Abstract
Intraperitoneal (IP) anesthesia is commonly used for laboratory animal experiments including rat islet isolation. However, the direct effects of anesthetics on pancreatic islets have been neglected. This study compared the islet function and recovery yield from rats that were anesthetized using IP and intramuscular (IM) injection. In addition, the lag time required to lose deep pain was measured in the following anesthetics combinations. Lewis rats were anesthetized using ketamine and xylazine (K/X) or zoletil and xylazine (Z/X). A glucose challenge test was performed on each group of prepared islets. The effect of the anesthetic agents (e.g., ketamine, zoletil, xylazine alone, and the combination of K/X and Z/X) on cell lines (rat insulinoma; RIN-5F) was investigated by determining their effect on the cell viability, the amount of insulin, and insulin mRNA expression levels of RIN-5F. The time needed for deep anesthesia in IM anesthesia was significantly shortened in comparison to IP [K/X (IM: 313 ± 66 s, IP: 371 ± 84 s) and Z/X (IM: 206 ± 76 s, IP: 245 ± 92 s)]. In addition, number of isolated islet yield by IM anesthesia was significantly improved [K/X (IM: 1530 ± 242, IP: 1245 ± 149) and Z/X (IM: 1136 ± 226, IP: 511 ± 154)]. The functions of fresh islets, indicated by the stimulation index, acquired under IM anesthesia was better preserved than that of IP. The viability and the insulin secretion of RIN-5F were decreased at 24 and 48 h. Insulin gene expression levels were decreased at 24 h as well. Anesthetics may be absorbed through the pancreas surface to the islets and have a direct effect, resulting in islet exposure and deterioration during isolation. In conclusion, for rodent islet isolation, IM anesthesia is simpler and safer in comparison to IP anesthesia.
Introduction
Apart from inhalation anesthesia with diethyl ether, anesthesia via intraperitoneal (IP) injection is most commonly used for anesthesia in laboratory animal experiments. Although diethyl ether is preferred because of simplicity and low cost, it is quite volatile and potentially irritates both animals and researchers. When other volatile agents for inhalation anesthesia are used in rodents, problems such as high cost and technical difficulty of airway preservation are still present.
The combination of ketamine/xylazine (K/X) is a well-established and commonly used anesthetic mixture (4, 23). Occasionally, a zoletil (tiletamine-zolazepam)/xylazine (Z/X) cocktail is selected for rat anesthesia (19, 26). IP injection is usually chosen by most researchers because it is more convenient and easier than intramuscular (IM) injection. Trained professionals are able to handle small laboratory animals independently when administering IP injection. On the other hand, IM injection is discouraged because of the difficulty in restricting unexpected movements of the rodents, which frequently results in animal bites.
One of authors, who had used a restrainer for IM injection, previously proposed (11–13) that use of IM anesthesia may be superior to that of IP anesthesia for islet yields from rats that underwent two different types of anesthesia, in comparison to earlier studies (6, 7). In experiments involving the abdominal organs, such as islet isolation, IP anesthesia may have disadvantages because the organs are exposed directly to the anesthetic drugs. Therefore, the direct exposure of the pancreas to anesthetic agents may affect islet isolation, thus resulting in changes in yield, function, and viability of the islets.
The aim of the present study was to explore the utility of IM anesthesia for rodent islet isolation and compare the function and yields of pancreatic islets isolated following IM and IP anesthetics treatment.
Materials and Methods
Animals and Anesthesia
Adult male Lewis rats (SLC Japan Co., Hamamatsu, Japan; body weight 300–350 g) were used for islet isolation. The rats were maintained according to the ethical guidelines of the institution, and the Committee on Animal Investigation of the Yonsei University approved the experimental protocol. This study was performed in accordance with the institutional guidelines for animal research. The rats were anesthetized using a combination of 50 mg/kg of ketamine (Yuhan Co., Seoul, South Korea) and 6.7 mg/kg of xylazine (Bayer Korea Ltd., Seoul, South Korea) (K/X) or 15 mg/kg of zoletil (Virbac Lab. Carros, France) and 9.3 mg/kg of xylazine (Z/X). These drugs were injected intramuscularly or intraperitoneally. A specially designed restrainer (12) was used to administer the IM injection. The lag time required to lose deep pain reaction by negative pedal withdrawal responses (supposed deep anesthesia) was measured by adding physical stimulation of pain with forceps.
