Abstract
This retrospective study reviews the results of our experience with the occurrence of CMV DNAemia in islet cell transplanted cynomolgus monkeys subjected to different immunosuppressive protocols, including induction treatment with thymoglobulin (TMG), with a combination of thymoglobulin and fludarabine (FLUD), with cyclophosphamide, or with daclizumab. CMV DNA in the peripheral blood (CMV DNAemia) of 47 monkeys was quantified by real-time PCR on a weekly to biweekly basis. As compared to other immunosuppressive regimens, and in association with greater decreases in WBC, lymphocyte, CD3+CD4+, and CD3+CD8+ lymphocyte counts, frequent CMV DNAemia occurred earlier (within the first month posttransplant), and was of greater severity and duration in recipients of TMG ± FLUD. Treatment of recipients with alternative induction agents that resulted in less dramatic reductions in WBC and lymphocyte counts, however, resulted in occurrence of CMV DNAemia after postoperative day 60. The frequency, average intensity, duration, and area under the curve (AUC) for CMV DNAemia in animals receiving TMG ± FLUD were 75–100%, 4.02 ± 1.75 copies/ng DNA, 23.0 ± 5.3 days, and 367.0 ± 121.1 days x copies/ng DNA, respectively; corresponding values in animals receiving other treatments (0–44%, 0.19 ± 0.10 copies/ng DNA, 0.5 ± 0.3 days, and 75.4 ± 40.2 days x copies/ng DNA, respectively) were significantly different. The value of WBC, T and B cells at the nadir of cell depletion greatly affects the occurrence of CMV DNAemia. No animals developed CMV DNAemia within the next 3 weeks when the lowest value of WBC, lymphocyte, CD3+, CD3+CD4+, CD3+CD8+, or CD20+ cells was above 4500, 1800, 300, 200, 150, or 300 cells/μl, respectively. Oral valganciclovir prophylaxis did not completely prevent the appearance of CMV DNAemia.
Introduction
Cytomegalovirus (CMV) is a member of the beta class of herpesviruses and is found in humans, nonhuman primates (NHP), and other mammals (5,11). It is easily transmitted through contact with body fluids (2,5,11,33,57), infecting individuals early in life and remaining latent thereafter (5,11). Primary infection in immunocompetent individuals is usually benign, and does not result in clinical signs of disease (3,5). However, in immunocompromised individuals, such as in immunosuppressed transplant recipients, CMV infection can produce life-threatening complications (20,31). The occurrence and severity of CMV infection depend on the type of transplant, the immunosuppressive regimen, and CMV serostatus of the donor and recipient. Clinical manifestations of CMV can vary from completely asymptomatic infection, defined by active CMV replication in blood in the absence of clinical symptoms or abnormalities in organ function, to fulminant CMV disease, characterized by CMV infection with overt clinical symptoms or organ dysfunction (38). Incidence of CMV infection in humans has been reported in solid organ (9,13,39), bone marrow (14,43), islet after kidney, simultaneous islet with kidney (17,58), and islet alone transplantation (IAT) (16). The incidence of CMV sero-conversion and transmission in IAT is low, with few cases of diseases reported to date (6,16,19,24,49,59).
Nonhuman primates (NHP) and humans share close developmental and evolutionary relationships. Results from NHP models can provide invaluable aid to accelerate the clinical development of promising new therapies for treating human disease. There are many similarities in the nature, history, and course of simian and human CMV infections (5). CMV infection was reported in the first week posttransplantation in cynomolgus monkeys (Macaca fascicularis) receiving kidney transplants along with humanized MHC class II-specific monoclonal antibody CDP855 (35). Detectable CMV viral load in blood by PCR assay (1) and CMV disease was reported (44) in rhesus monkey recipients of islet cell or renal allografts treated with anti-CD40 monoclonal antibody. CMV DNAemia was found in immunosuppressed baboons (40) and cynomolgus (29) and rhesus (12) monkeys in pig-to-primate models of xenotransplantation.
We have utilized a number of different agents in allogeneic islet and islet/bone marrow transplant studies in a cynomolgus monkey model. Sirolimus (Rapamune), tacrolimus (Prograf), and daclizumab (Zenapax), as well as thymoglobulin (26,27), the lymphocyte immunosuppressive agent, fludarabine (7,23,53,56), and 153Samarium-EDTMP, a bone-seeking radioactive compound (22,34,55) have been employed in various combinations. The present study is a retrospective analysis of the frequency, severity, and duration of CMV viral load in cynomolgus monkey recipients of islet allografts treated with various immunosuppressive regimens that differentially affect white blood cells (WBC) and T-cell levels.
Materials and Methods
Cynomolgus Monkeys
Donor and recipient cynomolgus monkeys (Macaca fascicularis) were obtained from Charles River BRF, Inc. (Houston, TX) or from The Mannheimer Foundation, Inc. (Homestead, FL). All animals were negative for tuberculosis, herpes B, SRV, SIV, and STLV-1 and were negative for CMV DNA by quantitative PCR. Recipients were 3–6 years old and weighed between 2.8 and 5.3 kg at the time of islet transplantation. Donors were 4–12 years old and weighed between 3.7 and 12 kg. Donor–recipient pairs were ABO compatible (41). We followed CMV DNA levels posttransplant in 47 recipients.
Monkeys were single housed, supplied with water ad libitum, and fed twice daily. All study protocols were approved by The Institutional Animal Care and Use Committee of The University of Miami.
Islet Isolation and Transplantation
Islets were isolated from the donor pancreas using a modification of the automated digestion-filtration method for human islet isolation (37,46) followed by density gradient purification in a COBE 2991 centrifuge (COBE, Lakewood, CO).
Recipients were rendered diabetic by streptozotocin administration (10) and received an intrahepatic islet cell infusion [4,000–16,400 islet equivalent (IEQ)/kg body weight] under general anesthesia on postoperative day (POD) zero.
