Abstract
Many studies have reported the contribution of bone marrow-derived cells (BMDC) to the CNS, raising the possibility of using them as a new source to repair damaged brain tissue or restore neuronal function. This process has mainly been investigated in the cerebellum, in which a degenerative microenvironment has been suggested to be responsible for its modulation. The present study further analyzes the contribution of BMDC to different neural types in other adult brain areas, under both physiological and neurodegenerative conditions, together with the mechanisms of plasticity involved. We grafted genetically marked green fluorescent protein/Cre bone marrow in irradiated recipients: a) the PCD (Purkinje Cell Degeneration) mutant mice, suffering a degeneration of specific neuronal populations at different ages, and b) their corresponding healthy controls. These mice carried the conditional lacZ reporter gene to allow the identification of cell fusion events. Our results demonstrate that BMDC mainly generate microglial cells, although to a lesser extent a clear formation of neuronal types also exists. This neuronal recruitment was not increased by the neurodegenerative processes occurring in PCD mice, where BMDC did not contribute to rescuing the degenerated neuronal populations either. However, an increase in the number of bone marrow-derived microglia was found along the life span in both experimental groups. Six weeks after transplantation more bone marrow-derived microglial cells were observed in the olfactory bulb of the PCD mice compared to the control animals, where the degeneration of mitral cells was in process. In contrast, this difference was not observed in the cerebellum, where Purkinje cell degeneration had been completed. These findings demonstrated that the degree of neurodegenerative environment can foster the recruitment of neural elements derived from bone marrow, but also provide the first evidence that BMDC can contribute simultaneously to different encephalic areas through different mechanisms of plasticity: cell fusion for Purkinje cells and differentiation for olfactory bulb interneurons.
Introduction
In recent years, it has been shown that bone marrow-derived cells (BMDC) owe their plasticity to changes in their fate, which allows their contribution to different cell populations in diverse organs (7,19,22,26,40,41,48), including the central nervous system (CNS) (2,8,9,34,54). This surprising capacity has been considered a potential tool for the development of innovative therapeutic strategies to treat degenerative, posttraumatic, and hereditary diseases (3,9). Thus, the contribution of BMDC to diverse tissues has been described under normal and pathological conditions. The contribution rate was always very scarce (2,42), but this low “basal” frequency, described under physiological conditions, was significantly increased when the target organs underwent injury (2,27,34,56), especially when accompanied by inflammation (23,38).
In the CNS, the arrival of BMDC at the brain has mainly been studied in the cerebellum, where they contribute to the Purkinje cell population in animal models (2,43,50,54) and humans (35,53). Similar to other organs, this contribution of BMDC seems to be increased by a neurodegenerative or injured microenvironment (3,30). The underlying mechanism responsible for this process has long been a very controversial issue, and two main possible plasticity mechanisms have been proposed: differentiation (24,40,42) and cell fusion (2,39, 47,51,54). They are not mutually exclusive. However, despite this, the lack of studies in which both processes are analyzed together and discerned under exactly the same conditions has reinforced the controversy. Recent evidence supports the cell-fusion mechanism for the generation of Purkinje cells (23,30,38). However, very little is known about the possible plasticity mechanisms for other neuronal types. Thus, encephalic regions other than the cerebellum require a more extensive review. This is the case of the olfactory bulb (OB), where previous works studying the contribution of BMDC are scarce and have provided confusing or inconsistent data (8,11,32,43). This region is characterized by a continuous recruitment of new neurons along adulthood, which also may influence the behavior, differentiation, and arrival of BMDC. Moreover, no previous studies have been performed in animals suffering from neurodegeneration in the OB, and hence an exhaustive study of the behavior of BMDC after their arrival at this highly plastic encephalic region, in comparison with others, under both healthy and diseased conditions should help us to better understand the contribution of BMDC to the brain.
Accordingly, in this work we further analyzed the effect of different neurodegenerative stages on the contribution of BMDC to several brain regions, together with the plasticity mechanism(s) involved. As in previous work in the field, we performed intravenous transplantation of genetically marked bone marrow in lethally irradiated mice. The recipients were both wild-type and mutant mice from the Purkinje Cell Degeneration (PCD) mouse line. Homozygous recessive mice (pcd/pcd) undergo a selective postnatal neurodegeneration in several encephalic areas at different ages (16,36,44). They lose the Purkinje cells around postnatal day 20, which causes severe ataxia (36). In addition, they have a selective loss of OB mitral cells, starting around 60 days after birth (18). Prior to this age, the cellular and synaptic organization in the OB seems to be normal (4,17,18). Thus, analysis of the cerebellum and the OB of these mutant animals grafted with wild-type bone marrow allowed us to study the possible effect of different neurodegenerative environments on the arrival and contribution of the BMDC to these areas.