Rat Islet Isolation
Lewis rats were anesthetized using K/X and Z/X via IM or IP injection and an abdominal midline incision was made. The subjects were divided into four groups (K/X-IM, K/X-IP, Z/X-IM, and Z/X-IP) of three rats each numbered at random. Pancreatic islets were prepared using a modification of the technique described in previous publications (10, 14, 15). The pancreatic duct was cannulated with a 24-gauge catheter. Subsequently, the pancreata was injected and expanded with 10 ml of 2 mg/ml collagenase P (Roche, Indianapolis, IN, USA) dissolved in Hank's balanced salt solution (HBSS; Invitrogen Co., Carlsbad, CA, USA) and digested for 16 min. The enzyme activity was blocked by cold HBSS containing 5% fetal bovine serum (FBS, v/v). Therein, the islets were passed through a 500-μm mesh and islets were purified by Histopaque (Sigma Chemical Co., St. Louis, MO, USA) density gradients, collected, and were purified twice by hand-picking. Individual groups of islets were counted and their diameters were measured, following islet staining with diphenylthiocarbazone (dithizone; Sigma Chemical Co.). Isolated islets was counted and expressed as number of islet yield per each rat.
Glucose Challenge Test of Isolated Islets
Within 30 min of purification, the fresh isolated islets underwent a glucose challenge test. Prepared islets of each experimental group (50 each by hand-picking, 100–150 μm in diameter) were divided for static incubation. Two different media were prepared and used for glucose the challenges as follows. A mixed culture medium containing 50% of Ham's F-12 medium (Invitrogen Co.) and 50% of RPMI-1640 without glucose (Invitrogen Co.) were used and 25 mmol/L HEPES (Sigma Chemical Co.), 1% antibiotic/antimycotic mixture (Invitrogen Co.), 50 μg/ml ascorbic acid (Sigma Chemical Co.), and 10% heat-inactivated FBS were added. Glucose (Sigma Chemical Co.) was adjusted to 100 mg/dl in the low media and 400 mg/dl in the high media. Under standard culture conditions (37°C, 5% CO2), each batch of islets was incubated for three successive periods of 1 h in the low media, then 1 h in the high media, and additionally 1 h in the low media again. In vitro islet functions of the four groups were evaluated and insulin contents were measured under different glucose levels. Islet function was represented by the stimulation index (SI). The SI of each group was calculated using the following formula: SI = insulin content in the high media/insulin content in the low media. The insulin content of the media samples was measured using an Enzyme-Linked ImmunoSorbent Assay (ELISA, ALPCO, Salem, NH, USA).
Cell Line Culture and Anesthetics Treatment
Rat insulin-secreting cells (RIN-5F) were purchased from the American Type Culture Collection (ATCC). Cells were maintained on tissue culture plates in RPMI-1640 supplemented with 10% FBS, 100 U/ml penicillin, and 100 μg/ml streptomycin at 37°C in a 5% CO2 humidified incubator. At 90% confluence, the cultured cells were incubated with serum-free RPMI-1640 overnight to arrest and synchronize cellular growth. The media were then replaced with fresh serum-free RPMI-1640 containing anesthetic agents. RIN-5F cells were treated with 5 μg/ml of ketamine, zoletil, and xylazine separately, and combinations of K/X (5 μg/ml of K and 2 μg/ml of X) and Z/X (5 μg/ml of Z and 3.3 μg/ml of X). Nontreated cells were prepared and tested as control groups at the same time.
