Abstract
Diabetic patients suffer from impaired wound healing, characterized by only modest angiogenesis and cell proliferation. Stem cells may stimulate healing, but little is known about the kinetics of mobilization and function of bone marrow progenitor cells (BM-PCs) during diabetic wound repair. The objective of this study was to investigate the kinetics of BM-PC mobilization and their role during early diabetic wound repair in diabetic db/db mice. After wounding, circulating hematopoietic stem cells (Lin-c-Kit+Sca-1+) stably increased in the periphery and lymphoid tissue of db/db mice compared to unwounded controls. Peripheral endothelial progenitor cells (CD34+VEGFR+) were 2.5- and 3.5-fold increased on days 6 and 10 after wounding, respectively. Targeting the CXCR4—CXCL12 axis induced an increased release and engraftment of endogenous BM-PCs that was paralleled by an increased expression of CXCL12/SDF-1α in the wounds. Increased levels of peripheral and engrafted BM-PCs corresponded to stimulated angiogenesis and cell proliferation, while the addition of an agonist (GM-CSF) or an antagonist (ACK2) did not further modulate wound healing. Macroscopic histological correlations showed that increased levels of stem cells corresponded to higher levels of wound reepithelialization. After wounding, a natural release of endogenous BM-PCs was shown in diabetic mice, but only low levels of these cells homed in the healing tissue. Higher levels of CXCL12/SDF-1α and circulating stem cells were required to enhance their engraftment and biological effects. Despite controversial data about the functional impairment of diabetic BM-PCs, in this model our data showed a residual capacity of these cells to trigger angiogenesis and cell proliferation.
Introduction
Wound healing integrates multiple biologic processes involving several types of adult and stem cells (3,17,40). Although there is an increasing prevalence of chronic wounds in the diabetic population, the exact cellular and molecular mechanisms underlying the pathogenesis of diabetic healing have not been fully elucidated (33,36). In addition, the mobilization and role of endogenous bone marrow progenitor cells (BM-PCs) during wound repair in diabetes have not yet been investigated.
In diabetic patients, a number of hyperglycemia-dependent mechanisms have been identified to help explain the defects in skin repair ability, but effective strategies to correct such defects are still far from being available (33,36). Large randomized clinical trials have concluded that offloading and prevention are still the best treatment option for diabetic ulcers (10). Experimental preclinical studies of diabetic wounds have found decreased numbers of fibroblasts, expression of aberrant growth factor, altered inflammatory responses, and enhanced glycosylation of proteins with progressive accumulation of advanced glycation end-products (33,36). All these factors may contribute to the altered wound-healing process in diabetes (33,36) and may influence the migration and function of BM-PCs during tissue repair.
Among BM-PCs, endothelial progenitor cells (EPCs) may have a role in the revascularization of diabetic wounds, as they actively participate in adult neoangiogenesis (7,45,47). Another pool of BM-PCs, hematopoietic stem cells (HSCs) committed to lymphoid/myeloid differentiation, may also enhance wound healing by stimulating the inflammatory phases (5,6,11,25,37,38) and the proliferation phase of diabetic wounds. While these cells have mostly been studied in healthy conditions, little is known about their kinetics of mobilization and recruitment during diabetic repair in vivo.
The leptin receptor-deficient diabetic (db/db) mouse has been found particularly suitable for studying diabetic wound healing, among the experimental models for diabetes type 2 (20). Compared to wild-type littermates, full-thickness wounds on db/db mice are characterized by delayed closure and reduction in angiogenesis and collagen deposition (20), similar to healing in diabetic patients, making the model particularly suitable for studying stem cells in diabetic conditions.
Several authors have claimed that a defect in stem cell regenerative capability in diabetic subjects and in diabetic mice may contribute to impaired wound healing (5,6,11,25,37,38). In this study, we aimed to characterize the kinetics of mobilization and recruitment and the main functions of endogenous BM-PCs during diabetic wound healing.
Materials and Methods
Mice
Twelve-week-old diabetic male mice (strain C57BL/KsJ-Leprdb, db/db; n = 10 per group, 30—40 g; Jackson Laboratories, Bar Harbor, ME), which carry a single autosomal recessive mutation on chromosome 4 and display leptin deficiency that causes characteristics similar to those of human type 2 diabetes, were used to study wound healing (15). Fasting blood glucose levels were measured by a Glucometer (Elite XL 3901E; Bayer, Tarrytown, NY) in blood extracted from the tail vein on a weekly basis. Diabetic db/db mice developed diabetes at 7—8 weeks of age and had fasting serum glucose levels >250 mg/dl for a minimum of 20 days before experiments began. Their glucose levels during the experimental period were typically 400—450 mg/dl.
