Abstract
The goals of our study were to determine the possible association of interleukin (IL)-31 with Th17 cytokine profile in serum and to quantify retinoic acid-related orphan receptor C (RORC) mRNA expression in psoriatic arthritis (PsA) patients. This cross-sectional study was conducted in 50 patients with PsA and 30 control subjects (CS) matched by age and gender. The cytokine serum levels were quantified by magnetic bead–based assay using the Bio-Plex MAGPIX system, and RORC mRNA expression was determined by quantitative polymerase chain reaction (qPCR). As a result, significant differences in IL-31 were observed between study groups (77.23 pg/mL in PsA vs 64.4 pg/mL in CS, P < 0.001) and Th17 cytokine profile serum levels (IL-17A: 6.36 pg/mL in PsA vs 2.97 pg/mL in CS, P = 0.02; IL-17F: 44.15 pg/mL in PsA vs 23.36 pg/mL in PsA, P = 0.01; IL-17E: 3.03 pg/mL in PsA vs 0.82 pg/mL in CS, P < 0.001; IL-21: 36.45 pg/mL in PsA vs 12.44 pg/mL in CS, P = 0.02); however, significant differences were not observed for IL-23 (31.2 pg/mL in PsA vs 53.26 pg/mL in CS, P = 0.58). Furthermore, positive correlations between IL-31 and Th17 cytokine profile serum levels were found (IL-17A: rs = 0.64, P < 0.001; IL-17F: rs = 0.73, P < 0.001; IL-17E: rs = 0.70, P < 0.001; IL-21: rs = 0.54, P = 0.002; IL-23: rs = 0.5, P < 0.01). Regarding RORC gene expression, the PsA group showed an increase of 6.85-fold compared to the CS group. We did not find any association between the serum levels of cytokines and RORC gene expression. In conclusion, in PsA, there are increased serum levels of IL-31, IL-17A, IL-17F, IL-17E, and IL-21, but not IL-23. Moreover, there was a positive correlation of IL-31 with the Th17 cytokine profile and a high RORC gene expression. Altogether, these findings suggest a proinflammatory contribution of IL-31 in close association with the Th17 cytokine profile in PsA.
Introduction
Psoriatic arthritis (PsA) is a highly heterogeneous systemic autoimmune disease, characterized by severe manifestations that involve psoriasis, defined as the presence of erythematous, scaly, well-demarked plaques, with thickening of skin associated to musculoskeletal illness as spondylitis, enthesitis, or peripheral arthritis. 1 The PsA is classified into spondyloarthropathies, usually negative for rheumatoid factor. 2 Currently, it is identified by two clinical subtypes: first, oligoarticular PsA that can evolve conforming severity of disease, antinuclear antibodies’ positivity, uveitis, and dactylitis; second, polyarticular PsA that may present enthesitis, nail pitting, onycholysis, and spondylitis. 3 The worldwide prevalence of PsA is highly heterogeneous; however, in Mexico, it was estimated in 0.02% 4 with a 1:1 ratio between males and females. Likewise, several comorbidities have been associated with PsA as obesity, metabolic syndrome, and cardiovascular disease. 5 The etiology of PsA is still elusive, but several factors including the environment, genetic factors, and the immune dysfunction are associated with the development of this disease. 3 In the pathogenesis of PsA, immune dysfunction is the main player in the development of pathological manifestations in the skin, enthesis, and synovium, driven by distinct T helper (Th) cell subsets. 6 Particularly, a strong deregulation of Th17 cytokine profile has been implicated with an increase in epidermal and joint damage in PsA. On the other hand, other cytokines such as interleukin (IL)-31 have been implicated in skin inflammatory diseases such as atopic dermatitis, 7 but there are no existing reports regarding PsA. IL-31 is a cytokine produced mainly by the immune system cells such as Th2 and mast cells, and it has been demonstrated that interferon γ (IFNγ) and the cathelicidin LL-37 can upregulate IL-31 expression. 8 IL-31 has been shown to induce the secretion of cytokines such as IL-6, IL-8, and IL-32 as well as chemokines and matrix metalloproteinases (MMPs) in diseases involving chronic skin inflammation, suggesting a proinflammatory role. 8 Based on this knowledge, this study was focused on the evaluation of IL-31 and Th17 cytokine profile serum levels in PsA, as well as the retinoic acid-related orphan receptor C (RORC) gene expression, which represents the main transcription factor associated to Th17 cell differentiation in autoimmune diseases.9,10
Materials and methods
Study population
A cross-sectional study was conducted including 50 PsA patients classified according to the 2006 Classification Criteria for Psoriatic Arthritis (CASPAR); the PsA patients were recruited from psoriasis clinic of Dermatological Institute of Jalisco “Dr. José Barba Rubio,” Health Ministry, Mexico. Physical assessment, medical record, and evaluation of 28 joints to determine the clinical activity of disease according to the Disease Activity Score-28 (DAS28) were realized in all patients. Also, the body surface area (BSA) was used for the evaluation of skin disease extension. The cut-offs to determine severity categories were assigned as follows: mild (BSA < 3%); moderate (BSA: 3%–10%); and severe (BSA > 10%).
