Abstract
Recently, animal studies have demonstrated the efficacy of endothelial progenitor cell (EPC) therapy for diabetic wound healing. Based on these preclinical studies, we performed a prospective clinical trial phase I/IIa study of autologous G-CSF-mobilized peripheral blood (PB) CD34+ cell transplantation for nonhealing diabetic foot patients. Diabetic patients with nonhealing foot ulcers were treated with 2 × 107 cells of G-CSF-mobilized PB CD34+ cells as EPC-enriched population. Safety and efficacy (wound closure and vascular perfusion) were evaluated 12 weeks posttherapy and further followed for complete wound closure and recurrence. A total of five patients were enrolled. Although minor amputation and recurrence were seen in three out of five patients, no death, other serious adverse events, or major amputation was seen following transplantation. Complete wound closure was observed at an average of 18 weeks with increased vascular perfusion in all patients. The outcomes of this prospective clinical study indicate the safety and feasibility of CD34+ cell therapy in patients with diabetic nonhealing wounds.
Keywords
Introduction
Diabetic patients with nonhealing chronic ulcer are increasing yearly. Most nonhealing diabetic ulcers with peripheral vascular disease are difficult to cure, and once all conventional treatment modalities are exhausted, amputation is the final solution. More than 40–60% of nontraumatic lower extremity amputations are related to diabetic foot. It is also reported that individuals with diabetes have 15 to 46 times greater risk of high-level lower extremity amputations than those without diabetes (29). In addition, 5-year mortality rates after lower extremity amputation for diabetics, critical limb ischemia, and peripheral artery disease range from 39% to 68% (15). Furthermore, the economic burden of diabetic foot ulcer is estimated to be $98 billion per year (26). These data suggest the importance and necessity of alternative and more effective treatment option for diabetic patients with nonhealing ulcers.
After the discovery of endothelial progenitor cells (EPCs) in 1997, these vascular stem cells became the subject of intense experimental and clinical investigation for angiogenesis and wound healing. EPCs are an immature cell population that possesses an enhanced potential to differentiate into mature endothelial cells (2). EPCs can be isolated as cluster of differentiation 34-positive cells (CD34+) and CD133+ mononuclear cells (MNCs) from adult bone marrow (BM) and peripheral blood (2,9). EPCs mainly reside in the bone marrow and are mobilized into the peripheral blood with tissue ischemia or systematic administration of granulocyte colony-stimulating factor (G-CSF), vascular endothelial growth factor (VEGF), or estrogen (22,31). Mobilized EPCs will be home to ischemic sites for vascular repair. Preliminary studies support the potential of EPC therapy for angio-genesis and wound healing, and systemic (27) and local transplantation of EPCs (30) has become an alternative therapeutic option for diabetic ulcers. In murine diabetic ulcer, human EPCs are incorporated into the wound bed of diabetic mice following local injection and participate in neovascularization with recipient's endothelial cells, resulting in enhancement of wound vascular density and higher wound closure rate (28). Lin et al. also demonstrated that topical application of bone marrow-derived progenitor cells accelerates diabetic wound healing and increases wound vasculogenesis (19). These promising results encouraged clinical application of EPC transplantation for improvement of nonhealing diabetic ulcer. Until now, several researchers have investigated and reported the efficacy of autologous bone marrow and peripheral blood MNCs on patients with diabetic ulcers; however, the efficacy of purified EPC transplantation is not yet investigated (13,18).
Here we report a phase I/IIa clinical trial of transplantation of autologous and G-CSF-mobilized CD34+ cells in patients with intractable diabetic ulcer. G-CSF was used to efficiently mobilize bone marrow-derived EPCs to peripheral blood, and the mobilized CD34+ cells were isolated as the EPC-enriched fraction.
Materials and Methods
Ethical Conduct of Research
The authors have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human experimental investigation. In addition, appropriate informed consent has been obtained from all patients by the investigator in charge.
