Abstract
To evaluate whether further improvement in porcine islet xenotransplantation is feasible, a number of questions were addressed. Earlier we showed significant improvement in the nude mouse of the porcine islets by selection through long-term culture. Now these islets were tested in the stringent pig-to-rat model. Islets were isolated from adult pigs, cultured for 1.5–3 weeks and transplanted to rats. Possible rejection mechanisms were assessed by interference of the cellular response with cyclosporine A (CsA), blocking macrophages with gadolinium chloride (GdCl), and suppressing the humoral response with cyclophosphamide. Modifications in graft size and condition were analyzed. Untreated control recipients showed primary nonfunction (PNF). CsA treatment could fully overcome PNF and resulted in graft survival from 10 to over 134 days. Rejection was the main cause of function loss. Although rejection could not be prevented by intensifying the induction therapy, increased maintenance immunosuppression effectively blocked rejection, albeit at the expense of toxicity. Blocking the humoral response was ineffective; all grafts showed PNF. In contrast, depletion of macrophages fully prevented PNF. Combination of GdCl and CsA gave no additional effect, and grafts were rejected between 57 and 162 days. Generally, graft survivals were similar to those reported in the literature; however, long-term cultured islets required much less maintenance immunosuppression. Cessation of graft function was not always due to rejection; in some cases “islet exhaustion” was found, possibly caused by discrepancy between the graft size and the rapidly growing recipient. Neither the presence of damaged islet tissue in the graft nor the size of the graft exerted any influence on graft survival. On rejection, no real infiltration of the graft was seen; destruction gradually processed from the outside. The good functional capability of the cultured islets was illustrated by disappearance of the clinical symptoms and increase in body weight, which almost doubled in the long-term survivors.
Introduction
The pig is widely considered a potentially interesting donor in islets of Langerhans xenotransplantation. Unfortunately, porcine islets have proven very difficult to handle. This becomes evident during their isolation, but also when they are cultured and functionally assayed. Moreover, for a long time these problems have probably been jointly responsible for the variable results after transplantation of porcine islets in different recipient species (5, 7, 12, 14, 29). In most studies on adult porcine islet xenotransplantation to the rat, islets are transplanted directly after isolation (7, 21, 30) or after a few days of culturing (29, 32).
Recently, we showed that grafts of adult porcine islets isolated from commercially slaughtered pigs, although seemingly of good quality, actually contained large numbers of islets of poor quality. Most likely a substantial part of the islets was severely damaged or killed by the violence of the isolation procedure, resulting in a digest consisting of islets of highly different “quality.” Long-term culture improved the overall viability of the graft by providing the opportunity for a part of the damaged islets to recover, whereas the most severely damaged and dead islets fragmented and were eliminated during the washing steps. In this way culturing selected for high-quality islets, which showed superior function when transplanted in diabetic nude mice compared to freshly isolated and short-term cultured islets (26).
The aim of the present study was to evaluate the transplantation of these adult porcine islets selected by long-term culture in the stringent pig-to-rat model. Further, a series of factors considered of potential relevance for successful long-term graft function was evaluated.
The role of graft rejection was explored by interference in three of the major segments of the immune system, all generally considered of potential significance in both organ and tissue xenotransplantation. The cellular immune response was blocked with cyclosporine A (CsA), the humoral immune response with cyclophosphamide (Cyphos), and the role of macrophages was studied by depletion through gadolinium chloride (GdCl). Although several studies histologically investigated the immune response at the cellular level, little information is available on the way these immune cells attack the graft. Therefore, it was evaluated in eventual rejection if immune cells actually invaded the graft from the newly formed vasculature or from the periphery. It was further assessed how the size and condition of the islet transplant were related to graft function.
Materials and Methods
Donor Pigs
Pancreata from retired breeder sows weighing 200–250 kg were obtained from a local slaughterhouse. Warm ischemia times ranged from 10 to 15 min and organs were stored in Hanks balanced salt solution without any additional intraductal infusion and transported on ice to the laboratory.
Recipient Rats
Six- to 10-week-old male Lewis rats (Charles River Laboratories, Lyon, France) were made diabetic by intravenous injection of 65 mg/kg streptozotocin (Roche Diagnostics, Almere, The Netherlands). Diabetes was defined by two measurements of nonfasting blood glucose levels ≥ 20 mM/L. Animals were kept under standard conditions with free access to water and standard rodent chow. All experiments were approved by the Institutional Animal Experimentation Committee.