The control subjects (CS) group comprised 30 clinically healthy subjects with no history of allergic, autoimmune, or chronic inflammatory diseases, with normal weight, recruited from the general population in the same geographic area of PsA patients. Body mass index (BMI) in PsA and CS was determined taking into account the body weight (kg) divided by squared height (m2), and the classification of obesity (30 < BMI < 34.9), overweight (25 < BMI < 29.5), and normal weight (18.5 < BMI < 24.9) was made following the World Health Organization cut-offs for BMI. The PsA patients under biological or steroidal treatment were not included in this study. All recruited PsA patients presented active psoriasis and swollen painful joints.
Due to the low prevalence of PsA in Mexico (0.02%), for this study, all patients who attended dermatologist consultation and fulfilled the CASPAR inclusion criteria were recruited in the period of 2012–2013. In addition, for evaluating the statistical power of the study, a post hoc statistical test was performed; the Wilcoxon–Mann–Whitney test was performed through the G*Power 3.1.9.4 software, finding a minimum of 80% power for the statistical comparisons.
Quantification of IL-31 and Th17 cytokine profile
The serum levels of IL-31 and the Th17 cytokine profile (IL-17A, IL-17F, IL-17E, IL-21, and IL-23) were measured using a magnetic bead–based multiplex assay (Bio-Plex Pro™ Human Th17 Cytokine Panel, Cat. No. 171AA001 M, BIO-RAD, Hercules, CA, USA); the measurements were performed in duplicate according to the manufacturer’s protocol. The serum samples were stored at −80°C until the assay was performed.
Quantification of RORC target gene transcript
We conducted the quantification of RORC mRNA expression from total peripheral blood leukocytes by quantitative real-time polymerase chain reaction (PCR) using the FastStart Essential DNA Green Master kit (Roche Applied Science, Penzberg, Germany). For total leukocyte isolation, dextran reagent (Sigma-Aldrich Co, St. Louis, MO) was used and the total RNA was extracted through the modified Chomczynski and Sacchi 11 method. The RNA purity and concentration were quantified by spectrophotometry (NanoDrop 2000; Thermo Scientific, Waltham, MA, USA). Later, 1 μg of total RNA employing primer oligo-dT and Moloney Murine Leukemia Virus Reverse Transcriptase (Promega Corp., Madison, WI, USA) was used to synthesize complementary DNA (cDNA). Quantitative PCR reaction was normalized using actin-β (ACTB) gene mRNA expression as the reference gene. The primers used for cDNA amplification of RORC target gene were obtained from Luan et al. 12 and those for ACTB reference gene from Liu et al. 13 The primer sequences are as follows: RORC (NM_05060.3) forward 5′-CTGCAAGACTCATCGCCAAAG-3′ and reverse 5′-TTTCCACATGCTGGCTACACA-3′; ACTB (NM_001101.4) forward 5′-CATGTACGTTGCTATCCAGGC-3′ and reverse 5′-CTCCTTAATGTCACGCACGAT-3′. The PCR reactions were run in duplicate and performed in a LightCycler® 96 System (Roche Applied Science). The mRNA expression analysis was performed through the Pfaffl 14 and 2−ΔCq comparative methods after the validation of reaction efficiency for the target and reference genes by running serial dilutions.