Methylthiasoletetrazolium Assay
To examine islet cell death, RIN-5F cells were cultured in 96-well culture plate (3 × 104 cell/well each) and phenol red-free medium (RPMI-1640). Methylthiasoletetrazolium (MTT; 1 mg/ml) was added to each well after the experimental culture periods of anesthetic treatments (24 and 48 h), and incubated at 37°C in humidified 5% CO2 in air for 2 h. Then an extraction buffer (20% SDS, 50% N,N-dimethylformamide, pH 4.7) was added and further incubated overnight at 37°C. The optical density (OD) of samples was measured with a microplate reader (SpectraMax 340, Molecular Devices Co., CA, USA) at a wavelength of 562 nm.
Insulin Secretion of RIN-5F Cells
To measure the insulin secretion of RIN-5F cells, they were incubated for 24 h under the condition mentioned above. After incubation, the media were transferred to a new tube and centrifuged for sedimentation of extra debris. The supernatants were maintained at −70°C before undergoing the insulin measuring test by ELISA according to the manufacturer's protocol.
RNA Isolation and Real-Time Polymerase Chain Reaction
To extract mRNA from RIN-5F cells, the cells were rinsed twice with cold PBS and TRIZOL reagent (Invitrogen Co.) was added to the culture dish for lysis. The samples that contained cells in TRIZOL reagent were transferred to a microtube and chloroform was added. After 15 min of incubation, samples were centrifuged at 13,000 × g for 15 min at 4°C and the aqueous phase was mixed with isopropyl alcohol for RNA precipitation. The samples were then incubated at room temperature for 10 min and centrifuged at 13,000 × g for 10 min at 4°C. The supernatant was discarded after centrifuge. The extracted RNA was washed twice with 75% ethanol in RNase-free water. Finally, the isolated RNA were dissolved in 20 μl of RNase-free water and stored at −70°C before experimental treatments. The quantities of total RNA were measured by NanoDrop spectrophotometer (NanoDrop Technologies Inc., DE, USA) and 5 μg of total RNA was used for reverse transcriptase (RT) reaction (AccuPower RT premix kit, Invitrogen Co.) to synthesize cDNA. The cDNA synthesized from RNA were quantitated by real-time Polymerase Chain Reaction (PCR) using cDNA specific primer of GAPDH: forward 5′-TCCCTCAAGATTGTCAGCAA-3′, reverse 5′-AGA TCCACAAACGGATACATT-3′, and insulin: forward 5′-CCCAAGATTTTGTGCAGTGGTT-3′, reverse 5′-GT TGCAGTAGTTCTCCAGGTG-3′. Real-time PCR was performed using Power SYBR Green PCR Master Mix (Applied Biosystems, CA, USA) on the ABI PRISM 7500 Current Real-Time PCR machine (Applied Biosystems) according to the manufacturer's protocol. The gene expression levels of insulin were accessed as fold change relative to GAPDH.
Statistical Analysis
All results were calculated as the ratios of anesthetics-treated versus control group, and expressed as the means ± SE. The mean values for each group were compared by ANOVA and then by using Fisher's least significant difference method. Values of p < 0.05 were considered to be statistically significant.
Results
Time Needed for Deep Anesthesia
The lag time required to lose deep pain by negative pedal withdrawal responses was counted to compare the availability between IM and IP anesthesia. The lag time to deep anesthesia was significantly reduced with IM anesthesia regardless of the combination of anesthetics (Table 1).
Lag Time Required to Lose Deep Pain and the Islet Yield Using Different Administration of Anesthesia
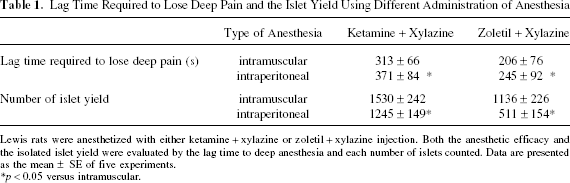
Lewis rats were anesthetized with either ketamine + xylazine or zoletil + xylazine injection. Both the anesthetic efficacy and the isolated islet yield were evaluated by the lag time to deep anesthesia and each number of islets counted. Data are presented as the mean ± SE of five experiments.
p < 0.05 versus intramuscular.
