Abstract
Induced pluripotent stem (iPS) cells have recently been generated by Yamanaka's group, and then followed by others. iPS cells are expected to have clinical applications including an important role in regenerative medicine. This study focused on the cell-penetrating peptides (CPPs) for differentiation or functional application of iPS cells, because several transduction domains can deliver a large size-independent variety of molecules into cells. Two CPPs, Texas Red-R8 and Rhodamine-TAT, were generated as representative CPPs and these CPPs were tested to determine their ability to penetrate the membrane of iPS cells. Both CPPs were transduced in iPS cells through macropinocytosis classified in endocytosis within 2 h in a manner consistent with many other cells, and no cytotoxicity and influence on their undifferentiated state was observed. In conclusion, CPPs can be utilized for their differentiation or functional application in iPS cells.
Keywords
Introduction
Embryonic stem (ES) cells have multilineage potential and provide numerous applications in regenerative medicine (17, 22). However, there are some problems associated with transplantation therapies using ES cells such as immune rejection and ethical controversies (10, 36). As a result, it is difficult to generate the patient or disease-specific ES cells that are required for their effective application. Mouse and human skin fibroblasts can be transformed into a novel ES cell-like pluripotent cells with capacity for self-renewal, termed induced pluripotent stem (iPS) cells (2, 32). The iPS cells were generated by ectopic expression of four transcription factors, Oct3/4, Sox2, Klf4, c-myc (14, 17). Yamanaka et al. showed that mouse iPS cells could not be distinguished from ES cells in morphology, proliferation, gene expression, and teratoma formation (1, 6, 8, 12, 23, 29). The iPS cells may play a key role in overcoming the problems associated with cell transplantation of ES cells, and differentiation and selection methods for target cells are needed for regenerative medicine with use of iPS cells (30).
This study focused on the cell-penetrating peptides (CPPs), also known as protein transduction domains (PTDs), for differentiation or functional application in iPS cells, because it is well known that several transduction domains can deliver a large size-independent variety of molecules into cells, including proteins, peptides, antisense oligonucleotides, large metal beads, quantum dots, and liposome. Small CPPs from the Tat protein of human immunodeficiency virus (HIV-1) (4, 35), VP22 protein of herpes simplex virus (3), and Antennapedia (Antp) homeoprotein of Drospholia (27) fuse to biologically active proteins, and are capable of transducing the proteins across the membrane of many mammalian tissues (18, 21). These CPPs consist of arginine and lysine-rich sequences. Recently, poly-arginine (PolyR), especially 8–11-arginine peptides, and poly-lysine have been shown to exhibit an even greater efficiency in the delivery of several peptides and proteins (20). Biological studies have shown that CPP fusion proteins or peptides can elicit phenotypic changes in live cells both in culture and in vivo. On the other hand, the mechanism of CPP transduction has become clear, and the main mechanism is an electrostatic interaction with the plasma membrane, penetration into cells by macropinocytosis (15, 16, 38), and release to cytoplasm and nuclei by retrograde transport (5, 7). Macropinocytosis is independent of clathrin-mediated and caveolin-mediated endocytosis, and the size of the uptake particle is more than 1 μm, and it requires dynamin GTPase activity (19). Cell membrane heparin sulfate proteoglycans (HSPG) mainly act as receptors for extracellular uptake.
This study investigated whether CPPs including 8-arginine straight chain (R8) and Tat peptides (TAT) conjugated with fluorescent labels can penetrate the cell membrane of iPS cells. Moreover, the time course and dose dependency of CPPs transduction into iPS cells were examined.
Materials and Methods
iPS Cells Culture
The iPS cells (iPS-MEF-Ng-20D-17) established by Prof. S. Yamanaka in Kyoto University were purchased from Cell Bank of Riken Bioresource Center.
MEF feeder cells (Chemicon International Lot 70915-9) were prepared. The MEF cells were cultured at 37°C with 5% CO2 in MEF culture medium [Dulbecco's modified Eagle medium (DMEM) with 10% heat-inactivated fetal bovine serum (FBS) and 1% penicillin-streptomycin], and maintained until they reached confluence. The cells were treated by 50 μl 100x mitomycin-C and incubated for more than 2 h. Thereafter, they were cultured in the 0.1% gelatin-coated plate for more than 5 h.
The iPS cells were maintained at 37°C with 5% CO2 in ES culture medium [DMEM containing 15% FBS, 1x NEAA, 1 mM sodium pyruvate, 2 mM 2-mercaptoethanol, 1% penicillin-streptomycin and 1000 U/ml mouse leukemia inhibitory factor (LIF)] on feeder layers of already mitomycin-treated MEF cells. The medium was changed to a fresh ES culture medium every day and a passage culture was conducted every 3–4 days (22, 31, 33).
