Abstract
In this study, we examined the effects of systemic administration of rat or human bone marrow stromal stem cells (MSC) at early and later times following middle cerebral artery occlusion (MCAO) on blood cytokines/growth factors, brain glia, and motor behavior in rats. Rats were tail vein injected with rat (r) and human (h) MSCs at 1 or 7 days post-MCAO. In some rats (N = 4) MSCs isolated from transgenic GFP rats were used to track the migration of cells peripherally and centrally at 2.5 and 28 days. Motor behavior was assessed using the modified Neurological Severity Score/climbing test at various time points before and after MCAO and transplantation. Prior to sacrifice at 1, 7, or 28 days post-MCAO, blood serum was collected for cytokine array analysis. Brains were analyzed for markers of activated microglia (CD11) and reactive astrocytes (GFAP). Administration of either allogeneic (rMSCs) or xenogeneic (hMSCs) stem cells produced a significant recovery of motor behavior after MCAO, with cells delivered at 1 day having greater effect than those at 7 days. Correlated with recovery was an amplification in activated microglia, reactive astrocytes, and new blood vessels in the infarct region, resulting in greater preservation in brain integrity. Concomitantly, expression of blood cytokines/chemokines (IL-13, MMP2, MIP) and growth factors/receptors (VEGF, neuropilin, EPOR, TROY, NGFR, RAGE) were modified following MSC administration. Because only rare GFP-labeled MSCs were observed in the brain, these effects did not depend on the central incorporation of stem cells. The early systemic administration of allogeneic or xenogeneic MSCs soon after experimental stroke produces a structural/functional recovery in the brain which is correlated with an increase in activated brain glia and changes in circulating cytokines and growth factors. Stem cells therefore induce an important neuroprotective and/or regenerative response in the host organism.
Keywords
Introduction
Stroke remains the leading cause of disability and the third leading cause of death in the US. Although there has been a decline in the stroke-associated mortality rates in the last decade, its overall incidence continues to climb as does the burden on society (48). While great strides have been made in antithrombosis treatment, it is estimated that relatively few (<5%) ischemic patients benefit from tissue plasminogen activator (16,30). Therefore, expanding our arsenal of ways to treat stroke remains a major priority.
The notion that stem cells might ameliorate the devastating effects of ischemic stroke or repair the brain damage that accompanies it has opened a new and important frontier in stroke research. Indeed, a variety of stem cell types have been tested in experimental stroke models, most resulting in functional improvement (3,35,55). The vast majority of studies have utilized either bone marrow stromal cells (MSCs) (6,17,26,28) or umbilical cord blood cells (5,32,33,37). Irrespective of their means of delivery (intraparenchymal, intraventricular, or IV), most studies have reported improved function after ischemic stroke despite the near total absence of surviving transplant cells in the infarcted brain (27,28,49). Thus, transplanted MSCs, while short-lived, have long-term consequences for brain recovery. Moreover, a recent clinical trial has further established that MSCs can be safely transplanted into the brains of stroke patients (42). Although this study was small and not powered for efficacy, many patients experienced improved motor function.
Amazingly, we know little about the mechanisms through which stem cells mediate this recovery of function. Thus far, most evidence suggests that stem cells work indirectly to reduce apoptosis in the stroke injured brain (15). In almost all studies, the injection of stem cells was accompanied by a reduction in infarct size, mediated possibly through the enhanced production of growth factors such as brain-derived neurotrophic factor (BDNF), fibroblast growth factor (FGF), etc. (21,24,29). In addition to these neuroprotective mechanisms, stem cell treatment appeared to augment the host brain repair response by increasing local synaptogenesis (7,8,14,41) and promoting the recruitment of endogenous progenitor cells from the subventricular zone to the injury, although the functional significance of the latter has yet to be determined (1,19,20,33,46,56). In addition, most studies of stem cell transplants noted an increase in angiogenesis and vasculogenesis in the area surrounding the infarct, which was associated with neurological recovery after stroke (11,13,15,18,34,39,40,46).
