Abstract
The transplantation of neural stem cells (NSCs) is a challenging therapeutic strategy for the treatment of neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS). To provide insight into the potential of the intravenous delivery of NSCs, we evaluated the delivery of NSCs marked with green fluorescent protein to the central nervous system (CNS) via intravenous tail vein injections in an ALS model. The injected cell fates were followed 1, 3, and 7 days after transplantation. The highest efficiency of cell delivery to the CNS was found in symptomatic ALS (up to 13%), moderate in presymptomatic ALS (up to 6%), and the lowest in wild-type animals (up to 0.3%). NSCs injected into ALS animals preferentially colonized the motor cortex, hippocampus, and spinal cord, and their differentiation was characterized by a decrease of nestin expression and the appearance of MAP2-, GFAP-, O4-, and CD68-positive cells. Tumor necrosis factor (TNF) administration increased the CNS delivery of transplanted cells in wild-type and presymptomatic, but not ALS symptomatic animals. Moreover, a TNF-related increase in NSC differentiation and survival was detected. Apoptosis was detected as the main cause of the loss of transplanted cells and it was influenced by TNF. Although 3 days after TNF treatment cell death was accelerated, TNF slowed down apoptosis after 7 days. This study provides elementary facts about the process occurring after NSCs leave the blood stream and enter the nervous tissue affected by inflammation/degeneration, which should help facilitate the planning of future bench-to-bedside translational projects.
Keywords
Introduction
Neural stem cell (NSC) transplantation provides new prospects for the treatment of neurodegenerative diseases. However, compared with hematology, for which the use of mesenchymal stem cells is grounded in convincing experience, the fates and beneficial potential of intravenously delivered NSCs to an organism affected by a chronic neurodegenerative disease, such as amyotrophic lateral sclerosis (ALS), has only started to be investigated (24).
ALS is a fatal and still incurable neurodegenerative disease characterized by the loss of both upper and lower motor neurons, leading to paralysis and death, which occurs on average 3–4 years after the onset of the first symptoms. Its highly predominant form is sporadic, with only 10% of the cases classified as genetically linked familial ALS (fALS), which in 20% of the patients is caused by mutations in the Cu/Zn superoxide dismutase (SOD1) gene (32). The complex pathology at the level of motor neurons includes the accumulation of neurofilaments in the perikaryon and axon, impaired axonal transport, oxidative stress, and dysfunction of mitochondria. “The bad neighborhood” concept has revealed astrocytes as cells responsible for producing a toxic environment for motor neurons [pathology of ALS reviewed by (5, 18, 19), which has been confirmed in the opposite direction as well, because the injection of wild-type (wt) astrocytes increases the survival of motor neurons in ALS (23). Moreover, several proinflammatory molecules, including tumor necrosis factor (TNF), are substantially increased in the advanced stage of the disease (6, 17). The recent discovery of mutation within genes TDP-43 and FUS/TLS suggested impaired RNA processing as the common starting mechanism leading to neurodegeneration (18). Therefore, recent advances in understanding ALS pathology currently drive researchers to look for new therapeutic strategies aiming to restore impaired tissue homeostasis, by the substitution of lost cells and/or secretion of factors beneficial for the host tissue.
Until now, research on transplantation therapy in ALS has focused on the injection of cells into localized regions of the spinal cord (23, 36). However, intravenously injected NSCs seem to yield promising effects in central nervous system (CNS) affected by stroke (20), Huntington disease (21), multiple sclerosis (30), and compression spinal cord injury (33). In other studies, therapeutic efficiency was improved using TNF, which, as in the case of mesenchymal cells-based therapy of infarcted myocardium, increased the permeability of the blood vessel wall and cell engraftment (14).
The main aim of this study was to use the blood delivery route to explore the fates of engrafted NSCs in the CNS affected by the neurodegenerative process. Moreover, the potential of TNF in increasing cell delivery efficiency and its possible influence on NSC behavior in the ALS environment were analyzed. After injection of 107 green fluorescent protein (GFP)-marked NSCs, their migration to the preferential sites—the brain cortex, hippocampus, and spinal cord—was detected. Unlike in wt animals, where grafting efficiency was very low, injected NSC colonized the CNS of ALS-affected animals, and the process was more efficient in symptomatic than in presymptomatic animals. Successfully engrafted cells started to differentiate, and the process showed a tendency to be faster in ALS-affected animals. Treatment with TNF increased cell delivery in presymptomatic and wt animals as well as cell survival and differentiation in all tested groups. The unexpected failure of TNF to increase cell delivery in ALS symptomatic animals suggests a role of this molecule in attracting NSCs to the CNS affected by ALS.