Study Design
This phase I/IIa study was conducted from July 2005 to March 2010 at Tokai University School of Medicine Department of Plastic and Reconstructive Surgery. The protocol was reviewed and approved by the Ethics Committee of the Tokai University School of Medicine, Kanagawa, Japan. The primary end point of this trial is safety, and the secondary end point is primary efficacy. Patients providing informed consent had type 2 diabetes with a nonhealing chronic ulcer. If wounds were on both feet, the foot with a more severe wound was treated with autologous G-CSF-mobilized peripheral blood CD34+ cells. Inclusion criteria included (1) type 2 diabetic patients of ages 20 to 70 years with a nonhealing chronic wound deeper than the subcutaneous layer of the skin. The wound was determined as nonhealing and chronic when the wound was treated with current standard care for diabetic foot ulcer by a wound care specialist for at least 3 months prior to the therapy with less than 40% of wound closure. There were no limitations of the wound size. (2) Patients with strict diabetic control of glycated hemoglobin (HbA1c) below 6.5%. The exclusion criteria included (1) collagen tissue disease or malignant disease, (2) an ejection fraction lower than 50%, (3) interventional treatment required for coronary or cerebral artery stenosis within 6 months, (4) diabetic retinal bleeding, (5) hematological disorder, (6) onset of myocardial infarction or cerebral infarction within the last 6 months, (7) side effects arising from G-CSF pharmaceutical or apheresis procedures, and (8) wound infection. Written informed consent for participation was obtained from all subjects. All patients did not receive any medication changes pre- and post-EPC injection therapy. Since the study was a phase I/IIa clinical trial, controls for the study were not established.
Study Procedures
Screening assessments within 14 days before treatment included medical history, review of inclusion and exclusion criteria, review of medications, vital signs, physical examination, chest X-ray (Shimadzu, Kyoto, Japan), urinalysis, blood collections for hematology [complete blood count (CBC) and differential], clinical chemistry [bilirubin, alkaline phosphatase, alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehy-drogenase (LDH), urea nitrogen, creatinine, glucose, uric acid, calcium, phosphorus, total protein, albumin, electrolytes, amylase, cholesterol, triglycerides], and HbA1c. All blood-related measurements were performed by the blood testing central laboratory at the Hospital of Tokai University, Kanagawa, Japan. Patients underwent cardiac echography, abdominal echography (Toshiba, Tokyo, Japan), and cerebral, chest, and abdominal computed tomography (CT; Siemens, Tokyo, Japan) to rule out any conditions listed in the exclusion criteria. During the 3-month screening period, patients continued to receive the standard care for diabetic feet. Foot ulcers were photographed using a digital camera with a 1-cm2 size sticker marked near the wound. The area of the ulcer was calculated by measuring the size of the photographed ulcer divided by the photographed 1-cm2 size marker using VH analyzer (Keyence Corp, Osaka, Japan). Vascular perfusion was evaluated by using the following parameters: ankle brachial pressure index (ABI), skin perfusion pressure (SPP), and transcutaneous oxygen pressure (TcO2). Angiographic analysis was performed for cases with severe peripheral vascular disease. TcO2 was measured with an oxymonitor (PO-850, Sumitomo-Hightechs, Tokyo, Japan). The probe was placed at the dorsum pedis, and the skin was heated to 42°C (4). SPP was measured with the PAD3000 (Vasamedics, St. Paul, MN, USA) proximal to the wound with an exclusive cuff (5,6). These physiological examinations were performed while the patient was in a supine position after more than 30 min of rest in a temperature-controlled room. Angiographic examinations were performed with the intra-arterial digital subtraction angiography (IA-DSA) technique. The standard Seldinger approach was used as follows: the top of the 4 Fr catheters was placed at the external iliac artery, and iohexol (Omnipaque, 300 mg of iodine/ml; Daiichi Pharmaceutical, Tokyo, Japan) was injected automatically using an infusion pump at a speed of 20 ml/s for 2 s. Wound care was standardized throughout the entire study by using several different dressing types dependent on the type of the wound (e.g., dry, wet, and intermediate) with daily dressing changes and use of an offloading device by a wound care specialist. Minor debridement of necrotic tissue without general anesthesia was performed as regular wound care. Major debridement was performed only at the time of cell transplantation under general anesthesia.
Treatment Period
Collection of Peripheral Blood CD34+ Cells
Patients who met the criteria were admitted to the hospital, and 10 μg/kg/day of G-CSF (Filgrastim; Gran®, Kyowa Hakko Kirin, Co. Ltd., Tokyo, Japan) was injected subcutane-ously for 5 days. Blood counts, peripheral leukocyte differential counts, and peripheral CD34+ cell counts were determined daily. If leukocyte count exceeded 50,000/ μl, the dose of G-CSF was reduced to one half. On day 5, peripheral blood MNC collection was performed with the COBE SPECTRA apheresis system (Gambro BCT, Lakewood, CO, USA). MNC collection was followed by purification of CD34+ cells by means of a magnetic-activated cell sorting system (CliniMACS, Milteny Biotech, Bergisch Gladbach, Germany).