Finally, and most intriguingly, is the possibility that systemically delivered stem cells mediate their effects by modifying the inflammatory and immune response. While the inflammatory/immune reaction of the brain to ischemic stroke injury has been well described (22), little is yet known about how stem cells modulate that response. There is a growing literature suggesting that stem cell transplants can attenuate the inflammatory response while inhibiting T-cell activation (38,47,50). How these cells and their products (cytokines, chemokines, growth factors) contribute to the recovery of function seen after stem cell transplantation remains one of the most important puzzles to be solved.
In this study, we systematically compare the efficacy of rat and human MSCs administered at various times after experimental stroke. In addition, we examine host brain and blood for the cellular and molecular correlates that accompany the improved behavior observed after MSC administration. These studies use a well-established rat model of experimental stroke in which focal ischemia is caused by transient occlusion of the middle cerebral artery (MCAO) (31). We will show that the early systemic administration of rat or human MSCs soon after experimental stroke produces a structural/functional recovery in the brain, which is correlated with an increase in activated brain microglia and astrocytes and changes in circulating cytokines/growth factors, consistent with the notion that stem cells induce a neuroprotective and/or neuroregenerative response in the host organism.
Materials and Methods
Animals and MCAO Lesion
All experimental procedures were approved by the Thomas Jefferson University Institutional Animal Care and Use Committee. Male Sprague-Dawley rats (Harlan) 300–325 g were subjected to MCAO lesions. Using methods described previously (31), anesthetized (sodium pentobarbital, 50 mg/kg, IP) rats were subjected to transient intraluminal occlusion of the MCA. Briefly, the common carotid (CCA) and external and internal carotid arteries (ECA, ICA) were exposed through a 2-cm incision in the neck on the right side. The proximal CCA and ECA were ligated. A microvascular clip was used to temporarily occlude the pterygopalatine artery. A 4.0 surgical nylon suture was inserted into the lumen of the CCA through a small hole and advanced until the tip had gone 18 mm beyond the bifurcation, at which point the tip occluded the MCA origin. Two hours later, the suture was removed for reperfusion of the brain.
Tissue Culture
Rat MSCs were harvested from SD rats (Harlan) or GFP (green fluorescent protein) transgenic SD rats [SD-Tg(GFP)Bal] from the NIH-funded Rat Resource and Research Center (RRRC) University of Missouri, Columbia. In brief, bone marrow cells were flushed from the shaft of the femur with PBS. Cells were then plated into medium containing 15% fetal calf serum (FCS), 1% nonessential amino acids, 1% glutamine, 1% pen-strep in DMEM. Cells were passaged 6–8 times before use in transplantation studies. In addition, hMSCs from the hMPC 32 F line (9) were grown in culture as described previously (44,45).
Transplantation Procedures
Rat or human MSCs were harvested from culture, centrifuged (1000 rpm) and the pellet resuspended in sterilized PBS. Cells were injected into the rat tail vein, at a concentration (5 × 105 in 0.5 ml) previously shown to be effective (10,11). Rats receiving hMSC transplants were treated with the immunosuppressive agent cyclosporin A (10 mg/kg, IP) daily, beginning the day before transplantation.
Behavioral Tests
To evaluate neurological function before and after stem cell transplant, rats were subjected to a battery of tests before and after MCAO and transplantation. Motor deficits were evaluated using a modified Neurological Severity Score (mNSS) (10,43), which includes fore and hind limb flexion, walking, righting, placing, balance, body resistance, paralysis. Using this scale, one point was given for the inability to perform a test. Consequently, the higher the score, the more severe the motor deficit (maximum score of 8; eliminating tests used in the original NSS that do not show consistent variation after stroke). In addition, motor function was assessed using a climbing test, wherein rats were placed in a Plexiglass rotometer and the number of touches of each forepaw to the sidewalls in 15 min was counted. Behavior was assessed at regular intervals as described in the Results section.