Materials and Methods
Experimental Animals
For the purpose of this study three rat strains were used: 1) SOD1G93A rat model for ALS, characterized by the presence of multiple copies of the mutated human SOD1 gene (SOD1G93A) on the Sprague-Dawley background {Taconic Farms Europe ]002148-T, NTac:SD-Tg (SOD1G93A) L26H]}, 2) Sprague-Dawley line with inserted eGFP {Rat Resource and Research Center University of Missouri, USA ]RRRC:0065, SD-Tg(GFP)-Bal/2Rrr]}, and 3) wild-type Sprague-Dawley line. The starting pool of three groups of animals (presymptomatic, symptomatic, and wt) were divided in two additional groups: those treated by TNF and those not TNF treated. This made a total of six groups with nine animals in each (Table 1). Presymptomatic ALS rats were on average 14 weeks, whereas symptomatic rats were 26 weeks old. Control wt animals were also divided into two groups, so they matched the presymptomatic and symptomatic animals in age. The number of male and female animals in all compared groups was carefully balanced. All lines were bred and housed in our animal facility (ULB, Campus Erasme) according to the institutional guidelines (Animal welfare guidelines of the Universite Libre de Bruxelles), and all experiments were performed according to the ethical regulations (Animal welfare guidelines of the Universite Libre de Bruxelles) (http://www.ulb.ac.be/facs/medecine/comiteethiqueanimal.html). Transgenic rats were geno-typed by PCR using specific primers for the human SOD1 transgene as described (29). To obtain GFP expressing NSC, SD-eGFP rats were mated and embryos were isolated on day 16 of pregnancy under deep anesthesia using 30% ketamine (Ketalar, Pfizer) and 20% xylazine (Rompun, Bayer) in PBS (1 ml/500 g of body mass). After obtaining the embryos, animals were euthanized by decapitation.
Comparison of the Total Number of Cells and the Cell Densities 1, 3, and 7 Days After Systemic Cell Delivery

Data are expressed as the mean ± SEM. S ALS, symptomatic ALS-affected animals; S ALS TNF, symptomatic ALS-affected animals treated by TNF; PS ALS, presymptomatic ALS-affected animals; PS ALS TNF, presymptomatic ALS-affected animals treated by TNF; WT, wild-type animals; WT TNF, wild-type animals treated by TNF. Statistical comparisons have been made using the ANOVA test. Post hoc analysis was conducted using the Bonferonni's method.
S ALS and PS ALS versus WT (p < 0.001).
WT TNF versus WT (p < 0.001).
PS ALS TNF versus PS ALS (p < 0.05).
S ALS and PS ALS versus WT (p < 0.01).
NSCs were injected into the rats' tail vein. Before injection, rats were anesthetized using the same procedure mentioned above (ketamine/xylazine). To decrease the blocking of pulmonary veins by NSCs and reduce subsequent cell loss, before treatment with NSCs all animals received 25 ng of the potent pulmonary vein dilatation drug sodium nitroprusside (Merck, M-6541). It was diluted in 100 μl of physiological solution (0.9% NaCl) and injected in the tail vein 5 min before NSC delivery. In order to open the blood-brain barrier (BBB), half of the animals (N = 27) received 150 ng of TNF (Peprotech, 400–14) diluted in 100 μl of physiological solution into the tail vein 3 h before NSC injection (8). Immediately before injection, cells were harvested from tissue flasks by centrifugation, dissociated using the described trypsine procedure (see Neural Stem Cells section), twice washed with fresh stem cell medium, and dissolved in a volume of 1 ml. Finally, animals received 107 of cells in 1 ml of physiological solution with 0.2% glucose, using a Dual Syringe Pump injector (Harvard Apparatus, MA1 70-2209). The speed of the injection was 100 μl/min. To obtain tissue for histological analyses, animals were transcardially perfused with 4% paraformaldehyde (Merck). The procedure led to euthanasia.