Animal Interventional Studies
On day 0 (baseline), 20 diabetic db/db mice received a full-thickness wound (through to the level of the panniculus carnosus muscle) on the dorsum. They were then randomly allocated to receive treatment with the CXCR4 antagonist (NIBR1816, labeled CXCR4Ant) or vehicle (saline, labeled wounded). A group of diabetic db/db mice (n = 10) received NIBR1816 for 5 days at a dose of 30 mg/kg/day IP (~1 mg/day) diluted in 0.25 ml of saline, following a protocol previously established in islet transplantation. In another group of diabetic db/db mice (n = 10), vehicle (saline 0.25 ml daily injected IP) was injected for the same period. We also performed a clinically relevant experiment in which CXCR4 antagonist treatment was given in association with an agonist of BM-PC mobilization: the recombinant murine granulocyte-macrophage colony-stimulating factor (rmGM-CSF), 1 μg diluted in 100 μl saline, administered IP daily for 5 days. In addition, diabetic db/db mice (n = 10) were injected with CXCR4 antagonist together with ACK2 mAb (anti-CD117 mAb or anti-c-Kit mAb, 250 μg at 0 and 5 days), which has been shown to abrogate BM-PC mobilization in various models by blocking stem cell function and mobilization (24).
Wound Model and Treatment Procedure
The db/db mice were used under an approved animal protocol in an AAALAC-accredited facility. The day before surgery, hair at the wound site was clipped and depilated (Nair®, Church & Dwight Co., Princeton, NJ). On the day of surgery, animals were weighed and anesthetized with 60 mg/kg Nembutal (pentobarbital). A dorsal 1.0-cm2 area of skin and panniculus carnosus was excised, and the wounds were photographed (32).
Wound Closure Analysis
Digital photographs captured on day 7 were compared with initial photographs by two independent treatment-blinded observers using planimetric methods (Image J, NIH, Bethesda, MD). Wound closure was quantified by measuring contraction, reepithelialization, and open wound as a percentage of the original wound area.
Contraction is measured at the level of the initial excision line (which progressively becomes smaller by contraction), reepithelialization is measured between the contraction line and the inner edge of the translucient layer (which corresponds at the epithelium sliding onto the wound), and open wound is the inner raw surface (red, not covered with epithelium macroscopically). The sum of contracted (C%), reepithelialized (E%), and open wound (O%) areas any given day equals 100% of the original wound size (32).
Wound Watch Staging System: Quantification of Vascular Area and Cell Proliferation
High-powered images of stained sections were used to quantify the degree of proliferation and vascularization and to compare treatments with spontaneous healing. Three digital images of PECAM-1- and Ki-67-stained slides were captured for each sample, one in the middle and two at the edges of the wound bed. Pictures were viewed with Adobe Photoshop CS Software (Adobe Systems Inc., San Jose, CA), and blood vessels and proliferating nuclei in each high-powered field were marked and counted and expressed as a ratio of proliferating nuclei (Ki-67 positive) to total nuclei. Ratios were calculated between results obtained from the center of the lesions and from the edges of each treatment group to the plasma-treated group. Fifteen microscopic fields at 40x magnification were used in each treatment arm (32).
Microscopic Analysis
Central wound cross sections (n = 5 per group) were embedded in paraffin, sectioned, and stained according to routine hematoxylin and eosin (H&E) protocols. Panoramic cross-sectional digital images of each wound were prepared using Adobe Photoshop CS Software. These sections were analyzed with digital planimetry (Image J, NIH) by two independent treatment-blinded observers to quantify wound tissue. Granulation tissue in plasma-treated wounds was arbitrarily set to 100%.
Immunohistochemistry
PECAM-1 and Ki-67 Staining. Paraffin-embedded sections (n = 5 per group) were rehydrated, and antigen was retrieved for Ki-67 analysis by microwaving in 10 mM sodium citrate (pH 6.0) for 10 min. Sections for platelet endothelial cell adhesion molecule-1 (PECAM-1) were treated with 40 μg/ml proteinase K (Roche Diagnostics Corp., Basel, Switzerland) for 25 min at 37°C. PECAM-1 (Pharmingen, San Jose, CA) and Ki-67 (Dako Corp., Carpinteria, CA) primary antibodies were incubated at 4°C overnight. PECAM-1 and Ki-67 signals were intensified with the tyramide amplification system (PerkinElmer, Boston, MA). In PECAM-1-stained slides, the total number of blood vessels (positive for PECAM-1) was counted under 40x magnification. In Ki-67-stained slides, the ratio of proliferating nuclei (positive for Ki-67) to total nuclei was quantified under 40x magnification; 10 microscopic fields at 40x magnification were used in each treatment arm and immunolocalization condition.
