Abstract
Previous studies suggest that neural crest (NC)-derived stem cells may reside in NC derivatives including the human periodontal ligament (hPDL). The isolation and manipulation of autologous NC-derived cells could be an accessible source of adult neural stem cells for their use in cell replacement and gene transfer to the diseased central nervous system. In this study, we examined the expression of NC markers and neural differentiation potential of hPDL-derived cells both in vitro and in vivo. In vitro we found that hPDL-derived cells expressed stem cell markers (Oct3/4, Nestin, Sox2, and Musashi-1) and a subset of NC cell markers (Slug, p75NTR, Twist, and Sox9). hPDL-derived cells differentiated into neural-like cells based on cellular morphology and neural marker expression (TUJ1, MAP2, MAP1b, GAD65/67, GABA, NeuN, ChAT, GAT1, synaptophysin, GFAP, NG2, and O4). In vivo, hPDL-derived cells survive, migrate, and give rise to DCX+, NF-M+, GABA+, GFAP+, and NG2+ cells after grafting the adult mouse brain. Some of the grafted hPDL-derived cells were located in stem cell niches such as the ventricular epithelium and the subventricular zone of the anterolateral ventricle wall as well as in the subgranular zone of the hippocampal dentate gyrus. Thus, the hPDL contains stem cells that originate from the NC and can differentiate into neural cells. The engraftment and differentiation properties of hPDL-derived cells in the adult brain indicate that they are a potential stem cell source to be used in neuroregenerative and/or neurotrophic medicine.
Keywords
Introduction
Embryonic and adult human stem cells have recently received a large amount of attention and interest due to their therapeutic potential for neurological disorders (5,20,31). However, the application of human embryonic stem cells in regenerative medicine is limited by legal and ethical restrictions (3,8,16). On the other hand, human adult stem cells represent an attractive alternative source of cells since they lack the above-mentioned problems and are suitable for autologous cell therapies, thus avoiding any important or long-term immune rejection complications (4,13,21).
Several studies have revealed the existence of stem cells in various human tissues including dental structures (14,17,27,29,38,44). The periodontal ligament (PDL) is a connective tissue that surrounds the tooth root and maintains homeostasis as well as repairing damaged tissue caused by mechanical trauma or periodontal disease (7). PDL contains a heterogeneous cell population that includes fibroblasts, osteoblasts, cementoblasts, endothelial cells, and epithelial cell rests of Malassez (22).
Isolation and characterization of plastic-adherent, mesenchymal stem cell-like cells from the human PDL (hPDLSCs) have been previously described (37). hPDLSC cultures are highly proliferative, form fibroblast colony-forming units, and express mesenchymal and embryonic markers (41). In vitro, under defined cultured conditions, hPDLSCs differentiate into cementoblast-like cells, adipocytes, and collagen-forming cells. When transplanted into rodents and dogs, these cells are capable of generating a cementum/periodontal ligament-like structure and contribute to periodontal tissue repair (32,36).
Recent findings suggest that the PDL may contain stem cells that originate from the neural crest (NC) (40). The NC is a migratory population of stem cells that can give rise to neurons and glial cells of the peripheral nervous system, endocrine, connective tissue, muscle, as well as pigment cells during embryonic development (6,10,26).
Previous reports demonstrated that hPDLSCs expressed mRNA of various NC-derived stem cell markers and suggested that hPDLSCs can be induced in vitro to differentiate into cells with neural phenotypes in response to basic fibroblastic growth factor (bFGF) and human recombinant epidermal growth factor (EGF) (9,19). hPDLSCs, when grafted onto organotypic hippocampal slice cultures, are capable of surviving, integrating, and expressing several neural markers (43). However, there are no reports that study the in vivo features of hPDLSCs transplanted into the adult mammalian brain and their potential for gene therapy and cell replacement in the central nervous system.
Therefore, the neural crest origin and differentiation plasticity make hPDL interesting as a cellular population to be used as experimental and cell therapy platforms, which requires one first to deeply explore their neural potentiality in vitro and in vivo. Here we report the isolation, propagation, genetic modification, and neural differentiation of human PDL-derived cells. Our data show that these hPDL tissue and isolated cells express several stem cell and neural crest markers. DNA transfection by electroporation in hPDL-derived cells allows stable expression of enhanced green fluorescent protein (eGFP), both in vitro and in vivo. Moreover, these cells, under certain culture conditions, could differentiate into neural lineages both in vitro and in vivo. Finally, we demonstrate, for the first time, that hPDL-derived cells can survive, integrate, and migrate when transplanted into adult mouse brain.
