Abstract
The expression of phosphatidylserine-specific phospholipase A1 (PS-PLA1) is most upregulated in the genes of peripheral blood cells from chronic rejection model rats bearing long-term surviving cardiac allografts. The expression profile of PS-PLA1 in peripheral blood cells responsible for the immune response may indicate a possible biological marker for rejection episodes. In this study, PS-PLA1 mRNA expression was examined in human THP-1-derived macrophages. The effects of several immunosuppressive agents on this expression were also examined in in vitro experiments. A real-time RT-PCR analysis revealed that PS-PLA1 mRNA expression was found in human THP-1-derived macrophages. This expression was enhanced in the cells stimulated with lipopolysaccharide (LPS), a toll-like receptor (TLR) 4 ligand. Other TLR ligands (TLR2, 3, 5, 7, and 9) did not show a significant induction of PS-PLA1 mRNA. The time course of the mRNA expression profiles was different between PS-PLA1 and tumor necrosis factor-α (TNF-α), which showed a maximal expression at 12 and 1 h after LPS stimulation, respectively. Among the observed immunosuppressive agents, corticosteroids, prednisolone, 6α-methylprednisolone, dexamethasone, and beclomethasone inhibited PS-PLA1 expression with half-maximal inhibitory concentrations less than 3.0 nM, while methotrexate, cyclosporine A, tacrolimus, 6-mercaptopurine, and mycophenoic acid showed either a weak or moderate inhibition. These results suggest that the expression of PS-PLA1 mRNA in THP-1-derived macrophages is activated via TLR4 and it is inhibited by corticosteroids, which are used at high dosages to suppress chronic allograft rejection.
Introduction
The control of chronic rejection is important for long-term graft survival in organ transplantation, because this is a significant cause of graft loss during the first year following transplantation (11). Therefore, specific and sensitive markers predicting the chronic rejection are required for improving the clinical outcome. The current study used a high-throughput microarray analysis to demonstrate that phosphatidylserine-specific phospholipase A1 (PS-PLA1) showed the highest upregulated expression among all the observed genes in peripheral blood cells from chronic rejection model rats with long-term surviving cardiac allografts (12).
PS-PLA1, an enzyme of the lipase family, acts specifically on phosphatidylserine (PS) and hydrolyzes fatty acid from the sn-1 position of PS to produce lysophosphatidylserine (lysoPS) and fatty acid (6, 7, 16, 20, 24). Because both PS and lysoPS are associated with the activation of immune cells, including macrophages via PS and lysoPS receptors (15, 18, 19, 21), the activity of PS-PLA1 might be important for the immune response in regulating the production or elimination of PS and lysoPS (1, 2). A marked elevation of the PS-PLA1 expression is observed in rats treated with lipopolysaccharide (LPS) (4), thus suggesting that PS-PLA1 plays a role in the activated immune system (1), which is associated with chronic rejection (11). The current study focused on the expression profile of PS-PLA1 in immune cells as sample specimens for clinical application.
This study examined PS-PLA1 mRNA expression in human THP-1-derived macrophages stimulated by toll-like receptor (TLR) ligands, including LPS. The effects of immunosuppressive agents on the expression were also examined using this in vitro system.
Materials and Methods
Materials and Reagent Solutions
LPS (ultra-pure lipopolysaccharide, from Escherichia coli K12), heat-killed Listeria monocytogenes (HKLM), polyinosine-polycytidylic acid [poly(I:C)], flagellin (from B. subtilis), loxoribine and oligonucleotide 2006 (ODN2006) were purchased from InvivoGen (San Diego, CA) and prepared according to the manufacturer's instructions. RPMI-1640 medium, dimethyl sulfoxide (DMSO), prednisolone (PSL), 6α-methylprednisolone (MPSL), 6-mercaptopurine monohydrate (6MP), tacrolimus (TAC), dexamethasone (DEX), and mycophenoic acid (MPA) were purchased from Sigma-Aldrich (St. Louis, MO). Beclomethasone dipropionate (BDP) was purchased from MP Biomedicals (Solon, OH). Methotrexate (MTX), cyclosporine A (CYA), and phorbol 12-myristate 13-acetate (PMA) were purchased from Wako Chemicals (Osaka, Japan). Fetal bovine serum (FBS) was purchased from JRH Biosciences (Lenexa, KS). All other reagents and solvents for the assay were of either analytical or regent grade and they were purchased from Wako Chemicals or Sigma-Aldrich.