CXCL12 and phosphoMAPK Stainings. Immunohistochemistry reagents were peroxidase blocking reagent, DAB chromagen, and substrate buffer (Dako Cytomation), citrate buffer, pH 6, and antibody diluent (Invitrogen, Carlsbad, CA), the avidin/biotin blocking kit, biotinylated horse anti-mouse and goat anti-rabbit antibodies (Vector Laboratories, Inc., Burlingame, CA), Mayer's hematoxylin solution (Sigma, St. Louis, MO), and Permount (Fisher Scientific, Pittsburg, PA). Primary antibodies were anti-CXCL12, clone 79018.111, and rabbit anti-phospho-p44/p42 MAP kinase (Thr 202/Tyr 204) (Cell Signaling Technology, Beverly, MA). Sections (4 μm) of formalin-fixed, paraffin-embedded mouse skin were deparaffinized and rehydrated in a xylene-ethanol series. For antigen retrieval, slides were heated in citrate buffer in a decloaking chamber (Bio-care Medical, Concord, CA) for 2 min. Peroxidases were quenched with peroxidase blocking reagent for 15 min. Blocking in 5% horse or goat serum in PBS with avidin block was performed for 1 h for SDF-1 and phospho-MAP kinase IHC, and these antibodies were used at 1: 200 and 1:100 dilutions, respectively, in PBS with biotin block. Without blocking, clone 1D11 was used in antibody diluent at a 1:150 dilution. All primary antibody reactions were at 4°C overnight. Incubations with biotinylated antibodies (30 min) and ABC reagents (15 min) were followed by addition of DAB chromagen. Slides were counterstained with hematoxylin, dehydrated, and cover-slipped with Permount.
OCT3/4 and CD34 Staining. For immunohistochemical stains, sections were mounted on poly-l-lysine-coated slides, deparaffinized, treated with 3% hydrogen peroxide for 10 min to inhibit endogenous peroxidase, and pretreated with 0.01 M citrate buffer, pH 6, for 10 min in a microwave oven at 650 W. Then sections were incubated with primary antibody (monoclonal anti-CD34 clone MEC14.7, 1:50 dilution; Novus Biologicals, Littleton, CO; polyclonal anti-OCT-3/4,1:500 dilution, Santa Cruz Biotechnology Inc., Santa Cruz, CA) at 4°C overnight followed by the avidin-biotin complex (ABC) procedure (22). Immunoreactions were developed using 0.03% 3,3′-diaminobenzidine tetrahydrochloride, and sections were counterstained with Harris's hematoxylin. Specificity controls consisted of absorption of antibodies with 10—20 nM of their related antigens, substitution of the primary antibody with nonimmune serum of the same species at the same dilution, and use of control tissues with or without the pertinent antigen.
Flow Cytometry (FACS)
KLS cells (Lin-c-Kit+Sca1+) were identified by FACS analysis by gating on lineage-negative cells [using a biotinylated cocktail of Gr1, CD8, CD4, CD11b, and B220 cells (BD Biosciences, San Jose, CA) followed by incubation with streptavidin FITC (BD Bisociences)] and then gating on APC anti-mouse CD117 (c-Kit+ (BD Biosciences) and PE anti-mouse Sca-1+ (eBioscience, San Diego, CA) cells. BM progenitors were analyzed in the blood and spleen; however, the spleen was chosen as the preferred examination site because of the very low percentage of BM progenitor cells in the blood. Endothelial progenitor cells (EPCs) were evaluated as CD34+VEGFR+ double-positive cells [using FITC anti-mouse CD34 (BD Biosciences) and monoclonal anti-mouse VEGF R2/Flk-1 (R&D Systems) followed by PE anti-rat IgG1 (BD Biosciences)]. Cells recovered from spleens were thus subjected to FACS analysis and were run on a FACSCalibur™ (Becton Dickinson, San Jose, CA). Data were analyzed with FlowJo software version 6.3.2 (Treestar, Ashland, OR).
Wound Harvesting and Tissue Dispersion for FACS Analysis
Wounds were harvested under a surgical microscope by excising the full wound and digesting the tissue with an overnight digestion procedure as previously reported (43). Briefly, harvested tissue was cut into 2-mm pieces and incubated overnight at 4°C in 5 ml of solution A for each gram of tissue [HBSS containing 1 mg/ml dispase I (Roche Molecular Biochemicals, Indianapolis, IN), 3% heat-inactivated FBS, and 10 mg/ml penicillin-streptomycin (Cambrex, Charles City, IA)]. The next morning, the remaining tissue was removed from solution A and further minced as finely as possible. Solution B (20 μl) [RPMI containing 1 mg/ml hyaluronidase I (Sigma), 1 mg/ml collagenase D (Roche Molecular Biochemicals), 150 U/ml DNase I (New England Biolabs, Beverly, MA), and 5 mg/ml penicillin-streptomycin] was added for each gram of tissue and incubated in a shaking 37°C water bath for 2 h, after which the suspension was agitated and combined with solution A. The combined solutions were passed through a 70-μm cell strainer, centrifuged, washed twice in PBS + 2% FBS, and resuspended in 1 ml PBS containing 2% FBS and 0.1% sodium azide. A manual cell count was performed, and cell viability was assessed by trypan blue exclusion. Cells extracted were then stained and run on a FACSCalibur.