Materials and Methods
Cell Culture
Human premolars were extracted and collected from normal adult patients undergoing orthodontic therapy in Murcia dental hospital (Spain). Before extraction, the patients were informed of the procedures to be performed and signed a written consent. hPDL was scraped from the middle third region of the root surface. After washing the extracted PDL with Ca and Mg-free Hank's balance salt solution (HBSS; Gibco, Gaithersburg, MD, USA), hPDL was digested with 3 mg/ml type I collagenase (Worthington Biochemical Corporation, Lakewood, NJ, USA) and 4 mg/ml dispase II (Gibco) in alpha modification minimum essential medium eagle (α-MEM; Sigma-Aldrich, St. Louis, MO, USA) for 1 h at 37°C. The reaction was stopped by the addition of α-MEM. The dissociated tissue was passed through a 70-μm cell strainer (BD Falcon, Bedford, MA, USA). Cells were centrifuged, and the pellet was resuspended in serum-containing media (designated as the basal media), composed of α-MEM supplemented with 10% calf serum (Sigma), 100 units/ml penicillin–streptomycin (Sigma), 50 mg/ml l-ascorbic acid (Sigma), and 2 mM l-glutamine (Sigma). The cell suspension was plated into six-well multiwell plates (BD Falcon) and incubated at 37°C in 5% CO2. To induce neural differentiation, cells were cultured in serum-free media (designated as the neural induction media), consisting in Dulbecco's modified Eagle's medium/F12 (DMEM/F12, Gibco) supplemented with bFGF (20 ng/ml, R&D Systems, Minneapolis, MN, USA), EGF (20 ng/ml, R&D Systems), glucose (0.8 mg/ml, Sigma), transferrin (100 μg/ml, Sigma), insulin (25 μg/ml, Roche Molecular Biochemicals, Indianapolis, IN, USA), putrescine (60 nM, Sigma), progesterone (20 nM, Sigma), sodium selenate (30 nM, Sigma), 2 mM l-glutamine (Sigma), and 100 units/ ml penicillin–streptomycin (Sigma). Neural induction media were changed every 3–4 days until the end of the experiment.
RT-PCR Analysis
Total RNA was extracted using RNeasy Mini Kit and DNAse I treatment (Qiagen, Germantown, MD, USA) to avoid genomic contamination. hPDL tissue and hPDL-derived cells were collected and frozen for further RNA extraction. cDNA was synthesized from 2 μg RNA, using AMV reversed transcriptase (Finnzymes, Vantaa, Finland) and random hexamers (Roche Diagnostics, Indianapolis, IN, USA). One-tenth of the RT reaction was subsequently processed for PCR using Red Taq genomic DNA polymerase (Sigma), and the following primers were used: octamer-binding transcription factor 3/4 (Oct 3/4; accession NM_002701.4) forward (5′-GAGGAGTCCCA GGACATCAA-3′), reverse (5′-TCTCCAGGTTGCCTCT CACT-3′); nestin (NM_006617.1) forward (5′-GAAGTA GGGGATGAGGAGGC-3′), reverse (5′-TAGAGACCTCC GTCGCTGTT-3′); Slug (accession NG_012130.1) forward (5′-CAGTCCAAGCTTTCAGACCC-3′), reverse (5′-CAG TGAGGGCAAGAAAAAGG-3′); sex-determining region Y box 9 (Sox9; accession NM_000346.3) forward (5′-AG AAGGACCACCCGGATTAC-3′), reverse (5′-AAGTCG ATAGGGGGCTGTCT-3′); Sox2 (accession NM_0031 06.2) forward (5′-CATCACCCACAGCAAATGAC-3′), reverse (5′-TTTTTCGTCGCTTGGAGACT-3′); Twist (accession NM_000474.3) forward (5′-ATTCAAAGAA ACAGGGCGTG-3′), reverse (5′-CTATGGTTTTGCAG GCCAGT-3′); Musashi-1 (MLS1, accession NM_0024 42.2) forward (5′-GGCTTCGTCACTTTCATGGACC-3′), reverse (5′-GGGAACTGGTAGGTGTAACCAG-3′); fibroblast growth factor receptor 1(FGFR1; accession NM_0011 74066.1) forward (5′-GATAGCAGACTTTGGCCTCG-3′), reverse (5′-AAAGCTGGGGGAGTACTGGT-3′). PCR products were electrophoresed in 2% agarose gels (Sigma) and photographed after ethidium bromide staining (Sigma). Internal negative controls confirmed the absence of human genomic DNA in RNA samples.
