results also suggest that dendrite formation occurs at a later stage than axon polarization in ES-derived motoneurons.
The GFP+ cells that adhered to the culture plate on day 8 initially extended several thin filopodia and formed immature minor cell processes. Most GFP+ cells developed bipolar cell morphology (21/24) and only one filopodia of the cell subsequently developed into an axon (16/21), as revealed by time-lapse confocal microscopy (Fig. 3A). In GFP+ cells that developed bi-axons (4/21), axonal growth was initiated following a state of bipolar organization and then both neurites further extended bilaterally (Fig. 3B). In these bi-axonal GFP+ cells, both axons were a similar length on day 10 (ratio of longer axon/shorter axon: 1.23 ± 0.48). Of interest, one cell with a bifurcate neurite developed a bi-axonal cell morphology following the migration of the cell soma along the neurite (1/21) (Fig. 3C). Only a few GFP+ cells (3/24) still were immature and possessed very short cell processes by day 10. Taken together, these results suggest that the morphogenesis of the motoneuron is initiated at a bipolar organizational stage and consequently develops either a uni-axonal or bi-axonal organization (Fig. 3D).
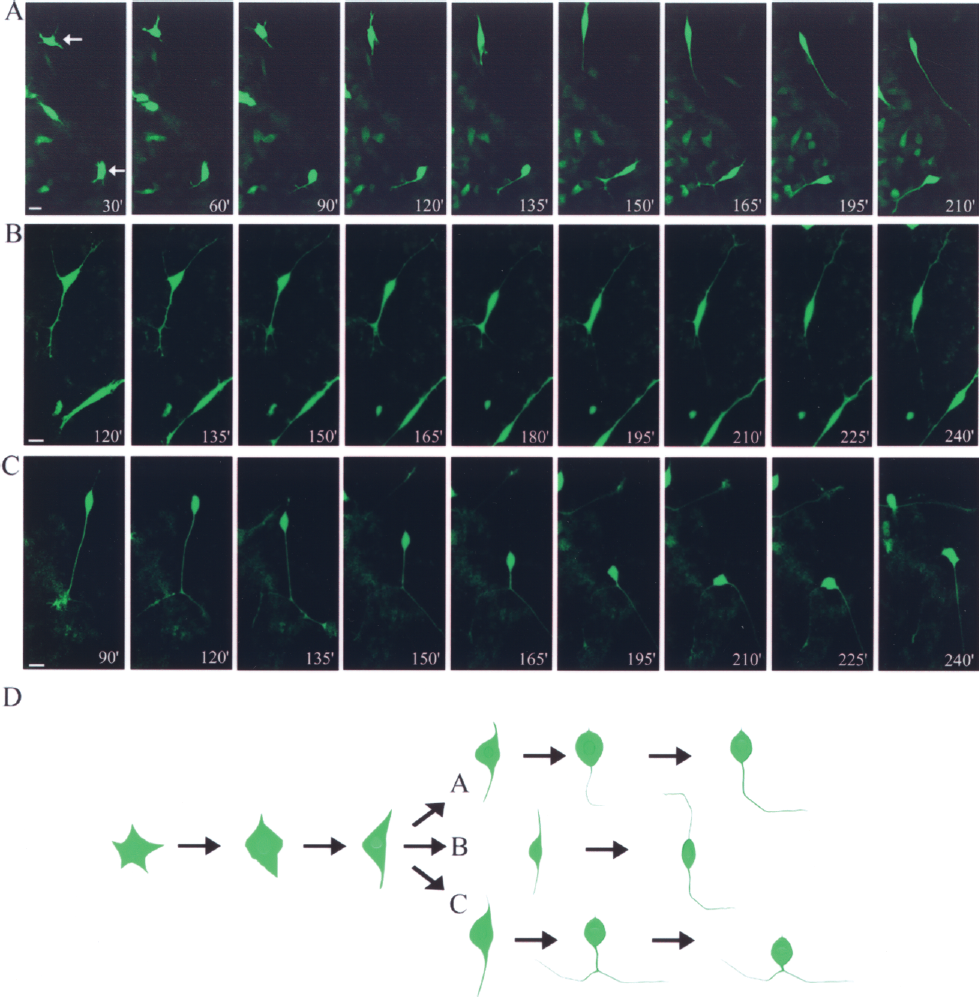
Axon formation in the HB9-GFP+ ES cells. (A–C) Time-lapse confocal microscopy shows three examples of axon morphogenesis. Following the adherence of dissociated GFP+ cells on day 8, serial pictures of axon morphogenesis were taken at 15-min intervals and representative differentiation stages of development are shown. (D) A summary of the neuronal polarity development depicted in (A–C). Scale bar: 10 μm.