Administration of Isolated CD34+ Cells
Debridement and transplantation of CD34+ cells were performed under general anesthesia on the same day of cell isolation. After debridement and irrigation of the wound, CD34+ cells were injected intramuscularly within 20 cm surrounding the wound with a 26-gauge needle (Nipro Corporation, Osaka, Japan). A total of 2 × 107 CD34+ cells/patient was administered by 20 injections, each injection containing 1 × 106 cells/0.25 ml saline (0.25 ml × 20 sites, 1.5–2.0 cm deep). Saline gauze dressing was placed over the treated wound immediately after the treatment to avoid cell damage, and standard of wound care was continued starting postoperative day 1. The patient was discharged from the hospital the following day unless any side effects due to the CD34+ cell therapy were seen. Current standard of care for diabetic foot was performed starting on the day of discharge.
End Points
The primary end point of this study is to evaluate the safety, and the secondary end point is to evaluate efficacy 12 weeks posttherapy. No gold standard efficacy end points have been established for small size, early phase clinical trials in patients with diabetic foot, so we originally pre-specified the efficacy score as a surrogate end point so that we could simultaneously evaluate subjective and objective parameters in the study. Twelve weeks posttherapy was determined as the time point of evaluation in reference to other clinical trial reports assessing diabetic wound healing because all patients included in the study had nonhealing wound for more than 3 months (20,37). The adverse effects were evaluated according to the National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE version 3 http://ctep.cancer.gov/protocol-Development/electronic_applications/docs/ctcaev3.pdf). Full physical examination, blood collection for hematol-ogy, clinical chemistry, chest X-ray, abdominal echo, cardiac echo, cerebral CT, chest CT, and abdominal CT were performed to find if any conditional change existed after the therapy. C-Reactive protein was measured by latex-enhanced immunoturbidimetric assay (Eiken Chemical Co. Ltd., Tokyo, Japan). The efficacy score was defined as the sum of four scores, each of which measured a difference in the parameters between baseline and 12 weeks after cell therapy: (1) percent wound closure calculated by VH analyzer as previously mentioned in the study procedure section (wound needing minor or major amputation was evaluated and performed at 12 weeks posttherapy; major amputation is an amputation of below the knee or above the knee or a procedure proximal to this level, whereas minor amputation is classified as all other partial foot or toe resection) (10); (2) skin perfusion pressure (SPP) in the treated foot; (3) Wong–Baker FACES Pain Rating Scale score (http://www.wongbakerfaces.org/), evaluation of pain in the treated leg; and (4) recurrence of the treated wound. Each score is given a range of plus 2 to minus 2 points; the best response is assigned plus 2, and the worst outcome is assigned minus 2. The efficacy score sum is in the range of +8 to −8 (Table 1). The patients visited the clinic 2 weeks, 4 weeks, 8 weeks, and 12 weeks postoperatively to evaluate safety and efficacy. The patients were still followed every 4 weeks when complete wound closure was not seen at 12 weeks posttherapy. The photographs were taken for percent wound closure, and ABI, SPP, and TcO2 were tested for vascular perfusion at each visit. Angiographic analysis was performed for cases with severe peripheral vascular disease 12 weeks postoperatively. Each patient evaluated pain level at baseline and at 12 weeks after transplantation using the Wong–Baker FACES Pain Rating Scale (7). A clinical research coordinator interviewed each patient regarding the level of psychroesthesia, paresthesia, and the required quantity of analgesic drugs at baseline and at 12 weeks after transplantation. Recurrence of the wound was evaluated every 4 weeks posttherapy at each clinical visit. The recurred wounds were treated by standard of wound care and evaluated for time of wound closure. The efficacy score was validated by two individual physicians not included in the study.
Efficacy Score

The efficacy score = (wound closure + SPP + Wong-Baker FACES Pain Scale) post-12 weeks therapy + Recurrence. Range: −2 to 2 for each category. SPP, skin perfusion pressure.
Quality Analysis of CD34+ Cells
CD34+ cells isolated for transplantation were labeled with fluorescein isothiocyanate (FITC)-conjugated anti-CD34 (BD Biosciences, San Jose, CA, USA) and phy-coerythrin (PE)-conjugated anti-KDR [kinase insert domain receptor or vascular endothelial growth factor receptor 2 (VEGFR2); BD Biosciences] antibodies for 20-min incubation at 4°C. Cells were then analyzed using FACSCalibur and CellQuest Pro software (Becton Dickinson, Franklin Lakes, NJ, USA).
EPC Colony-Forming Assay
The vasculogenic potential of isolated peripheral blood CD34+ cells was assessed using the EPC colony-forming assay (EPC-CFA) as previously described (17,21,32). Briefly, isolated 3,000 CD34+ cells were suspended in 300 μl of 30% fetal bovine serum (FBS; Nichirei Bioscience Inc., Tokyo, Japan)/Iscove's modified Dulbecco's media (IMDM; Gibco) and mixed with 3 ml of previously stored EPC-CFA working medium (Methocult, Stem Cell Technologies, Vancouver, BC, Canada). A total of 1,000 CD34+ cells/dish were seeded into a 35-mm hydrophilic tissue culture dish (BD Falcon, Bedford, MA, USA; 1 ml working medium to one dish, a total of three dishes per sample). After 18 days, the number of total EPC colony-forming units (EPC-CFU) was counted by two investigators who were blinded to the experimental conditions.