Expression Vectors and Cell Transfection
The expression vectors used in the present study were derived from pIRES1hyg (Clontech, Cambridge, UK). p(eGFP) IRES1hyg was generated by subcloning eGFP cDNA as a BamHI + NotI insert from pEGFP-N1 (Clontech) into the BamHI + NotI digested pIRES1hyg. Isolated hPDL-derived cells were electroporated using the Electroporador Gene Pulser-II (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Electroporation was performed in a sterile cuvette with a 0.4-cm electrode gap (Bio-Rad Laboratories, Inc.), using a single pulse of 270 V, 500 μF. Plasmid DNA (5 μg) was added to 1.5 × 106 viable hPDL-derived cells in 0.2-ml electroporation solution (α-MEM) before electrical pulsing. Drug selection of stable transfectants was performed with 50-100 mg/ml hygromycin B (hyg; Calbiochem, La Jolla, CA, USA).
Immunocytochemistry
Cells were plated onto poly-l-lysine (10 μg/ml, Sigma-Aldrich)-coated multichambers (BD Falcon) and maintained in basal media or neural induction media. Cells were fixed in freshly prepared 4% paraformaldehyde (PFA; Sigma), except for neurotransmitter detection, in which case 0.1% glutaraldehyde (Millipore, Billerica, MA, USA) supplemented 4% PFA was used. Fixed cells were blocked for 1 h in PBS containing 10% normal horse serum (Gibco) and 0.25% Triton X-100 (Sigma) and incubated overnight (O/N) at 4°C with monoclonal antibodies (mAb) against: cluster of differentiation 90 (CD90) (1:300, BD Pharmingen, San Diego, CA, USA), CD44 (1:300, BD Pharmingen), green fluorescent protein (GFP) (1/300, Molecular Probes, Eugene, OR, USA), β-III-tubulin (TUJ1; 1:1,000, Covance, Princeton, NJ, USA), glial fibrillary acidic protein (GFAP) (1:1,000, Calbiochem), microtubule-associated protein 1B (MAP1B) (1/300, Abcam, Cambridge, MA, USA), microtubule-associated protein 2 (MAP2) (1/300, Chemicon, Temecula, CA, USA), oligodendrocyte marker 4 (O4; 1/300, MAB228, Chemicon), neuronal nuclei (NeuN) (1/300, Chemicon), hNestin (1/300, Chemicon), or polyclonal antibodies (pAb) against choline acetyltransferase (ChAT; 1:1,000, Chemicon), γ-aminobutyric acid (GABA; 1:300, Sigma), high-affinity GABA plasma membrane transporter (GAT1; 1:300, Chemicom), glutamic acid decarboxylase (GAD65-67; 1/300, Sigma), chondroitin sulfate proteoglycan (NG2; 1/300, Chemicom), low-affinity neurotrophin receptor (p75NTR; 1/300, Chemicom), and synaptophysin (1/100, Zymed Laboratories, South San Francisco, CA, USA). Afterwards, cells were rinsed and incubated with the corresponding secondary antibodies: Alexa Fluor® 488 (anti-mouse or anti-rabbit; 1:500, Molecular Probes), Alexa Fluor® 594 (anti-mouse or anti-rabbit; 1:500, Molecular Probes), biotinylated anti-mouse (BA-2020 and BA-9200; 1:250, Vector Laboratories, Burlingame, CA, USA), biotinylated anti-rabbit (BA1000, 1:250; Vector Laboratories), biotinylated anti-goat (BA5000, 1:250, Vector Laboratories), biotinylated anti-rat (BA-9400, 1:250, Vector Laboratories), CY3-streptavidin (1:500, GE Healthcare, Little Chalfont, Buckinghamshire, UK). Cell nuclei were counterstained with DAPI (Molecular Probes, 0.2 mg/ml in PBS).