Islet Isolation and Culture
Cold ischemia times ranged from 3 to 5 h. In the laboratory the pancreas was distended maximally with a solution of 0.5 mg/ml Liberase PI (Roche) in University of Wisconsin solution (UWS). After incubation at 37°C, the organ was dispersed in UWS on ice. Islets were purified using OptiPrep discontinuous gradients (Nycomed, Oslo, Norway) in UWS. Purity, morphology, and islet numbers were determined in duplicate after dithizone staining and islet numbers were expressed as islet equivalents (IE). Islets were cultured at 37°C in 5% CO2 in air in bacteriological petri dishes (10 cm, Falcon, Micronic B.V., The Netherlands; 1,000–5,000 IE/dish) in 10 ml RPMI-1640 medium (Invitrogen, Breda, The Netherlands) supplemented with 10% (v/v) heat-inactivated adult porcine serum (Cellect Porcine serum hi, Invitrogen). Medium was changed at day 1 and subsequently only once weekly to minimize mechanical stress.
Transplantation
Islets were harvested after 1.5–3 weeks of culturing, washed twice in RPMI-1640 without serum, pelleted for 2 min at 50 × g, and aspirated in a silicone tubing connected to a 100-μl Hamilton syringe and gently infused under the renal capsule of the left kidney. Grafts consisted of 15,000–30,000 IE/kg unless indicated otherwise.
In two experiments dead islets were grafted. Islets were killed by 24-h storage in UWS at 4°C. As a control, killing was checked by reculturing these islets under normal conditions. After 1 day in culture no surviving islets were found. In experiments in which dead islets were transplanted mixed with normal islets, they were added to the normal graft directly before transplantation, gently mixed and pelleted, and transplanted as described above. In experiments where dead islets were transplanted separately, they were pelleted and transplanted under the capsule of the contralateral kidney.
Intraportal transplantation was performed after laparotomy. Transplantation was carried out through direct intraportal injection of 10,000–18,000 IE/kg suspended in 0.2–0.5 ml RPMI-1640 without serum.
Immunosuppression
CsA (Novartis Pharma, Arnhem, The Netherlands) was administered intramuscularly. GdCl (Sigma-Aldrich Chemie B.V., The Netherlands) was diluted in saline and administered intravenously in a dose of 20 mg/kg. Cyclophosphamide (Endoxan-Asta, Dagra Pharma B.V., Diemen, The Netherlands) was diluted in distilled water and administered intraperitoneally in a dose of 100 mg/kg on the day of transplantation.
Functional Outcome and Histology
Nonfasting blood glucose levels were determined at various intervals. Graft survival (cessation of graft function, return to hyperglycemia) was defined as the last day of two consecutive blood glucose measurements of ≥20 mM/L. Normoglycemia was defined as blood glucose level ≤11.1 mM/L. For glucose levels above the detection limit of 27.7 mM/L of the glucometer (Reflolux, Roche) a value of 30 mM/L was assumed for graphic representation. In some normoglycemic recipients the graft-bearing kidneys were removed to verify graft function by recurrence of diabetes. Kidneys from normoglycemic and hyperglycemic recipients were fixed in Bouin and stained with hematoxylin and eosin and aldehyde fuchsin/Halmi (AF, staining β-cells).
Statistics
The graft survival times of the groups were compared by one-way ANOVA using the Kruskal-Wallis test for nonparametric data with Dunn's multiple comparison test for post hoc differences between groups. Values of p < 0.05 were considered significant.
Results
Functional Outcome
Untreated diabetic control rats transplanted with long-term cultured islets showed PNF (Table 1, group 1). CsA therapy starting directly after transplantation resulted in rapidly decreasing blood glucose levels and normoglycemia on day 1 in all recipients (group 2), contrasting markedly with the blood glucose levels of the control animals (Fig. 1). Prolonged graft survival was observed in all recipients, followed by loss of graft function between 10 and 116 days in eight of nine rats. One animal remained normoglycemic and was nephrectomized on day 134 to verify graft function, which was confirmed by rapid recurrence of diabetes.