Cell-Penaetrating Peptides (CPPs) Conjugated with Fluorescent Labeling
All CPPs were chemically synthesized by Fmoc (9-fluorenylmethyloxycarbonyl) solid-phase peptide synthesis on a rink amide resin as previously reported. De-protection of the peptide and cleavage from the resin were performed by treatment with a trifluoroacetic acid/ethanedithiol mixture (95:5) at room temperature for 3 h. Fluorescent labeling of the CPPs was conducted by treatment with 1.5 equiv. of Texas red and Rhodamine C5 maleimide sodium salt in a dimethylformamide/methanol mixture (1:1) for 1.5 h followed by reverse phase high-performance liquid chromatography (HPLC) purification. The fidelity of the products was ascertained by matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOFMS). MALDI-TOFMS for Texas Red-R8 and Rhodamine-Tat is 1968.4 and 2246.7, respectively.
Evaluation of Dose Dependency of Each CPP Transduction by Flow Cytometry
iPS cells were seeded in each well of MEF-treated 12-well microplate (BD Biosciences) with 400 μl of fresh culture medium in the presence of 0, 1, 2, and 5 μM each CPP for 2 h. The cells were washed three times with PBS, treated with trypsin-EDTA in PBS, and washed again with PBS. Cells were centrifuged at 1000 rpm for 5 min. After the supernatant was removed, the cells were washed three times by PBS and centrifuged in PBS. After this washing cycle, the cells were suspended in 1 ml of PBS and then were used for a flow cytometric analysis on a FACS caliber (BD Biosciences) flow cytometer using 488 nm laser excitation and a 515 to 545 nm emission.
Evaluation of Each CPPs Transduction Time by Flow Cytometry
iPS cells were seeded in each well of MEF-treated 12-well microplate (BD Biosciences) with 300 μl of fresh culture medium in the presence of 5 μM each CPP for 0, 0.5, 1, 2, and 24 h. The cells were washed three times by PBS, treated with trypsin-EDTA in PBS, and washed again with PBS and evaluated the transduction by flow cytometry with same way as for the evaluation of dose dependency of CPPs.
Analysis of the Endocytosis Pathway
To confirm the mechanism by which the transduction of each CPP penetrates the membrane of iPS cells through endocytosis, the effects of an endocytosis inhibitor was examined at a low temperature (an inhibitor of internalization through endocytosis). The iPS cells were seeded in each well of MEF-treated 24-well microplate with 1 ml fresh culture medium for 1 day. The cells were pretreated at 4°C for 30 min. After the addition of 5 μM each CPP, the cells were maintained at the same temperature for 30 min. Next, the cells were washed three times by PBS and analyzed by conventional fluorescence microscopy in live (not fixed) conditions.
Analysis of the Macropinocytosis Pathway
To confirm that macropinocytosis is a major pathway, iPS cells were seeded in each well of MEF-treated 24-well microplate with 1 ml fresh culture medium for 1 day. The iPS cells were pretreated for 1 h in 200 μl serum-free medium with 5 mM amiloride (Sigma) or 10 μM cytochalasin D (Sigma), respectively. After the addition of 5 μM each CPP, the cells were maintained for 1 h in the presence of inhibitors. Thereafter, the cells were washed three times by PBS and incubated for 1 h in complete medium.
Results
Transduction of CPPs Into iPS Cells
The primary structures of CPPs conjugated with Texas Red and Rhodamine used are shown in Figure 1A. To investigate whether the CPPs can be transduced into iPS cells, iPS cells were incubated in MEF-treated 24-well microplate at 37°C with 5 μM of each CPP for 2 h. Thereafter, the cells were washed three times by PBS, and placed in serum-free medium. The red fluorescence of Texas Red and Rhodamine was confirmed by conventional fluorescence microscopy in live conditions (Fig. 1B). In addition, the iPS cells transduced with CPPs maintained their undifferentiated state and no cytotoxicity was confirmed at this concentration. These data suggested that both CPPs could penetrate iPS cells membrane safely within 2 h.