Molecules Involved in Axonogenesis
In primary hippocampal neurons, axon specification is intrinsically initiated by the polarization-regulating molecules, including the Par3/Par6/PKC complex, PI3K, and MAPK signals (3). It is intriguing to investigate whether the same polarization-regulating molecules also contribute to the aberrant bi-axon formation observed in motoneurons. While Par3 is an axon-determining factor in hippocampal neurons (Fig. 4A), we failed to detect the Par3 polarization in the ES-derived motoneurons (Fig. 4B, 0/21). In the GFP+ cells displaying a single axon, phosphorylated AKT (pAKT) and pERKs (18/23 and 14/18, respectively) (Fig. 4C, D), but not pJNK and pp38 kinase (2/32 and 3/45, respectively) (Fig. E, F), were often detected in the axon and the cell body proximal to the axon. Statistical analyses supported that polarized distribution of pAKT (10/12, 8/11) or pERK (8/10, 6/8) is significantly correlated to the axonogenesis, comparing to that of pJNK (0/13, 2/19) or pp38 kinase (2/24, 1/21) (p < 0.01). Notably, in the bi-axonal motoneurons, pAKT and pERKs were found in both of the bilateral neurites (9/11 and 14/17 respectively) (Fig. 4G, H), emphasizing the correlation between pAKT/pERK and axon formation in motoneurons.

Compartmentalization of axon specification molecules in motoneurons. (A, B) The distribution of Par3 on day 9 in primary hippocampal neurons (A) and in the GFP+ cells (B). (C–H) The localization of phosphorylated MAPK-related kinases, including pAKT (C), pERKs (D), pJNKs (E), and pp38 kinase (F) in both the uni-axonal GFP+ cells (C–F) and the bi-axonal GFP+ cells (G–H). Arrows highlight the dendrites of GFP+ cells. Scale bar: 15 μm.
Engrafting ES-Derived Motoneurons Into the Spinal Cord
To investigate the influence of microenvironment on the differentiation of motoneurons, HB9-GFP+ cells were injected into different milieu including the anterior horn (AH) and white matter (WM) of spinal cord (Fig. 5A). Around 30% of grafted cells, either in the location of gray mater or anterior horn, developed into differentiating neurons by 1 week after transplantation. Most of engrafted cells showed the typical pattern of single axons (Fig. 5B); however, 31.6 ± 2.4% and 31.3 ± 1.5% of the engrafted GFP+ cells over the anterior horn of T8 and T9, respectively, showed the aberrant pattern of bi-axon morphology (n = 1,501) (Fig. 5C, E). The engrafted cells over WM also showed the same trends (32.6 ± 1.8% and 31.3 ± 1.5% in T8 and T9 sections, respectively) (n = 1,441) (Fig. 5D, E). Furthermore, there was no statistical difference either for the ratios of bi-axon neurons or those in different engrafting area (p > 0.05) (Fig. 5E). Similar to the in vitro result of ES differentiation, we did not observe multiaxonal motor neurons after grafting. From this in vivo study, we found that the aberration morphology of motor neuron differentiation from HB9-GFP ES was not altered by the mature microenvironment of spinal cord.

Bi-axonal GFP+ cells were detected in the engrafted spinal cord. (A) Cryosectioned spinal cord tissue with PBS (mock) (left panel) or ES cell engraftment (right panel) was immunostained using GFP (green) antibody on posttransplantation day 7. The arrowheads indicate the engrafted sites of anterior horn (AH) and the white matter (WM). (B) A typical uni-axonal GFP+ cell was stained with Tau1 (red). (C, D) The bi-axonal GFP+ cells were found in AH (C) and in WM (D). Figures were illustrated using compiled fluorescent intensity of confocal z-stacks of captures. (E) The statistic ratio of bi-axon neurons in the engrafted GFP+ cells was estimated in the T8 and T9 section of the spinal cord. Scale bar: 10 μm (B, C, D).
Discussion
ES-derived neurons can be continually traced in vitro from the stages of postmitosis to axon initiation, elongation, and maintenance, providing a new system for studying de novo axon formation and the effects of intrinsic neural polarization factors. In contrast, the isolated hippocampal neurons from neonatal rat are polarized prior to dissociation and reinitiate cellular polarization after being plated in vitro (3). The new axon determination among immature neurites may be affected by the restricted distribution of preexisting axon-regulating factors, which are already localized in the axon tip of hippocampal neuron before the cell isolation.