Immunocytochemistry
Transplanted rat brains were perfused with 4% cold (4°C) periodate-lysine-paraformaldehyde, sectioned at 30 μm on a freezing microtome, and processed for immunocytochemistry using the immunofluorescence staining method described previously (54). Cultures or sections through brain, spleen, liver, and heart were stained with one or more of the following antibodies: mouse anti-human nuclear antigen (HNA) from Millipore at 1: 40; rabbit antibodies to glial fibrillary acidic protein (GFAP), 1:1000; monoclonal mouse antibodies to CD11, 1:25; and Chemicon chicken antibodies to GFP, 1:200. All secondary antibodies were Alexa Fluor antibodies from Invitrogen used at 1:200–1:300. Cultures or sections were analyzed using a Nikon-Scanalytics Image System or an Olympus IX81 Image System.
TTC Staining Method
Following sacrifice, the fresh (unfixed) brain was removed from the skull, washed in cold PBS, and placed in a brain mold. Coronal sections 1 mm in thickness were cut through the cerebrum and placed in 1% 2,3,5-triphenyltetrazolium chloride (TTC; Sigma) for 30 min in a 3°C incubator. TTC solution was drained and sections were immersed in 4% formalin in 0.1 m PB at 4°C and stained as described previously (2).
Cytokine Array
Blood (0.2–0.4 ml) was collected from the tail vein of rats before MCAO, 1 day after MCAO surgery (prior to stem cell transplantation), and 1 week and 4 weeks after transplantation of rMSC or PBS into the ischemic MCAO rat. Samples were spun at 2000 rpm (10 min room temperature, clear serum removed and stored at −80°C for further analysis). Blood serum (80 μ l) from duplicate samples was then analyzed by Raybiotech using a Rat L-series 50 cytokine custom array. Background (blank) levels were subtracted, and specific cytokine expression was quantified by densitometry, averaged, and expressed in OD units relative to one another.
Statistical Analysis
Data were statistically analyzed by one-way analysis of variance (ANOVA). When p < 0.05, then the F-test was followed by the two-tailed Student's t-test to compare the statistical significance between control and experimental groups (ANOVA with subsequent post-hoc analysis using Bonferroni correction). Differences were considered significant only when the value was p < 0.05.
Results
The MCAO Model of Experimental Stroke
Rats were subjected to experimental stroke using an established published protocol (31) described in Materials and Methods. In brief, rats were subjected to a transient (2-h) intraluminal occlusion of the middle cerebral artery that produced a reproducible ischemic lesion of the striatum and cortex on the ipsilateral side. Contralateral motor function was assessed before and after MCAO using a modified neurological severity (mNSS) scale (12). As shown in Figure 1A, the magnitude of functional decline after MCAO varies in relative proportion to the size of the infarct, with severe ischemia producing a score equal to or greater than 6 using the mNSS.