Additionally, in order to discern the plasticity mechanism involved in this process, the PCD mouse line was crossed with the conditional Cre reporter mouse line, R26R (2,31). In this line, the lacZ reporter gene is expressed exclusively after CRE recombinase excision of a loxP-flanked stop cassette. As bone marrow donors we used mice constitutively expressing CRE recombinase and green fluorescent protein (GFP) under the control of the β-actin promoter. This transplant strategy has been used previously with success to discern between cell fusion and differentiation events (2,31).
Our data indicated that the contribution of BMDC to the formation of new neuronal elements in the encephalon of adult mice was not restricted to the Purkinje cells of the cerebellum, but also took place in OB interneurons. Unexpectedly, the neurodegenerative environment had no effect on the behavior of BMDC. In contrast, and very interestingly, the contribution of BMDC occurred through two different plasticity mechanisms, depending on the region and cellular type: cell fusion for the Purkinje cells of the cerebellum, and differentiation for the OB interneurons. This strongly suggests that plasticity mechanisms may be modulated by region- and cell type-specific factors.
Materials and Methods
Animals
Recipients were obtained from the PCD-R26R mouse colony. This colony was established at the Animal Research Facility of the University of Salamanca in two steps. First, in order to be able to genotype the pcd1J mutation, heterozygous C57BL/6J mice (+/pcd) from a colony originally obtained from Jackson Laboratories were mated with wild-type DBA/2J females, establishing the PCD colony. Thus, the pcd1J allele was associated with the genetic background of the C57BL/6J strain, and the wild-type one with the genetic background of the DBA/2J strain. Mice were genotyped by PCR amplification of specific microsatellite sequences that show intraspecific divergences. Second, in order to obtain wild-type and pcd/pcd animals carrying the CRE reporter lacZ gene (2,31), +/pcd animals (from the PCD colony) were crossed with R26R mice (JAX 003474).
Donors were obtained from the PCD-(CRE-GFP) mouse colony. Homozygous mice expressing CRE recombinase under the control of a hybrid regulatory element of the cytomegalovirus enhancer β-actin promoter (28), and homozygous mice expressing GFP under the same promoter (20) were bred to generate β-actin CRE-GFP mice. Furthermore, to prevent allograft rejection, the mice were crossed with +/pcd animals (from the PCD colony), increasing the consanguinity between donors and recipients (Fig. 1). The experiments were carried out in accordance with the guidelines of the European Community Council (86/609/EEC) and current Spanish legislation for the use and care of laboratory animals (RD 1201/2005). The protocols for animal care and manipulation used were approved by the Bioethics Committee of the University of Salamanca.

Chart depicting the work schema to demonstrate both the destination and cell mechanism for BMDC to be integrated into nervous tissue. Donor mice contain GFP, the Cre transgene, or both, while the recipient mice carry the conditional floxed reporter gene lacZ in the R26R loci. Recipient mice were wild-type or exhibit a natural neurodegenerative process (PCD). After irradiation, all the recipient mice were transplanted with donor bone marrow, and the brains of the resultant chimeric mice were analyzed and compared.
Bone Marrow Transplantation
Wild-type bone marrow cells were collected from 4–8-week-old donor mice by flushing tibias and femurs with Iscove's modified Dulbecco's medium (IMDM; Invitrogen). Red blood cells were subsequently lysed by 5 min of incubation in ice-cold ammonium chloride (140 mM in Tris 17 mM). After a double wash and mild centrifugation, 7.5 × 106 bone marrow cells were injected intravenously into the tail vein in both the wild-type and pcd/pcd R26R mice. Animals undergoing bone marrow transplantation (BMT) were lethally irradiated to remove their own bone marrow 24 h before grafting. Owing to their particular characteristics (fairly young—P19—and some of them PCD mutant animals), we previously tested different doses of irradiation (unpublished data) in order to select the minimum lethal dose for these P19 animals (7.5 Gy). Moreover, to ensure the survival of the transplanted PCD mutant mice, we performed whole BMT, without sorting its populations.
Flow Cytometry
Donor engraftment was determined by flow cytometry of peripheral blood (FACS Calibur, BD Biosciences). Peripheral blood was subjected to red blood cell lysis by treatment with ammonium chloride (see above), washed, and resuspended in phosphate buffered saline (PBS). Cytometric analysis was performed twice: 2 weeks after grafting and on the day of sacrifice (see below). Recipients showing a donor chimerism <50% in the first sample were rejected. As indicated in Table 1, all transplanted mice analyzed in this study showed significant levels of multilineage chimerism of peripheral blood leukocytes.