Neurons with different morphologies may utilize distinct molecular mechanisms to organize their cellular architecture. While the establishment of neuronal polarity has been extensively studied using rat hippocampal neurons, this process has been largely unexplored in mammalian motoneurons. When examining at axonogenesis in ES-derived motoneurons, our study revealed that the immature cellular processes extended by the GFP+ cells were highly dynamic and typically not preserved after the first 3 h of tracing. Later the GFP+ cell retracted many of its initial cellular processes and developed a bipolar organization. In most of the GFP+ cells, one of the immature neurites underwent axonal specialization, which was accompanied by the compartmentalization of intrinsic polarizing factors into the axon. This mechanism for axon formation in the ES-derived motoneuron is similar to the model for bipolar definition (7), but distinct from the stochastic model (2), in the primary hippocampal neurons.
We showed that two thirds of the total GFP+ cells were precisely configured and correctly programmed to form the normal uni-axonal conformation, indicating that the appropriate intrinsic regulators were present and likely play a critical role in the morphogenesis of motoneurons. In hippocampal neurons, an inhibitory feedback signal is necessary to prevent the respecification of additional axons from dendrites and maintain the correct axon configuration (1,2). In the HB9-GFP+ cells, one third of the nascent motoneurons extended bilateral cell processes that developed into aberrant bi-axonal configuration, suggesting the similar inhibitory mechanism may be absent in ES-derived motoneurons. However, regulatory factors contributing to appropriate bipolar motoneuron development are not well understood, and we cannot exclude the possibility that environmental cues, such as an extracellular matrix or diffusible chemoattractive factors, must be precisely integrated with intrinsic signals to orchestrate axon formation.
In this study, the ES-derived cells were dissociated with collagenase and were seeded at low cell density to record single GFP+ cells. Notably, we observed that bi-axonal GFP+ cells also developed when other neural-inducing approaches were utilized, including the SDIA (15) and neurobasal/N2B27 methods (25) (data not shown). However, precisely estimating the ratio of bi-axonal formation in GFP+ cells in either of these adherent culture systems is difficult because the attached ES-derived colonies showed extensively tangled neurites and were highly vulnerable to trypsin digestion on day 8 or day 9.
The effects of bi-axon motoneuron after transplantation still require further exploration. We did not observe the dendrite formation in the bi-axon motoneuron, implying that these cells may not transmit functional action potential to their connecting cells. The bi-axon motoneurons may serve as bridging cells to establish a stable connection of engrafted cells with host tissue and help the survival of engrafted neurons. However, we also did not rule out the possibility that the bi-axon motoneuron might be activated by adjacent neurons and consequently transmit inappropriate electric signals to sensory neurons or interneurons. The resulting misconnection might lead to allodynia-like neuropathy, thereby restricting the application of this engraftment for treating spinal cord injury (12).
Taken together, here we discovered that ES-derived motoneurons developed bi-axonal processes in vitro and vivo. Understanding the detailed cellular mechanisms underlying axonal and dendritic specification will allow us to rectify the aberrant bipolar morphogenesis of motoneurons and thereby increase the safety of stem cell-based therapy.
Footnotes
Acknowledgments
This work was supported by the National Science Council (H.L.S., NSC 96-2321-B-005-007-MY3) of Taiwan, the grant from the Taichung Veterans General Hospital and National Chung Hsing University (TCVGHNCHU-9776614 and −977602; to H.L.S and H.C.P.), Taichung, Taiwan, and by Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH99-TD-B-111-004). We thank Dr. Thomas Jessell and Austin Smith for the gift of HB9-GFP ES cells and Sox1-GFP knock-in ES cells, respectively. Dr. Yi-Ling Lin and the Affymetrix Gene Expression Service Lab (http://ipmb.sinica.edu.tw/affy/) in Academia Sinica, supporting the cDNA microarray assays, are highly appreciated. Author contributions were: P.-H.C., Y.-T.W., H.-L.S. (conception and design, collection, and/or assembly of data, data analysis and interpretation, manuscript writing, final approval of manuscript); S.-C.S., C.-C.W., M.-S.T., F.-C.C., S.-Z.L., C.-W.C., C.-S.L. (collection and/or assembly of data, data analysis and interpretation; conception and design, collection and/or assembly of data, data analysis and interpretation, manuscript writing).