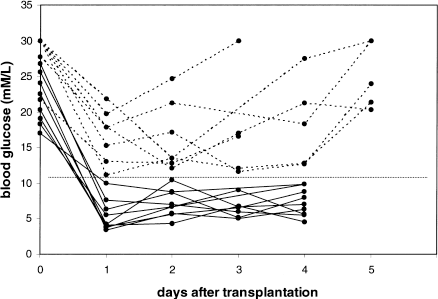
Transplantation of cultured adult porcine islets to rats. Controls and treatment with CsA: blood glucose levels directly after transplantation. The dotted lines show untreated control recipients in group 1. The solid lines show the CsA-treated recipients in group 2 (blood glucose levels after day 4 were measured only on indication). The horizontal line is drawn at 11.1 mM/L.
Graft Survival and Histology After Transplantation of Cultured Adult Porcine Islets to Rats: Controls and Treatment With CsA

Graft survival was defined as the last day of two consecutive blood glucose measurements of ≥20 mM/L. MST, median survival time. Statistical analysis showed significant difference of the medians (p < 0.0002). Survival in group 1 was significantly different from groups 2, 4, and 6 (p < 0.05) and group 8 (p < 0.01). The greater then symbol (>) indicates death or sacrifice without cessation of graft function.
CsA doses are given in mg/kg; 3× wk = Monday, Wednesday, and Friday; the last dose (15 mg, except group 4, 25 mg) is the maintenance dose.
Histological analysis was carried out on AF-stained slides. “Functioning graft” and “nonfunctioning graft” refer to animals sacrificed normoglycemic or hyperglycemic, respectively. The presence of islets and infiltrate was scored and the average per group is given semiquantitatively from - (no islets or infiltrate present) to +++ (large amount of islets or infiltrate); where appropriate the range is given in parentheses.
Recipients were sacrificed with rising blood glucose.
Three grafts were not included (rejected at days 21, 49, and 49).
Only from graft survival day 8.
The standard islet dose was mixed with 10,000–15,000 IE dead islets prior to transplantation.
10,000–15,000 IE dead islets were grafted under the capsule of the contralateral kidney.
Cessation of immunosuppression, resulting in rejection at later time point.
When the induction therapy was extended, recipients showed graft survival comparable to group 2, varying from 27 to 117 days (group 3). Increasing the maintenance dose from 15 to 25 mg/kg resulted in improved graft survival, although substantial CsA toxicity was seen (group 4).
To assess whether recipient pretreatment could improve graft survival, induction therapy was started 2 days before grafting (group 5). However, most animals in this experiment showed PNF.
Although the high purity of the islets after the isolation procedure combined with the long culture period resulted in a very “clean” graft, it could not be excluded that small rests of apoptotic or dead islets were still present or might have been formed by the physical stress of the transplantation procedure. To investigate whether the substantial spreading in the survival times seen in groups 2–4 might have been caused by varying amounts of such islet debris in the grafts, purposely killed islets were co-transplanted, either together with the graft or under the capsule of the contralateral kidney (groups 6 and 7). The graft survival times of both groups were similar and comparable to those of groups 2–4 and ranged from 21 to more than 108 days and 27 to 130 days, respectively.
To assess a possible role of the graft size on graft survival, a group was transplanted with 50,000 IE/kg, representing a 100–200% addition to the standard dose used in this study. However, no further prolongation of graft survival was seen (group 8).
Although the T-cell-directed immunosuppression used in the previous groups was highly effective in prolongation of graft survival, finally most grafts were rejected. To investigate a possible contribution of macrophages and the humoral immune system, strategies targeting these modalities were applied. The role of the macrophage was analyzed through depletion studies with GdCl. This fully prevented occurrence of PNF and resulted in normoglycemia during approximately 2–5 days (data not shown) after which cessation of graft function was observed several days later (Table 2, group 9). Starting GdCl administration 2 days before transplantation gave similar graft survival (groups 10 and 11). The combination of CsA with GdCl therapy (group 12) did not further improve graft survival compared to the previous groups with CsA monotherapy.
Graft Survival and Histology After Transplantation of Cultured Adult Porcine Islets to Rats: Macrophage Depletion, Cyclophosphamide Treatment, and Intraportal Transplantation

Graft survival was defined as the last day of two consecutive blood glucose measurements of ≥20 mM/L. MST, median survival time. Statistical analysis showed no significant difference of the medians of groups 9–11 (p > 0.06). Survival in group 12 was not significantly different from groups 2–4 and 6–8 (Table 1; p > 0.8). The greater than symbol (>) indicates death or sacrifice without cessation of graft function.