Summary of the Different Experimental Animals Employed and the Experimental Procedures Followed in the Present Study

Genotype: +/+, wild-type; pcd/pcd, PCD. Treatment: BMT, bone marrow transplantation; NT, not treated. Degenerative stage at sacrifice age of both Purkinje cells (cerebellum) and mitral cells (OB): N, normal; D, degeneration; PD, postdegeneration.
Tissue Collection
At P60, P110, and P150, groups of experimental animals were deeply anaesthetized intraperitoneally with a solution composed of ketamine [120 μg/g body weight (b.wt.)] and xylazine (10 μg/g b.wt.) and were perfused through the ascending aorta, first with 0.9% saline for 1 min and then with 50 ml of fixative containing 4% paraformaldehyde (for general immunofluorescence) or 2% paraformaldehyde + 0.25% glutaraldehyde (for X-gal histochemistry) in 0.1 M phosphate buffer (PB). Encephala were dissected out, cut into blocks (rostral, including the OB; medial and caudal, including the cerebellum) and immersed in the same fixative solution for 20 min at room temperature (RT). Then, the blocks were rinsed in PB and cryoprotected with 30% sucrose in PB overnight at 4°C. Rostral and caudal blocks were cut in 40-μm coronal or sagittal sections, respectively; serial sections were obtained with a freezing microtome and were collected in alternating series. Representative sections were stained with X-gal staining or immunohistochemistry. Medial blocks were frozen and stored at −80°C for future experiments.
X-gal Staining and Immunohistochemistry
To identify cell-fusion events, sections were processed with X-gal staining by placing them in PB containing 10 mM K3Fe(CN)6 and 10 mM K4Fe(CN)6 together with the β-gal substrate, 5-bromo-4-chloro-3-indolyl-β-D-galactopyranoside (X-gal; 1 mg/ml; Molecular Probes), at 37°C overnight. Then, to characterize the X-gal-positive cells, selected sections were processed immunohistochemically with anti-Iba1 antibody [goat (1:500), from Everest Biotech, or rabbit (1:750), from Dako], or anti-GFAP [rabbit (1:2,000), from Sigma], or anti-calbindin [CB; mouse (1:2,000) or rabbit (1:7,000), from Swant], or anti-Reelin [mouse (1:1,000), from Chemicon] in a medium containing 0.2% Triton X-100 (Probus) and 5% normal donkey serum (Vector) in PBS overnight at RT. Biotinylated donkey anti-goat, rabbit, or mouse IgG secondary antibodies (1:200) were from Jackson ImmunoResearch, and avidin-biotin-peroxidase complex (Kit Elite ABC; 1:200) was from Vector. The reaction product was visualized by incubating sections in 0.02% 3,3′-diaminobenzidine (DAB) and 0.003% H2O2 in 0.1 M Tris-HCl buffer.
Immunofluorescence
Double or triple immunofluorescence was used to identify GFP+ cells, along with other cellular antigens for both neurons (CB and Reelin) and glial cells (GFAP and Iba1). Sections were incubated with 0.2% Triton X-100 and 5% normal serum and were costained with anti-GFP antibody [goat (1:2,000) or rabbit (1:4,000), from AbCam] and the above described primary antibodies. Secondary antibodies (Cy-2, Cy-3, Cy-5 conjugated) were used at a dilution 1:500 (from Jackson Immunore-search). Nuclei were counterstained with DAPI (Gibco-BRL) or propidium iodide (PI; Sigma).
Microscopy Analysis
Processed sections were analyzed for the coexpression of the indicated markers under an Olympus Provis AX70 microscope (UPlanSApo lens up to 60× N.A. 1.35) equipped with an Olympus DP70 digital camera. Selected sections were also examined with a confocal microscope (Leica TCS SP spectral confocal microscope; pinhole opening up to 1.5 Airy; Ar/ArKr and He/Ne laser excitation and Planapochromatic objectives up to 63× N.A. 1.4). Digital images were processed with Adobe Photoshop 7.0 (Adobe Systems) to adjust contrast slightly and assemble the final plates.