Statistical analysis
The GraphPad Prism v5.0 software (GraphPad Software, San Diego, CA, USA) was used to perform the statistical analysis and graphics construction. The distribution of variables was analyzed with the Shapiro–Wilk normality test. Continuous variables that were normally distributed were expressed as mean and standard deviation (SD) and those non-normally distributed were expressed as median and 25th–75th percentiles. Differences were considered significant with a P value < 0.05.
Results
Clinical and demographic characteristics
In total, 50 patients diagnosed with PsA and 30 CS were enrolled in this cross-sectional study. The mean age was 52 ± 12 years and the median of disease evolution for psoriasis (skin only affected) was 14 (1–48) and 4 (1–33) in PsA. The mean of DAS28 was 4.29 ± 1.26 and the most frequent psoriasis type was plaque (66%), followed by scalp psoriasis (22%), guttate (6%), and palmoplantar and inverse (6%). The BSA was mild in 34% of PsA patients, moderate in 22%, and severe in 44%. Concerning nail change, the PsA patients showed dystrophy (36%), pitting (28%), hypertrophy (8%), and no changes (28%). Regarding pharmacological treatment, the patients were treated as follows: non-steroidal topical treatment (34%), methotrexate (MTX) 7.5–10 mg/week (22%), MTX 12.5–20 mg/week (14%), and a portion of PsA patients referred not to undergo any medical treatment (30%). The demographic and clinical parameters of PsA patients are shown in Table 1.
Clinical variables and serum levels of cytokines in PsA patients.

IL: interleukin; DAS28: Disease Activity Score-28; BSA: body surface area.
Data are provided as median (25th–75th percentile).
Data are provided as mean ± standard deviation.
Data are provided as percentage and n.
P < 0.05 (Kruskal–Wallis test with post hoc Dunn’s test for multiple comparisons).
Quantification of RORC gene expression in PsA and CS
For quantification of RORC mRNA, 20 PsA patients were selected according to DAS28: 10 patients with moderate activity (3.2 ⩽ DAS28 < 5.1) and 10 patients with severe activity (DAS28 ⩾ 5.1); in addition, 10 CS matched by age and gender were analyzed.
The relative expression of RORC mRNA was 6.85-fold higher in PsA compared to CS (Figure 1(a)). Moreover, we analyzed the RORC mRNA expression considering the disease activity of PsA patients and observed that those patients with moderate activity had an 8.4-fold higher RORC gene expression than CS, while the PsA group with high activity had only 3.4-fold more gene expression than CS (Figure 1(b)).

(a) RORC gene expression in PsA and CS. RORC gene expression stratified according to (b) Disease Activity Score-28 (DAS28), (c) pharmacological treatment, (d) evolution time of PsA, and (e) body mass index (BMI; kg/m2) in PsA patients. Analysis of relative expression was performed by the Pfaffl 14 method.
On the other hand, when we stratified the gene expression according to pharmacological treatment, we observed that the patients treated with non-steroidal topical and MTX 7.5–10 mg/week showed 20.23- and 5.89-fold increases in RORC gene expression, respectively, in comparison to CS, while the patients treated with MTX 12.5–20 mg/week showed a 0.61-fold decrease in comparison to CS (Figure 1(c)).
Taking into consideration the time of disease evolution, all PsA groups showed higher RORC mRNA expression compared with CS, with a 33.85-fold increase in subjects with PsA ⩽1 year, a 19.05-fold increase in subjects with 1 < PsA ⩾ 10 years, and a 1.38-fold increase in PsA >10 years (Figure 1(d)). Also, the PsA patients with obesity and overweight showed 5.85- and 4.32-fold increases compared with CS, respectively. Moreover, a similar expression between patients with regular weight and CS was found, with a slight decrease of 0.96-fold in patients with regular weight (Figure 1(e)).