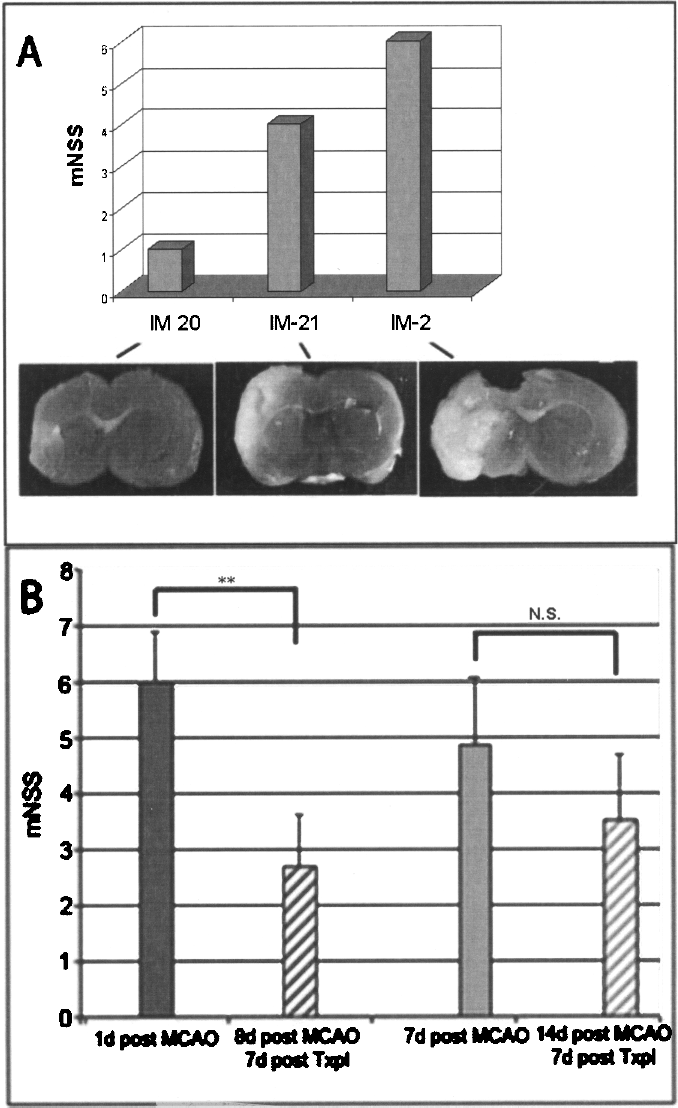
Functional deficits after middle cerebral artery occlusion (MCAO) are proportional to the size of the infarct and change with systemic administration of bone marrow stromal cells (MSCs). Shown in (A) are the individual modified neurological severity score (mNSS) scores for three MCAO rats (IM-2, IM-20, IM-21) and representative slices through the corresponding brains following staining with 2,3,5-triphenyltetrazolium chloride (TTC) (see Materials and Methods). Note the size of the infarcted tissue (white tissue) varies in relative proportion with behavioral deficits. Shown in (B) are motor behavior assessments before and after systemic administration of MSCs in the MCAO rat. Tail vein injections of hMSCs (N = 14) (shown) or rMSCs (N = 14) (data not shown) were administered to half of each experimental group either 1 day after MCAO or 7 days after MCAO; 7 days posttransplant (Txpl) motor behavior was assessed again using the mNSS scale. **p < 0.001, a comparison of scores at 1 day post-MCAO with scores 1 week later after hMSC transplantation.
Peripheral Administration of MSCs Into the MCAO Rat
In order to assess the effects of systemic injection of MSCs after experimental stroke and determine whether the timing and source of stem cell administration is critical to their effects, rats with moderate to severe infarcts (≥ 6 on the modified NSS scale) were with hMSCs or rMSCs administered via tail vein injection at 1 day or 7 days following MCAO. We found a significant improvement in behavior, as represented by a reduction in the mNSS score 7 days later (Fig. 1B). Moreover, both hMSCs and rMSCs administered at 1 day post-MCAO produced a more significant (p < 0.001) recovery than those administered at 7 days post-MCAO in which mNSS scores declined but did not reach significance.
When rats were sacrificed 4 weeks later and formalin-fixed brains examined at the gross level, we noted clear differences in the vascularization of the brain of rMSC, hMSC, and PBS transplanted rats (Fig. 2A), as others had seen previously (11,18,39,40,46). Despite the enormous damage that often accompanied MCAO injury, in rats with peripheral rMSC or hMSC transplants there was a marked increase in the number of vessels in the region of the infarct as compared to control (PBS-injected) rats. Because cardiac perfusion did not remove the contents of these blood vessels as elsewhere in the brain, it suggests that these may have been new vessels, yet unconnected to the systemic circulation.