Statistical Analysis
All data are presented as the mean ± standard deviation. Student's t test was performed to assess statistical significance between the two groups.
A Kruskal–Wallis one-way ANOVA with Tukey– Kramer post hoc analysis was performed when comparisons involved more than two groups. Significance was considered to be p < 0.05. The statistical program used for the analysis of all data was Graph Pad Prism 5 (Graph pad Software, Inc., La Jolla, CA, USA).
Results
Patient Characteristics
A total of five patients were enrolled in the trial. The characteristics of all patients enrolled in the trial are listed in Table 2. The age ranged from 41 to 70 years old. All patients had diabetes and chronic renal failure with hemo-dialysis as past medical history. Blood sugar levels were controlled for all patients, and hbA1C was below 6.5%. All of the wounds extended into bone or tendon and were located in the digits of the foot. The average size of the wound was 3.3 ± 1.1 cm2 × 2.92 ± 1.9 cm2. The average wound history prior to EPC therapy was 34 ± 23 weeks (240 days). Average ABI was 0.9 ± 0.17. All patients had low skin perfusion pressure (SPP: 14.8 ± 5.3) proximal to the wound, indicating peripheral vascular disease. Cases 4 and 5 had percutaneous transluminal angioplasty (PTA) 3 months prior to the therapy.
Exposure and Outcome for All Patients Treated With Autologous G-CSF-Mobilized Peripheral Blood CD34+ Cell Therapy

G-CSF, granulocyte colony-stimulating factor; CD34, cluster of differentiation 34; PMH, past medical history; HbAlc, glycated hemoglobin; SPP, skin perfusion pressure; ADL, activities of daily living; DM, diabetes mellitus; CRF, chronic renal failure; HD, hemodialysis; PAD, peripheral artery disease; Pop. A, popliteal artery.
Outcome of Mobilization, Harvest, and Isolation of CD34+ Cells
G-CSF was administered 10 μg/kg/day for 5 days for all patients. The frequency of CD34+ cells in the peripheral blood at day 5 post-G-CSF injection was 0.3 ± 0.1% by flow cytometry analysis. The average apheresis product number was 2.7 ± 1 × 1010, and the average of total CD34+ cells obtained after magnetic sorting by CliniMACS was 8.0 ± 4.0 × 107. Flow cytometry revealed that the purity and viability of the CD34+ cell fraction following magnetic sorting were 70 ± 15.8%. The number of CD34+ cells isolated was significantly low for Case 2 (1.23 × 105/kg) and high for Case 1 (9.0 × 105/kg) (Table 3).
Outcome of Mobilization, Harvest, and Isolation of CD34+ Cells

The frequency of CD34+ cells in the peripheral blood at day 5 post-G-CSF injection was 0.28 ± 0.1% by flow cytometry. The average apheresis product number was 2.68 ± 1 × 1010, and the average of total CD34+ cells obtained after magnetic sorting was 7.96 ± 4.0 × 107. Flow cytometry revealed that the purity and viability of the CD34+ cell fraction following magnetic sorting were 70 ± 15.8%. PB, peripheral blood; MNCs, mononuclear cells.
Safety Evaluation
Neither death (NCI CTCAE grade 5) nor life-threatening adverse events (grade 4) were observed during the 12-week follow-up after cell therapy. In contrast, mild to moderate adverse events (grades 1–2) were observed as G-CSF-related events in all patients. Symptoms such as bone pain (n = 5), headache (n = 1), fever (n = 1), and C-reactive protein (CRP) elevation (n = 1) seen were transient and disappeared without permanent damage. There were no adverse events following general anesthesia. No episodes of site infection following cell injections were noted. There was no incidence of pathogenic angiogen-esis after serial examination of fundus oculi.
Efficacy Evaluation
The efficacy score at 12 weeks was more than or equal to 2 in all patients, indicating the efficacy of transplantation of CD34+ cells.
Wound Closure
Only two out of five patients (Case 1 and Case 2; Figs. 1 and 2) had complete wound closure within 12 weeks of evaluation. Since the remaining sequestrum was obstructing wound healing, two out of five patients (Case 3 and Case 4; Figs. 3 and 4) received further debridement resulting as minor amputation. In Case 5, the patient had the largest wound pretherapy, which had healed within 16 weeks posttherapy (Fig. 5). Although only two had complete wound closure by 12 weeks posttherapy and two resulted as minor amputation, all patients had complete wound closure without any major amputation at an average of 18.8 weeks (131.6 days) (Fig. 6). During the trial, Case 4 had sudden stenosis of popliteal artery after 8 weeks of therapy, which had healed after percutaneous transluminal angioplasty (PTA).

