Abstract
Self-assembling peptide scaffold (SAPS) is well known to have very good bone conduction properties. However, the intensity of SAPS is too weak to actually use it for a clinical bone regeneration. Therefore, we have produced a hybrid scaffold system that involves fabricating a cage from polyetheretherketone (PEEK) that has high intensity, filling the interior of this cage with SAPS, and then transplanted this hybrid scaffold to bone defects in rat femurs. After 28 days, soft X-ray radiographs and histological assessment revealed that good new bone formation was clearly observed in the defects transplanted the PEEK cage with SAPS, but not in the PEEK cage only. The PEEK cage maintained a form and osteoconduction ability of internal SAPS, and SAPS promoted bone formation inside the PEEK; therefore, each was in charge of intensity and bone regeneration separately. The present study suggests that hybrid scaffolds made from PEEK cages and SAPS can be useful tools for the regeneration of load-bearing bones, based on the idea that it should be possible to develop ideal bone filler materials by combining the strength of artificial bone with the bone regeneration and bone conduction properties of SAPS.
Keywords
Introduction
Bone filler materials are needed to treat patients with many disorders, including bone defects and nonunion resulting from bone tumor excision or trauma, and in procedures such as spinal fusion (15). The most useful bone filler is the patient's own bone, but sometimes the patient's bone is too limited or cannot be reached without highly invasive surgery (18). Consequently, artificial bone materials such as hydroxyapatite and β-TCP are often used as fillers for bone defects (13, 14). These artificial materials can bond chemically to bone tissue in living organisms. Their constituent materials also have bone conduction properties and allow bone tissue to penetrate into their interior, although in practice the penetration of bone only occurs at the surface (3). There are measures that can be employed to achieve deeper penetration and promote remodeling, such as increasing the porosity of the material and ensuring that adjacent pores are interconnected. But because increased porosity causes reduced strength, it is difficult to combine strength and bone conduction with remodeling (19).
We have focused on mesenchymal stem cells and osteoblasts, which play a major role in bone regeneration, and on the cell components of vascular endothelial cells and the like, which promote the anchorage of these cells, and we have studied how to provide lesion sites with an environment where these cells can function more easily, particularly with regard to bone regeneration centered on an extracellular matrix (ECM). As a material that promotes the functioning of cells as an ECM, we have concentrated on self-assembling peptide scaffold (SAPS), and we have reported on the use of RAD-16-I (PuraMatrix™) to promote bone regeneration of calvarial bone defects in mice (16). In this study, we demonstrated that RAD-16-I has very good bone conduction properties. Because SAPS is a synthetic chemical product, it carries a lower risk of infection than other ECM materials. Furthermore, because it is normally a liquid and has the characteristic property of reacting with liquids containing cations to form a gel, it also makes it easy to fill defects with a complex shape, and can be combined with a wide variety of other materials. In animal experiments, it was found to have no adverse effects, and it is thought that SAPS is a useful material that is almost ready for clinical use (2, 7, 8, 10, 11, 16, 19).
The main issue with regard to the clinical use of SAPS is its strength. Although it has been reported that the strength of this material can be improved by combining it with granules of β-TCP or the like, but this strength is still insufficient for use in the regeneration of load-bearing bones (19). With regard to this issue, based on the idea that it should be possible to develop ideal bone filler materials by combining the strength of artificial bone with the bone regeneration and bone conduction properties of SAPS, we have produced a hybrid scaffold system that involves fabricating a cage from polyetheretherketone (PEEK), a material that is used in clinical applications such as spinal fusion procedures (5, 6), filling the interior of this cage with SAPS and forming a gel. We have investigated the applicability of this hybrid scaffold to bone defects in rat femurs.