CsA doses are given in mg/kg; 3× wk = Monday, Wednesday, and Friday; the last dose (15 mg) is the maintenance dose. For details on dosing of gadolinium chloride (GdCl) and cyclophosphamide (Cyphos) see Materials and Methods section.
Histological analysis was carried out on AF-stained slides. “Functioning graft” and “nonfunctioning graft” refer to animals sacrificed normoglycemic or hyperglycemic, respectively. The presence of islets and infiltrate was scored and the average per group is given semiquantitatively from - (no islets or infiltrate present) to +++ (large amount of islets or infiltrate); where appropriate the range is given in parentheses.
Recipients were heparinized peritransplant.
To evaluate the role of the humoral system, cyclophosphamide was added to the CsA regime. The induction dose of CsA was reduced to minimize toxicity of the combined treatment. All recipients showed PNF (group 13).
Although most experimental islet transplantation studies utilize the renal subcapsular space as graft location, it is likely that in future clinical xenotransplantation the liver will be the preferred site. To explore the applicability of this route, cultured porcine islets were infused intraportally in diabetic rats, resulting in a survival of 4 days to more than 70 days (group 14).
In all groups, successfully transplanted recipients with prolonged graft survival showed remarkable improvement of their clinical condition. Polydipsia and polyuria disappeared directly after islet grafting in conjunction with achieving normoglycemic blood levels. Moreover, the body weight increased in all recipients during the normoglycemic period, sometimes with doubling of the weight in long-term survivors (Fig. 2). Conversely, return to hyperglycemia was recognized by re-appearance of these symptoms and halting of growth.

Time course of body weights of 38 rats successfully transplanted with adult porcine islets. Body weights are given from the day of induction of diabetes; day 0 represents the day of transplantation. A substantial weight gain was seen in all recipients, in the longest survivors of more than a doubling of the original weight. Generally, return to hyperglycemia was preceded by weight loss.
Histology
To evaluate the rejection process in untreated diabetic recipients, rats were transplanted and sacrificed on fixed time points (Table 3, group a). As expected, all animals remained hyperglycemic. Histology taken on both days 1 and 3 after transplantation showed plentiful islets without infiltration, neither inside nor next to the graft. In contrast, on days 5 and 6 the graft was reduced or absent and a large infiltrate present. Two immunosuppressed recipients (group b) sacrificed on day 1 and being normoglycemic showed an identical histological picture as the untreated controls on the same day. The untreated control recipients from group 1 in the experimental series presented in Table 1, although never normoglycemic, were sacrificed after two consecutive blood glucose measurements of ≥20 mM/L (day 3–6 after transplantation). Also in this group histology showed only remnants of the graft and substantial infiltrates.
Histology of Controls, at Different Time Points, After Transplantation of Cultured Adult Porcine Islets to Rats

Diabetic recipient rats were transplanted with porcine islets under the renal capsule and did not receive any immunosuppression, except group b, which received the standard starting dose of CsA on day 0 (40 mg/kg). Animals were sacrificed for histology at days 1, 3, and 5–6. Histological analysis was carried out on AF-stained slides. The presence of islets and infiltrate was scored and the average per group is given semiquantitatively from - (no islets or infiltrate present) to +++ (large amount of islets or infiltrate).
Where histology was taken in the experimental groups with the graft still functioning, a large amount of islets was always found with no infiltration in most cases (Tables 1 and 2, groups 2, 4, 6, 7, 12). In the histology of the nonfunctioning grafts, a remarkable dichotomy was observed. On the one hand, the grafts that had failed rapidly (within 2 weeks) and that mostly had extensive infiltrates with few or no islets left (groups 1, 5, 9–11, 13). On the other hand, the relatively long functioning grafts (groups 2–4, 6–8, 12). Here in most groups infiltration was also seen; however, often less extensive and in all groups islets were found, sometimes considerable numbers. Especially in recipients with function loss after 50 days, a high percentage of the animals still showed substantial graft tissue: 9 of 11 of such recipients had an islet score of + (3×), ++ (4×) or +++ (2×) and 4 of these 11 recipients had no infiltration (individual data not shown).