Gross and histological examination of the brain in MCAO rats with MSC transplants. Inspection of the formalin-perfused brain 4 weeks after tail vein injection of rMSCs (N = 4), hMSCs (N = 5), or PBS (N = 8) 1 day post-MCAO revealed new blood vessels (arrow) in the area surrounding the infarct of stem cell recipients (A). Further histological examination of the brain after immunocytochemical localization of GFAP (green) and CD11 (red) revealed a reduction in the size of the infarcted area (circumscribed and labeled infarct) and greater brain integrity in rats adminstered hMSCs (C) than in PBS controls (B). As with other indices, both rMSCs (not shown) and hMSC administration at 1 day (C) had greater effect than at 7 days (D) post-MCAO. Scale bar (B–D): 250 μm.
Concomitantly, in the majority of cases (3 of 4 hMSC and 3 of 4 rMSC grafts), there was a greater preservation of tissue integrity (and a reduction in the size of the infarct) observed when MCAO rats were injected with MSCs compared to control (note the reduction in tissue damage in Fig. 2C of MSC-injected rats compared with the large circumscribed area of infarct damage in PBS-injected rats in Fig. 2B). As with behavior, MSC treatment at 1 day postinfarct (Fig. 2C) was more effective in this regard than transplants at 7 days postinfarct (Fig. 2D). Interestingly, xenogenic hMSCs injections in immunosuppressed rats produced results similar to those caused by allogeneic rMSCs (data not shown).
We next compared the long term changes in behavior in MCAO rats treated 1 day postinfarct with peripheral rMSC administration. Even in those rats with a PBS injection, the severe deficits in motor behavior caused by MCAO ameliorated somewhat over 28 days, as evidenced by a decline in the mNSS score in all rats (Fig. 3A). However, in rats that received rMSCs, we observed a greater and more sustained improvement in mNSS scores (Fig. 3A) at 28 days post-MCAO. Likewise, while MCAO rats exhibit compromised climbing behavior with both the affected (left) and the nonaffected (right) forelimbs (Fig 3B), only rats that received rMSCs (not PBS) showed significant recovery in climbing function 14 days later. Although performance with both limbs improved in these animals, it should be noted that the lesion-affected side always lagged behind the contralateral side.
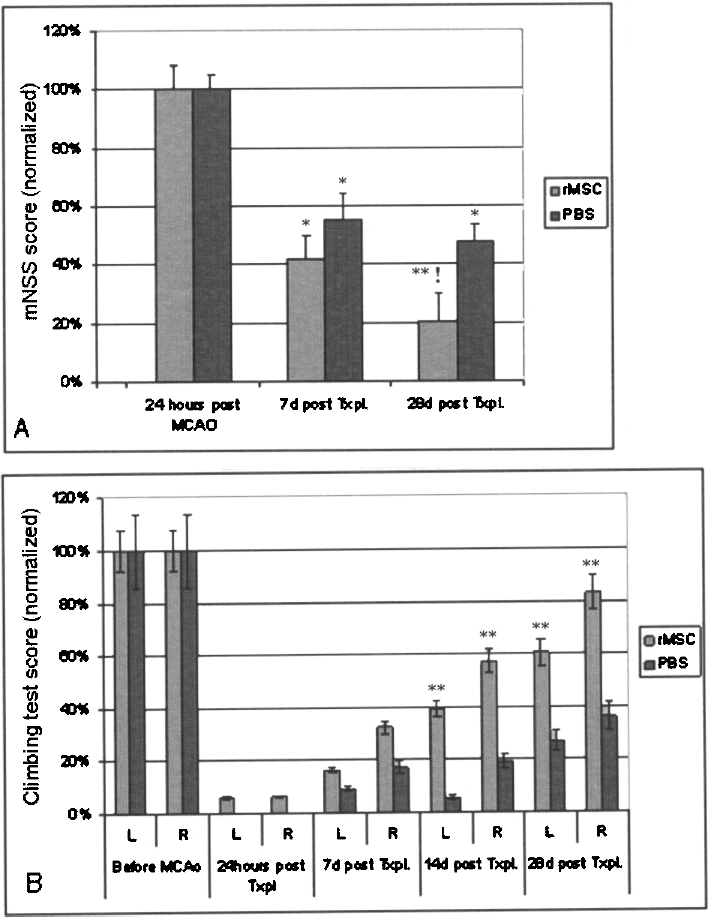
Evaluation of motor behavior in MCAO rats with PBS and rMSC transplants. Rats were administered rMSCs (N = 10) or PBS (N = 8) 1 day following MCAO and assessed for changes in NSS scores (A) and climbing test scores (B). (A) mNSS scores are normalized such that the score at 24 h after MCAO is 100%, and decreasing values represent an improvement of function. *p < 0.01 and **p < 0.001 compared to scores at 24 h post-MCAO. !p < 0.05 compared to PBS injected rats at 28 days post-MCAO. (B) Climbing scores are normalized such that the pre-MCAO score is 100%, and increasing values represent improvement of function. **p < 0.001 compared to climbing scores after MCAO.