Statistical Analysis
Data are expressed as mean ± SE. ANOVA (for parametric data) and Kruskal-Wallis test (for nonparametric data) were used. When two groups were compared cross-sectionally, two-sided unpaired Student' t-test (for parametric data) or Mann-Whitney test (for nonparametric data) were used according to distribution. Bonferroni correction was performed when multiple comparisons were performed. A value of p < 0.05 (by two-tailed testing) was considered an indicator of statistical significance. Data were analyzed using an SPSS statistical package for Windows (SPSS Inc., Chicago, IL). Prism software was used for drawing graphs (GraphPad Software, Inc., San Diego, CA).
Results
Peripheral BM-PCs in db/db and WT Mice
The percentage of Lin-Sca-1+c-Kit+ (KLS), the murine hematopoietic stem cell pool, as well as of CD34+VEGFR+ (EPCs), the endothelial progenitor cell pool, was analyzed by FACS on splenocytes and blood obtained from diabetic db/db mice and from regular WT C57BL/6 mice (n = 10 mice per group). No significant differences were evident as far as KLS or EPCs in the spleen or blood of unwounded db/db mice compared to unwounded WT (Fig. 1 and data not shown).

The percentage of hematopoietic stem cells (KLS cells, Lin-Sca-1+c-Kit+) and endothelial progenitor cells (EPCs, CD34+VEGFR+) in the spleens of diabetic db/db mice and from regular WT C57BL/6 mice (n = 10 mice per group) was analyzed by FACS. No major differences in the aforementioned cell populations were evident between the two groups at baseline.
Spontaneous and Enhanced Kinetics of Mobilization of BM-PC During Diabetic Wound Repair
We next sought to determine whether wounding would have an effect on the release of stem cells in db/db mice. On day 6 after wounding, the KLS population in the spleen increased approximately twofold in db/db mice compared to unwounded animals (wounded = 1.20 ± 0.39 vs. baseline = 0.65 ± 0.11%, p < 0.05) (Fig. 2A). On day 10, the KLS percentage continued to increase in the spleen and remained higher in wounded db/db animals compared to baseline (wounded = 1.65 ± 0.22% p < 0.05) (Fig. 2A).

The percentage of Lin-Sca-1+c-Kit+ (KLS) cells increased on day 6 in both wounded + CXCR4Ant-treated and wounded db/db mice compared with baseline (both p < 0.01 vs. baseline) (A). On day 10, a further increase in KLS percentage was evident in the wounded + CXCR4Ant-treated diabetic db/db mice but not in the wounded mice [D10-wounded + CXCR4Ant-treated vs. baseline (*p < 0.05) and vs. D10-wounded (**p < 0.01)] (A). An increase in the percentage of peripheral CD34+VEGFR+ cells (EPCs) was evident on day 6 in wounded + CXCR4Ant-treated diabetic db/db mice compared with wounded diabetic db/db mice (**p < 0.05) and with baseline (*p < 0.01) (B). On day 10, the number of EPCs continued to rise in wounded + CXCR4Ant-treated mice and in wounded diabetic db/db mice compared with baseline [D10-wounded + CXCR4 Ant-treated and D10-vounded vs. baseline (p < 0.01)] (B). Mice wounded and treated with CXCRAnt showed increased levels of stem cells (OCT3/4+ or CD34+ cells) in wounds compared to untreated mice (C, D, E, F). Corresponding to the increased levels of circulating and engrafted stem cells, wounds treated with CXCR4Ant exhibited elevated expression of MAP kinases and CXCL12 (SDF-1 α), the ligand of stem cells (G, H, I, J).
We then evaluated in the spleen the percentage of EPCs, which have been shown to have a role in angiogenesis in healthy subjects and are both fewer in number and functionally deficient in diabetes. On day 6, the percentage of EPCs in the spleen increased twofold in wounded animals compared with baseline (wounded = 2.25 ± 0.15 vs. baseline = 1.10 ± 0.14%; p < 0.05) (Fig. 2B). On day 10, the number of EPCs in the spleen continued to rise in the periphery compared with baseline (wounded = 3.56 ± 1.50%; vs. baseline, p < 0.01) (Fig. 2B). These findings were also confirmed in peripheral blood, but the rather low number of HSCs and EPCs makes such an evaluation uncertain.
BM progenitor cells express high levels of CXCR4, which anchors them to the CXCL12 (CXCR4 ligand)-rich BM stroma (8,9). CXCR4 antagonists mobilize BM-PCs to the periphery by blocking the interaction between the CXCR4 receptor expressed by BM-PCs and CXCL12 expressed by reticular cells (8,9,41). Here, by using a CXCR4 antagonist with a short half-life (~2—4 h) (42), we aimed to target the CXCR4—CXCL12 axis determine whether a further mobilization of autologous BM-PCs in the periphery of wounded diabetic mice was possible. We treated db/db mice for 5 days with 30 mg/kg/day (1 mg/day per mouse) of CXCR4 antagonist (NIBR1816, CXCR4Ant), as a short-term protocol to avoid excessive depletion of BM.