Neural Stem Cells
The embryonic brain cortex was staged (34) and isolated using scissors for micromanipulation (Fine Science Tools), and the tissue was kept in HBSS (Gibco). Dissociation of the tissue and obtaining of the cells was performed using trypsine solution (0.05% Try-Edta, Gibco). Tissue was incubated for 10 min at 37°C, after which reaction was stopped using 2 ml of NSC medium (DMEM/F12, Gibco; N2 and B27, Invitrogen; penicilin/streptomicin, Gibco) with 10% fetal calf serum (Gibco) and 0.001% DNAse (Roche). The solution was centrifuged, supernatant discarded, and cells resuspended in fresh NSC medium. Cells were cultured in NSC medium with 2 μg/ml of EGF and 1 μg/ml of FGF2 (both Peprotech) in Sarstedt tissue culture flasks. For the purpose of transplantation to experimental animals, a sufficient number of cells had been obtained by 2–3 weeks of expansion in the described medium. Cells were passaged once in a week by plating 2 million cells per one T75 flask. In parallel to transplantation, a small sample from the injected pool (105 cells) was transferred to a 96-well plate (Sarstedt) and tested for a nestin marker. In this way, it was ensured that experimental animals had been treated by undifferentiated multipotent NSCs.
Immunohistochemistry
For the purpose of this study the following primary antibodies and concentrations used were: glial fibrillar acidic protein (GFAP, Dako, M0761, 1:2000), oligodendrocyte marker O4 (O4, Millipore, MAB345, 1:200), nestin (Nes, Millipore, MAB353, 1:100), microtubule-associated protein 2 (MAP2, Brion, B-9, 1:100), antigen CD68 (CD68, AbD Serotec, MCA341R, 1:200), antigen Ki67 (Ki67, Millipore, AB9260, 1:400), synaptotagmin (Syp, DSHB, MAB30, 1:50), and green fluorescent protein (GFP, Molecular Probes, A6455, 1:200). Secondary antibodies used for immunofluorescence were from Alexa Fluor family (Invitrogen): 350 GAM (A11045, 1: 50), 350 GAR (A11046, 1:100), 488 GAM (A11001, 1: 200), 488 GAR (A11008, 1:200), 594 GAM (A31623, 1:100), and 594 GAR (A31631, 1:200).
Paraffin sections (10 μm thick) of analyzed tissue were deparaffinized and washed/permeabilized using wash buffer (1 × PBS + 0.5% Triton-X 100). Blocking was carried out for 2 h using a blocking solution (wash buffer plus 5% goat serum and 0.5% BSA). Primary antibodies were incubated overnight at 4°C at the appropriate dilution in blocking solution. Excess primary antibody was washed for 5 min (three times) and then 15 min (three times) and secondary antibodies were incubated for 2 h at room temperature before another round of washing. For the majority of slides, DAPI was used as a nuclear counterstain. Slides were mounted and images were taken using a Zeiss Axiovert 200M fluorescent microscope.
Detection of Apoptosis
Apoptosis was analyzed in two rats from every group using the Flivo Apoptosis Kit (Immunochemistry Technologies, #982). According to the manufacturer's protocol, 100 μl of freshly prepared solution was injected into the rat's tail vein. The rat was sacrificed after 1 h and 10-μm-thick paraffin sections were obtained using a standard procedure. Apart from routine immunohistochemistry for differentiation, proliferation, and GFP markers, this allowed direct fluorescent detection of caspase activity.
Cell Counting
For the purpose of this study, we analyzed the number of transplanted cells in the whole brain and the spinal cord. Cell counting was performed on 10-μm-thick paraffin sections, and cell numbers were expressed either as an absolute cell number (the brain cortex) or as average densities (all analyzed tissues). The absolute cell number in the brain cortex was assessed by counting GFP-positive cells in the cortex in every 20th section. An estimation of the total number of cells was then obtained using the following calculation: because the average diameter of half of the rat brain was 5 mm, this led to an average of 25 sections in an average distance of every 200 μm. Cell densities were observed by counting cells in 10 sections taken at every 100 μm distance. The percentage of cells expressing different markers was assessed by counting cells in the same sections used to count cell densities. All measurements of the tissue were assessed using AxioVision 4.7.1 software.
Statistical Analyses
The level of significant differences was analyzed using ANOVA and the Bonferroni correction methods.