Immunosuppression
Table 1 summarizes the various experimental protocols and immunosuppressive regimens used to treat the animals. All animals received daily intramuscular (IM) sirolimus (LC Laboratories, Woburn, MA) to achieve and maintain trough levels between 12 and 20 ng/ml.
CMV DNAemia in the Peripheral Blood of Islet Cell Transplanted Animals Treated With Different Immunosuppressive Protocols

With the exception of groups 9 and 10, all animals received at least one donor bone marrow infusion between POD -1 and 28.
Controls.
Animals received oral valganciclovir starting POD -14 to -7 twice a day (20 mg/kg marked as XX, or 10 mg/kg marked as X).
CMV DNAemia was defined as detection of CMV DNA loads of more than 0.1 copies/ng DNA in a sample.
Samarium-Lexidronam (Sm-Lex) Protocol
Fifteen animals were administered 153Samarium-Lexidronam (Quadramet, Berlex Laboratories, Wayne, NJ) intravenously (IV) at 3 mCi/kg on days 12–14 before islet transplantation. Among the 15 monkeys, five groups were divided based on the immunosuppressive induction treatment received as described in detail in Table 1 (groups 1–5). All 15 monkeys received either single or multiple doses of donor bone marrow infusions between PODs -1 and 28. One animal received anti-CD40L monoclonal antibody (murine 5c8 clone).
Anti-CD40L Protocol
Eight monkeys were administered IV anti-CD40L specific monoclonal antibody (IDEC 131, Biogen IDEC, Boston, MA) at 20 mg/kg on PODs -1, 0, 3, 10, 18, 28 and every 28 days for 6 months (groups 6–7, Table 1). Five of eight also received thymoglobulin at 10 mg/kg and fludarabine at 50 mg/m2 on PODs -5, -4, -3, and -2 (group 6, Table 1). All eight monkeys were given at least one dose of donor bone marrow infusion between PODs -1 and 28.
Thymoglobulin and Fludarabine Protocol
Six monkeys received thymoglobulin at 10 mg/kg and fludarabine at 50 mg/m2 on PODs -5, -4, -3, and -2; and 2–3 doses of donor bone marrow infusion between POD -1 and 11 (group 8, Table 1).
Marginal Mass Protocol
Eighteen monkeys participated in a marginal mass model protocol in which islets isolated from one or a pool of two donors were infused into two metabolically closely matched recipients at a dose of 5,000 IEQ/kg (10). One of the paired recipients received an agent designed to limit early islet loss or enhance engraftment, including inhibitors of coagulation, growth factors, or inhibitors of inflammation (group 9, Table 1). The matched recipients were used as controls (group 10, Table 1). This protocol included a steroid-free immunosuppression, consisting of IV daclizumab induction (five doses at 1 mg/kg starting POD -1 every 2 weeks), daily IM tacrolimus (Astellas Pharma US, Inc., Deerfield, IL; trough levels of 4–6 ng/ml), and daily IM sirolimus (trough levels of 12–20 ng/ml) starting POD -2 or -1.
CMV Prophylaxis and Treatment
Except for the animals in groups 7, 9, and 10, all other animals received prophylaxis treatment for CMV by giving valganciclovir (Roche) in a Fig Newton cookie (Kraft Foods, Inc.) starting on POD -14 to -7. Animals in groups 1, 4, 5, 6, 8, and 2/9 in group 9 received 20 mg/kg, twice a day (n = 22), and animals in groups 2 and 3 received 10 mg/kg, twice a day (n = 6). Seven of nine animals in group 9 and all animals in groups 7 and 10 (n = 19) received valganciclovir treatment only when a posttransplant blood sample tested positive for CMV DNA by PCR. When animals had CMV levels over 10 copies/ng DNA, IV ganciclovir (10 mg/kg; Cytovene, Roche) was given for a course of 7 days followed by oral valganciclovir at 20 mg/kg twice daily.
CMV Monitoring
CMV DNA was quantitated in peripheral blood by using real-time PCR (LightCycler, Roche) with the primers and probe described by Kaur et al. (36). DNA was extracted from EDTA-anticoagulated blood by using “QIAamp DNA Blood Mini” kit (Qiagen, Inc., Valencia, CA). Amplification of each sample was performed in a 20-μl mixture containing 1x LightCycler FastStart DNA Master Hybridization Probe (Roche), 4 mM MgCl2, 0.5 μM of each primer, 1 μM TaqMan Probe, and 50 ng genomic DNA. PCR amplification consisted of 95°C for 10 min, followed by 40 cycles of denaturation at 95°C for 15 s, and annealing and/or extension for 1 min at 60°C. Serial 10-fold dilutions of CMV DNA fragment (copy number 107–101) were included in every PCR run for the construction of the standard curve for quantitation of CMV DNA in the samples. CMV results were expressed as CMV copies/ng DNA. Each sample was analyzed in duplicate and β-actin amplification was used to ensure the quality of each sample (25).
All animals underwent CMV testing weekly for the first month posttransplant and biweekly thereafter. CMV DNAemia was defined as the detection of a CMV DNA load of more than 0.1 copies/ng DNA in a sample. If CMV DNAemia was detected in any animal, weekly samples were tested until CMV DNAemia was cleared. Various tissues were collected at the time of animal necropsy for CMV DNA determination when an animal was seriously ill or when CMV infection was suspected.
Immunophenotyping
Cell surface antigens on whole blood were analyzed using a Coulter Epics XL flow cytometry (Beckman Coulter) (48). Briefly, 100 μl of EDTA-anticoagulated blood was stained with the following monoclonal antibodies (mAbs): anti-CD3 FITC (clone SP34; BD Pharmingen, San Diego, CA), anti-CD4 PE (clone MT477; BD Pharmingen), anti-CD8 PC5 (clone B911; Beckman Coulter, Fullerton, CA), and anti-CD20 ECD (clone B9E9; Beckman Coulter), and the appropriate isotypic controls. Erythrocytes were lysed using an ImmunoPrep Reagent System and a Q-Prep Workstation (Beckman Coulter). Lymphocytes were gated on the forward and side scatter pattern and 10,000 events were collected. Data are represented as an absolute count (cells/μl).