On day 6, the KLS population increased in CXCR4Ant-treated db/db mice compared to baseline, but only to the same levels as seen in wounded, un treated mice (treated = 1.25 ± 0.37, p < 0.01) (Fig. 2A). On day 10, 5 days after the end of the treatment, a further increase in KLS percentage was evident in the CXCR4Ant-treated diabetic db/db mice but not in the wounded untreated mice, where KLS nevertheless remained higher than baseline (CXCR4Ant-treated = 2.34 ± 0.30 vs. wounded untreated = 1.65 ± 0.22%; CXCR4Ant-treated vs. baseline and vs. wounded untreated, p < 0.01 and p < 0.05, respectively) (Fig. 2A).
When we evaluated the percentage of peripheral EPCs on day 6 after wounding, the percentage of EPCs had increased to a greater degree in CXCR4Ant-treated diabetic db/db mice compared with wounded untreated diabetic db/db mice and unwounded baseline (CXCR4Ant-treated = 3.48 ± 0.43 and wounded untreated = 2.25 ± 0.15 vs. baseline = 1.10 ± 0.14%; CXCR4Ant-treated vs. baseline and vs. wounded untreated, p < 0.01 and p < 0.05, respectively) (Fig. 2B). On day 10, the number of EPCs continued to rise in CXCR4Ant-treated mice and also in the wounded untreated diabetic db/db mice compared with baseline (CXCR4Ant-treated = 3.60 ± 1.88 and wounded untreated = 3.56 ± 1.50%; CXCR4Ant-treated and D10-wounded untreated vs. baseline, p < 0.01) (Fig. 2B).
An Increased Level of HSCs and EPCs in the Wound of Treated Mice Is Associated with a Modulation of the Wound Bed
To better characterize the effects of the release of endogenous BM-PCs after wounding, we performed FACS and immunohistochemistry studies evaluating “stemness” marker expression in the wounds of db/db mice. Day 10 was selected as the intermediate stage of healing, as the primary difference between diabetic human and murine wounds is the natural tendency of murine wounds to heal spontaneously by contraction, regardless of treatment. As shown in previous studies (31,32), the db/db mouse reaches approximately 50% healing on day 10, and although macroscopic differences tend to fade, it is at this point possible to observe histological changes induced by different therapies (30). CXCR4Ant-treated diabetic db/db mice on day 10 showed increased levels of HSCs and EPCs in the wound compared with vehicle-treated mice when the same tissues were subjected to FACS analysis (data not shown). On day 10, the wounds of CXCR4Ant-treated db/db mice exhibited increased (up to 5%) numbers of OCT3/4+ and CD34+ cells, suggestive of stem cells (Fig. 2D, F), while after wounding only scarce spontaneous engraftment (lower than 2%) of OCT3/4+ or CD34+ cells was noted (Fig. 2C, E).
To further evaluate the effect of increased BM-PC engraftment in diabetic wounds, we performed immunohistological studies in which we analyzed the expression of two important pathways relevant to wound healing: (i) the pMAPKinase, common to many proliferative pathways highly activated during efficient tissue repair (21) and (ii) the chemoattractant factor CXCL12 (or SDF-1α) natural ligand of stem cells (19).
These studies revealed that increased peripheral levels of stem cells may profoundly influence wound healing. Wounds obtained from CXCR4Ant-treated diabetic db/db mice showed considerably increased levels of pMAPKinase (2.5-fold increase, p < 0.05), which is involved in cell proliferation and migration, compared with wounded db/db mice (Fig. 2G, H) (21). Surprisingly, the increased levels of stem cells in the periphery corresponded to an increased expression of CXCL12 (CXCR4 ligand, threefold increase, p < 0.01) in diabetic wound tissues compared with those wounded and left untreated (Fig. 2I, J). Some studies have implicated low levels of CXCL12 (19) as one of the potential mechanisms involved in the impaired wound healing of diabetic db/db mice. Increased levels of expression of CXCL12 may explain the local recruitment of stem cells.
Taken together, these results suggest a natural shedding of stem cells from the bone marrow during wound healing, with EPCs mobilized earlier than KLS cells as a response to tissue damage. In addition, spontaneous recruitment and engraftment of stem cells in diabetic repair is inefficient, and increased levels of stem cells in circulation corresponded to increased levels of these cells in wounded tissues.
BM-PC Peripheral Mobilization Enhances Neoangiogenesis and Cell Proliferation in Diabetic db/db Mice Wounds
To characterize the physiological effects of BM-PCs during tissue repair, wounds were harvested on day 10 from untreated and CXCR4Ant-treated animals.