Results
The Blood Stream Delivery of NSCs to the CNS of ALS-Affected Rats
One day after intravenous injection of 107 GFP-marked NSCs, they were detected in the CNS of all animal groups, but the number of cells and cell densities were significantly different (p ≤ 0.05) (Table 1). The three major sites of cell invasion in ALS rats were the brain cortex, dentate gyrus of hippocampus, and spinal cord (Table 1, Fig. 1). In wt animals, the highest density of cells was found in the hippocampus. The maximum efficiency of cell delivery in the CNS, reaching up to 13% was found in ALS symptomatic animals: 9 × 105 in the brain cortex, 2.5 × 105 in the spinal cord, 105 in the other parts of the brain, and 5 × 104 in the hippocampus. In presymptomatic animals, the average number was 50% lower, so grafting efficiency reached 6%. The difference in efficiency rates of cell delivery between presymptomatic and symptomatic ALS animals was because of the differences in migration to the cortex. Average numbers of cells found in the hippocampus, spinal cord, and the rest of the brain were not significantly different between presymptomatic and symptomatic ALS animals. A rather low rate of CNS invasion was revealed in wt animals, reaching 0.3% (Table 1, Fig. 2).

Visualization of GFP-marked intravenously delivered NSCs 1 day after injection in the brain cortex (A), the dentate gyrus of the hippocampus (B), and the spinal cord (C) of symptomatic ALS animals with examples of cell density assessment.

The efficiency of cell delivery and change in cell number in the brain cortex in presymptomatic and symptomatic ALS rats, and their wt counterparts 1, 3, and 7 days after intravenous delivery. The highest efficiency of delivery was observed in symptomatic, moderate in presymptomatic animals, and very low in wt animals. TNF increased cell invasion efficiency in wt and presymptomatic, but not in symptomatic animals. Three days after injection, cell loss was observed in all tested groups, and it was accelerated in TNF-treated animals. Although 7 days after injection the number of cells continued to decrease, the loss was decelerated in TNF-treated animals. Statistical analyses were made using the ANOVA test. On day 1, a significant difference was observed when wt was compared to TNF-treated wt group (p < 0.0001) and for presymptomatic ALS compared to presymptomatic ALS-TNF treated group (p < 0.01). On day 3, a significant difference was observed between the symptomatic and symptomatic/TNF-treated group (p < 0.05). On day 7, both presymptomatic and symptomatic groups compared to their TNF-treated counterparts revealed significant differences in the number of cells (p < 0.05). Therefore, the observed differences between tested groups as observed by ANOVA were significant (F = 451.57, p < 0.0001). The change in the number of cells during time in each tested group was significant (p < 0.0001), except for all three tested points in wt group and the difference between day 3 and 7 for the TNF-treated wt group. Therefore, the observed changes in the number of cells for each tested group over time as determined by ANOVA were significant (F = 319.19, p < 0.0001).
To increase the efficiency of cell delivery and test the role of TNF in attracting NSCs to the CNS affected by ALS, 150 ng of TNF was injected into the blood stream 3 h before NSC injection. TNF had a dramatic positive effect when administered to the wt animals (Table 1, Fig. 2). The cell densities significantly increased in the brain cortex (up 650%, p ≤ 0.05), and the hippocampus (up 500%, p ≤ 0.01), whereas in the spinal cord and the other parts of the brain the increase was not significant. Thus, the efficiency of cell injection in wt animals increased six times (reaching 2%). Administering TNF significantly increased (27%) cell invasion in presymptomatic ALS animals and had no significant negative effect on ALS symptomatic animals (Table 1, Fig. 2).
The Role of Apoptosis in Gradual Cell Loss of Grafted NSCs
To analyze the survival of grafted cells, we compared their total numbers 1, 3, and 7 days after cell injection (Table 1, Fig. 2). In addition, to enhance cell grafting, half the animals were treated by TNF. Programmed cell death was analyzed using caspase assay (Flivo) (Fig. 3).

Detection of apoptosis in the cortex region (A–C) reveals the death of neurons derived from the injected NSC GFP population (one neuron marked by arrow). In the hippocampal region (D–F), apoptotic cell death was found in both NSC-derived (gray arrow) and in neighboring cells (white arrow).
One day after injection of NSCs, the number of engrafted cells dying by apoptosis in the brain cortex, hippocampus, and spinal cord was similar in all untreated groups and was around 4%. TNF administration increased apoptotic cell death of transplanted cells, which reached 7%, and was observed in both wt and presymptomatic ALS animals. TNF did not influence the rate of cell death in symptomatic ALS animals.