Statistics and Data Analysis
All values <0.1 copies/ng DNA limit have been considered as 0 (no DNAemia) for the quantitative analysis. Duration was calculated as the longest continuous period of consecutive positive DNAemia observations. Comparisons of CMV DNAemia among different treatment groups were carried out by linear mixed models or two-sample t-tests (assuming equal variances) with α = 0.05. As a composite measure of severity and duration, an area under the curve (AUC) type descriptor has been calculated using the linear trapezoidal rule. The multiple linear regression type analysis has been performed using indicator variables (I) for the presence (I = 1) or absence (I = 0) of each medication using all available data (n = 706) for the CMV DNAemia intensity. Because of the wide-ranging distribution of variables, log-transformed data were used as customary to better normalize distribution, and a loge(y + 1) type transformation was employ when 0 values were present (50,54). For the regression, manual forward selection of descriptors has been performed until no more descriptors with statistically significant contributions could be found. Because intercepts were not significantly different from zero (p = 0.47), a zero intercept was enforced in all cases; this considerably improved all correlations, and it is in line with the assumption that DNAemia is present mainly because of the immunosuppressive regimen. Statistical analyses were performed using SPSS 12.0 (SPSS, Inc., Chicago, IL) and MS Excel 2007 (Microsoft, Seattle, WA).
Results
CMV DNAemia was detected in the peripheral blood of 27/47 or 57.4% of the monkeys studies (Table 1, Fig. 1A). Among them, 16 animals were positive for a period of time ranging from 7 to 210 days; the rest of the 11 animals had one positive sample point and went back to negative status within 7 days, once (n = 5/11) or multiple times (n = 6/11). The frequency, intensity, and duration of CMV viral load were affected by the choices of immunosuppressive treatment as discussed below.
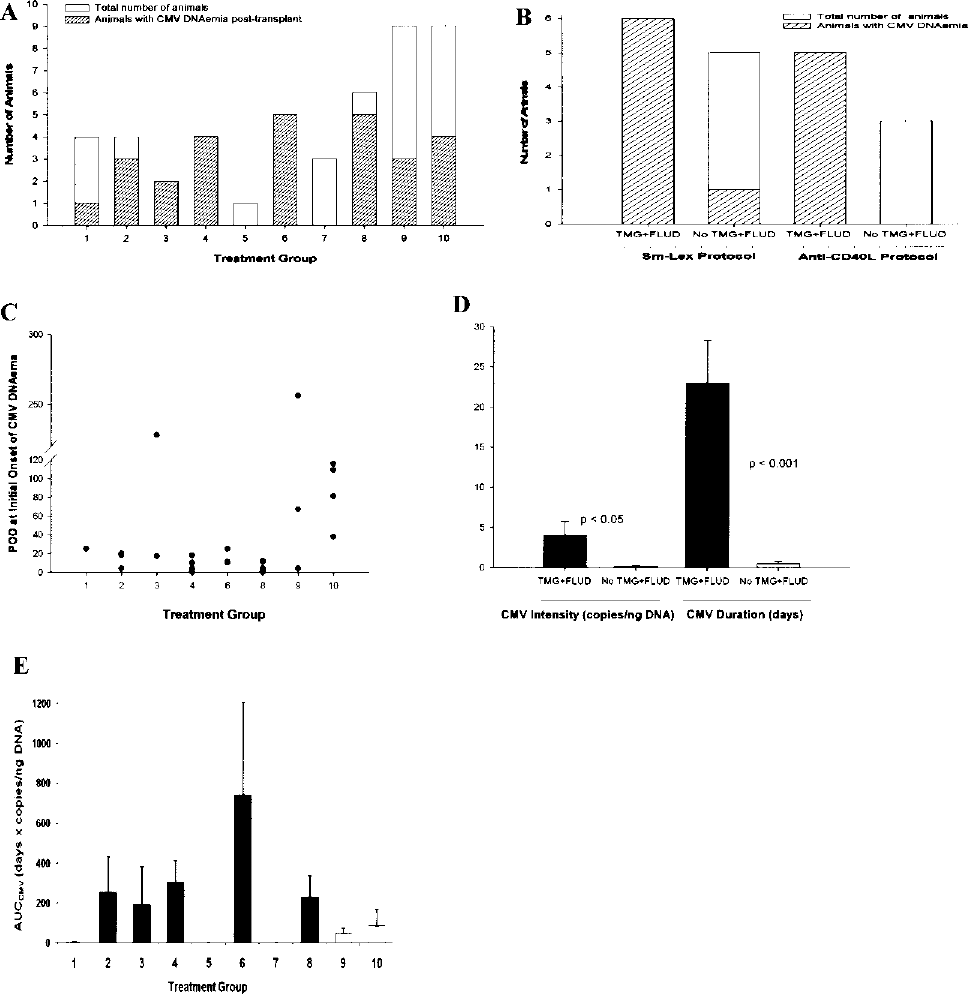
CMV DNAemia in allogeneic islet cell transplanted cynomolgus monkey recipients. (A) Independent treatment groups. (B) Effect of treatment with and without thymoglobulin (TMG) and fludarabine (FLUD) in Sm-Lex and anti-CD40L protocols. The striped boxes show the number of monkeys with CMV DNAemia (CMV DNAemia was defined as detection of CMV DNA loads of more than 0.1 copies/ng DNA in a sample). (C) POD of first positive peripheral blood sample. (D) The average intensity and duration of CMV DNAemia in the peripheral blood of animals with and without TMG and FLUD therapy. (E) Area under the curve (AUC) as a composite measure of intensity and duration of CMV DNAemia per treatment group, average per treatment groups shown, groups receiving TMG ± FLUD are shown in black.