To determine whether the observed changes were due to the direct effects of stem cell incorporation into the brain, GFP-labeled rMSCs were injected into the tail vein and tracked in the brain and peripheral organs at 2.5, 7, and 28 days later. At 2.5 days, we found only rare GFP-labeled cells (<0.01%) in the peri-infarct region of the brain and scattered labeled cells in the spleen but not other organs (data not shown). By 7 or 28 days posttransplantation, no GFP+ cells were present in the brain or in peripheral organs (heart, liver, spleen) (data not shown), suggesting that IV injected cells do not take up long-term residence in host tissues despite their long-lasting effects on tissue integrity, vascularization, and behavior in the brain.
Host Response to Systemic Administration of MSCs After MCAO
We next explored the cellular responses that ensue in the brain following peripheral injection of MSCs. Rats were administered rMSCs 24 h after MCAO, sacrificed various times later, and the fixed frozen brains sectioned and stained for markers of reactive astrocytes (GFAP) and activated microglia (CD11). Although MCAO alone results in an increase in these indices, we found marked differences in those animals also administered IV rMSCs. In all cases, we observed a striking number of reactive GFAP+ astrocytes in rMSC-treated versus PBS-treated rats (Fig. 4). Moreover, by 4 weeks postinjection, the infarcted area was bounded by abundant GFAP+ reactive astrocytes in stem cell recipients (Fig. 4). In contrast, in MCAO brains without stem cells astroglia assumed a more tortuous morphology typical of scar formation. In addition, brains were stained for the microglial marker CD11. As with astrocytes, there was a marked increase in the number and the activation of CD11+ cells following MSC administration (Fig. 5). Importantly, as with other parameters of recovery (behavior, tissue integrity), the microglial response was greater in rats that had received MSCs 1 day as compared to 7 days after MCAO and endured for many weeks (4 weeks) longer than that seen after MCAO alone, suggesting a possible neuroprotective/regenerative role for these cells.

Immunocytochemical analysis of GFAP+ reactive astrocytes in the MCAO rat brain 4 weeks after rMSC injection. Note the expanded area of GFAP+ reactive astocytes adjacent to the infarct in MSC-injected (A) versus PBS-injected (B) MCAO rats where large hypertrophic astrocytes appear to form a scar (N = 6). Higher power view of white rectangular areas in (A) and (B) are shown in (C) and (D) and their respective insets, demonstrating the difference in the morphology of the reactive astrocytes in the presence of stem cells. Scale bar (A, B): 250 μm; (C, D): 50 μm; inset: 25 μm.

Immunocytochemical localization of CD11 in the MCAO rat 10 days after transplantation of hMSCs. Note that some activated CD11+ microglia are present in MCAO rats without transplants (A) but that there are distinctly more activated cells in rats that received a hMSC transplant 1 day (B) or at 7 days (C) after MCAO (NM = 6 in each group). Importantly, activated microglia remain long after the usual inflammatory response caused experimental stroke. Scale bar (A–): 50 μm.