Cell Counting and Statistical Comparison
The same animals were used for analyzing both the olfactory bulb and cerebellum. All GFP+ Purkinje cells and OB interneurons were counted immediately after the observation of all sections, since their numbers were low. However, to compare the quantities of GFP+ microglial cells anatomically comparable sections were selected. Both the OB and cerebella of PCD mice undergo a dramatic shrinkage (4,14,25,33), which would make the comparison of cell density values unfounded for analyses. Thus, we chose the number of cells per section as the morphometric value for comparisons between experimental groups. For the cerebellum, two purely sagittal sections were chosen (where the 10 cerebellar lobules where clearly seen). Regarding the OB, a central coronal section was chosen for comparisons, as previously described (14,55). Four animals from each experimental group were analyzed. Therefore, these values represent an estimation of the arrival of BMDC to specific regions, without the influence of their shrinkage due to the pcd1J mutation. GFP+ cells were counted with the Neurolucida and Neuroexplorer programs (MicroBrigh-Field, Magdeburg, Germany).
All counts were performed by the same person (D.D.), following the same criterion and using a doubleblind study. Once homoscedasticity had been checked with the Kolmogorov-Smirnov test, a one-way ANOVA test was employed to analyze possible differences among different graft survival times within each genetic group; Bonferroni post hoc test was performed to make the simple comparisons. Following this, Student's t-test was performed in order to assess possible differences between PCD and wild-type mice within each survival time.
Results
In order to analyze the effect of different neurodegenerative stages on the contribution of BMDC to the cerebellum and olfactory bulb, we performed BMT in 20-day-old normal and PCD mice (see Materials and Methods and Fig. 1). At this age, Purkinje cell degeneration in PCD animals has just started (36), while OB mitral cells are still alive (4,17). After transplantation, the animals were sacrificed at three different ages: P60, P110, and P150. At P60 (40 days posttransplantation), Purkinje cells were mostly lost in PCD mutants, whereas mitral cell degeneration had not yet begun. At P110 (90 days posttransplantation), Purkinje cell death had already finished and the neurodegenerative process in the OB was taking place. Finally, at the last age (130 days posttransplantation), both degenerative processes had been already completed.
Each of the transplanted animals selected for this study displayed substantial engraftment of GFP+ donor cells in the peripheral blood, with chimerisms ranging from 58% to 85% of total cells (Table 1), including contributions to both the myeloid and lymphoid lineages (unpublished data). No significant difference (Student's t-test, p ≥ 0.05) was found in the percentage of GFP+ cells incorporated into the peripheral blood of PCD and wild-type transplanted mice. This demonstrated that the capacity of the PCD mice to host a wild-type bone marrow was not affected by the PCD mutation and allowed a comparison of the results obtained from both experimental groups.
Generation of Glial Elements From Transplanted Bone Marrow Under Physiological and Neurodegenerative Conditions
Gross observations of GFP+ donor-derived cells in both the cerebellum and the OB of transplanted mice revealed that the vast majority of these cells had the typical morphology of resting microglial cells. To confirm this observation, we performed double immunofluorescence colocalization for GFP and Iba1 (a typical microglial marker) (Fig. 2). The overall contributions of double-positive GFP/Iba1 microglial cells to the whole resident microglial populations (Iba1 single-labeled cells) were quite weak, about less than a tenth part (as previously described) (32), even for neurodegenerative conditions in both encephalic regions. However, the analysis revealed a time-dependent increase in double-positive GFP/Iba1 microglial cells in both the wild-type and PCD animals along the ages studied, this increase being significantly different at P150 (Table 2, Fig. 3). Most of these cells were located peripherally, close to the meninges, but also in perivascular locations of the cerebellum and the OB (Fig. 4). Furthermore, in the transplanted PCD mice, the neurodegenerative environment of the OB at P60, and to a lesser extent at P110, led to significant differences in the contribution of BMDC to the generation of microglial cells in comparison with age-mated transplanted wild-type animals (p < 0.01 at P60) (Fig. 3), coinciding with the time window of mitral cells degeneration (18,49).

Epifluorescence microscopy images from encephalic sections of a P150 wild-type mouse transplanted with GFP bone marrow. These images show the contribution of the bone marrow to the microglial cell population, both resting microglial cells (A–C) and activated ones (D–F). Resting and activated cells were identified by their morphological characteristics. Upper images (A, D) show GFP labeling, which allows the identification of the newly generated cells coming from the transplanted bone marrow. Images in the central portion (B, E) exhibit the Iba1 signal (microglial cell specific marker). Lower photographs (C, F) correspond to the merging of both previous images for each kind of microglial cell. Scale bar: 20 μm.