Materials and Methods
Fabrication of Hybrid Scaffolds
The PEEK cages were made by Yasojima Proceed Co. Ltd, (Kobe, Japan), using TECAPEEK CLASSIX (Ensinger, Nufringen, Germany). These cages were tubular structures of outer diameter 5 mm, inner diameter 3 mm, and height 5 mm. Four elliptical holes were formed in the side wall of these cylinders (Fig. 1A). Compression strength tests of these PEEK cages were performed using a Strograph V10-β (Toyoseiki, Amagasaki, Japan) (n = 10) (Fig. 1A). Using a pipette, we filled the cages with 1% PuraMatrix™ (3-D Matrix, Ltd., Tokyo, Japan) (represented in Fig. 1B) in a 100cm dish. A liberal quantity of phosphate-buffered saline (PBS; pH 7.4) was then poured over the scaffolds, which were left to soak overnight. 3D-Matrix, Ltd. provided all the PuraMatrix™ we used for this experiment.

Schematic views of PEEK cage and macroscopic aspect of hybrid scaffold of SAPS with PEEK cage. (A) The axial and side schematic views of PEEK cage. (B) Photographs of hybrid scaffold of SAPS with PEEK cage. SAPS was gelated with PBS in the real experiment, but in the photograph culture, medium with Phenol Red was used to make the SAPS gel in the cage visible. (C) Transplant and external fixation of the hybrid scaffold to a rat left femur bone defect. The characteristics of PEEK cage: tubular structure, outer diameter 5 mm, inner diameter 3 mm, height 5 mm, four elliptical holes 3.0 × 1.5 mm, inner volume 51.791 mm3. Uniaxial compression test: maximum stress 901.5 ± 2.4 N, maximum displacement 0.677 ± 0.017 mm.
Segmental Defect Induction and Surgical Implantation of Scaffolds
Ten-week-old female Wistar rats (300–350 g) (Nippon CLEA, Tokyo, Japan) were used for all transplantation procedures, which were approved by the Okayama University Institutional Animal Care and Use Committee (OKU-2008075). The rats were anesthetized with sodium pentobarbital (40 mg/kg). Perioperative anesthesia was maintained by inhalation of isoflurane. Under sterile conditions, a posterolateral incision was made in the left femur, and the thigh muscles were divided. The midshaft of the femur was exposed and fixed using an external fixator and four pins of diameter 1.4 mm (Meira, Gifu, Japan), and a 5-mm-long section of diaphysis was then removed together with the periosteum using a microcutting saw (IMPLATEX, Tokyo, Japan). The bony defect was either left intact or filled with a prepared scaffold and compressed by external fixation (Fig. 1C). The wounds were closed with size 5–0 nylon sutures. The rats were divided into three groups: G1, transplanted PEEK cages with SAPS (n = 10); G2, transplanted PEEK cages with autologous bone as a positive control (n = 10); G3, transplanted empty PEEK cages as a negative control (n = 10). The rats were then kept for 28 days.
Gene Expression Analysis
Seven days after transplanting the scaffolds, some of the rats were killed (n = 3 in each group) and total RNA samples were extracted from the tissues inside PEEK cage by using an RNeasy® minikit (QIAGEN Science, ML) according to the manufacturer's instructions. All procedures were performed with i-cyclor (Bio-Rad, Tokyo, Japan), as previously reported (16). Briefly, 1.0 μg of total RNA was transcripted with River-Tra Ace (Toyobo, Tokyo, Japan), Oligo(dT) 20, RNase inhibitor, 5 × RT buffer, and dNTP mixture. RT-PCR was performed at 42°C for 40 min, and then at 95°C for 5 min, using 1.0 μg of RNA per reaction, to ensure that the amount of cDNA amplified was proportional to the mRNA present in the original samples as previously reported using specific primers. cDNA was amplified with TaKaRa Taq (TaKaRa, Ootsu, Japan) according to the manufacturer's instructions. The PCR products were separated on 2% agarose gel containing ethidium bromide. The following specific primers were used: Runx2—sense, 5′-ccacctctgacttctgcctct-3′ and antisense, 5′-gggatgaaatgcttgggaac-3′; osteocalcin—sense, 5′-agg gcagcgaggtagtgaag-3′ and antisense, 5′-ctgaaagccgatgtg gtcag-3′; VEGF-A—sense, 5′-ctggctttactgctgtacct-3′ and antisense, 5′-caattggacggcaatagctg-3′; GAPDH—sense, 5′-gcaccgtcaaggctgagaac-3′ and antisense, 5′-atggtggt gaagacgccagt-3′. After hybridization with labeled probes, the hybridized bands were resolved on 2% agarose gels and visualized by ethidium bromide staining. Rat GAPDH served as an internal control for the efficiency of mRNA isolation and cDNA synthesis. The data were confirmed by real-time PCR analysis. Real-time quantitative PCR reactions were performed on a Lightcycler (Roche Diagnostics, Mannheim, Germany) using a LightCycler-FastStart DNA Master SYBR Green I kit (Roche Molecular Biochemicals, Mannheim, Germany) according to the manufacturer's instructions. Gene expression levels are given as the percentage of the housekeeping gene GAPDH.