Islet Yield
The islet purity of all groups reached over 99% after hand-picking. The yield of the isolated islets of the IM groups was significantly greater in comparison to that of the IP groups regardless of the different anesthetic combinations. The yield difference of the Z/X groups was also greater than that of the K/X groups (Table 1).
Glucose-Stimulated Insulin Secretion
The mean SI values of the IM groups were significantly higher than those of the IP groups regardless of the different anesthetic combinations. The mean SI of K/X-IM and K/X-IP groups were 2.447 and 2.142, whereas mean SI values of the Z/X-IM and Z/X-IP groups were 1.292 and 1.097, respectively (Fig. 1).

Stimulation index of each islet group isolated under intramuscular (IM) or intraperotoneal (IP) anesthesia. (A) Ketamine + xylazine, (B) zoletil + xylazine. Primary islets were tested by the glucose challenge test. Stimulation indices were calculated as a ratio of insulin from islets in media containing 400 mg/dl glucose and that of insulin secretion in 100 mg/dl glucose. ∗p < 0.05 versus IM.
Viability of RIN-5F Cells Treated by Anesthetic Drugs: MTT Assay
Slow deterioration in the viability of the treated RIN-5F cells was noticed in all treated groups in comparison to the untreated control cells (regarded as 100% viability). The viabilities of samples treated with ketamine, zoletil, and xylazine declined 11.7%, 11.2%, and 13% after 24 h, respectively, and 14.9%, 12.5%, and 17.3% after 48 h, respectively. The viabilities of ketamine, zoletil, and xylazine were 88.3%, 88.8%, and 87% at 24 h, and 85.1%, 87.5%, and 82.7% at 48 h, respectively. The groups treated with anesthetic cocktail showed decreased viabilities in comparison to those treated with a single anesthetic agent. The viabilities of the groups under combination with K/X and Z/X declined to 83.1% and 85.5% after 24 h, and to 80.2% and 81.3% after 48 h. The data of all groups showed a time-dependent decrease (Fig. 2).
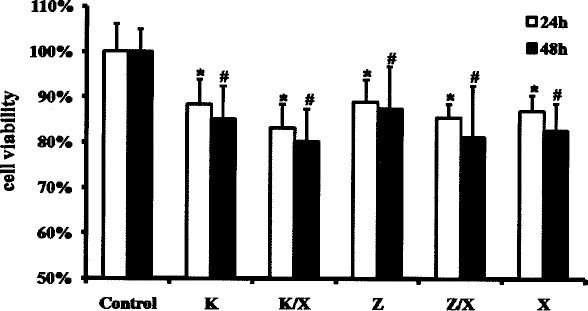
The effect of anesthetics on viability of RIN-5F evaluated with MTT assay. RIN-5F cells were treated with 5 μg/ml of ketamine (K), zoletil (Z), and xylazine (X) separately, and combination of K/X (5 μg/ml of K and 2 μg/ml of X) and Z/X (5 μg/ml of Z and 3.3 μg/ml of X). Nontreated cells as control groups were prepared and tested at the same time. The data were investigated at 24 and 48 h. ∗,#p < 0.01 versus control.
Insulin Secretion of RIN-5F Cells
The insulin secretion was decreased in all treated groups in comparison to untreated control cells (regarded as 100% insulin secretion). The groups treated with a combination of anesthetics showed greater decrease in secretion than those treated with a single agent after 24 h. Insulin accumulation from the cells treated with ketamine, zoletil. and xylazine decreased 9.8%, 18.5%, and 19.7%, respectively. The insulin contents of the groups treated with a combination of agents showed a remarkable decline by 44.8% and 57.5% in K/X and Z/X, respectively (Fig. 3).

The effect of anesthetics on insulin secretion of RIN-5F using ELISA. RIN-5F cells were treated with 5 μg/ml of ketamine (K), zoletil (Z), and xylazine (X) separately, and combination of K/X (5 μg/ml of K and 2 μg/ml of X) and Z/X (5 μg/ml of Z and 3.3 μg/ml of X). Nontreated cells as control groups were prepared and tested at the same time. The data were investigated at 24 h. ∗p < 0.05 versus control.