Cell Culture
The THP-1 human monocytic cell line (ATCC, TIB-202) was maintained in RPMI-1640 medium supplemented with 10% heat-inactivated FBS, 2 mM l-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin at 37°C in 5% CO2. Differentiated THP-1 cells were obtained by treatment with 5 nM PMA for 2 days and then were starved overnight in RPMI-1640 medium containing 0.5% FBS and 5 nM PMA. They were thereafter stimulated with 0-105 g/ml LPS in the presence of 10% heat-inactivated FBS for up to 34 h. In some experiments, differentiated THP-1 cells were stimulated with HKLM (108 cells/ml), poly(I:C) (25 mg/ml), flagellin (10−5 g/ml), loxoribine (1 mM), or ODN2006 (5 mM) instead of LPS. To investigate the effects of immunosuppressants, cells were treated with 10−5 M of PSL, MPSL, DEX, BDP, MTX, 6MP, CYA, or TAC together with/without LPS (10−5 g/ml) for 12 h. Dose dependency was also examined in 10−9-10−4 M of PSL, MPSL, DEX, and BDP.
Quantitative Real-Time RT-PCR Analysis
Primer sets used were synthesized at Invitrogen (Carlsbad, CA). The primer sets were: PS-PLA1 (sense, 5′-CCTTCCTTAGCAGTAACATCACC-3′; antisense, 5′-CAAACTCGGTTGGAAGACTGAA -3′), group IVA cytosolic PLA2 (cPLA2α) (sense, 5′-ACTCCAGATCC CTATGTGGAAC-3′; antisense, 5′-GCTGTCCCTAGA GTTTCATCCAT-3′), tumor necrosis factor-α (TNF-α) (sense, 5′-ATCTACTCCCAGGTCCTCTTCAA-3′; antisense, 5′-GCAATGATCCCAAAGTAGACCT-3′), β-glucuronidase (GUS) (sense, 5′-TGATGGAAGAAGTGGT GCGTAG-3′; antisense, 5′-AGTTGCTCACAAAGGT CACAGG-3′), glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (sense, 5′-GCCAAGGTCATCCATGA CAACT-3′; antisense, 5′-GAGGGGCCATCCACAGT CTT-3′).
Total RNA was extracted by Isogen (Nippon Gene, Tokyo, Japan) into TE according to the manufacturer's instructions. Single-strand cDNA was synthesized with SuperScript™ III Reverse Transcriptase and real-time PCR was performed with a dsDNA binding dye, SYBR Green I, using SuperScript™ III Platinum® Two-Step qPCR Kit with SYBR Green (Invitrogen, Carlsbad, CA). Fluorescence was detected with Mx3000P system (Stratagene, La Jolla, CA) and analyzed with MxPro software (Stratagene). The expression levels of mRNA for each gene were normalized by that of GUS.
All data are presented as the mean ± SD. Nonrepeated measures ANOVA followed by Dunnett's test was used to assess any significant differences between the absolute values. Significance was set at p < 0.05.
Results
PS-PLA1 mRNA Expression in Human THP-1-Derived Macrophages
The PS-PLA1 mRNA expression was compared with the expression of TNF-α mRNA (positive control for TLR response) in the THP-1-derived macrophages after treating the cells with several TLR ligands for 1 and 12 h. Among the TLR ligands [HKLM, poly(I:C), LPS, flagellin, loxoribine, and ODN2006 for TLR2, TLR3, TLR4, TLR5, TLR7, and TLR9, respectively], LPS induced PS-PLA1 expression at 12 h after treatment, while no other ligands exhibited such induction (data not shown). On the other hand, TNF-α mRNA was induced when the cells were treated with HKLM, LPS, and flagellin for 1 h (data not shown).
LPS treatment increased PS-PLA1 mRNA expression on THP-1-derived macrophages 800-fold, which was four times greater than that of cPLA2a (Fig. 1). The upregulation of PS-PLA1 mRNA was LPS dose dependent at the concentration of 10−9-10−5 g/ml (data not shown). The time course profiles of mRNA expression after LPS stimulation was different between PS-PLA1 and TNF-α. The maximum mRNA expression for PS-PLA1 and TNF-α was at 12 and 1 h after LPS stimulation, respectively (Fig. 1).

Time course profile of the LPS-mediated upregulation of mRNA expression for PS-PLA1, cPLA2α, and TNF-α in THP-1 macrophages. Differentiated THP-1 cells were incubated for 1–34 h with/without 10−5 g/ml of LPS. The relative mRNA levels of PS-PLA1 (upper panel, left axis: with LPS, filled circles; without LPS, open circles), cPLA2α (upper panel, right axis: with LPS, filled diamonds; without LPS, open diamonds) and TNF-α (lower panel: with LPS, filled squares; without LPS, open squares) to GUS were determined respectively by quantitative real-time RT-PCR. The results are shown as the mean ± SD of three independent experiments.