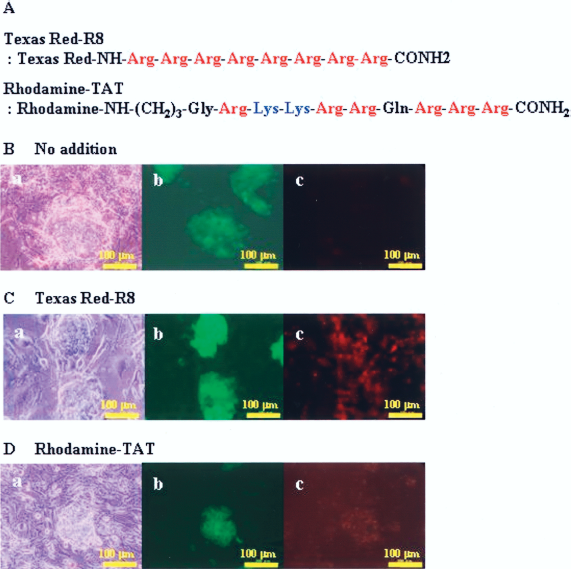
Sequence of cell-penetrating peptides (CPPs) and transduction of CPPs into iPS cells. Sequence of Texas Red-labeled R8 and Rhodamine-labeled TAT peptides are shown (A). Morphology of iPS cells incubated with 5 μM CPPs [Texas Red-R8 (B) or Rhodamine-TAT (C)] at 37°C for 1 h. Phase contrast of iPS cells (B, C-a), GFP fluorescence (B, C-b), and Texas Red and Rhodamine fluorescence (B, C-c) micrographs are shown. The iPS cells demonstrated GFP fluorescence.
Transduction of CPPs in a Dose-Dependent Manner
PDX-1 containing CPPs were transduced into isolated islet and pancreatic ductal cells and in a dose-dependent manner up to 1 μM. Similar results were obtained in the transduction of both CPPs (Texas Red-R8 and Rhodamine-TAT (Fig. 2A, B) into iPS cells. The dose-dependent transduction of both CPPs into iPS cells at 0, 1, 2, and 5 μM was confirmed by measuring the fluorescent intensity of the iPS cells by flow cytometry.

Dose-dependent transduction of extracellular CPPs. iPS cells were incubated with each CPP [Texas Red-R8 (A) or Rhodamine-TAT (B)] at 0, 1, 2, and 5 μM at 37°C for 2 h and washed with PBS three times. Next, the cells were analyzed by flow cytometry. The bar graphs indicate that CPPs were transduced in a dose-dependent manner up to 5 μM.
Uptake Kinetics of Extracellular CPPs
The internalization of both CPPs was visualized by a flow cytometry analysis of the treated cells. In the Texas Red-R8, almost all of the CPP was taken up by iPS cells within 2 h but the CPP uptake continued until 24 h (Fig. 3A). On the other hand, in the case of Rhodamine-TAT, the CPP was almost completely taken up within 2 h (Fig. 3B). These data therefore suggested that both CPPs were efficiently transduced by iPS cells within 2 h.

Uptake kinetics of extracellular CPPs. iPS cells were incubated with 5 μM each CPP [Texas Red-R8 (A) or Rhodamine-TAT (B)] at 37°C for 0, 0.5, 1, 2, and 24 h and washed with PBS three times. At the indicated time points, the cells were analyzed by flow cytometry. The line graphs indicate the CPPs to be efficiently taken up by iPS cells within 2 h.
CPPs Enter iPS Cells by Endocytosis
Endocytosis is an essential cellular process for the uptake of a wide variety of extracellular factors and occurs by functionally distinct mechanisms. To confirm whether CPPs transduction is dependent on endocytosis, iPS cells were treated with CPPs at low temperature (37). iPS cells were preincubated at 4°C for 30 min followed by the addition of 5 μM CPPs and further incubated at 4°C for 30 min. The iPS cells were washed three times by PBS and added fresh culture medium. The cells were observed by conventional fluorescence microscopy. Parallel incubations at 37°C in the absent of this inhibitor were performed as a control. Red fluorescence of Texas Red (Fig. 4A) and Rhodamine (Fig. 4B) in living iPS cells was markedly reduced in incubation at 4°C in comparison to at 37°C. These data suggested that endocytosis plays a key role in the transduction of CPPs into iPS cells.

Transduction of CPPs into iPS cells by endocytosis. iPS cells were incubated with 5 μM each CPP [Texas Red-R8 (A) or Rhodamine-TAT (B)] at 37°C for 30 min (a, b, c). To confirm the effect of the endocytosis, the cells were pretreated at 4°C for 30 min before addition of CPPs, and then treated with 5 μM CPPs at 4°C for 30 min (d, e, f). Phase contrast (a, d), GFP fluorescence (b, e), and Texas Red and Rhodamine fluorescence (c, f) micrographs were shown. These images were analyzed by conventional fluorescence microscopy under live conditions.