Immunosuppression and CMV DNAemia
Thymoglobulin and Fludarabine Are Associated with an Increased Risk of CMV DNAemia in Islet Cell Transplanted Monkeys
The incidence of CMV DNAemia in animals that received thymoglobulin induction therapy with or without fludarabine was significantly higher (p < 0.01) compared to that seen in animals that received other treatments (Table 1, Fig. 1A, B). The highest incidence of CMV DNAemia was observed in animals treated with both thymoglobulin and fludarabine (100%, groups 3, 4, and 6; Table 1, Fig. 1A, B). Monkeys that received thymoglobulin without fludarabine (group 2, Table 1) also had a higher incidence of CMV DNAemia (3/4, 75%) compared to those that did not receive thymoglobulin (p < 0.05).
Cyclophosphamide
Induction treatment with cyclophosphamide alone had no significant impact on the occurrence of CMV DNAemia. Four animals received cyclophosphamide only as an induction treatment in the Sm-Lex protocol (group 1, Table 1), and only 1/4 animals experienced a weak “one-point-CMV DNAemia” throughout the posttransplant monitoring period.
Steroid-Free Immune Suppression (SFIS) (Marginal Mass Protocol)
Animals that received SFIS were found to develop CMV DNAemia less frequently: in a total of 18 animals, only 33% of group 9 (3/9) and 44% of group 10 (4/9) had measurable CMV virus (Table 1, Fig. 1A), a marked contrast with recipients of thymoglobulin ± fludarabine (p < 0.001). Furthermore, there was no difference in CMV DNAemia between “controls” and the matched recipients treated with an additional agent (p > 0.05). The level of sirolimus in groups 9 and 10 was not associated with occurrence of CMV DNAemia (p = 0.99).
Time of Onset and Intensity of CMV DNAemia Level
Data regarding time of onset for all animals that tested positive is shown in Figure 1C. For 6/7 SFIS-treated, CMV positive animals, CMV DNAemia was detected in blood after the first month posttransplant (POD 38–256), but onset was much earlier for other treatment groups.
The distributions of intensity and duration of CMV DNAemia in the different treatment groups are summarized in Table 2. CMV levels and duration varied among individual animals even within the same treatment group. Animals that had received thymoglobulin alone or together with fludarabine induction therapy had a significantly higher average intensity (4.02 ± 1.75 vs. 0.19 ± 0.10 copies/ng DNA; p < 0.05) and longer duration (23.0 ± 5.3 vs. 0.5 ± 0.3 days; p < 0.001) of CMV DNAemia compared to those animals that did not receive thymoglobulin and fludarabine (Fig. 1D). As a composite measure of severity and duration, an AUC type descriptor was calculated. This also showed a very clear difference between the thymoglobulin ± fludarabine group and the rest of the animals (367.0 ± 121.1 vs. 75.4 ± 40.2 days x copies/ng DNA; p < 0.05); a corresponding comparison for all groups is shown in Figure 1E.
Duration and Intensity of CMV DNAemia in the Peripheral Blood of Islet Cell Transplanted Animals Receiving Different Protocols and Immunosuppressive Treatments

Data are expressed as mean ± SEM. AUC, area under the curve.
CMV data for one animal were not included in group 10. That animal had CMV DNAemia starting POD 38 and did not receive valganciclovir treatment until POD 294, because samples in that animal were archived and CMV was measured later.
In an attempt to quantitatively assess the relative contribution of each treatment to CMV DNAemia, a multiple linear regression type analysis using indicator variables for the presence or absence of each medication was performed (see Materials and Methods). Analysis of all log-transformed CMV DNAemia data (n = 706) resulted in the following equation, which incorporates only the statistically significant contributions (i.e., only those treatments that had a statistically significant nonzero contribution in increasing DNAemia intensity):
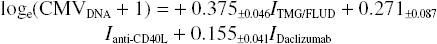
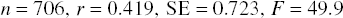
This indicates that animals receiving thymoglubolin ± fludarabine had, on average, an increase in their DNAemia of 0.45 copies/ng DNA (e0.375 − 1 = 0.45) (despite many of them receiving effective ganciclovir therapy) (p = 2.5 × 10−15) and those receiving daclizumab had an average increase of 0.17 copies/ng DNA (p = 0.0002). Those receiving anti-CD40L had an average increase of 0.31 (p = 0.002); however, the group receiving anti-CD40L alone (group 7, n = 3) showed no DNAemia.
Donor Tissues and CMV DNAemia
Before transplantation, all donors were CMV negative in peripheral blood. The effect of donor islets on the occurrence of CMV DNAemia was examined in recipients that received only islets and no bone marrow (groups 9 and 10, Table 1). Animals in group 9 (agent-treated recipients) and their matched control recipients (group 10) received the same amount of islets from the same donor. Among the nine pairs of animals in the two groups, seven animals experienced CMV DNAemia and only one pair was positive for CMV DNA in both treated and control animals. The rest occurred randomly among different recipients. To assess the effect of donor bone marrow, we compared CMV DNAemia in groups 6 and 7 (Table 1). Animals in both groups were under anti-CD40L protocol and received multiple doses of bone marrow infusions between PODs -1 and 28, but with different immunosuppressive treatments. All five animals that received thymoglobulin and fludarabine (group 6) experienced CMV DNAemia, as opposed to none of the three animals that received other immunosuppressive treatment in group 7.