Graph showing the number of GFP+ microglial cells in the recipient animals after bone marrow transplantation. The number of cells was quantified in comparable sections in both wild-type and mutant animals in order to avoid the incorrect evaluation of cell densities because the natural shrinkage of the tissue in mutant mice. Within each studied encephalic regions, statistical differences were founded among survival times for both wild-type animals (##p < 0.01, ANOVA-Bonferroni post hoc test) and PCD mutant mice (§p < 0.05, ANOVA-Bonferroni post hoc test). Moreover, in the olfactory bulb, statistical differences were demonstrated between both experimental groups at P60 (**p < 0.01 Student's t-test).
Number of GFP+ Microglial Cells After Bone Marrow Transplantation
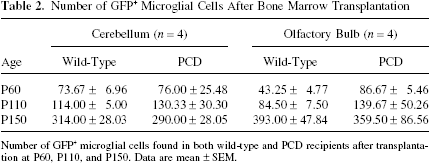
Number of GFP+ microglial cells found in both wild-type and PCD recipients after transplantation at P60, P110, and P150. Data are mean ± SEM.

Epifluorescence microscopy images of cerebellum sections from a P150 wild-type mouse (A, B) and a P150 PCD mouse, both grafted with GFP bone marrow, showing the GFP+ cells. (A) Panoramic image showing the distribution and abundance of the cells newly generated from the transplanted bone marrow in the cerebellum. A GFP+ Purkinje cell can be observed. (B) Magnification of the previous image and allows the assessment in greater detail of both the GFP+ Purkinje cell and the surrounding microglial cells. This GFP+ Purkinje cell has a very complex dendritic tree and a long axon (indicated with arrows), presenting direct evidence of its integration in the cerebellum. Moreover, the axon presents the typical recurrent collaterals (arrowheads) that characterize Purkinje cells. Furthermore, microglial cells were basically located in a peripheral position. Some of these microglial cells are indicated with asterisks. (C) The same region of a transplanted PCD mouse, showing the location of the newly generated cells in the cerebellum of these mutant animals. Scale bars: 200 μm (A) and 100 μm (B, C).
To investigate whether GFP+ microglial cells were generated by cell fusion, we performed X-gal staining to detect the lacZ expression resulting from the recombination of floxed sequences after a fusion event. We never observed GFP+ microglial cells colocalized with X-Gal staining in any brain region of either experimental group. Accordingly, the contribution of BMDC to microglial cells must occur through a differentiation process.
Finally, the possible formation of astrocytes from the BMDC was also analyzed. No GFP+ cells coexpressing GFAP (considered as a specific marker for astrocyte lineage) were observed in any region studied (including the subventricular zone, SVZ) under any of the experimental conditions tested (data not shown).
Contribution of BMDC to Neuronal Elements Under Normal Conditions
To analyze the contribution of donor BMDC to neuronal elements under physiological conditions, we immunocolocalized GFP+ donor-derived cells with glial markers (Iba1 or GFAP) and neuronal region-specific markers, either CB in the cerebellum (Purkinje cells marker) or Reelin in the OB (mitral cell marker). As criteria for characterizing neurons, we required (a) the absence of immunostaining for glial markers and (b) positive immunostaining with CB or Reelin, or a display of distinctive typical neuronal morphology.
With these criteria, in the cerebellum of transplanted wild-type animals the only neuronal elements found were Purkinje cells (Fig. 4). As previously reported, their frequency was very scarce (Table 3), their maximum number observed per cerebellum being 4 at P150. Owing to the low number of these cells, the statistical comparison between groups would be untrustworthy. These cells clearly exhibited the characteristic morphology of Purkinje cells and expressed CB (Fig. 5). Furthermore, it was possible to confirm their integration in the cerebellar system through the observation of long axons, even showing recurrent collaterals (Figs. 4 and 5). Consistent with previous work (2,23,30,38,54), the plasticity mechanism involved in their generation was clearly cell fusion, as confirmed by the expression of the lacZ reporter gen (X-Gal staining) (Fig. 5) and the observation of GFP+ binucleated Purkinje cells (Fig. 5).

Fluorescence microscopy images of cerebellum sections from wild-type mice grafted with wild-type bone marrow from CRE-GFP donors showing some examples of Purkinje cells originated from the transplanted bone marrow. (A, D) Two perfectly differentiated different GFP+ Purkinje cells. The arrowhead in (A) indicates the axon. (B, E) The calbindin (CB) labeling for the previous images, and (C, F) the corresponding merging. Purkinje cells produced from the grafted bone marrow not only show the typical morphology of this specific cell type, but also express CB, like the other Purkinje cells. (G–I) The X-gal staining of a cerebellar section from a wild-type R26R mouse grafted with CRE-GFP bone marrow. (G) A Purkinje cell that has undergone a fusion cell event with a BMC. (H) Magnification of the fused cell in (G), and (I) the double staining of that Purkinje cell with X-gal and CB (with the DAB immunohistochemical technique). Images at the bottom (J–L) represent high magnifications of the soma from a GFP+ Purkinje cell (J), the staining of this region with the nuclear marker propidium iodide (PI; K), and the merging of both stainings (L). Arrowheads in (K) indicate the two nuclei belonging to the previous GFP+ Purkinje cell, suggesting that it is a binucleate and presumably polyploid cell. Scale bar in (C): 100 μm for (A–F), 200 μm for (G), and 50 μm for (H, I). Scale bar in (L): 20 μm for (J–L).