In order to investigate the systemic changes that accompany the brain's response to MSC transplant, we examined blood levels of 50 cytokines/chemokines/growth factors of potential importance by Cytokine Array Analysis (Raybiotech Rat L-series) in rats with MCAO lesions and transplants, as shown in Figure 6. Because the array used rat-specific antibodies, cytokines released by any residual transplanted human MSCs were not detected. Instead, only host-derived factors were measured. Blood was collected from the tail vein of rats before MCAO, 1 day after MCAO surgery, and 1 and 4 weeks after transplantation of rMSC or PBS injections immediately following behavioral evaluation of the rats. Each sample (80 μl serum) was analyzed and changes in factor expression were quantified by densitometry. A dynamic picture emerged as some cytokines and growth factors [i.e., cytokine IL-13, vascular endothelial growth factor (VEGF) and its receptor neuropilin], which declined after MCAO and PBS injection rebounded after rMSC injection (Fig. 7). Interestingly, these factors continued to be expressed at elevated levels even 4 weeks later. Other cytokines and chemokines such as macrophage inflammatory protein 1 (MIP) and matrix metalloproteinase (MMP2) and cytokine receptors such as erythropoietin receptor (EPOR) and tumor necrosis factor receptor (TROY) were increased after rMSC injection even though there was no immediate response following ischemic injury (Fig. 7). In addition, certain growth factor receptors [i.e., nerve growth factor receptor (p75 NGFR) and the receptor for advanced end products (RAGE)] were increased in response to MCAO and were subsequently maintained at higher levels after rMSC but not PBS injection (Fig. 7).

Timeline for experimental procedures (MCAO, transplant, behavioral assessment, cytokine array analysis) in rats with MCAO lesions and transplants.

Systemic cytokine levels in MCAO rats with and without peripheral stem cell transplantation. Blood was drawn from the tail vein of intact rats (prior to surgery), 24 h after MCAO (prior to stem cell transplantation), and at 1 and 4 weeks following systemic injection of PBS (sham) or rMSCs and analyzed by cytokine array after sacrifice at 4 weeks. Values were quantified by densitometry (RayBio) and expressed in optical density units after subtraction of background expression. Relative levels are shown for the cytokines Neuropilin2, Il-13, and vascular endothelial growth factor (VEGF), matrix metalloproteinase (MMP2), the chemokine macrophage inflammatory protein 1 (MIP), cytokine receptors erythropoietin receptor (EPOR) and tumor necrosis factor receptor (TROY), and growth factor receptors nerve growth factor receptor (NGFR) and receptor for advanced glycation end products (RAGE).
Discussion
This study demonstrates that the peripheral administration of MSCs following occlusion of the middle cerebral artery produces profound changes in the host animal, resulting in preserved brain structure and long-lasting improvement in motor function. Importantly, the timing of stem cell treatment was critical to the magnitude of the observed effects, with MSCs administered soon (1 day) after the ischemic event producing greater recovery in function than treatment 1 week later. Because systemically injected GFP-labeled MSCs did not take up residence in the brain, it suggests that their beneficial effects occurred secondary to the modification of other host cells/factors. Consistent with this notion, we observed a long-lasting amplification in activated microglia, reactive astrocytes, and blood vessel formation in the region surrounding the infarct in rats receiving MSCs compared to PBS-injected MCAO rats. Concomitantly, blood levels of specific cytokines/chemokines and growth factors/receptors were significantly modified by stem cell administration in a manner consistent with a neuroprotective and/or regenerative host response. That both rMSCs and hMSCs stem cells were equally effective in this regard validates the continued study of xenotransplanted hMSCs in an MCAO rat as needed for the development of therapies suitable for the treatment of stroke patients.