Effect of WBC, Lymphocyte, T- and B-Cell Counts on Incidence of CMV DNAemia
Decreased levels of WBC, and T and B cells were highly associated with the occurrence of CMV DNAemia in transplanted animals. In animals receiving thymoglobulin ± fludarabine, the WBC, lymphocyte, and T-cell nadirs occurred within a week before or after islet transplant, with reductions of WBC, lymphocyte, total CD3+, CD3+CD4+, and CD3+CD8+ subsets greater than 77%, 93%, 95%, 97%, and 95%, respectively, compared to preimmunosuppression levels (Table 3). CMV DNAemia (75–100%) occurred 1–3 weeks later. B-cell reduction ranged from 84% to 94% and occurred within the first 2 months posttransplant. Although not as dramatic as the aforementioned data, SFIS-treated animals also experienced reduction of WBC and lymphocytes, and onset of CMV DNAemia occurred later, with a reduced intensity of disease. Figure 2A shows the relationship between CMV DNA levels and WBC, lymphocyte counts posttransplant in three animals in the Sm-Lex protocol. Figure 2B shows CMV DNAemia in relation to the levels of CD3+, CD3+CD4+, CD3+CD8+, and CD20+ lymphocyte subsets in different treatment groups. The nadirs of WBC, lymphocytes, T and B cells for animals that developed CMV DNAemia within the next 3 weeks were all significantly lower than the respective levels for animals without CMV DNAemia (p < 0.05). We observed that 66.7% of animals with a lowest WBC level less than 4,500 cells/μl and 55.2% of animals with a lowest value of lymphocyte less than 1,800 cells/μl developed CMV DNAemia within the 3 weeks after the nadir; while no animal with a lowest WBC level greater than 4,500 cells/μl or with a lowest lymphocyte count greater than 1,800 cells/μl developed CMV within the next 3 weeks (Fig. 3). When the lowest levels of CD3+, CD3+CD4+, CD3+CD8+, or CD20+ were less than 300, 200, 150, or 300 cells/μl, respectively, 85%, 88.9%, 80%, or 69.6% animals, respectively, developed CMV DNAemia within the next three weeks; however, no animal developed CMV DNAemia within the three weeks when the lowest levels of CD3+, CD3+CD4+, CD3+CD8+, or CD20+ were above the respective levels (Fig. 3).
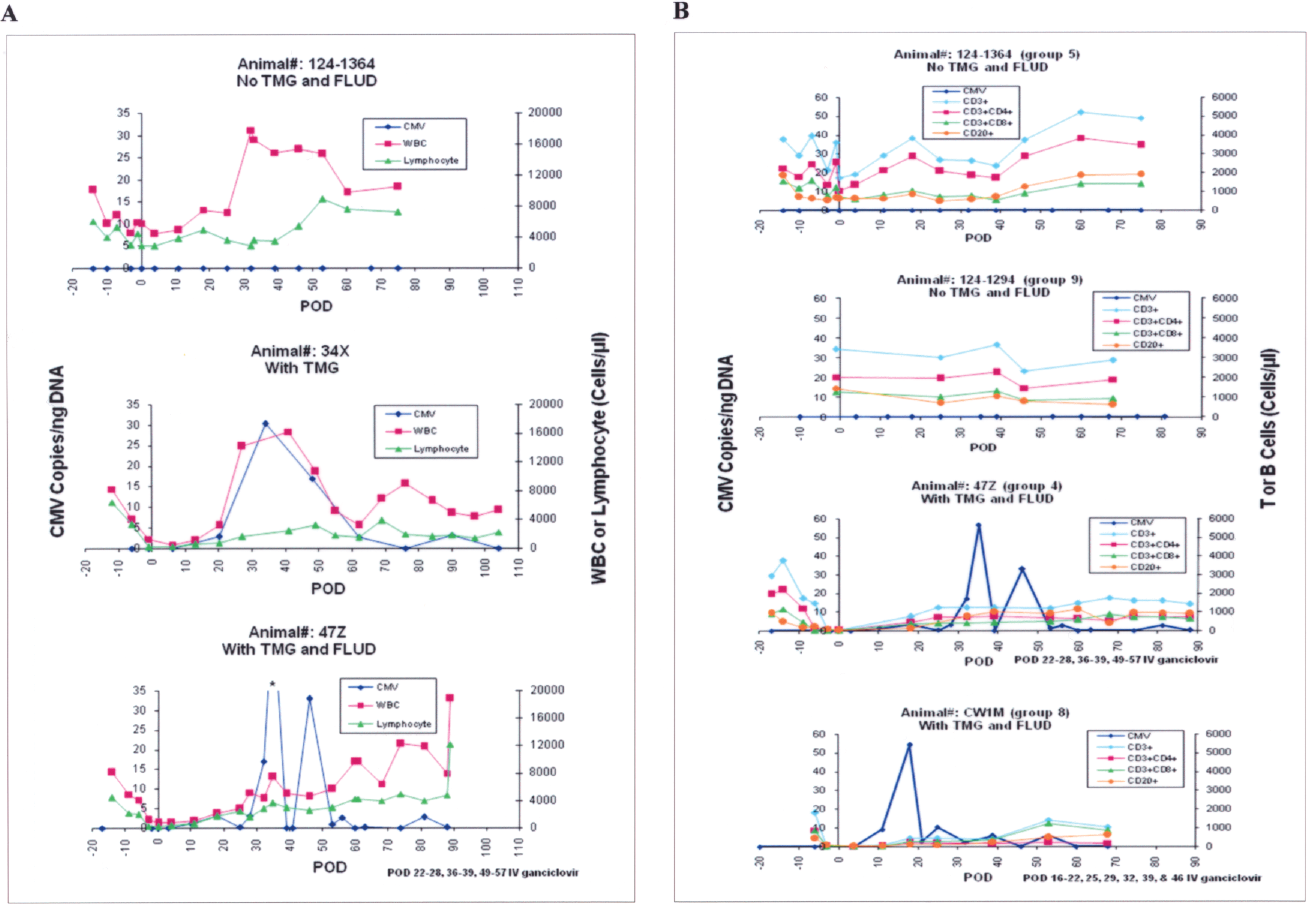
(A) CMV gene levels in peripheral blood samples versus WBC and lymphocyte counts in three animals participating in the Sm-Lex protocol. (B) CMV gene levels in peripheral blood samples versus CD3+, CD3+CD4+, CD3+CD8+ T cells, and CD20+ B cells in animals received different immunosuppressive treatment (group 4: Sm-Lex protocol with TMG and FLUD; group 5: Sm-Lex protocol without TMG and FLUD; group 8: TMG and FLUD; group 9: steroid-free immunosuppression). *Out of range. Sm-Lex, samarium-lexidroram; TMG, thymoglobulin; FLUD, fludarabine.