Number of GFP+ Neurons After Bone Marrow Transplantation
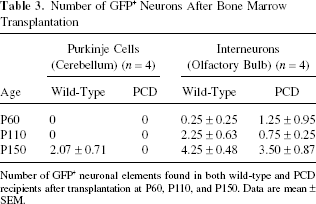
Number of GFP+ neuronal elements found in both wild-type and PCD recipients after transplantation at P60, P110, and P150. Data are mean ± SEM.
Interestingly, in the OB of the transplanted wild-type animals we found mature olfactory interneurons expressing GFP (Fig. 6). These newly generated neurons, derived from donor cells, were very scarce (Table 3). They were located in different layers of the OB, with no preferential distribution. Owing to the strong neuro-chemical diversity of OB interneurons (5,13), we determined the OB neuronal subtype by means of their characteristic localization and very distinctive morphology. We unmistakably characterized both juxtaglomerular cells and granule cells (Fig. 6C). All of them displayed the typical morphology of neurons belonging to the layer where they were found. No double-positive Reelin/GFP cells were found in the OB, indicating that BMDC do not contribute to OB mitral cells (Fig. 6G–J).

Fluorescence microscopy images of olfactory bulb (OB) sections from wild-type (A–C, G–J) and PCD (D–F) mice grafted with wild-type bone marrow from a CRE-GFP donor showing some examples of the typical neuronal types of this region originated from the transplanted bone marrow. (A, D) The newly generated cells (GFP+) in the different OB layers in both wild-type (A) and PCD (D) recipients. (B, E) The same sections counterstained with DAPI to display the location of GFP+ cells and the laminar organization of this region in both experimental groups. (C) A GFP+ cell from a grafted wild-type mouse that can be classified as a granule cell, considering its typical morphology and location in the OB. The white arrow points to its soma and the white arrowheads to its typical prolongation. (F) A GFP+ cell from a grafted PCD mouse that can be sorted, by its location and morphology in the OB, as a juxtaglomerular cell. The white arrow points to its soma and the white arrowheads to its typical prolongation towards an olfactory glomerulus. (G–J) Laser scanning confocal images from the OB of a mouse grafted with GFP bone marrow. (G) The GFP labeling to identify donor cells. (H) The mitral cell layer (Reelin positive). (I) The Iba1 labeling to show the microglial cells. (J) The merging of the previous confocal images. Taking into account its characteristics, this newly generated cell can be classified as a granule cell. MCL: mitral cell layer, GL: glomerular layer, GCL: granule cell layer, EPL: external plexiform layer. Scale bars: 100 μm (A, B, D, E), 50 μm (C, F), 25 μm (G–J).
Finally, contrary to what was seen for the Purkinje cells, none of these newly generated OB cells had two nuclei, strongly suggesting a differentiation mechanism as their possible underlying origin. To check this assumption, we looked for the expression of the lacZ reporter gene in the OB of transplanted mice. We did not find any cell fusion event in this region. Moreover, we carefully looked into the SVZ or along the rostral migratory stream, since these structures support the arrival of new interneurons to the adult OB (14): no positive GFP+ elements were found neither in wild-type animals nor in the PCD mice. Therefore, all the evidence points to differentiation as the plasticity mechanism involved in the generation of new OB interneurons from BMDC.
Contribution of BMDC to Neuronal Elements Under Neurodegenerative Conditions
Analysis of the cerebellum and OB regions in transplanted PCD mice allowed the discrimination of: a) the effect of a neurodegenerative environment on the contribution of BMDC to these regions; and b) the effect of the transplantation of healthy BM in the damaged brain. The markers and criteria used to examine these processes were identical to those employed for physiological conditions. No Purkinje cells with markers from donor BMDC were observed in the cerebellum of the transplanted PCD mice, even in the cerebellar vermal lobule X, where the neurodegenerative process starts later [(25); our own observation]. This result was similar for the three different neurodegenerative stages studied and indicates that the PCD neurodegenerative process did not modify the arrival or plasticity of the BMDC. These data contrast with findings published previously (3,23,30), in which the ability of BMDC to fuse with this neuronal population was reported to be increased after the Purkinje cells had been damaged, suggesting that differences in the nature of the injury may affect the fusion process. Moreover, histological comparison of the cerebellum from transplanted PCD animals and PCD mice not receiving treatment did not reveal any difference or improvement in the degenerative process (unpublished data). This was confirmed by the absence of Purkinje cells (CB positive) in all the mutant mice analyzed, with or without BMDC transplantation.