Radiographical Analysis
The remaining rats were killed at 28 days after transplantation and the femoral samples were subjected to soft X-ray examination using a Softex type CMB (Softex, Kagawa, Japan). The degree of bone regeneration was evaluated by measuring the length of the newly formed bones. Specifically, we measured the length of the bone defects and newly formed bones, and calculated the ratio of the newly formed bone length to the defect length. This ratio is referred to as the bridging ratio. Due to its radiolucency, it was easy to distinguish the newly formed bone from the PEEK in the central portion of the defect.
Histological Assessment
The femoral samples were fixed with 4% paraformaldehyde for 24 h, and then demineralized in ethylenediamine tetra-acetic acid for another 7 days. After demineralization, the PEEK cages were removed, and the femoral bones were set in paraffin and cut into 6-μm sections. To facilitate staining, the paraffin was removed by immersion in xylene, and the tissue sections were stained with hematoxylin-eosin and Masson trichrome (21).
Statistical Analysis
Mean values are presented with SDs. A Mann-Whitney U-test was used to calculate the significance of difference in median values. Bonferroni correction was conducted. A value of p < 0.05 was considered statistically significant.
Results
Compressive Strength of PEEK Cages
Uniaxial compression strength tests of these PEEK cages were performed using a Strograph V10-β (Toyoseiki, Amagasaki, Japan) (n = 10). The average maximum stress was 901.5 ± 2.4 N, so the average maximum stress was 71.8 ± 0.18 MPa. The average maximal displacement was 0.677 ± 0.017 mm (Fig. 1A).
The Expression of Runx2, VEGF-A, and Osteocalcin in SAPS
Total RNA was extracted from the tissue inside the PEEK cages 7 days after transplantation. The expression of Runx2, VEGF-A, osteocalcin, and a housekeeping gene (GAPDH) were analyzed with RT-PCR (Fig. 2). The quantification of the expression of Runx2, VEGF-A, and osteocalcin was performed with real-time RT-PCR analysis (Fig. 2B). The expression of these genes in the tissues of G1 were all higher in that of G3. Especially, the expression of VEGF-A in G1 was high; the ratio of VEGF-A to GAPDH was 6.87-fold for G1, 1.81-fold for G2, and 0.95-fold for G3. Also, the ratio of Runx2 to GAPDH was 0.93-fold for G1, 0.84-fold for G2, and 0.31-fold for G3. The ratio of Runx2 to GAPDH was 0.80-fold for G1, 3.80-fold for G2, and 0.16-fold for G3.

The expression of Runx2, VEGF-A, and osteocalcin in PEEK cages at 7 days after implantation. (A) The expression of these makers in G1 was higher than in G3. (B) The result of the quantitative PCR analysis. In particular, the expression of VEGF-A was high in G1. A housekeeping gene, GAPDH, was as internal control.