Wounds from CXCR4Ant-treated diabetic db/db mice showed increased levels of vascularization (as demonstrated by PECAM-1 staining), with up to 3.5fold increases in angiogenesis compared to untreated wounded db/db mice (*p < 0.05) (Fig. 3A—C). We next evaluated the possibility of increasing the effects of CXCR4Ant treatment by using recombinant murine granulocyte-macrophage colony-stimulating factor (rmGM-CSF), which may synergize with the CXCR4 antagonist in mobilizing BM-PCs (39). Therefore, if successful, rmGM-CSF could be important in clinical applications, given the availability of long-acting compounds for human use (1,2,4). Finally, we used the ACK2 mAb (an anti-CD117 mAb or anti-c-Kit mAb), known for its ability to abrogate BM-PC mobilization, to demonstrate a link between BM-PC mobilization and improved wound healing.

On day 10, wounds from CXCR4Ant-treated diabetic db/db mice showed increased vascularization (as shown by PECAM-1 staining) compared to wounded mice (*p < 0.05) (A—C). RmGM-CSF and ACK2 mAb added to CXCR4 decreased angiogenesis in the wounds, although vascularization levels tended to remain higher than in wounded mice in the CXCR4Ant + ACK2-treated mice (**p < 0.05) (C). Arbitrary units means fold increase over wounded animals.
At day 10 spleens were harvested from the three different groups and BM-PCs were evaluated by FACS. Surprisingly, adding rmGM-CSF did not elicit mobilization of an increased number of BM-PCs (data not shown), while, as expected, ACK2 antagonized BM progenitor cells' mobilization and decreased HSCs and EPCs peripheral levels at day 10. Particularly, HSC (p = 0.03) and EPC (p = 0.02) percentages were reduced in the AKC2/CXCR4 antagonist cotreated group compared to the group treated with CXCR4Ant alone, but remained higher compared to untreated mice (data not shown). The addition of GM-CSF did not seem to have a specific effect on angiogenesis in diabetic wound repair, neither enhancing nor abrogating the effects induced by CXCR4Ant (Fig. 3C); however, CXCR4Ant + ACK2-treated mice still retained higher vascularization compared to controls (**p < 0.05) (Fig. 3C).
Analysis of cell proliferation by Ki-67 expression showed that treatment with CXCR4Ant induced a 2.5fold increase over wounded untreated db/db mice (*p < 0.05), (Fig. 4A—C). While the addition of GM-CSF did not further stimulate cell proliferation but maintained a proliferation higher than wounded controls (*p < 0.05) (Fig. 4C), ACK2 decreased the level of proliferating cells compared to that found in CXCR4Ant-treated db/db mice, although levels remained higher than in untreated wounded db/db mice (**p < 0.05) (Fig. 4C).

On day 10, wounds from CXCR4Ant-treated diabetic db/db mice showed increased cell proliferation (as shown by Ki-67 staining) compared to wounded mice (*p < 0.05) (A—C). When rmGM-CSF was added to CXCR4Ant treatment, no changes in cell proliferation were measured on day 10, but maintained a proliferation higher than wounded controls (*p < 0.05) (C). ACK2 mAb added to CXCR4 decreased the levels of cell proliferation, although levels remained higher than in untreated wounded db/db mice (**p < 0.05) (C). Arbitrary units means fold increase over wounded animals.
Results suggest the stimulation of angiogenesis and cell proliferation through the mobilization of KLS and EPC cells by CXCR4Ant, while combination therapies did not produce clinically relevant improvements. As a consequence, we studied the effects of the mobilization of stem cells by CXCR4Ant by doses lower and higher doses than the ones used in the study thus far. In addition, we monitored the effects of the CXCR4Ant on healthy wild-type mice.
The administration of the CXCR4Ant for 5 days, to shed high numbers of stem cells from the bone marrow and avoiding its depletion, resulted in the highest stimulation of angiogenesis and cell proliferation compared to a 1-day and a 10-days injection schedule (data not shown). Because all our results illustrate a natural recruitment of stem cells associated with defective engraftment during diabetic wound repair, it was not surprising to find that the CXCR4Ant had no effect in healthy mice, whose recruitment and engraftment mechanisms are naturally highly efficient.