Additionally, analysis of the OB of transplanted PCD mice (Fig. 6) suggested that the sparse formation of neurons from BMDC was not influenced by mitral cell degeneration either, since the results were similar to those obtained in wild-type transplanted mice (Table 3): in normal mice mature olfactory interneurons expressing GFP were also observed in the OB of some transplanted PCD mice at similar ages (Fig. 6C, F). However, none of these newly generated cells expressed Reelin or displayed the typical mitral cell morphology. Therefore, despite the mitral cell degeneration in the OB of the PCD mice, BMDC did not contribute to this neuronal population under either normal or degenerative conditions.
Similar to the results observed in wild-type grafted animals, none of the newly generated GFP+ OB cells had two nuclei nor expressed the lacZ reporter gene, supporting their origin through a differentiation mechanism. Finally, histological comparison of the OB of transplanted PCD mice and PCD mice not receiving treatment revealed no difference in the remaining mitral cell population at the different ages analyzed. This suggested that the transplantation of wild-type BM had no effect on the degenerative process of the OB, as in the cerebellum.
Discussion
The Formation of Microglial Cells From the Bone Marrow Is Influenced by Selective Postnatal Neurodegeneration
Analysis of the contribution of BMDC to the cerebellum and the OB of both wild-type and PCD transplanted animals clearly demonstrated a stable generation of new neural elements from donor bone marrow under normal physiological conditions and along the different stages of the selective postnatal neurodegenerative processes. The most common cell type generated from the BMDC was microglia, in accordance with previous studies (1,6,12,15,21,32,43,45,50). All the experimental groups underwent the same procedure (irradiation), were grafted with the same wild-type bone marrow, and had similar percentages of GFP+ cells in their peripheral blood. Along time, the number of engrafted GFP+ microglial cells increased in both the wild-type and PCD recipients, suggesting that donor-derived bone marrow generates permanent new microglial cells for the encephalon throughout the life span of the animals, according to previous findings (6,15,21,32).
The PCD animals showed difference in the degree of neurodegeneration in each of the encephalic regions studied (16,18,27). Interestingly, the percentage of contribution of BMDC to the microglial population was higher in the OB neurodegenerative environment of P60 PCD mice, but not later on. In contrast, this increase in the arrival of BMDC was not found in the cerebellum. This observation fits with the degenerative time course in both encephalic regions: while mitral cells in the OB undergo degeneration at P60, Purkinje cells in the cerebellum have almost completely disappeared at that age. Previous reports have claimed that microglial cells, which proliferate after injury to the CNS, do not originate from the bone marrow but are resident in the encephalon (1,32,46). Alternatively, the neurodegenerative microenvironment would also attract more BMDC increasing the resulting microglial population (21,32). Notwithstanding these observations, the significant rise of microglial cells observed in the ongoing boiling degeneration in the OB at P60 could be the result of both phenomena.
Regarding the plasticity mechanism(s) involved in the formation of microglial cells, the absence of X-gal staining in this cell type clearly showed that this phenomenon occurred through differentiation. Nonetheless, we cannot discard a silencing or an uncompleted CRE recombination process of the conditional reporter gene in some cell types, which would mask some of the fusion events (10,29).
Finally, no astrocytes were observed expressing any marker from donor bone marrow. This absence is in accordance with most previous studies (8,32,37,42,52) and demonstrates that BMDC do not contribute to astrocyte formation under either physiological or neurodegenerative conditions.
BMDC Contribute to Different Neuronal Types Through Two Different Plasticity Mechanisms
Our results concerning the contribution of BMDC to neuronal elements under physiological conditions corroborate previous studies addressing this phenomenon in the cerebellum (2,12,23,30,38,43,50,54). The frequency of Purkinje cells expressing donor markers and their relationship with the posttransplantation time lie within the values described previously in works using a similar BMT procedure (irradiation for removing the recipient's bone marrow, intravenous graft, and no additional conditioning). In addition, here we report new anatomical information (the existence of long axons with recurrent collaterals) concerning the integration of Purkinje cells expressing BMDC markers in the cerebellum. Moreover, we can confirm that BMDC fuse with Purkinje cells (2,23,30,38,54).