Case 1: A 41-year-old male who was diagnosed with diabetes at age 30 and currently undergoing dialysis. (a) Pretherapy: the ulcer seen in the picture was not healing for 22 weeks. (b) Three months posttherapy. After cluster of differentiation 34-positive (CD34+) cell therapy, the wound healed rapidly, and wound closure had occurred by 12 weeks after the therapy. No recurrence was seen for 4.8 years posttherapy. Skin perfusion pressure (SPP) measured proximal to the wound showed 13 mmHg pretherapy and 75 mmHg 12 weeks posttransplant.
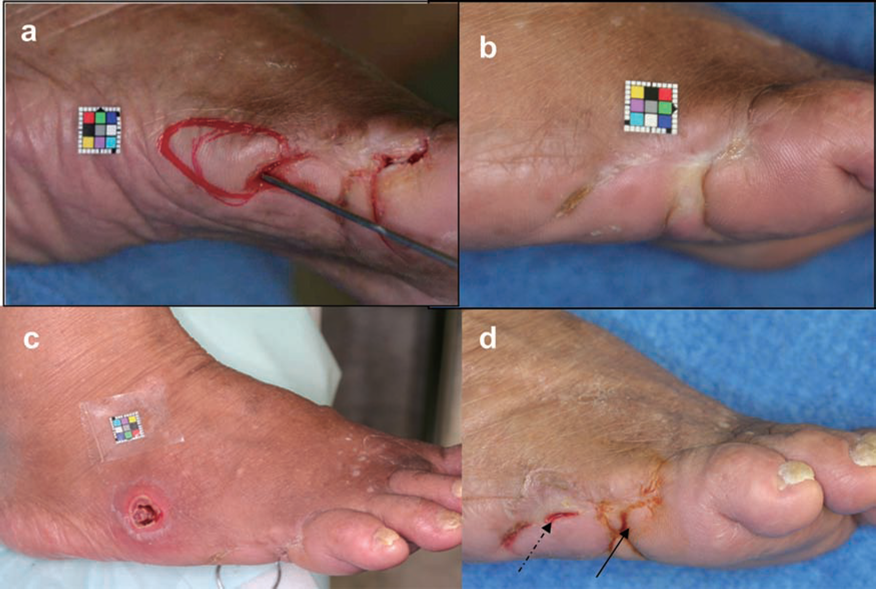
Case 2: A 70-year-old female with 48 years of diabetes and 28 years of chronic renal failure (CRF) on hemodialysis. (a) Pretherapy: the ulcer with a deep pocket reaching to the fifth metatarsal bone located in the lateral side of the right foot was nonhealing for 74 weeks. SPP was 10 mmHg pretherapy. (b) Posttherapy: the ulcer healed completely, and SPP increased to 45 mmHg after 12 weeks posttherapy. (c) One year posttherapy. (d) The dotted arrow shows cured heterotrophic ulcer 2 years posttherapy. The solid arrow shows ulcer recurrence.

Case 3: A 63-year-old male with diabetes and CRF on hemodialysis. (a) The ulcer on the left fifth toe seen in the picture was nonhealing for 14 weeks. Dorsal SPP was 10 mmHg at this point. (b) The ulcer after 12 weeks posttherapy; the ulcer did not heal at this point, but SPP increased to 45 mmHg. (c) The patient underwent minor amputation of the fifth toe, and the wound completely healed 28 weeks after the therapy. (d) Angiography pretherapy; circled area is avascular. (e) Angiography 12 weeks posttherapy showed increased vascularity in the avascular area pretherapy.
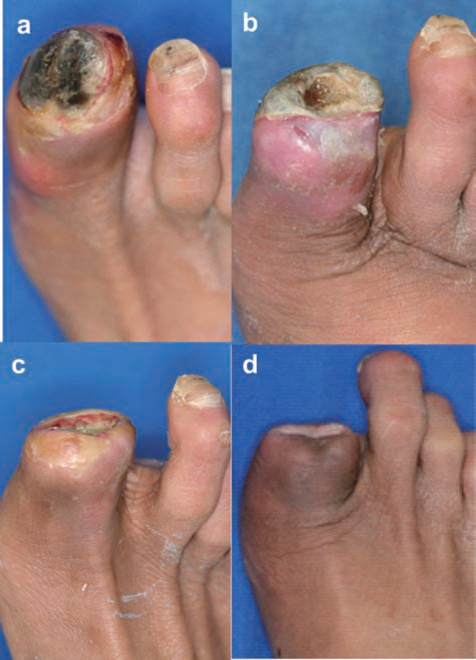
Case 4: A 53-year-old male with 15 years of diabetes and 9 years of CRF on hemodialysis. (a) The ulcer on the right toe pretherapy, which did not heal for 33 weeks. (b) Eight weeks after the therapy, SPP proximal to the wound, which was 14 mmHg, suddenly dropped to 2 mmHg. Angiogram showed stenosis of popliteal artery, and emergency percutaneous transluminal angioplasty (PTA) was performed. (c) At 12 weeks post-therapy. SPP increased to 58 mmHg with increased granulation. (d) At 26 weeks posttherapy. The wound completely healed at this point, and the patient was wound free for more than 2 years with an average SPP of 42 ± 11 mmHg.