The Effects of the Mobilization of BM-PC on Wound Closure in Diabetic db/db Mice
We next evaluated the effect of endogenous BM-PCs on wound closure by comparing the clinical and histological aspects of wounds in untreated animals to those in CXCR4Ant-treated mice. On day 4, CXCR4Ant-treated diabetic db/db mice showed improved wound closure compared with controls. CXCR4Ant-treated mice reached 14.8 ± 4.3% wound closure compared with 2.3 ± 3.4% wound closure in vehicle-treated diabetic db/db mice (*p = 0.04) (Fig. 5A). The primary mechanism contributing to enhanced wound closure on day 4 in CXCR4 antagonist-treated animals was found to be reepithelialization (Fig. 5B), which was increased by twofold on day 4 in the wounds of CXCR4Ant-treated db/db mice compared with vehicle-treated animals (*p = 0.04). On the same day, wound contraction was similarly superior in treated animals (*p = 0.03) (Fig. 5C). On day 7, CXCR4Ant-treated animals showed a similar tendency toward more rapid wound closure than vehicle-treated controls (vehicle-treated = 25.4 ± 4.0 vs. CXCR4Ant-treated = 42.6 ± 6.0%, **p = 0.03) (Fig. 5A). Similar to what was observed on day 4, CXCR4 antagonist-treated diabetic db/db mice showed more reepithelialization than vehicle-treated diabetic db/db mice (*p = 0.04) (Fig. 5B), while wound margins in the two groups reached comparable levels of contraction (ns) (Fig. 5C). By day 10, differences in wound closure between the two groups diminished (Fig. 5A, D). Between day 7 and 10, untreated animals showed increased stimulation of wound contraction compared with an earlier period, similarly to CXCR4Ant-treated mice (Fig. 5C, D), probably due to the natural tendency of these animals to heal.

Diabetic (db/db) mice received a full-thickness wound (through to the level of the panniculus carnosus muscle) on the dorsum. On day 4 CXCR4Ant-treated diabetic db/db mice showed improved wound closure compared with controls (*p = 0.04) (A). On day 4 in CXCR4 antagonist-treated animals reepithelialization (*p = 0.04) (B) and contraction (*p = 0.03) (C) appeared to be increased compared with vehicle-treated animals. On day 7, CXCR4Ant-treated animals showed a similar tendency toward more rapid wound closure than vehicle-treated controls (**p = 0.03) (A), with more reepithelialization than vehicle-treated diabetic db/db mice (*p = 0.04) (B), but similar levels of contraction (ns) (C). On day 10, the only persistent difference between the groups was a more evident reepithelialization in CXCR4Ant-treated wounded mice (**p = 0.03) (B, D). A superficial epithelial layer covering the centre of the wounds was evident in CXCR4Ant-treated mice (E, F, arrows). This layer was not found on the wound surface of untreated controls (E, F).
On day 10, the only persistent difference between the groups was a more evident reepithelialization in CXCR4Ant-treated wounded mice (**p = 0.03) (Fig. 5B, D). Morphological-histological correlations showed a superficial epithelial layer covering the center of CXCR4Ant-treated wounds (Fig, 5D, E, F). This layer was not found on the wound surface of untreated controls (Fig. 5D, E, F).
In summary, these results suggest a role in early wound reepithelialization of endogenous BM-PCs during healing, with nonsignificant clinical overall effects on wound closure, at least in this animal model, but a significant increase in wound coverage at the histologic level.
Discussion
In this study, we show that (i) mobilization of endogenous BM-PCs occurs naturally after wounding in diabetic mice with a unique profile characterized by vascular progenitors mobilized earlier and to higher levels than hematopoietic progenitors; (ii) mobilization of diabetic BM-PCs can be enhanced by targeting the CXCR4—CXCL12 axis; (iii) higher numbers of peripheral endogenous BM-PCs corresponded to higher numbers of engrafted stem cells and higher levels of expression of MAP-Kinases and SDF-1α levels in the wound beds; and (iv) wound reepithelialization was found to be the main mechanism contributing to wound closure in the presence of higher levels of BM-PCs.
Impaired wound healing is a major cause of increased morbidity and mortality in diabetic patients (36). Diabetic wounds often show critically impaired revascularization, making surgical treatment, including skin grafting or more complicated free transfers of tissues, unsuccessful (16). Our results show that circulating BM-PCs increase in number in diabetic mice after wounding, suggesting that these cells play a role in modulating the physiological response to damage. Because during healing, particularly in the beginning, the majority of the cells come from the circulation, our results suggest a rather important role of stem cells in repair.
To dissect out the role of these cells during diabetic wound repair, we were able to specifically increase their mobilization and wound recruitment by the use of a CXCR4 antagonist. Results show that increased release of these cells in the periphery corresponded to increased engraftment in healing tissues due to higher levels of expression of their main ligand SDF-1α and MAP kinases, markers of effective cell migration and activation. Interestingly, it has recently been shown in the same animal model that diabetic wounds exhibit defective CXCL12 expression (28), and this has been related to deficient wound repair. The resulting enhanced angiogenesis and cell proliferation of diabetic wounds appear to be the two main outcomes of this increased BM-PC release and engraftment.
Some studies have recently suggested that BM-PCs in diabetic patients may be dysfunctional or be subject to dysfunctional recruitment (5,6,11,12,14,25,34,37,38). Our results in vivo showed that wounding mobilizes BM-PCs, while indicating that the main defect during diabetic wound healing may be in relatively low release and engraftment. The presence of an increased number of stem cells in the wound of CXCR4Ant-treated mice, suggested that during the mobilization higher levels of circulating endogenous BM-PCs in this animal model retained their capacity to migrate into healing tissues and directly and indirectly participated in or stimulated wound revascularization and proliferation.