Likewise, we analyzed the possible correlation of RORC gene expression with the clinical variables of PsA patients; nevertheless, no associations were found (data not shown).
Quantification of IL-31 and Th17 cytokine profile
As shown in Figure 2(a)–(f), the serum levels of IL-31 and the Th17 cytokine profile were increased in PsA compared to CS, with the exception of IL-23 (IL-31: 77.23 pg/mL (65.48–117) in PsA vs 64.37 pg/mL (39.3–74.7) in CS, P < 0.001; IL-17A: 6.4 pg/mL (3.5–13.6) in PsA vs 2.97 pg/mL (1.68–6.44) in CS, P = 0.02; IL-17E: 3.04 pg/mL (1.45–9.78) in PsA vs 0.82 pg/mL (0.3–3.86) in CS, P < 0.001; IL-17F: 44.15 pg/mL (18.38–73.2) in PsA vs 23.36 pg/mL (8.72–40.3) in CS, P = 0.01; IL-21: 36.45 pg/mL (12.44–105.5) in PsA vs 12.44 pg/mL (5.51–31.74) in CS, P = 0.02; IL-23: 31.2 pg/mL (15.99–71.43) in PsA vs 53.26 (8.59–89.75) in CS, P = 0.58).

Quantification of IL-31 and Th17 cytokine profile in PsA and CS. Comparison of serum levels of (a) IL-17A, (b) IL-17F, (c) IL-17E, (d) IL-21, (e) IL-23 and (f) IL-31 between PsA and SC. Horizontal lines indicate median and 25th–75th percentiles of the cytokine serum levels. Statistical analysis was performed using Mann–Whitney U test. PsA= Psoriatic arthritis; CS = Control subjects.
We also stratified the serum levels of the cytokines according to the clinical characteristics of PsA patients and found that the patients with scalp psoriasis had higher levels of IL-17A (Figure 3(a)) and IL-17E (Figure 3(b)) in comparison with the other clinical presentations of psoriasis (plaque, guttate, and palmoplantar and inverse). Furthermore, an increase in IL-17F serum level in the PsA patients with severe psoriasis extension was found compared with those with moderate extension (58.7 pg/mL (37.2–110) vs 14.5 pg/mL (10.8–43.2), P = 0.041; Table 1). In addition, an analysis of cytokine levels in PsA patients stratified according to the presence versus the absence of nail involvement was carried out; however, no significant differences were found (data not shown).
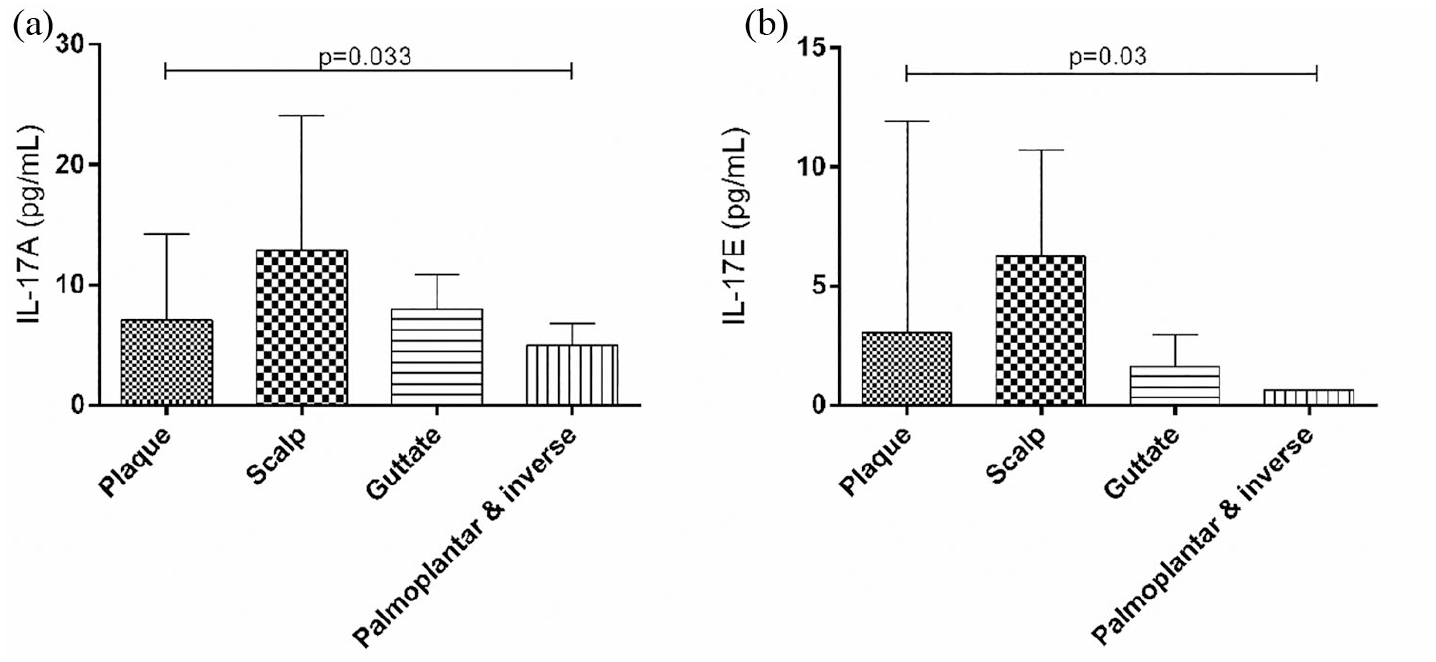