Insulin mRNA Expression
The insulin gene expression declined in all treated groups in comparison to the untreated control cells (regarded as 100% insulin mRNA expression level). The groups treated with a combination of anesthetics showed a more decreased level of gene expression than those treated with a single agent after 24 h. The insulin gene expression of the cells treated with ketamine, zoletil, and xylazine decreased 33.8%, 34.9%, and 47.8%, respectively, and the insulin mRNA expression of the groups treated with combinations of K/X and Z/X were remarkably decreased by 45.7% and 54.9%, respectively (Fig. 4).

The effect of anesthetics on insulin gene expression levels of RIN-5F using real-time PCR. RIN-5F cells were treated with 5 μg/ml of ketamine (K), zoletil (Z), and xylazine (X) separately, and combination of K/X (5 μg/ml of K and 2 μg/ml of X) and Z/X (5 μg/ml of Z and 3.3 μg/ml of X). Nontreated cells as control groups were prepared and tested at the same time. The data were investigated at 24 h. ∗p < 0.05 versus control.
Discussion
Inhalation anesthesia, such as diethyl ether, halo-thane, isoflurane, and sevoflurane, and IP anesthesia are most commonly adopted by the investigators when conducting rodent experiments. With the exception of diethyl ether, anesthetic gases in inhalation anesthesia are the optimal protocol in rodents. However, there are many associated problems, including the high cost of equipment such as vaporizers and ventilators, and the expense of drugs themselves and the technical difficulty of intubation. Diethyl ether is a very cheap agent; however, it presents an extremely dangerous explosion hazard. The obstacles associated with diethyl ether gas include its highly volatile, inflammable, and detrimental nature, which irritates both animals and researchers at the same time. Therefore, the use of diethyl ether requires safety measures, such as ventilation hoods, in the research center. Moreover, it is difficult to control and monitor the depth of the anesthesia and has bronchial irritation, which induces both a marked secretion from the airway epithelium and salivation. Therefore, unpredictable accidents regarding anesthetic procedures frequently occur. Inhalation anesthesia with diethyl ether may be accessible and cost-effective, but when considering animal rights, diethyl ether is not recommendable.
Most investigators prefer to use IP anesthesia because of its simplicity and the lower cost. However, this method may allow the absorption of anesthetics directly through the organ surface when the drug is directly injected in the peritoneal cavity and may have detrimental effects on the organs in abdominal cavity. In conformity with the Guide to the Care and Use of Experimental Animals (17) and the 3Rs (reduction, refinement, and replacement) (20, 24), optimal anesthesia is necessary to reduce the pain and stress to animals and to guarantee the safety of investigators for successful islet isolation from pancreas of abdominal cavity in small animals.
Although inhalation anesthesia with diethyl ether is accompanied by the risks mentioned above, ether has been widely adopted and used in laboratory animal research. On the other hand, injection anesthesia using ketamine, zoletil, nembutal (pentobarbital sodium), and urethane are applied via IP injection in rodent experiments. Among these agents, nembutal and urethane are almost always administered as a single drug. Xylazine (alpha 2 adrenaline receptor agonist) is usually combined with ketamine (4, 23) and zoletil (1, 19) to prolong anesthetic and analgesic effects; furthermore, they also provide a muscular relaxation effect. The dissociative anesthetic, ketamine, may be the most widely used injectable anesthetic in veterinary practice. Ketamine is the one of the famous “Club drugs,” which are substances commonly used at nightclubs, music festivals, raves, and dance parties to enhance social intimacy and sensory stimulation (9, 27). Therefore, regulations on the use ketamine are growing strict, which limits their availability to researchers. Due to the serious side effects (22) of ketamine, especially the psychomimetic side effects, this institution and the Committee on Animal Investigation of the Yonsei University recommends the use of zoletil instead of ketamine.