Case 5: A 63-year-old male with 20 years of diabetes and 4 years of CRF on hemodialysis. (a) Pretherapy: The ulcer located on the left third, fourth, and fifth toes to metatarsus did not heal for 26 weeks. SPP was 10 mmHg at this point. (b) The ulcer at time of debridement and CD34+ cell transplant. (c) At 12 weeks posttherapy with an SPP of 67 mmHg. (d) At 16 weeks posttherapy; time of complete wound closure. Currently, patient is ambulant for 2 years posttherapy without any recurrence and heterotopic ulcer with stable SPP of 66 ± 23 mmHg. (e) Angiography pretherapy; avascular area is marked with circle. (f) Angiography 12 weeks posttherapy; avascular area pretherapy has increased vascular perfusion after CD34+ cell therapy.

Improvement of efficacy parameters following CD34+ therapy. Serial changes of subjective and objective parameters of wound healing and vascular perfusion of the treated foot. Percent wound closure was calculated by VH analyzer. Cases 1, 2, and 5 had near-complete wound closure after 12 weeks. Ankle brachial pressure index (ABI), SPP, and transcutaneous partial oxygen pressure (TcO2) were performed to evaluate peripheral vascular perfusion. There was no significant change in resting ABI pretherapy and posttherapy (0.9 ± 0.2 vs. 0.9 ± 0.2), but SPP (14.5 ± 5.3 vs. 53.6 ± 16.3; p < 0.01) and TcO2 (27.8 ± 8.2 vs. 56.0 ± 7.2; p < 0.01) proximal to the wound showed significant increase 12 weeks after the therapy in all patients. Pain level was evaluated using the Wong–Baker FACES Pain Rating Scale. All patients with pain pretransplantation showed a significant decrease in Pain Rating Scale (3.2 ± 2.1 vs. 0.6 ± 0.9; p < 0.05). SPP, skin perfusion pressure.
Peripheral Vascular Perfusion
There was no significant change in resting ABI pretherapy and post-therapy (0.9 ± 0.2 vs. 0.9 ± 0.2), but SPP (14.5 ± 5.3 vs. 53.6 ± 16.3; p < 0.01) and TcO2 (27.8 ± 8.2 vs. 56.0 ± 7.2; p < 0.01) proximal to the wound showed a significant increase 12 weeks after the therapy in all patients (Fig. 6). Angiography performed 12 weeks posttherapy showed increased vascularity in the deep plantar artery for Cases 3 and 5 (Figs. 3 and 5).
Pain Scale
Four out of five patients had limb and foot pain before the therapy, which began to be relieved 4 weeks after the therapy. The average pain level evalu ated using the Wong–Baker FACES Pain Rating Scale of 3.2 ± 2.1 at baseline decreased to 0.6 ± 0.9 at 3 months after therapy (p < 0.05) (Fig. 6).
Recurrence
The ulcer of Cases 1 and 5 had not yet recurred after more than 2 years of cell therapy. Cases 3 and 4 showed recurrence of the ulcer within 1 year after the therapy, but it was rapidly cured in a few weeks (3 weeks for Case 3 and 2 weeks for Case 4) after standard care. However, the recurred ulcer for Case 2 did not heal even after 3 years of standard care (Fig. 2).
Vasculogenic Potential of Transplanted CD34+ Cells
Case 1 with wound closure at 12 weeks posttherapy and no incidence of heterotopic ulcers or recurrence demonstrated a significantly higher number of total EPC-CFU compared to the other four cases (Case 1: 25 ± 6 vs. Case 2: 4 ± 2, Case 3: 11 ± 1, Case 4: 11 ± 1.5, Case 5: 16 ± 1; p < 0.01) (Fig. 7). Flow cytometry of CD34 and KDR double-positive cell percentages within the transplanted CD34+ cells showed similar results to EPC-CFU. Case 1 showed a significantly higher number of CD34/KDR double-positive cell percentage compared to the other four cases (2.8 vs. 0.5, 0.37, 1.49, 1.67), suggesting that transplanted CD34+ cells with higher vasculogenic potential exhibit accelerated wound healing and better prognosis (Fig. 7).