Wounds exposed to higher levels of BM-PCs exhibited greater reepithelialization during the first week of healing. Because reepithelialization is the main mechanism in regenerative healing, while contraction predominates in repair and hypertrophic scars (46), there may be a link between the presence of stem cells and improved wound regeneration. Some studies have shown the direct participation of stem cells in wound reepithelialization as well as in other conditions where regeneration is an issue (18,23,26,35). Further studies with longer follow-up should be conducted to better understand the mechanism of increased reepithelialization induced by stem cells.
Results suggest that higher levels of endogenous BM-PCs were essential to stimulate diabetic wound healing. We have previously shown in the db/db model that a topical graft of side population hematopoietic stem cells accelerate the healing of excisional wounds (13). Clinically, adequate numbers of endogenous BM-PCs are difficult to obtain because of their small numbers in peripheral blood, and because of our limited ability to extract them from BM (44). Allogeneic approaches are also limited by immunological reactions (27), including possible graft-versus-host disease. The addition of GM-CSF did not further mobilize BM and, not surprisingly, did not further stimulate wound healing. The reduced mobilization induced by the association CXCR4 antagonist-ACK2 reduced wound cell proliferation as BM-PCs are highly cycling cells. Because levels of BM-PCs remained higher with CXCR4 antagonist-ACK2 compared to untreated wounds, proliferation rates still remained higher. From the combination studies, angiogenesis appeared to be less directly affected by peripheral BM-PCs, suggesting that additional research should be performed to elucidate this aspect.
Taken together, our results point to a clinically relevant strategy to harness the regenerative properties of endogenous BM-PCs in diabetic patients by CXCR4 antagonists. Optimization of the dose and duration, as well as strategies to improve the engraftment of mobilized HSCs, is necessary next steps.
The murine model we chose is characterized by leptin receptor deficiency. As leptin is a known contributor to wound healing, this is a confounding variable that could bias our data. However, performing our experiments in a different model of diabetes (i.e., the low-dose streptozotocin-diabetic mouse) would have encountered the same problem, given that STZ also interferes with the immune system. Of note, despite the fact that leptin has been shown to have an essential role in regulating hematopoiesis and lymphopoiesis (29), we did not observe any baseline deficit in peripheral stem cell percentages in diabetic db/db mice.
In conclusion, BM-PCs appear to play physiologic roles during diabetic wound healing. We found that normal (comparable to wild-type) levels of stem cells are anchored in the bone marrow and circulate in the periphery of diabetic mice. The use of a CXCR4 antagonist increases the number of circulating stem cells in the periphery and at the site of injury due to an increased expression of SDF-1α. Increased levels of angiogenesis and cell proliferation resulted from the higher levels of stem cells and were associated with a stimulation of reepithelialization of the wound bed (Fig. 6).
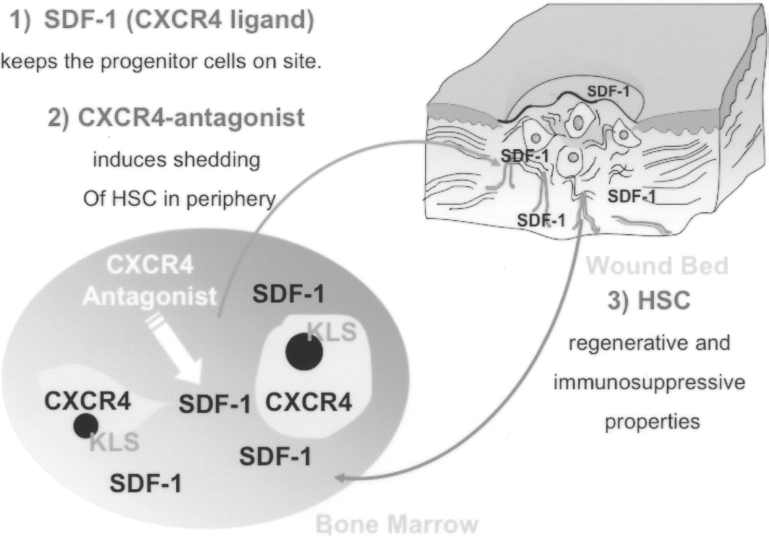
Hypothesized mechanism of action of CXCR4Ant in diabetic wounds: 1) normal levels of stem cells are anchored in the bone marrow of diabetic mice; 2) CXCR4Ant increases the levels of circulating stem cells in the periphery; and 3) increased levels of angiogenesis and cell proliferation, associated with stimulation of reepithelialization, corresponded to increased levels of SDF-1α in diabetic wounds.
Footnotes
Acknowledgments
Paolo Fiorina is the recipient of an AST-JDRF Faculty Grant and a JDRF-Career Development Award. Reza Abdi is the recipient of a JDRF Regular Grant. Andrea Vergani is the recipient of an AST-JDRF fellowship grant.