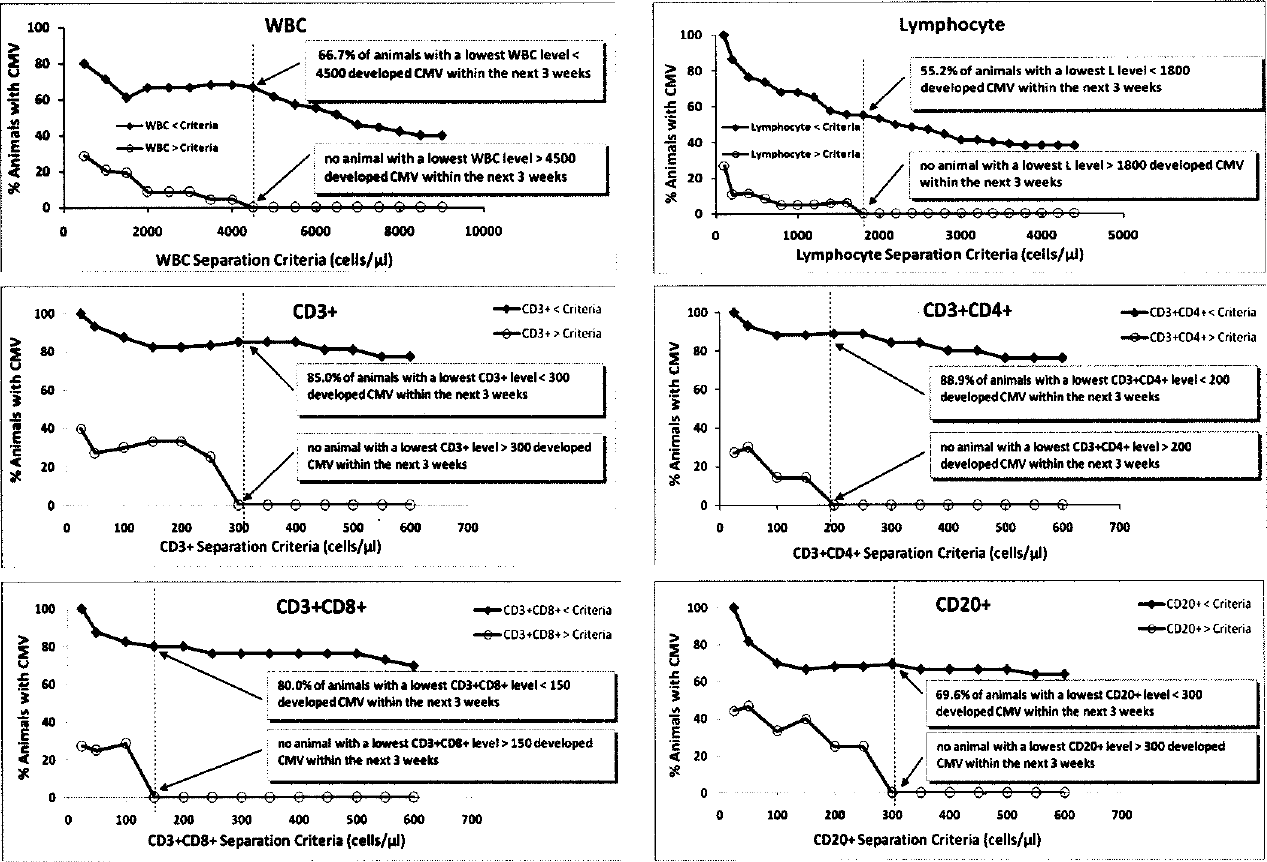
Likelihood of developing CMV DNAemia as a function of WBC and lymphocyte (n = 44), and CD3+, CD3+CD4+, CD3+CD8+, and CD20+ (n = 26) criteria used to separate animals with respective lowest level, below or above selected criteria (lymphocyte data was obtained after omission of one animal that developed CMV within 3 weeks despite a lowest lymphocyte level of 4028 cells/μl).
Lowest Level and Percentage of Reduction of WBC, Lymphocyte, CD3+, CD3+CD4+, CD3+CD8+ T Cells, and CD20+ B Cells and the Incidence of CMV DNAemia in the Peripheral Blood of Islet Cell Transplanted Animals That Received Different Immunosuppressive Treatments
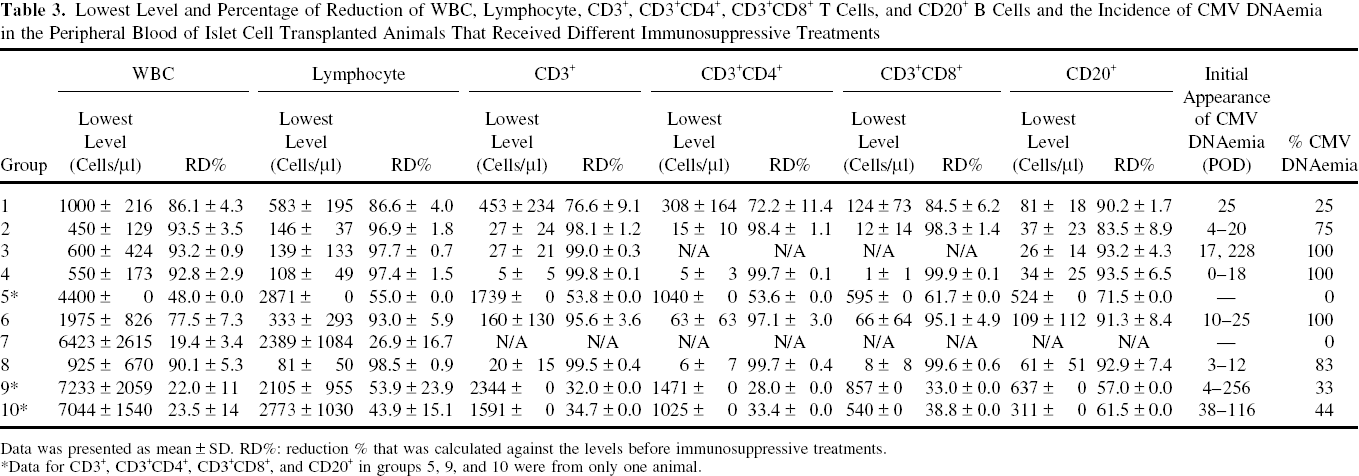
Data was presented as mean ± SD. RD%: reduction % that was calculated against the levels before immunosuppressive treatments.