Although return to hyperglycemia is probably caused by rejection, especially in the early failing grafts, other mechanisms may be involved as well. This is clearly illustrated in two identically treated recipients from group 4 (Fig. 3). One of these animals was scored hyperglycemic on day 8 and sacrificed; the other animal had an almost identical course but did not become formally hyperglycemic and was pursued further. After subsequent decline of the blood glucose to normoglycemic levels it was sacrificed on day 71 with a last recorded glucose of 5.4 mM/L. Remarkably, histology of both grafts showed a similar picture in that an intact graft was seen, almost without infiltration. However, the islets of the early hyperglycemic recipient did not stain with AF, in contrast to the long-surviving graft, which demonstrated strongly AF-positive islets.
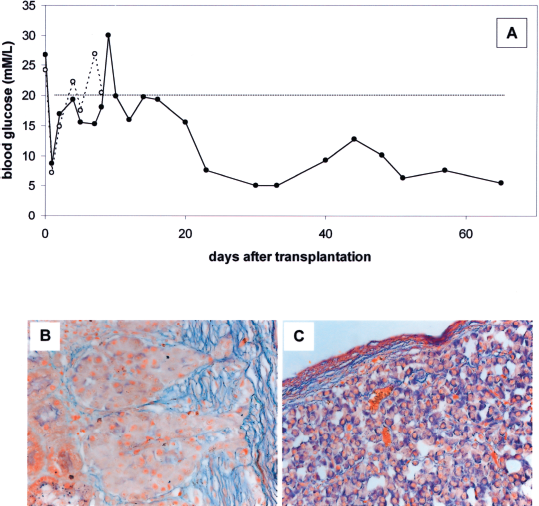
Transplantation of cultured adult porcine islets to rats. Cessation of graft function and long-term graft survival in two recipients with identical treatment. Functional outcome and histology of two recipients from group 4. (A) Blood glucose levels. One recipient had returned to hyperglycemia on day 8 after two consecutive blood glucose measurements of ≥20 mM/L (dotted line). The second one followed a similar course, but just “missed” the criterion for cessation of graft function, returned to normoglycemic levels, and was sacrificed on day 71 with a normal blood glucose (solid line). (B) Histology (AF staining) of the nonfunctioning graft on day 8, showing intact islets, not staining with AF. (C) Histology (AF staining) of the functioning graft on day 71 (last blood glucose recorded on day 65), showing intact islets, strongly staining with AF. Magnification: (B,C) 40×.
Throughout all experimental groups it was noticed that when infiltration was present at the graft site, infiltration inside the islets was never observed. The observation in several recipients of a single small infiltrate, directly apposed to the graft, suggests that rejection might originate from a singular event. Subsequently, expansion takes place and the infiltrate spreads along the borders of the graft and destroys it gradually from the periphery (Fig. 4).

Transplantation of cultured adult porcine islets to rats. Histology (AF staining) of islet grafts and infiltration in rats treated with CsA, taken on the time point of cessation of graft function. isl, islets; inf, infiltrate. (A) Islet graft (not staining with AF) with a very small focal infiltrate between graft and kidney parenchyma (arrow). (B) Islet graft with an infiltrate located subcapsularly next to the graft and clearly separated from the islets. No infiltration inside the islet graft. (C) Two uninfiltrated islet graft rests in a massive infiltrate. Magnification: 10×.
Discussion
A series of factors of potential relevance for the successful transplantation of porcine islets in the stringent pig-to-rat model was assessed in this study. The quality of the graft is generally considered of crucial importance in islet transplantation. In a recent study we optimized the conditions to obtain a high-quality islet graft (26). Adult porcine islets cultured for 1–2 weeks proved superior in establishing long-term and stable graft function in diabetic nude mice.
In the present study these long-term cultured islets were tested in diabetic rat recipients. In the control group without immunosuppression all animals showed PNF. This was also observed in animals sacrificed at defined time points for histology (Table 3, groups a and b). Application of CsA monotherapy proved very effective; PNF was completely overcome and already on day 1 all animals were normoglycemic. Graft survival varied from 10 to well over 100 days.
It is not formally proven that the extended graft survival in this model is due to the long-term culture of the islets. However, the results in our earlier studies clearly demonstrated that freshly isolated islets are not able to give adequate function in a nonimmunogenic model (26). Also two pilot transplantations were carried out with freshly isolated islets transplanted to rat recipients with identical CsA schedule, both resulting in PNF (data not shown). Three additional transplantations were performed under even more severe immunosuppression of 10 Gray total body irradiation, also without any graft function (data not shown).