Effects of Immunosuppressive Agents on PS-PLA1 mRNA Expression
Among the immunosuppressive agents, corticosteroids (PSL, MPSL, DEX, and BDP) showed marked inhibition of PS-PLA1 mRNA expression, while other agents, MTX, CYA, TAC, 6MP and MPA, showed weak or little effects (Table 1). The inhibitory effects of corticosteroids on PS-PLA1 mRNA expression were dose dependent (Fig. 2). The half-maximal inhibitory concentration (IC50) for PSL was 3.0 nM (Fig. 2). The inhibitory effects of corticosteroids on mRNA expression for cPLA2α were similar to those for PS-PLA1 (Fig. 2). Other agents inhibited cPLA2α mRNA expression, although the inhibitory effects were weaker than that for corticosteroids (Table 1).

Dose-dependent inhibition of the mRNA expression for PS-PLA1 and cPLA2α by corticosteroids. Differentiated THP-1 cells were incubated for 12 h at different concentrations of PSL (open bars), MPSL (dotted bars), DEX (shaded bars), or BDP (hatched bars) in the presence of 10−5 g/ml of LPS. The relative mRNA levels of PS-PLA1 (a) and cPLA2α (b) to GUS were determined, respectively, by quantitative real-time RT-PCR. The results are shown as the mean ± SD of three independent experiments.
Effects of Immunosuppressive Agents on mRNA Expression
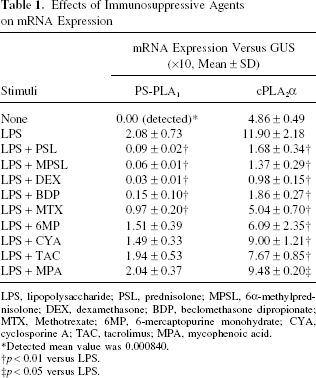
LPS, lipopolysaccharide; PSL, prednisolone; MPSL, 6a-methylprednisolone; DEX, dexamethasone; BDP, beclomethasone dipropionate; MTX, Methotrexate; 6MP, 6-mercaptopurine monohydrate; CYA, cyclosporine A; TAC, tacrolimus; MPA, mycophenoic acid.
Detected mean value was 0.000840.
p < 0.01 versus LPS.
p < 0.05 versus LPS.
Discussion
The present results revealed the expression of PS-PLA1 mRNA in THP-1-derived macrophages to be activated by treating the cells with LPS, a typical TLR4 ligand, but not with other TLR ligands. The induction of PS-PLA1 is also observed in rats after the administration of LPS (4). These findings suggest that PS-PLA1 mRNA is specifically activated via the TLR4 signaling pathways both in vitro and in vivo. On the other hand, cPLA2α mRNA was expressed on THP-1-derived macrophages in nature and it was little affected by LPS treatment. This means that these two phospholipases play different roles in the immune response with regard to TLR4 activation.
TLR4, a well-known receptor for LPS, interacts with a variety of other ligands including heat shock proteins (HSPs), and it rapidly activates the innate immune system (9, 17). The current findings suggest that innate immune events play a critical role in allograft rejection as well as host defense. In the initial allograft injury induced by reactive oxygen species, which are generated during allograft ischemia-reperfusion in implantation (10, 14, 22), the upregulated HSPs activate TLR4-bearing vascular cells, thus contributing to developing chronic rejection (9). The role of TLR4 in the development of chronic rejection has been also observed in rat recipients of cardiac transplantation (13). Therefore, the PS-PLA1 mRNA expression via TLR4 specific activation is consistent with the previous finding that PS-PLA1 is one of the most upregulated expression genes in a rat chronic rejection model, thus suggesting that it can also be possible biomarker for rejection episodes.
Another finding in the present study was that the PS-PLA1 mRNA expression was strongly inhibited by corticosteroids among the several known immunosuppressive agents (Table 1). cPLA2α, a key enzyme for prostaglandin biosynthesis (3, 23), is strongly inhibited by corticosteroids in their anti-inflammatory action (5). The magnitude of the inhibitory effects for PS-PLA1 mRNA expression was almost the same as those for cPLA2α (Fig. 2). This means that corticosteroids may exert different immunosuppressive effects on the status of PS-PLA1 expression via TLR4 activation. High-dose corticosteroids for chronic rejection (8) may therefore be a reasonable therapeutic strategy if PS-PLA1 plays an important role. Further study is thus required to confirm this speculation.
In conclusion, PS-PLA1 mRNA was specifically expressed via TLR4 activation in THP-1-derived macrophages. This expression was inhibited by corticosteroids, but not by other immunosuppressive agents. The clinical application of this potential treatment modality in allograft immunity therefore requires further investigation.
Footnotes
Acknowledgment
This work was supported in part by a Grant-in-Aid for the Encouragement of Scientists (No. 18923053) from the Japan Society for the Promotion of Science.