Data for CD3+, CD3+CD4+, CD3+CD8+, and CD20+ in groups 5, 9, and 10 were from only one animal.
Effectiveness of Valganciclovir on Prevention of CMV DNAemia
Valganciclovir prophylaxis (20 or 10 mg/kg) with induction immunosuppressants did not consistently prevent the occurrence of CMV DNAemia in monkeys that received thymoglobulin alone or together with fludarabine. In fact, the aforementioned regression analyses suggested no preventive effect for this treatment (data not shown). IV infusion of ganciclovir was effective in reducing or eliminating CMV DNAemia in all treated animals; the average slope calculated for the periods immediately surrounding the duration of IV ganciclovir treatments was −1.68 copies/ng DNA/day for the total of 10 treatments in 7 different animals.
Recurrence of CMV DNAemia
Recurrence of CMV DNAemia was found in 18 out of 27. Most of the animals (14/18) were receiving oral valganciclovir therapy, but none were on IV ganciclovir therapy at the time of recurrence. Average intensities between the recurrent and initial CMV DNAemia were similar (7.3 ± 2.3 vs. 7.1 ± 1.5 copies/ng DNA), but durations were much shorter in the recurrent CMV DNAemia than in the initial onset (3.1 ± 0.7 vs. 23.3 ± 6.1 days).
CMV DNAemia and CMV Disease
Six animals showed clinical evidence of CMV disease posttransplant. Two of these animals each were from groups 6, 8, and 10 (Table 1). Common clinical signs included weight loss, anorexia, soft feces, and lethargy. Laboratory abnormalities included increased liver enzymes (AST), thrombocytopenia, and occasionally pancytopenia (data not shown). Of the six animals that died or were euthanatized from overt CMV disease, three received IV ganciclovir therapy for 7–14 days. These animals experienced a decrease in the level of CMV DNAemia; however, they invariably had a recurrence after IV therapy was discontinued. Oral valganciclovir was given to four animals, and a therapeutic benefit was not seen. The incidence of CMV disease in animals that received thymoglobulin ± fludarabine was 19%, whereas it was 7.7% in the animals without thymoglobulin ± fludarabine therapy.
CMV Distribution in Postmortem Tissues
Table 4 shows CMV DNA levels in different tissues for all animals that had at least one positive tissue. In the animals diagnosed with CMV disease, the tissues most frequently positive for CMV DNA were kidneys and heart (6/6), followed by lung and liver (4/5), spleen and mesenteric lymph nodes (4/6), jejunum (3/6), stomach, duodenum, and inguinal lymph nodes (2/5), and ileum (2/6). When an animal was severely infected with CMV, all the tissues tested were positive as is shown for animal 13–1005. In the animals with positive CMV DNA, but no CMV disease (animal #93–353, 111X, 124–1349), kidney and mesenteric lymph nodes were CMV positive. Pancreas and spleen were CMV positive in one of the three animals, and all other tissues tested were CMV negative.
CMV DNA Levels in Postmortem Animal Tissues

CMV was expressed as copy numbers/ng DNA; UD, undectable.
Results were the average of left and right kidney samples.
Results were the average of one or more data from liver: left central lobe, left lateral, right central lobe, right lateral lobe, and caudate lobe.
Results were the average of one or more data from lung: left lingular, left lower, left upper, right lower, right middle, and right upper.
Results were the average of one or more data from pancreas body, head, and tail.
Discussion
Similar to CMV infection in humans, CMV is ubiquitous in NHP populations (5). Of the many CMV strains isolated from numerous species of NHP, rhesus CMV (RhCMV), isolated from rhesus macaques, is the best characterized. It has been reported that almost all the rhesus macaques, whether in the wild or in captivity, are seropositive by 1 year of age (5,57). Seropositive animals persistently secret infectious virus during their lifetime (4,33), and CMV is probably transmitted from infected to uninfected animals via excretion of virus in breast milk, saliva, and urine (2), the same transmission routes as in humans. CMV infection is asymptomatic in immunocompetent NHP (52); however, just as in human transplants (8,28), in immunosuppressed NHP recipients after organ transplantation CMV infection can be reactivated and lead to CMV disease, which can be life threatening. CMV reactivation has been reported in cynomolgus (29,35) and rhesus (1,12,44) monkeys and baboons (40) after organ transplantation. Immunosuppressive regimen used in transplantation has a major impact on the frequency and severity of CMV reactivation. In this study, we demonstrate that CMV DNAemia not only occurred more frequently and earlier, but was also more severe in islet cell transplanted cynomolgus monkeys that had received thymoglobulin alone or together with fludarabine. CMV DNAemia in thymoglobulin-alone animals was slightly, but not significantly, lower than in animals treated with both thymoglobulin and fludarabine. The appearance of CMV DNAemia within the first month posttransplant in animals receiving thymoglobulin induction therapy with or without fludarabine may be a consequence of extensive WBC and lymphocyte depletion. CMV DNAemia occurred after POD 60 in most of the animals with other induction treatments. Despite the limited number of animals included in each group in the study, these findings have potential relevance in CMV prevention in cell transplantation in NHP as described below.