Experiments to investigate if the early graft loss could be overcome by changes in the CsA induction dosing and schedule failed. In contrast, intensifying the maintenance dose largely eliminated early function loss, albeit at the cost of significant toxicity. Together these data demonstrate that substantial prolongation of graft survival can be obtained in this model when islets of defined quality are used together with CsA immunosuppression. With exception of the mouse as a recipient, it has historically appeared very difficult to achieve reasonable graft survival in discordant islet xenotransplantation models (4, 5, 19, 28, 29, 34) and clinical xenotransplantation (12). Undoubtedly, the problems in obtaining a good quality graft, especially of porcine islets, have in many instances contributed substantially. Apart from a series of sparsely documented studies (13, 20, 30), two recent articles reported successful transplantation of adult porcine islets in the rat (21, 32). In both studies the graft survivals were alike and remarkably similar to the present study. Although several variables were identical to the present study, there were also some important differences. Culture was only for 1 day and there were major differences in the immunosuppression used. In the present study high-dose CsA was given, whereas in the studies mentioned above CsA was used in combination with mycophenolate mofetil (MMF) + leflunomide (32) or FTY720 (21). Although different, both are very intense and similarly successful in overcoming early rejection. However, maintenance therapies were strikingly different, in that the Swedish studies continued the “induction” therapy, whereas in the present study the CsA dose was reduced substantially. Recalculated on a per day basis, rats in the present study received ±6.5 mg/kg/day, which is less than half of the dose in the Swedish studies (15 mg/kg/day) in which additionally substantial dosages of MMF and leflunomide (32) or FTY720 (21) were given daily.
It is not immediately clear how these large differences in maintenance therapy yielded similar long-term graft survivals. An important factor might be the condition of the graft. In the reported studies, islets were cultured for only 1 day and it was indicated in one of these studies that in vitro function was not optimal at that moment and clearly better on day 8 (29, 32), suggesting that prolonged culturing might have improved their results, which would be in line with our earlier observations (26). Also a recent article showed very limited graft function of a high dose of noncultured adult porcine islets transplanted to the nude rat (7). In a different study a very high dose of 10,000 IE/rat (80,000–100,000 IE/kg) was transplanted to untreated nude rats (3). Yet only 7 out of 11 animals were still normoglycemic on day 16. Remarkably, both Swedish studies and the present one showed strong similarity in their inexplicably wide range of graft survival times. To study whether the relatively short survival times seen in some of these recipients were caused by a possibly immunogenic role of islets damaged during the transplantation procedure, killed islets were added to the graft, which is normally largely devoid of remnants after the long-term culturing. However, no effect on graft survival was found.
It was also assessed if the size of the graft was of importance for the survival time. In pilot studies graft sizes of 10,000–15,000 IE/kg gave a clearly lower graft survival time compared to higher doses, which is in line with earlier reports (29, 32), and led to our standard dose of 15,000–30,000 IE/kg. However, transplanting an excess of islets (50,000 IE/kg) did not enhance graft survival.
It might be interesting to compare these data with experiments in a skin transplantation model in the same species combination (2). Porcine skin grafts were transplanted to rats immunosuppressed with CsA monotherapy. Interestingly, both induction dose (25 and 15 mg/kg) and maintenance dose (7.5 mg/kg 3 times/week) were significantly lower compared to the present study. Yet all grafts survived long term, notwithstanding that skin grafts are generally considered highly immunogenic. Probably the substantially larger size of the skin graft (at least 5–10 times larger) compared to the islet graft and the capacity to grow and recover from damage are causal. Lastly, islets might be more sensitive to the noxious effects of mediators generated through ischemic and immunologic events. This suggests that additional improvement might be obtained by further modification of the graft or the transplantation conditions.
Although no functional studies were carried out, the impressive weight gain demonstrates that porcine islets can sustain growth in such distantly related recipients. This is in marked contrast to previous reports, in which no weight gain was found (7, 20, 31–33) or even substantial weight loss was seen (31, 33).