Serum levels of (a) IL-17A and (b) IL-17E according to psoriasis type in PsA patients. Horizontal lines indicate median and 25th–75th percentiles of the cytokine serum levels. Statistical analysis was performed using Kruskal–Wallis test.
On the other hand, based on previous findings in spondyloarthropathies, 15 we tested the correlation between serum levels of IL-31 and the Th17 cytokine profile. Interestingly, the levels of IL-31 showed a positive correlation with IL-17A (rs = 0.64, P < 0.001), IL-17E (rs = 0.70, P < 0.001), IL-17F (rs = 0.73, P < 0.001), IL-21 (rs = 0.54, P < 0.01), and IL-23 (rs = 0.50, P < 0.01) (Figure 4). These correlations were not observed in CS. Likewise, no correlation between RORC gene expression and the Th17 cytokine profile was found.

Positive correlations between serum levels of IL-31 and the Th17 cytokine profile in PsA patients. Correlation between serum levels of IL-31 with (a) IL-17A, (b) IL-17F, (c) IL-17E, (d) IL-21, and (e)IL-23 in PsA.
Discussion
PsA is an arthritic inflammatory disease associated with psoriasis that can affect the skin and peripheral joints as well as the axial skeleton.2,5 It is well established that PsA is an IL-17/IL-23 axis–mediated disease with a strong deregulation of proinflammatory cytokine production.1,16 IL-17 is a cytokine family of six members named IL-17A to IL-17F, synthesized mainly by Th17 cells, which are differentiated from naïve T cells through several cytokine combinations such as IL-1β and IL-23 in the presence of IL-6, tumor necrosis factor α (TNFα), IL-21, and transforming growth factor β (TGFβ).17–19 The expression of the Th17 cell effector molecules depends on their master regulator, the transcription factor RORγt, encoded by the RORC gene, which is activated by the signal transducer and activator of transcription 3 (STAT3), under the stimulation of polarizing cytokines. 1
On the other hand, IL-31, a new cytokine associated with the Th2 cytokine profile, has been related to inflammatory skin diseases such as atopic dermatitis.8,20 In this cross-sectional study, the aim was to analyze the serum levels of IL-31 and the Th17 cytokine profile along with RORC gene expression in PsA.