Three days after injection, the total number of cells significantly decreased in all six analyzed groups of animals. In presymptomatic and wt animals, cell loss was significantly larger when treated by TNF (Fig. 2). Surprisingly, caspase assay revealed a significantly higher ratio of cells expressing the apoptotic marker in untreated (17%) than TNF-treated animals (12%). Moreover, the coexpression of caspase activity and nestin was not found, suggesting a correlation between the onset of differentiation and programmed cell death.
Although 7 days after injection, the numbers of cells continued to decrease in all analyzed groups, TNF-treated animals significantly slowed their rate of cell loss (Fig. 2). The most pronounced effect was observed in symptomatic TNF-treated animals that, after 7 days, had more detectable injected cells than their untreated counterparts. Slowing of the decrease in cell number was observed in TNF-treated presymptomatic and wt animals as well. Moreover, TNF-treated animals exhibited a lower ratio of programmed cell death, thereby continuing the trend observed at 3 days after injection. Changes in the cell death ratio in the spinal cord and the rest of the brain were the same as those observed in the cortex, whereas the ratio of cell death found within the hippocampus was rather low (1%) and constant.
Differentiation of NSCs Delivered to the CNS by Blood Stream Route
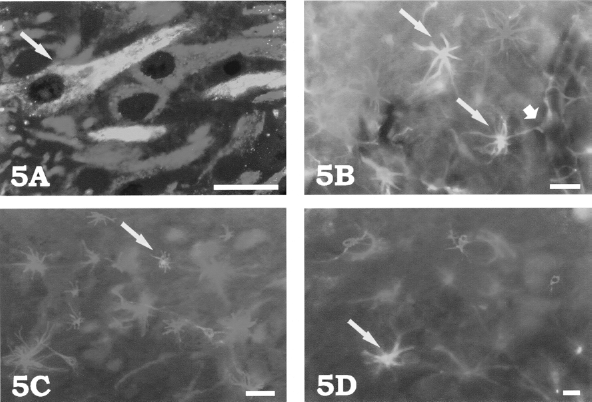
To gain an insight into the differentiation of injected cells, the following markers were used: nestin for undifferentiated NSC, MAP2 for neurons, GFAP for astrocytes, O4 for oligodendrocytes, and CD68 for microglia (Fig. 4). Results are visualized in Figure 5. Before injection (day 0), all NSCs were uniformly expressing nestin and no other differentiation markers. One day after injection, the majority (more than 85%) of injected cells continued to express nestin. The rest of the cells started to express other differentiation markers. The simplest pattern was found in untreated wt animals, where 10% of cells started to express MAP2, and in half of them coexpression with nestin was also found. The main difference between ALS and wt animals after 1 day was that in ALS animals, besides early observed neuronal fate, GFAP- and O4-positive cells were already present. None of the tested groups showed an expression of CD68 1 day after injection.

Expression pattern of blood stream-injected NSCs within the brain cortex: wt and TNF-treated wt (A) and ALS and TNF-treated ALS (B). Animals affected by ALS and TNF-treated animals exhibited a trend towards accelerated differentiation at days 1 and 3.
Differentiation of GFP-marked NSCs resulted with cells that coexpress MAP2 (A), GFAP (B), CD68 (C), and O4 (D). Particular NSC-derived cells are marked by long arrows. The short arrow in (B) denotes cell projection from NSC-derived astrocytes, which forms end feet just like other astrocytes residing in that region do (notice the blood vessel in the right part of the photo). Scale bar: 20 μm.
Three days after injection, levels of nestin substantially decreased in all analyzed groups of animals. The majority of cells started to express either MAP2 or GFAP, while the expression of O4 was rather low and uniform. CD68-positive cells were also found. Similar to 1 day after injection, 3 days after injection the ALS-affected animals and wt animals treated by TNF showed a tendency towards a larger ratio of differentiated cells, concomitant with a lower ratio of nestin expression. Thus, 3 days after injection more MAP2-, GFAP-, and CD68- positive cells were found in ALS-affected and TNF-treated wt animals than in wt animals (Fig. 4). Statistically significant differences (p ≤ 0.05) were observed when the wt and ALS animals were compared for levels of GFAP at days 1 and 3, and for MAP2 at day 1.