Soft X-Ray Radiography
Soft X-ray radiograph examination showed excellent bone formation inside the PEEK cages in group G1 at 28 days after transplantation; however, callus did not bridge the gap. In group G3, bone formation was observed only around the periphery of the osteotomy line. In the soft X-rays, the average bridging ratios of femoral bone defects was 78.9 ± 11.8% for PEEK cage with SAPS transplant (G1), 96.5 ± 4.7% for PEEK cage with autologous bone transplant (G2), and 29.5 ± 9.7% for empty PEEK cage transplant (G3) (p < 0.01 for G1 vs. G2 or G3 and G2 vs. G3) (Fig. 3).

Representative soft X-ray radiographs of specimens at 28 days after implantation. G1, PEEK cage with SAPS. New bone formation is seen inside the PEEK cage. G2, PEEK cage with autologous bone. The transplanted bone was left behind and some degree of contact was obtained between the transplanted bone and the new bone. G3, PEEK cage only. Little to no bone formation is noted. The average bridging ratios of femoral bone defects was 78.9% for G1, 96.5% for G2, and 29.5% for G3. Scale bar: 5 mm. ∗p < 0.01.
Histological Findings
In group G1, good new bone formation was observed inside the PEEK cage at 28 days after transplantation (Fig. 4A–C). In contrast, in group G3, bone regeneration was not almost detected but occupied by fibrous tissues. (Fig. 4D–F). The Masson trichrome staining clearly showed that the periphery and ends of new bone growth in the samples from group G1 were made of cartilage, demonstrating good enchondral bone formation had taken place. In group G3, we observed cartilage formation and a small amount of bone formation around the edges (Fig. 4C, F). And in group G2, the transplanted bone was nearly all left behind and some degree of contact was obtained between the transplanted bone sections (data not shown). There was no inflammatory reaction detected and SAPS did not remain in the center of the PEEK cages.
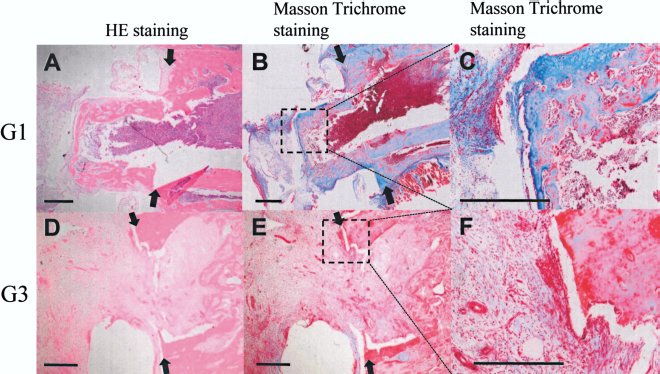
Representative histological images of segmental defects in G1 (A, B, C) and G3 (D, E, F) at 28 days after implantation. Sections are stained with hematoxylin-eosin (A,D) and Masson trichrome (B, C, E, F). Good new bone formation and enchondral bone repair were seen in G1. In contrast, there is little newly bone formation in G3. Arrow marks the cut end of rat femur. Scale bar: 1 mm.
Discussion
The factors necessary for bone regeneration include cells such as bone marrow mesenchymal stem cells and osteoblasts, an extracellular matrix to support the adhesion and movement of these cells, and various intercellular functions such as storage of materials, a supply of nutrients (e.g., by angiogenesis) necessary for tissue formation, cytokines to promote and control cell growth, and dynamic factors such as strength and stability (1, 17). The SAPS we used in these experiments was developed as an artificial extracellular matrix (ECM) (19, 22–24). It not only works as a support for cells and growth factors, but has also been reported to be highly conductive for the vascular system (9).