Efficacy score and vasculogeneic potential of transplanted CD34+ cells. (a) Efficacy score at 12 weeks following CD34+ transplantation. Efficacy score was positive for all cases indicating efficacy of the therapy. However, Cases 2, 3, and 4 showed less improvement compared to Cases 1 and 5. (b) Vasculogenic potential of transplanted CD34+ cells was evaluated by endothelial progenitor cell colony-forming assay (EPC-CFA). Number of total EPC colonies were counted per well of 500 CD34+ cells; n = 3 per patient. The total colonies for all cases were as follows: Case 1: 25 ± 6, Case 2: 4 ± 2, Case 3: 11 ± 1, Case 4: 11 ± 1.5, Case 5: 16 ± 1. CFU, colony-forming units. (c) Flow cytometry of CD34+ and kinase insert domain receptor [KDR or vascular endothelial growth factor receptor 2 (VEGFR2)] double-positive cell percentage of transplanted EPCs is graphed. Similar to EPC-CFU, Case 1 demonstrated the highest CD34+/KDR percentage. Cases 1 and 5 with high efficacy score demonstrated a significantly higher number of total EPC-CFUs and CD34 and KDR double positivity, indicating that transplanting EPCs with higher vasculogenic function leads to better therapeutic outcome.
Discussion
To the best of our knowledge, the present study is the first clinical trial of transplantation of autologous and purified CD34+ cells into diabetic patients on hemodialy-sis who were suffering from nonhealing chronic wounds for more than 3 months. Previous studies on autologous transplantation of bone marrow or peripheral blood stem cells for diabetic ulcers were focused on administrating mononuclear cells to more acute or subacute wounds as cell population including EPCs (13). Since application of peripheral blood or bone marrow mononuclear cells may contain cells not related to angiogenesis and wound healing, we believe that purified CD34+ cells are a more suitable source of stem cell therapy for angiogenesis and wound healing. Regarding this, although many investigators identify CD34+/CD133+/KDR+ cells as EPCs in basic research (5), isolating the rare cell population is clinically impractical due to the absence of a clinical-grade anti-KDR antibody. Therefore, we have chosen CD34+ cells as an enriched population of EPCs for cell isolation that was technically and clinically applicable.
Furthermore, animal studies have reported that purified CD34+ cells exhibit improved healing and vasculogenesis compared to nonpurified mononuclear cell transplantation (33). In a clinical randomized trial of direct intramyocar-dial injection of autologous mononuclear bone marrow cells during coronary artery bypass graft (CABG) to improve left ventricle function, it was reported that the “responder” group was transplanted with a cell population containing a significantly higher percentage and absolute number of CD34+ cells than nonresponders (11). Recently, Kawamoto et al. reported the safety and efficacy of G-CSF-mobilized CD34+ cell transplant to patients with critical limb ischemia (16). Therefore, we hypothesized that the autologous transplantation of purified CD34+ cells as enriched population of EPCs may be effective in the treatment of nonhealing chronic diabetic wounds with peripheral vascular disease. Since uncontrolled diabetes and high blood sugar level influence diabetic wound healing, our trial confirmed that the wounds were untreatable even after diabetic control and standard wound care. We designed this small size, phase I/IIa clinical trial as a prospective, uncontrolled, single-blinded study to obtain useful information for a future phase IIb/III trial. In this pilot study, we observed many clinical manifestations that significantly improved after autologous peripheral blood CD34+ cell transplant, such as wound closure, SPP, TcO2, and lower limb pain. Measurements of ambulatory blood pressure (ABP) are reported to fail to reflect the severity of peripheral ischemia if the underlying vessels are calcified in patients who have diabetes or are receiving hemodialy-sis (25,35). In contrast to ABP, measurements of SPP and TcO2 provide more accurate information even in noncompressive vessels, and false-positive results are rare (4,12). Since all patients in this trial were receiving hemodialysis due to chronic renal failure, skin perfusion pressure and TcO2 measurements were used as a more reliable evalua-tor of peripheral vascular perfusion. Consequently, we did not observe significant changes in ABP in our cases, but a significant increase in SPP and TcO2 was observed after receiving the therapy. Furthermore, patients of peripheral vascular disease with end-stage renal failure are reported to be less responsive to peripheral or bone marrow mono-nuclear cell therapy (24). Although all patients included in our trial were receiving hemodialysis, all patients had complete wound closure at an average of 18 weeks post-therapy and increased peripheral perfusion with no major amputations. This result suggests that purified CD34+ cells may be the feasible cell therapy of these patients.