Cellular Uptake of CPPs Into iPS Cells by Lipid Raft Macropinocytosis
CPPs are transduced into cells by macropinocytosis. Macropinocytosis is a rapid, lipid raft-dependent and receptor-independent form of endocytosis. It requires actin membrane protrusion that envelopes vesicles that formed macropinosomes. To test the involvement of macropinocytosis classified into endocytosis, iPS cells were preincubated with 5 mM amiloride, a specific inhibitor of the Na+/H+ exchange needed for macropinocytosis (19), or cytochalasin D, an F-actin elongate inhibitor (19) for 1 h. Pretreated cells were washed three times with PBS followed by addition of 5 μM CPPs and incubated for 30 min. Thereafter, the cells were washed by fresh culture medium. The treatment of the cells with both macropinosome inhibitors resulted in a decrease in CPPs transduction into vesicles with no cytotoxicity in living iPS cells (Fig. 5). These data suggested that the internalization of CPPs occurs mainly through lipid raft-mediated macropinocytosis.

CPPs enter iPS cells by lipid raft macropinocytosis. iPS cells were incubated with 5 μM each CPP [Texas Red-R8 (A) or Rhodamine-TAT (B)] at 37°C for 30 min (a, b, c). To confirm the effect of the macropinocytosis, cells were pretreated with 5 mM amiloride, a specific inhibitor of the Na+/H+ exchange required for macropinocytosis, for before addition of CPPs for 1 h (d, e, f). Moreover, cells were pretreated with 10 μM cytochalasin D, an F-actin elongation inhibitor, for before addition of CPPs for 1 h (g, h, i). Phase contrast (a, d, g), GFP fluorescence (b, e, h), and Texas Red and Rhodamine fluorescence (c, f, i) micrographs are shown. These images were analyzed by conventional fluorescence microscopy under live conditions.
Discussion
iPS cells are expected to have applications for regenerative medicine. General differential methods, such as the addition of inducers of differentiation to the culture medium and gene therapy with virus vectors and so on, therefore contribute to the differentiation of objective cells. CPPs can be transduced in most cell types and this allows for the cellular uptake of conjugated (or fused) biomolecules (11). A wide variety of biomolecules such as antigenic peptides (28), peptide nucleic acids (24), antisense oligonucleotides, full-length proteins (13, 26), or even nanoparticles (9) and liposomes (34) have been delivered effectively using this method.
Two CPPs, Texas Red-R8 and Rhodamine-TAT, were generated as representative CPPs to determine whether these CPPs could penetrate into iPS cells in a manner consistent with many other cells. Both CPPs were internalized in iPS cells and the undifferentiated state of iPS cells was maintained. Moreover, no cytotoxicity was confirmed within 10 μM (data not shown). The low cytotoxicity of CPPs has been observed in many cells and the similar results were confirmed in the iPS cells. Moreover, the short uptake time is very important for clinical applications, including the field of regenerative medicine. Both CPPs were efficiently transduced into iPS cells within 2 h in this study.
Previous studies with R8 and TAT have demonstrated that even the mild cell fixation methods used to evaluate uptake by fluorescent microscopy can give rise to erroneous images in comparison to the experimental mode using unfixed cells because of the highly cationic nature of the peptides (25). In addition, the strong binding of iPS cells to the cell membrane also caused the overvalued uptake of the peptides by flow cytometry because of membrane-bound (not incorporated) peptides unless trypsin digestion of the cell membrane is used to eliminate the attached peptides (25). In this report, the iPS cells treated with CPPs were thus analyzed in live (not fixed) conditions and strongly washed three times by PBS to avoid the need for trypsin treatment.
Furthermore, the CPPs transduction mechanism into iPS cells was determined. CPPs transduction into other cell types is dependent on macropinocytosis (18, 38). The mechanism of CPPs internalization into iPS cells was investigated by incubation at 4°C or with inhibitors of macropinocytosis such as amiloride and cytochalasin D. The CPPs internalization was suppressed by these methods, thus suggesting that CPP transduction into iPS cells occurred through macropinocytosis.
In conclusion, CPPs can effectively and rapidly penetrate iPS cell membranes with no cytotoxicity, and the undifferentiated state of iPS cells was maintained. These results suggested that CPPs could be utilized for either the differentiation or functional application of iPS cells. However, it is generally known that more than 90% of the peptides transduced by the macropinocytosis pathway are degraded in lysosomes after transportation from macropinosomes. For effective clinical application, however, the matter of degradation and retention in lysosomes must be overcome.
Footnotes
Acknowledgments
We thank the Kobayashi Pharmaceutical Co., Ltd and Rina Yokota (Nagoya University) for their assistance.