Consequently, the biggest drawbacks of SAPS in bone regeneration are its strength and stability. In the hybrid scaffold we produced from a PEEK cage filled with SAPS, the PEEK cage makes up for the shortfall in the strength of SAPS, and is thus able to satisfy all the above-mentioned requirements for bone formation. Furthermore, there have hitherto been no proposals of the concept where separate materials are used for parts responsible for regeneration and parts responsible for conferring strength, such as by using a PEEK cage for strength and SAPS for bone regeneration and bone conduction. This method may make it possible to develop the ideal bone filler.
The compressive strength of the PEEK cage allows it to withstand a maximum load of 901.5 ± 2.4 N with a maximum displacement of 0.677 ± 0.017 mm (Fig. 1A). This value is considerably inferior to the intensity of the cortical bone of the human, but it is a much higher value than intensity of the cancellous bone (20). None of the PEEK cages used in the transplantation examinations underwent any deformation or breakage. Also, the SAPS inside these cages was still left behind 2 weeks after being transplanted into the rat femur bone defects (data not shown), and it seems that the PEEK cages were strong enough for use in rat femur diaphysis bone defects. However, it cannot be said that they are strong enough for use in the regeneration of load-bearing bones in humans, and further study is needed with regard to the formation and materials of PEEK cages in the future.
The 5-mm rat femur diaphysis bone defects used in this study are considered to be critical size (4, 21). We already showed a utility of SAPS in the regeneration of the stable bone defect (16). So in this study, we selected an unstable bone defect model that was easy to become the problem in a clinic. The PEEK cage was transplanted so as to make contact with the cortical bone tissue of the excised bone and so that the space inside the cage made contact with the bone marrow, and was then fixed in place with a compressive force applied by an external fixator.
In group G3 where only the PEEK cage was used, we only observed slight bone formation at the cut ends of the bone because the PEEK cage on its own has no bone conduction abilities. On the other hand, in group G1 where the cage was filled with SAPS, we obtained good bone formation inside the PEEK cage (Figs. 3 and 4). With SAPS in the gel state, pores of 50–400 nm were formed by fibers with a diameter of 7–10 nm to form an artificial ECM with good bone conduction performance (9, 10, 19, 23, 24). This structure is fragile and can cause the bone defect to persist. However, by filling the interior of a sufficiently strong PEEK cage with SAPS and forming a gel, it seems that it was possible to maintain the structure even in the bone defect parts, and to achieve good bone conduction performance. It also seems that cells such as vascular endothelial cells, bone marrow mesenchymal stem cells, and osteoblasts penetrate into SAPS from an early stage, allowing angiogenesis and osteogenesis to proceed (9, 12). To evaluate this theory, we investigated the expression of VEGF-A, Runx2, and osteocalcin at 7 days after transplantation. All three of these genes were found to be highly expressed in groups G1 and G3. It is thought that early neovascularization and osteogenesis are promoted as for the expression of VEGF-A and Runx2 in group G1 in particular more highly than groups G2 and G3 (Fig. 2).
However, in group G1 complete bone union was not achieved even though good bone formation was observed inside the PEEK cage. Possible causes for this include the fact that the SAPS was absorbed for approximately 3 weeks before the tissue had fully penetrated into the central part, instability caused by loosening of the external fixator pins, and the possibility that the bone fusion was still at an intermediate stage on the 28th day after transplantation. In the future, we plan to investigate bone regeneration where bone marrow mesenchymal stem cells and growth factors such as BMPs are included into SAPS, and to study more ideal shapes and larger size for artificial scaffolds, and as a result we can expect further promotion of bone regeneration.
SAPS is a material that is highly likely to be used in clinical applications because it has several benefits. These include its favorable bone conduction properties, its ability to act as a support for various different cells and growth factors, its lack of infection risk compared with materials of animal origin such as ECM, and the ease with which it can be used to fill defects with complex shapes and combined with a wide range of other materials. In this study we used a PEEK cage as a scaffold for SAPS to produce a hybrid scaffold that com-bines high strength with good bone conduction performance, and we were able to confirm these important benefits. Hybrid scaffolds made from PEEK cages and SAPS can be useful tools for the regeneration of load-bearing bones.