Generally, the recommended dosage and protocol of these anesthetic drugs for both IP and IM anesthesia are noted in most textbooks (2, 18); however, IP injection is favored over IM injection by many veteran investigators, because they are well trained and feel comfortable performing such IP injections. IP injections are easy to perform; namely, injection inside the abdominal cavity requires only manual restraint (3). On the contrary, despite injection into gluteus muscle where fewer pain nerves are distributed, IM injection can result in the investigators being bitten during the experimental animals when animal restraint is incomplete. Consequently, it is necessary to undertake IM anesthesia with at least two investigators or to completely restrain the animal to prevent unpredictable accidents.
In experiments using abdominal organs such as islet isolation, IP anesthesia may pose many potential risks because the organs are directly exposed to the anesthetics and these drugs are inevitably absorbed through the surface of organs. In vitro experiments of cortical neurons (21) with ketamine have been reported, but these used an indirect cell line test. A previous report (11) compared the function and yield of islets from rats when anesthetized with zoletil and xylazine combination by different routes. However, no other study has compared the functions of isolated islet or cell lines anesthetized by IM versus IP administration routes.
There are no previous studies comparing the two different routes (IM versus IP) for anesthesia. Direct exposure of the pancreas to anesthetics agents may affect the cell line (RIN-5F) and islet isolation including yields, function, and viability of islets. This study is the first report to demonstrate the systematic difference between IM and IP anesthesia, showing their influence on isolated rodent islet viability and the function of isolated islets. According to the results of this study, the effects of the two routes of anesthetics were definitive. There was reduced islet yield in the IP groups. It is possible that the islets were exposed to and diminished by the anesthetic drugs during the isolation process. This may have been caused by the absorption of anesthetics through the pancreatic surface, affecting the islets, resulting in diminished islet function. The physical nature of these agents, such as their pH, may interact with collagenase during digestion in the isolation procedure. Further molecular and genetic studies are required in the future to evaluate the actual mechanism.
In general, the rate of absorption is IV > IP > IM > SC > per oral (2, 18). However, the current finding was contradictory to this pattern (Table 1). The lag time of IM anesthesia was significantly shorter than that of IP, which reverses the rate of drug onset to IM > IP. The yield of isolated islets with the use of K/X was higher than Z/X, which may imply that the zoletil and xylazine combination had a more harmful effect on islet recovery yield. This phenomenon may be a result of the high concentration of xylazine in the Z/X regimen or the toxic characteristics of zoletil itself may be greater than that of ketamine.
To eliminate differences in the insulin response among groups owing to the variation of islet size, stimulation indices based on glucose challenge tests were adapted. The results suggested a significant difference to exist in the glucose-stimulated insulin release following IM and IP administration. Insulin secretion was stimulated in the IM groups by low- and high-glucose concentrations and the SIs were greater than that of IP groups.
To compare the effects on primary islets and cell lines, the effects were investigated on a commercially available established cell line (rat insulinoma; RIN-5F). To perform the tests of RIN-5F, concentrations of anesthetic substances were determined based on previous reports of cell lines (5, 16, 25). The concentrations of anesthetics were lower than the concentration within the range of clinical relevance (8). Further studies are needed including in vitro experiments with cell lines or other isolated cells from the abdominal cavity such as liver cells to evaluate single anesthetic effects separately, including viability and functions of tissue exposed by the agent.
If a reliable method of restraint can be adapted in rodent anesthesia, it is likely that IM injection will become a general method for anesthesia, and the special restraint (patent pending by Jeong-Ik Lee) used in the current study would be useful for such animal experiments (12). Consequently, it is suggested that IM injection is a simple and superior method to IP injection for obtaining accurate data concerning intra-abdominal organs in small animal laboratory experiments, when it is difficult to use inhalation anesthesia.
Footnotes
Acknowledgments
This work was supported by a grant (No. A080058) of the Korean Healthcare Technology R & D Project, Ministry of Health, Welfare & Family Affairs, Republic of Korea. Jeong-Ik Lee was a research scholar supported by the research grant for the in vitro cell line (RIN-5F) investigation from the 1st Mandom International Research Grants on Alternative to Animal Experiments.