RORC encodes the RORγt transcription factor, which induces the transcription and expression of the Th17 gene signature in naïve Th cells and it is necessary for their development. 9 We found that the RORC gene expression was significantly increased in total leukocytes from the peripheral blood of PsA patients in comparison with the CS group. Similar findings were found by Muntyanu et al., 21 in the synovial fluid from PsA patients in comparison to those with other diseases that affect the musculoskeletal system. Likewise, an increased RORC expression was found in patients with moderate activity in comparison with CS, but this finding was not observed between patients with high activity and CS; this result was paradoxical because we expected that the patients with high disease activity would show the highest expression. Hence, we focused on the clinical characteristics of PsA patients, for which the RORC gene expression was stratified according to pharmacological treatment, the evolution time of the disease, and BMI. The patients had been treated with non-steroidal topical treatment and MTX in two different doses (7.5–10 and 12.5–20 mg/week), and we found a decreased RORC gene expression with the increasing MTX dose. MTX is a drug, the mechanism of action of which leads to a decrease in proinflammatory cytokines, including IL-1β 22 which has polarizing effects on Th17 cells; 18 in this regard, the patients with moderate activity who showed higher RORC expression had the lower doses of MTX and topical treatment, because of which this may be a possible explanation for the relationship observed between RORC expression and pharmacological treatment.
Another characteristic associated with disease severity in PsA is obesity. 23 We observed that RORC gene expression is directly proportional to the BMI increase in the PsA patients. It has been shown that in obesity the adipocytes contribute to the inflammatory process through the secretion of adipokines such as leptin, IL-6, and TNFα; 24 particularly, IL-6 can induce the expression of RORC, 18 and thus obesity could influence the severity of the disease.
On the other hand, we found that PsA patients with <1 year of evolution had increased RORC gene expression, which suggests that RORC could be a great biomarker with relevance on the opportune diagnosis. Particularly, a high expression of proinflammatory cytokines (including IL-17A) has been observed in the synovial tissue of PsA patients with early disease, which suggested that an important pathogenic mechanism succeeds in this stadium. 5
As for the Th17 cytokine profile, the IL-17 cytokine family, IL-17A and IL-17F, is the most studied. 25 In this study, an increase in IL-17A was found in PsA patients; this is a potent proinflammatory cytokine, for which the primary sources are CD4+, CD8+, and γδ T cells, natural killer T (NKT) cells, innate lymphoid cells (ILCs), and mast cells and, in certain circumstances, it can also be produced by neutrophils.26,27 IL-17A drives a powerful immune response that may lead to bone erosion and inflammation in both the skin and the synovium. 19 In fact, on keratinocytes, IL-17A can stimulate a positive feedback of proinflammatory cytokine production like IL-8 and β-defensins, synergistically with TNFα. 27 This positive correlation between IL-17A and TNFα has previously been observed at the systemic level in PsA patients in our population. 16 In inflammatory arthritis, IL-17A can induce MMP production driving the degradation of the extracellular matrix in the joint. 28 In addition, IL-17A can upregulate the RANK (receptor activator of nuclear factor kappa-Β) ligand on osteoblasts and T cells, promoting osteoclast activation and consequently bone destruction. 28
Pertaining to IL-17F, an increase in PsA was found. In addition, when comparing this cytokine in the PsA group, according to psoriasis extension, the patients with severe psoriasis showed higher levels of IL-17F. Thus, we suggest that IL-17F could be a key cytokine in skin injury.
IL-17F shares around 50% of the structural sequence with IL-17A, and there are overlapping functions between them. 29 It has been demonstrated that IL-17F is expressed in the lesioned skin and synovium of PsA patients. 30 IL-17F can induce proinflammatory mediators and synoviocyte activation, but to a lesser degree than IL-17A in PsA. 29 However, it has been suggested that, together, IL-17F and IL-17A can promote a more potent proinflammatory effect. 29
IL-17E (also known as IL-25) shares only 17% of structural sequences with IL-17A and does not have overlapping biological functions with IL-17A or IL-17F; instead, it has been involved in Th2-mediated disease and it is considered that IL-17E has an immunoregulatory role. 31 It has also been suggested that IL-17E acts as an attenuator of inflammation in inflammatory autoimmune diseases like inflammatory bowel disease, type 1 diabetes, and multiple sclerosis. 31 In autoimmunity, the role of IL-17E is still controversial due to some findings: in chronic inflammation as atopic dermatitis, IL-17E has been observed to be overexpressed and contributing to skin barrier dysfunction. 32 Specifically, IL-17E in keratinocytes from psoriatic plaques is increased, promoting inflammation via macrophages causing neutrophilia through IL-8 and MCP-1 (monocyte chemoattractant protein-1). 33 Furthermore, it has been observed that IL-17E on keratinocytes induces cell proliferation as well as cytokine and chemokine production via STAT3. 34 Moreover, increases in IL-17A and IL-17E were found in scalp psoriasis, because of which probably these cytokines are involved in this clinical presentation of psoriasis.