The role of G-CSF administration in the healing process in diabetic patients is not clear. There have been reports that G-CSF administration itself may accelerate wound healing (8). Some investigators reported that treatment by G-CSF improves symptoms but not signs of ischemic heart disease (36). However, in our cases, increase in SPP and TcO2 was not seen in the nontreated side of the foot. In addition, the velocity measurement of dorsal artery by Doppler showed an increase only in the treated side of the foot (data not shown). The contralateral foot could be identified as an internal control; therefore, this observation suggests that CD34+ cells might have an improved neovascularization effect regardless of G-CSF administration.
One of the limitations of autologous EPC therapy for diabetic patients is impaired mobilization and function of diabetic EPCs (7). We and others have previously reported that both bone marrow and circulating diabetic EPCs have significantly lower vasculogenic potential compared to healthy EPCs (6,34). For that reason, the number of isolated peripheral blood CD34+ cells in our trial was lower compared to the number reported in a similar trial conducted by Kawamoto et al. with nondiabetic patients (16). In our trial, the vasculogenic potential of isolated and transplanted CD34+ cells was evaluated by EPC-CFU and a number of CD34/KDR double-positive cells. The patient with accelerated wound healing and positive prognosis without recurrence or heterotopic ulcers had a higher total number of EPC-CFU and CD34/KDR double-positive cells, indicating that the vasculogenic potential of transplanted cells is an important factor of effective autologous EPC therapy. These outcomes suggest the necessity to future investigate the relationship between patient background, EPC potential, and its efficacy.
The role of transplanted CD34+ cells in wound healing and postnatal neovascularization was not investigated in our study; however, we believe that accelerated wound healing and increased vascular perfusion were promoted with involvement of direct and indirect CD34+ cell contribution to neovascularization. In the context of EPC biology, CD34+ cells that contain enriched population of EPCs can promote vasculogenesis by migration, proliferation, differentiation, and/or incorporation of bone marrow-derived EPCs into newly forming vasculature (1). This has been previously demonstrated by several studies using a well-established model using tunica interna endothelial cell kinase-dependent β-galactosidase reporter gene (Tie-2/LacZ) transgenic mice, allowing the detection of bone marrow EPCs in the targeted tissue (14,22). Transplantation of bone marrow-derived EPCs from Tie-2/LacZ mice accelerated wound healing and hind limb perfusion in wounded hind limb ischemia model by direct incorporation of EPCs into the neovessels in the granulation tissue using transgenic mice (3). We believe that not all EPCs incorporate into vasculature formation, but many of these cells greatly contribute to indirect contribution to vascular regeneration. EPCs activate the preexisting endothelial cells by producing various cytokines and other secreting proangiogenic factors in EPCs, such as VEGF, hepatocyte growth factor (HGF), insulin-like growth factor-1 (IGF-1), endothelial and induced nitric oxide synthase (eNOS/iNOS), angiopoietin 1 (Ang-1), and stem cell-derived factor-1 (SDF-1) (14,23).
As for the safety evaluation, there were no severe adverse events seen during and after cell therapy. Although mild adverse events were frequent, these were transient and expected. Exacerbation of diabetic retinopathy caused worry due to pathogenic angiogenesis such as arterio-venous shunt due to G-CSF administration and stem cell therapy. However, fundus oculi examinations demonstrated no pathogenic angiogenesis following CD34+ cell transplant. There was no malignant tumor or angina pecto-ris and embolism identified during this trial. These results indicate that the autologous transplantation of mobilized peripheral blood CD34+ cells is an effective and safe therapeutic approach for nonhealing chronic diabetic wounds.
In conclusion, we demonstrated that this prospective clinical trial of autologous peripheral blood CD34+ cell transplant may be an alternative therapeutic option for non-healing chronic diabetic wound for patients with peripheral vascular disease and chronic renal failure with hemodi-alysis. Future studies are needed to reveal its safety and efficacy in a larger number of patients and by comparison with an appropriate control group receiving G-CSF only or placebo.
Conclusions
Autologous peripheral blood CD34+ cell transplant is a safe and effective therapy for nonhealing diabetic wound patients. Patients treated with CD34+ cells with higher vasculogenic potential tend to have higher efficacy score and better prognosis. Furthermore, larger clinical studies with appropriate control groups are necessary to establish the safety and efficacy of this procedure.
Footnotes
Acknowledgments
This work was supported by Health and Labor Sciences Research grants from the Japanese Ministry of Health, Labor, and Welfare (20890227, 22791737), Funding Program for Next Generation World Leading Researchers LS113, and Tokai University Research Aid grant awarded to Rica Tanaka. The authors declare no conflict of interest.