The mechanism through which MSCs achieve these remarkable effects remains elusive. Although a number of studies have reported the presence of a small fraction of surviving MSCs in the MCAO rat brain weeks after their systemic administration (11,13,26,28,34), as in our study, others have not observed the central incorporation of these cells (49,52,53). Whether cell labeling methods (BrdU, LacZ, GFP), cell concentration (5 × 105–3 z 106), or some other methodological differences account for this discrepancy remains unclear. However, the fact that even MSCs delivered directly into the brain parenchyma only rarely express neuronal markers (4,24,45) suggests that stem cells mediate their effects via secondary changes in the host rather than through direct neuronal replacement.
While the cascade of effects, particularly inflammatory/immune effects, caused by experimental stroke has been well documented (3), until now there has been little exploration into the ways in which stem cell transplantation modifies those events. We found that MSC administration into MCAO rats causes an amplification in activated CD11+ microglia and reactive GFAP+ astrocytes in the peri-infarct area far greater and more long lasting than that seen after stroke alone. Normally, following stroke or other traumatic brain damage, microglia become transiently activated, but return to a quiescent state within days of injury (23). Likewise, soon after the initial damage of brain tissue, reactive astrocytes participate in the formation of a scar (22,23). In stem cell-treated MCAO rats, the response of these cells and the time course of their activation are quite distinct. Even at 4 weeks and beyond following stem cell treatment, microglia and astroglia remain in an activated state that has previously been associated with a regenerative response (22,25). Although the microglial and astroglial response may be unrelated to the regenerative process, it is possible that activated glia play some role in the regenerative response. For example, they might partition off the infarcted region, limiting the spread of ischemic brain damage without inducing scar formation (although further evidence is needed to clarify what, if any, role they might play). These modifications combined with the observed increase in new blood vessels in the peri-infarct region suggest that stem cell-induced changes in host cells and/or their factors plays a critical role in circumscribing brain injury, preserving tissue integrity, and/or enhancing regeneration after stroke.
As with stroke patients, local brain factors of potential interest in this regard were not easily accessed. Therefore, we began by sampling blood serum and tracking changes in the expression of 50 circulating inflammatory/immune and/or growth factor/receptors using a custom array. As documented previously (23,51), experimental stroke causes significant changes in cytokines and growth factors, many of which are thought to play an anti-inflammatory role. Importantly, we found that several of those factors that declined after MCAO, including IL-13, VEGF, and its receptor neuropilin, were recovered in rats following MSCs administration. This rebound phenomenon not only occurred rapidly (by 1 week post-MSCs) but factors continued to rise, reaching higher than normal levels at 4 weeks. Others have also reported a rise in VEGF, consistent with neovasculogenesis and reduced neuronal apoptosis after stem cell administration to the MCAO rat (15,57). In contrast, other cytokines/chemokines (i.e., MMP2, MIP) and cytokine receptors (i.e., EPOR, TROY) that were unchanged from normal at 24 h after experimental stroke and that declined after sham transplant, nonetheless increased after rMSCs treatment. Importantly, many of these factors are thought to be important in mitigating the inflammatory response and inducing neovascularization (13,36). In addition, several growth factor receptors (NGFR, RAGE) that were increased by MCAO remained elevated after rMSC injection, possibly in an attempt to sustain a neuroprotective and/or regenerative host response. Of course, the causal role that these blood-borne factors play in improving brain structure and function after MSC injection in the MCAO rat remains to be validated. It is possible that systemic changes are epiphenomenal and local brain factors are responsible for the beneficial effects seen after MSC treatment in stroke. Future experiments using Q-PCR to track the temporal response of host brain factors to MSC treatment will be needed to begin to distinguish between these possibilities.
In summary, there is now little doubt that the peripheral administration of stem cells of various types can modify the cellular and molecular landscape of the brain and blood, allowing the host animal to limit the spread of damage and sustain efforts to protect and repair the stroke-injured brain. Defining the mediators of these beneficial effects, while experimentally challenging, will be necessary to pave the way to new treatments for stroke.
Footnotes
Acknowledgments
This work was supported by a grant from the Joe and Marie Field Foundation and the Farber Institute for Neurosciences at Thomas Jefferson University.