To our knowledge this is the first work reporting the contribution of BMDC to the olfactory interneurons. This important result clarifies those reported in previous publications in which the contribution of BMDC to the OB appeared controversial, since the neural nature of the differentiated GFP+ elements remained unclear (8, 11,32,43); our observations provide a direct and consistent demonstration of the contribution of BMDC to perfectly differentiated OB neurons. Moreover, we also discard the possibility that the newly formed interneurons might be generated in the SVZ, since we did not detect GFP+ neural precursors in this neurogenic region or in the rostral migratory stream.
Finally, we show for the first time how BMDC contribute to the CNS in different ways in the same animal, depending on the region and cell-specific factors: cell fusion for Purkinje cells and differentiation for olfactory interneurons. Future studies should clarify whether the plasticity mechanism involved depends on a) the type of participating BMDC or the resulting cellular type; b) different plasticity properties of the BMDC or fusing partner; and/or c) the niche where the phenomenon occurs.
Despite Their Neurodegeneration, Wild-Type BMDC Do Not Contribute to the Cellular Populations Affected by the pcd1J Mutation
We never observed Purkinje cells, either originated from grafted bone marrow or belonging to the recipients, in any of the transplanted PCD animals at the three different ages studied. However, previous work has reported that neurodegeneration has an important effect in promoting the arrival of BMDC and their fusion with Purkinje cells (3,23,30,38). This difference in the results may be the consequence of differences in the nature and time course of the neurodegenerative processes and in the transplantation system (intracranial vs. intravenous). An alternative explanation could be the early onset of Purkinje cell degeneration in PCD mice, or its high rate of progression (25,27). It is likely that when transplantation is performed (P20; earlier transplantation is not viable because it strongly increases the mortality and morbidity of PCD mutant mice and induces morphological abnormalities in the cerebellum) the neurodegenerative process is already very advanced and cannot be reversed. This is similar to many human neurodegenerative diseases, which are often diagnosed when the degenerative process is very advanced, thus limiting possibilities for improvement. Consistent with this, and since the plasticity mechanism involved in the contribution of BMDC to the Purkinje cell population is cell fusion, the absence of one of the partners in the fusion process did not produce any fusion event. Accordingly, the therapeutic use of BMDC in neurodegenerative pathologies should be considered with caution, depending on the etiology of the disease studied, the age/stage of the disease, and the type of degenerative process to be treated. Moreover, the encephalic area affected must be taken into account. Our results concerning the OB of transplanted PCD animals, where BMDC did not contribute to the mitral cell population despite their degeneration, show that there must be region-specific factors that modulate the plasticity properties of BMDC.
Similarly, Purkinje cells might have a special propensity to undergo cell fusion events with BMDC (38,50). Further analyses of what renders them fusogenic would provide new guidelines for exploiting this mechanism in regenerative medicine.
The results of the present study also demonstrate that the formation of new interneurons in the adult OB from the bone marrow seems not to be affected by mitral cell degeneration. Instead, it seems to be connected to the plasticity mechanism involved, and does not depend on cells other than BMDC.
In light of our observations, we conclude that BMDC do not only contribute to the cerebellar Purkinje cell population, but also to the OB interneurons. Both contributions are limited but take place under physiological conditions. The former occurs through cell fusion and hence depends on the existence of the cellular type generated and its fusogenic capability (39). The latter takes place by differentiation and is not affected by the neurodegeneration of surrounding cells. Future experiments should be performed to discern the physiological importance of both processes in normal homeostasis and in cell replacement therapies for the adult CNS.
Moreover, despite the strong neurodegenerative environment in both the cerebellum and the OB of PCD mutant mice, neither Purkinje cells nor mitral cells were formed from the transplanted bone marrow. Therefore, the scarce neural elements generated from BMDC, the restriction of the neuronal and glial types formed, and the lack of response against selective neuronal loss, as is observed in many neurodegenerative diseases, recommend that further basic studies be continued with experimental models that will clearly differentiate regional- and neural-type variables before planning clinical applications.
Footnotes
Acknowledgments
This work was supported by the Ministerio de Ciencia e Innovación (BFU2010-18284), the Ministerio de Sanidad, Política Social e Igualdad (Plan Nacional Sobre Drogas), the Junta de Castilla y León, “MMA,” “Samuel Solórzano Barruso,” and the “Alicia Koplowitz” Foundations, and Centre for Regenerative Medicine and Cell Therapy of Castilla y León.