Seven days after injection, in all analyzed groups, a decrease in the MAP2 ratio and increase in the GFAP ratio was found (Fig. 4). Although in wt animals the proportion of the astrocytic population was kept low, in all other groups the dominance of GFAP prevalence become substantial. The number of CD68 cells remained close to the levels observed at 3 days and its levels in wt rats was lower than both ALS- and TNF-treated groups of wt animals. The expression of nestin in the brain cortex and spinal cord almost completely disappeared. However, in all groups of analyzed animals the levels of nestin in the dental gyrus of hippocampus remained at the same level as 3 days after injection.
To test whether transplanted cells proliferated and contributed to neurogenesis, the expression of Ki67 was tested. The majority of GFP-injected cells in all tested animals expressed Ki67, but only 1 day after injection. In the later tested stages (3 and 7 days), some level of mitotic activity was found in the hippocampus. Moreover, signs of tumorigenicity were not observed in any part of the CNS.
Presence of Grafted NSCs in the Spleen, Liver, Lungs, and Kidney
Cells injected into the blood stream invaded different tissues. The majority were found 1 day after injection in the spleen (3 × 106), liver (2 × 106), lungs (3 × 105), and kidney (4 × 104) (Fig. 6). There were no signs of neuronal or glial differentiation or caspase-mediated cell death 1 day after injection. However, 3 days after injection, the number of cells substantially decreased and the majority were dying by apoptosis. Seven days after injection, only several remaining cells dying by apoptosis were found.

GFP-expressing NSCs were transiently found in other organs. (A) The presence of some green cells in the liver is accompanied by apoptosis. (B) In the spleen, the majority of GFP-expressing cells were found at the edge of the white pulp (W) or in the marginal zone (M). R marks the red pulp. The majority of the injected cells exhibit signs of apoptotic death. Small amount of cells were found in the lungs (C) and the kidney (D). Scale bars: 100 μm.
Discussion
Cell transplantation using the blood stream represents a promising approach in the therapy of neurodegenerative diseases (13, 25). The main goal of this work was to analyze the fates of NSCs after they leave the blood stream and enter the neural tissue affected by ALS. This study analyzed intravenously engrafted NSCs in the CNS of ALS-affected animals and their wt counterparts and assessed their fate in terms of cell differentiation and survival. We focused on the quantity of cell colonization, cell differentiation, and cell death, which enabled us to provide the elementary facts about the behavior of NSCs in the ALS-affected and wt CNS. In other similar studies, cell fates and animal survival rates were obtained using human umbilical cord blood cells (10), or NSCs in the mouse model of experimental encephalomyelitis (30). In those studies, cell engraftment and improvement in animal condition was reported.
One of the main results of this study was that the blood stream injection efficiency of cell grafting was much higher in ALS than wt animals. This result is comparable to the report of Pluchino (30), who declared that no injected cells were found in the wt brain when the blood stream route was used (we report an efficiency of 0.3%), and showed that in an experimental autoimmune encephalitis model, NSCs have the ability to cross the BBB and populate the CNS with cell densities comparable to ours (10–30 cells/mm2). The first indication that the BBB might be affected in ALS came from Leonardi et al., who noticed abnormal IgG and albumin levels in patients' cerebrospinal fluid (22), whereas Garbuzova-Davis et al. found Evans blue leakage around spinal cord microvessels in SOD1G93A mice (9). Zhong et al. (38) suggested that a disrupted BBB was a possible independent factor in ALS pathology, which was recently demonstrated in an ALS rat model (26). However, one should be careful when correlating leakage of solutes (e.g., Evans blue), which suggest a disrupted BBB and cell invasion into the neuropil. It is indeed probable that (neural stem) cells migrate thorough the basement membrane of postcapillary venules using the same mechanism as leukocytes do [reviewed in (2)]. In vitro endothelial systems revealed that NSCs specifically interact with the endothelium and migrate into the subendothelial space where their differentiation is supported (7).