Likewise, we demonstrated an increase in IL-21, another cytokine from the Th17 profile, associated with the severity of PsA. IL-21 is a pleiotropic cytokine produced by Th17 cells, T follicular cells, and natural killer (NK) cells, and affects immune and non-immune cells. 35 In skin psoriasis, IL-21 levels correlate with the Psoriasis Area and Severity Index (PASI). In inflammatory arthritis, IL-21 promotes proliferation and cytokine production in T cells, amplifying damage in autoimmune diseases in several ways. First, IL-21 could bias the balance of the immune response through blocking regulatory T cells and the enhancement of Th17 cells. Second, IL-21R is found in synovial macrophages and fibroblasts, in which IL-21 can drive MMP secretion which leads to joint destruction. 36
On the other hand, we did not find any significant differences in IL-23 levels between PsA and CS. However, when we stratified the IL-23 serum levels in PsA patients according to DAS28, a tendency to the decrease in IL-23 was observed in patients with remission or low disease activity, without significant differences.
In this study, high serum levels of IL-31 in PsA patients were found. IL-31 is produced by Th2 cells, skin homing CD4 + CD45RO + CLA+ T cells, mast cells, keratinocytes, and dermal fibroblasts. 37 In recent years, IL-31 has been associated to skin inflammatory disease through the activation of dermal dendritic cells and increase in inflammatory mediators, controlling cell proliferation and tissue remodeling.38,39
The most consistent findings in this study were the strong positive correlations between IL-31 and the Th17 cytokine profile, so we suggest that IL-31 could induce a Th17 response directly or indirectly. IL-31 can signal through IL-31R, which is a heterodimer composed of IL-31RA and oncostatin M receptor β (OSMRβ). OSMRβ is expressed ubiquitously, while IL-31RA expression is stimulated mainly by IFNγ and TGFβ.37,39
The engagement of IL-31 with its receptors drives cell signaling that involves the activation of three signaling pathways: STAT3/5, PI3K/Akt, and MAPK (mitogen-activated protein kinase).8,15 STAT3 represents a signaling pathway associated with the generation of Th17 cells through promoting RORC gene expression, 1 but it is not yet clear if T cells express IL-31R.
On the other hand, IL-31 has been correlated with proinflammatory cytokines such as IL-6 and TNFα in spondyloarthropathies, which are considered Th17-polarizing cytokines. To the best of our knowledge, this the first report suggesting a potential inflammatory role of IL-31 through the induction of Th17 immune response in PsA.
A possible limitation of this study is that the measurement of cytokines and RORC gene expression was realized in a transversal way. This is important considering the fact that the cytokine serum levels in autoimmune patients are very dynamic, partly because of the high plasticity of T cells, treatment adherence, and the heterogeneity of this disease. Nevertheless, the variables were minimized through homogeneous selection of PsA patients as much as possible.
In conclusion, in PsA, there are high RORC expression and increased serum levels of IL-31, IL-17A, IL-17F, IL-17E, and IL-21, but not IL-23. Moreover, there is a positive correlation between IL-31 and the Th17 cytokine profile. Altogether, these findings suggest a proinflammatory contribution of IL-31 in close association with the Th17 cytokine profile in PsA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Research Ethical Committee of Centro Universitario de Ciencias de la Salud, Universidad de Guadalajara (C.I. 084/2012).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from Universidad de Guadalajara through Fortalecimiento de la Investigación y el Posgrado 2018 Grant (No. 244159), assigned to J.F.M.V.
Informed consent
Written informed consent was obtained from all participants before enrollment in the study according to the ethical guidelines of the 2013 Declaration of Helsinki.