Histological analysis revealed some intriguing findings. As expected, in the untreated control animals, sacrificed when hyperglycemic, almost no graft could be detected and large mononuclear infiltrates were present. However, in the untreated recipients sacrificed on specific time points, no infiltrates were found either on day 1 or day 3 after grafting. Still PNF occurred in all five animals, suggesting the deleterious effects of toxic mediators acting over substantial distance instead of by direct cell–cell interaction. Likely candidates such as IL-1, TNF, and nitric oxide (NO) have been suggested in the literature (16). These results are remarkably similar to those of Tze et al. (29), who also mention relatively late appearance of lymphocytes (day 3) and conclude that direct contact between host lymphoid cells and the graft was apparently not required for killing.
Although PNF has been associated with poor graft quality, this is not the case in the present study because CsA fully prevents PNF. PNF has been reported to occur both after grafting to the renal subcapsular site and intraportally (3, 16, 24, 29). Reported percentages of PNF in islet xenotransplantation vary widely, sometimes even within a model (pig-to-rat) in one institute: 0% (13), 54% (25), and 90% (24). PNF is reported as the prevailing outcome in untreated discordant islet xenotransplantation with exception of the mouse as a recipient (19); however, recent studies in the pig-to-rat and monkey showed no PNF in control animals (17, 32). The effective inhibition of PNF by GdCl in the present study emphasizes the role of macrophages and confirms similar findings in other islet xenotransplantation models (1, 6, 9, 14). The efficacy of CsA in the suppression of PNF in this study is remarkable also after intraportal transplantationm which generally results in PNF in the nude rat (3, 18), and is most likely caused by mediators produced by macrophages and can be prevented by NO inhibitors (3, 16). These data strongly point out the macrophage as the primary cell responsible for graft destruction, and support earlier observations that CsA can act directly on macrophages (8, 15).
B-cell-directed immunosuppression is the mainstay of organ xenotransplantation with cyclophosphamide as one of the most effective agents. However, its usefulness in nonvascularized xenografting is less unambiguous and has been tested only in a few discordant islet xenotransplantation studies (22, 32). In the present study no clear-cut effect of cyclophosphamide was seen, suggesting no prominent role for the humoral immune system.
The notion that T cells and macrophages are crucial in islet xenograft destruction in this model confirms previous observations (10, 27, 35). However, it is not entirely clear how these cells actually attack and destroy the graft. In contrast to typical organ graft rejection, islet grafts in the present study never contained focal infiltrates originating from the blood vessels. Graft destruction always took place from the periphery of the graft, the infiltrate progressing into the graft, in a more final stage resulting in graft rests (still uninfiltrated) as islands in a massive infiltrate. Although rejection of islets is mostly described as infiltration, suggesting mechanisms similar to those in organ graft rejection, it is not unlikely that mechanisms are operational like the ones described in the present study (32). One study explicitly mentions that no lymphoid cells were detected within such islets (29) and a second study reports a sharp border between the infiltrate and the porcine xenograft during the entire process of rejection (31).
In contrast to reports where well-functioning islet grafts were seen closely associated with large infiltrates, considered to play a role in maintaining the tolerogenic state (11, 23), in the present study all histologically documented cases with good graft function showed no or only minimal infiltration. It is unclear whether these differences might be explained by the nontolerogenic nature of our immunosuppression, as illustrated by the immediate rejection of all grafts in long-term survivors in which immunosuppression was stopped (data not shown).
At variance with these observations, some recipients of which graft function had stopped still contained a substantial amount of graft tissue, sometimes with relatively little infiltration, a finding also reported by the Swedish group (32). In several of these cases AF staining was considerably reduced or even absent, indicating hyperfunction of the β-cells (illustrated in Fig. 3). Here the recipient sacrificed because of cessation of graft function showed virtually no infiltrate and a normal amount of graft tissue, albeit without AF staining, probably caused by hyperfunction due to insufficient capacity. The other recipient was sacrificed much later while normoglycemic and showed plentiful AF-positive islets without infiltrate. Together these data suggest a temporary functional deficit probably caused by loss of damaged graft tissue or due to the rapid weight gain of the growing recipients and confirms the regenerative capacity of adult porcine islets (26).
It can be concluded that long-term cultured adult porcine islets are able to cure diabetic rats, requiring substantially lower amounts of immunosuppression compared to short-term cultured islets. Recipients lose the symptoms of diabetes and show impressive weight gain. Eventual rejection in this model is probably macrophage and T-cell mediated and different in mode of attack compared to organ graft rejection.
Footnotes
Acknowledgment
The authors wish to thank Mr. K. G. van der Ham for preparing the figures.