Higher CMV incidence and infection was reported coincided with a significant reduction of CD3+ and CD4+ cells in AIDS patients (21,47) and in kidney transplant recipients (15,18). In this study we found that the numbers of WBC, lymphocytes, CD3+, CD3+CD4+, CD3+CD8+, and CD20+ cells were drastically decreased in islet transplanted NHP after thymoglobulin treatment, with an even greater reduction in lymphocyte, T and B cells when combined thymoglobulin/fludarabine therapy was used. Levels of WBC, lymphocytes, T and B cells were highly associated with the frequency, intensity, and duration of CMV DNAemia in the transplanted animals. The value of WBC, T and B cells at the nadir of cell depletion greatly affect the occurrence of CMV DNAemia. We found that no animals developed CMV DNAemia with the next 3 weeks when the lowest value of WBC, lymphocyte, CD3+, CD3+CD4+, CD3+CD8+, or CD20+ cells was above 4500, 1800, 300, 200, 150, or 300 cells/μl, respectively. To our knowledge, this is the first time that threshold levels of WBC, lymphocyte, T and B cells for CMV infection in immunosuppressed transplanted animals are reported. These threshold levels should serve as a guideline for transplanted animals undergoing immunosuppressive protocols.
In monkeys that had undergone SFIS therapy, the incidence of CMV DNAemia was much lower. These results are in agreement with previous findings from our Clinical Islet Transplant Program; only one recipient with CMV disease was observed despite CMV positive donors and negative recipients (n = 29 patients) (16,24).
In humans, administration of antiviral prophylaxis during the first 3 months following solid organ transplantation has been shown to have a significant impact on the frequency, timing, and outcome of CMV infection and disease (13,32,42,51). Both preemptive valganciclovir therapy and valganciclovir prophylaxis were found equally effective in the prevention of CMV disease after renal transplantation (45). However, two cases of CMV infection and one case of CMV disease were reported during the 3-month period of ongoing CMV prophylaxis with oral valganciclovir posttransplant in islet transplantation, in which induction therapy with antithymocyte globulin was used (59). Prophylactic ganciclovir (2.5 mg/kg/day, intramuscular) treatment was used in immunosuppressed cynomolgus macaque recipients of porcine islets (30) or embryonic pig pancreatic tissue (29). However, even under ganciclovir prophylaxis, CMV reactivation was reported in one fourth of animals that received thymoglobulin induction treatment (29). In our study in cynomolgus monkeys, oral valganciclovir prophylaxis did not consistently prevent the onset of CMV DNAemia and CMV disease; however, treatment of animals with newly detected CMV DNAemia (IV ganciclovir) was effective in reducing or eliminating CMV DNAemia. It should be noted that both oral and intravenous administration of medication is much more difficult in NHP than in clinical patients. Oral medications must be broken or crushed and added to a palatable treat, such as a Fig Newton cookie, which adds variability in the amount of drug actually consumed by the animal.
Donor tissues, islets, and/or bone marrow did not appear to affect CMV DNAemia in transplanted recipients. This was clearly observed in pairs of recipients that received the same amount of islets from the same donor under the same immune suppression, and one animal received the experimental agent, while the other served as control. In only 1/9 pairs of such animals both experimental and control recipients developed CMV DNAemia, while CMV DNAemia in the rest of the recipients occurred randomly. In addition, in recipients of several doses of bone marrow under a similar protocol, only those animals that received thymoglobulin and fludarabine developed CMV DNAemia.
No correlation was observed between the occurrence of CMV DNAemia and the decrease in graft function (assessed by the c-peptide value within 3 weeks of occurrence of CMV DNAemia) in the studied monkeys (data not shown). However, CMV infection had a significant negative impact on the survival of transplanted animals. Six recipients had poor appetite, diarrhea, and weight loss in association with CMV disease and expired or had to be euthanatized.
In conclusion, the prevalence of CMV infection in animals treated with SFIS was significantly lower than in animals treated with thymoglobulin ± fludarabine. Induction treatment with thymoglobulin and fludarabine significantly increased the frequency, intensity, and duration of CMV infection with drastic reduction of WBC, lymphocytes, CD3+, CD3+CD4+, CD3+CD8+, and CD20+ cells. Even though the effect of the use of thymoglobulin in CMV reactivation and disease in clinical transplantation has been reported before, here, for the first time, we show the levels of WBC, T and B cell above which there is a minimum risk of CMV infection in NHP recipients of islet cell transplants under various immunosuppressive protocols. One of the significant points of this study is that when testing novel immunosuppressants that may cause drastic reductions in WBC, T and B cells in NHP, prophylactic CMV treatment of recipients with ganciclovir (IV or IM) should be used, together with a close monitoring of WBC, T and B cell, and CMV levels, especially within the first month posttransplant. Oral valgancyclovir prophylaxis cannot prevent CMV infection in this large-animal model. Therefore, once CMV DNAemia is detected, IV ganciclovir should be administered immediately to prevent the development of CMV disease.
Footnotes
Acknowledgments
This work was supported by the Diabetes Research Institute Foundation, Hollywood, FL and the following grants: National Institute of Health (U19AI43900, U19AI51728, R01DK55178), and Juvenile Diabetes Research Foundation International (2004-361, 2004-808). The authors gratefully acknowledge the expert assistance of Ms. Xiumin Xu, Ms. Dalina Alvarez, Ms. Gazelle Rouhani, Ms. Julie Zutel, Ms. Melissa Burgos, Ms. Mariana Khawand, Mr. Waldo Diaz, Mr. James Geary, Mr. Daniel Ortiz, Mr. Alexander Rabassa, Ms. Ana Hernandez, and Ms. Ena Poumian-Ruiz. Special thanks to Ms. Cynthia Healy for editing the manuscript. Dr. Joshua Miller's lab performed CBC and liver enzyme tests.