To test the influence of TNF on NSC engraftment and its role in attracting NSCs in ALS-affected animals and their wt counterparts, half the animals were treated by this molecule. The rationale for that was reported TNF-caused increased endothelial permeability via a tissue factor-dependent mechanism during which the cytoskeleton rearranges (8). In addition, it has been shown that TNF increased both the adhesiveness and migration of polymorphonuclear leukocytes (35) and mesenchymal stem cells (14). Here, we have shown that TNF influences NSC delivery, survival, and differentiation. In wt and presymptomatic ALS animals, TNF facilitated the invasion of NSCs within the brain tissue, which was not apparent in symptomatic ALS animals, probably because of an already high level of TNF in the symptomatic stage of the disease (6). This suggests an important role of this molecule in attracting NSCs to the CNS affected by ALS. A surprising result was obtained when cell survival was analyzed. Although during the first day after injection cell death in TNF-treated animals was increased, the rate of cell death after 3 and 7 days slowed down, which resulted in increased cell survival in TNF-treated animals. One possible explanation for this effect is that TNF can act through the p75 receptor, promoting cell survival [reviewed by Kraft et al. (16)]. Moreover, cell death exclusively affected cells that were starting to express differentiation markers. This is in congruency with the finding that undifferentiated NSCs do not express caspase 8, which is crucial for initiating the extrinsic death pathway (31). The possible beneficial effect of TNF on long-term regeneration has been suggested: compressive brain injury in TNF KO mouse caused prolonged motor deficits when compared with wt animals (27), and levels of TNF increased during the late phase of sciatic nerve regeneration (4). Although Grassi et al. (11), in in vitro conditions, reported that TNF increased synaptic activity and synaptogenesis, which is one of the prerequisites in the survival of NSC-derived neurons, we did not detect an increase in the ratio of synaptic markers in TNF-treated animals.
Our work also showed that injected cells do not distribute equally in the CNS of ALS animals. This can be explained by the fact that for successful engraftment and survival, chemoattractive cytokines, and/or a supportive neurogenic niche (hippocampus) is required. The molecular mechanism of NSC attraction by cytokines, including TNF, were elucidated (3, 28), and it was reported that hematopoetic stem cell transplantation resulted in an accumulation of cells exclusively in the sites affected by neurodegeneration (1). Therefore, NSCs probably do invade regions of the CNS following chemoattractive signals, and the disrupted BBB in ALS possibly acts as an additional precipitating factor of this process.
To detect specific fates of grafted cells, differentiation markers were analyzed. As expected, and comparable to in vitro observations, the majority of cells presented 1 to 3 days of latency before starting to express differentiation markers. Generally, the two main fates observed were neuronal and astrocytic. Moreover, the tendency towards an increased rate of differentiation in ALS-affected animals was observed. An increased rate of differentiation was also found in animals treated by TNF, suggesting accelerated differentiation in the CNS affected by inflammation. Indeed, in some in vitro studies TNF promoted cell communication via TNF-R1 (15).
The described pattern of cell migration, differentiation, and survival was obtained using allogeneic cell transplantation. In order to be able to relate these observations to the specific cellular and humoral niche present in ALS, and not to graft rejection, we used rats with the same genetic background (Sprague-Dawley). We did not use immunosuppression because that would influence inflammatory process, which has an important role in ALS pathogenesis. Although the immune response of the host brain could influence behavior of the graft (12), some articles suggested avoidance of immunosuppression in similar studies, as the immune reaction after allogeneic transplantation does not necessarily lead to graft rejection (37).
In conclusion, analyses of the efficiency of NSC intravenous delivery, cell differentiation, and survival in an ALS rat model provided elementary facts about the process occurring after NSCs leave the blood stream and enter the nervous tissue affected by inflammation/degeneration. The highest efficiency of cell delivery to the CNS was found in symptomatic ALS, moderate in presymptomatic ALS, and the lowest in wt animals. NSCs injected into ALS animals preferentially colonized the motor cortex, hippocampus, and spinal cord, and their differentiation was detected. These data should encourage the use of this easy cell delivery route and facilitate planning for future bench-to-bedside translational projects.
Footnotes
Acknowledgments
This work was supported by FNRS postdoctoral research grants awarded to R.P. and D.M. (grant No. 3.4.545.05 F). S.G. was awarded grant 108-1081870-1902 from the Ministry of Science, Education and Sports of the Republic of Croatia and grant 35/08 from Unity Through Knowledge Fund of the Republic of Croatia. The authors thank M. Thierry Latran and La Jeune Chambre Internationale 3 frontières for their financial support. We thank Prof. Christian M élot for statistical analyses, Robert De Decker for animal care, and Michele Authelet for help in obtaining paraffin sections.