Western Blotting
hPDL-derived cells proliferating (in basal media) were harvested using trypsin/EDTA (Gibco), washed twice with PBS, resuspended in lysis buffer (1% Triton X-100, 150 mM NaCl, 20 mM Tris-HCl, pH 7.5, 0.5 mM EDTA, 1 mM EGTA) in the presence of protease inhibitors (complete mini EDTA-free protease inhibitor cocktail tablets, Roche) for 30 min at 4°C, and centrifuged at maximum speed of 20 min at 4°C. Protein concentration was determined using the BCA protein assay (Bio-Rad). Thirty micrograms of protein were separated in a 8% polyacrylamide gel electrophoresissodium dodecylsulfate (PAGE-SDS) gel (Life Technologies, Grand Island, NY, USA) and transferred onto a nitrocellulose membrane (Whatman, Maidstone, Kent, UK). Blots were then incubated overnight at 4°C with pAb against β-actin (1:7,000, Sigma) and mAb anti-nestin (1:1,000, Chemicon). Secondary antibodies were used at 1:5,000 for peroxidase antimouse Ab (PI-2000, Vector) and 1:7,000 for peroxidase anti-rabbit Ab (PI-1000, Vector). Immunoreactivity was detected using the enhanced chemiluminescence (ECL) Western blot detection system (Amersham Biosciences Europe, Freiberg, Germany) using Fujifilm Bioimage Analyzer LAS-1000 Plus Gel Documentation System (Valhalla, NY, USA).
Transplantation Procedures
Experiments were carried out according to the guidelines of the Spanish and European Community (Council Directive 86/609/ECC) and in accordance with the Society for Neuroscience recommendations. Animals used in this study were 8-week-old immune-suppressed mice (Hsd:Athymic Nude-Foxn1 nu/nu; Harlan Laboratories Models, S.L., Castellar, Spain), housed in a temperature and humidity controlled room, under a 12-h light/ dark cycles, with ad libitum access to food and water. Cells for transplantation (eGFP+ hPDL-derived cells in neural induction media) were dispersed and resuspended in HBSS (Gibco) at a density of 105 cells/μl. Cell suspension (1 μl) were injected (using a 10-μl Hamilton syringe; Reno, NV, USA) into the hippocampus at the following coordinates: anterioposterior (AP) = −2.1 mm, mediolateral (ML) = −1.9 mm, dorsoventral (DV) = −2.1 mm.
Immunohistochemistry
One month after the transplantation procedure, the animals were anesthetized with an overdose of chloral hydrate (Sigma) and intracardially perfused with freshly prepared, buffered 4% PFA (in 0.1 M PB, pH 7.4). Brains were removed, postfixed for 12 h in the same fixative at 4°C, and dehydrated in 30% sucrose solution at 4°C overnight. Thirty-micrometer-thick coronal sections were collected using a freezing microtome (Mic rom, Walldorf, Germany). Serial sections were used for immunohistochemistry with monoclonal antibodies (mAb) against: GFP (1/300, Molecular Probes), GFAP (1:1,000, Calbiochem) or polyclonal antibodies (pAb) against GABA (1:300, Sigma), NG2 (1/300, Chemicon), doublecortin (DCX; 1:300, Santa Cruz Biotechnology, Dallas, TX, USA), neurofilament medium (NF-M; 1:300, Millipore), and GFP (chicken IgY-FICT conjugate; 1/300, Sigma). Briefly, free-floating sections were incubated O/N at 4°C with the primary antibodies diluted in PBS with 1% horse serum. Sections were rinsed four times in PBS during a 1-h period, then incubated for 2 h with the secondary antibodies in PBS (see Immuno-cytochemistry section for details), and mounted onto Superfrost Plus glass slides (Thermo Scientific, Waltham, MA, USA). The slides were dried O/N and coverslipped with mowiol-NPG (Calbiochem).
Cresyl Violet Staining
Microtome sections mounted on Superfrost plus glass slides were stained with cresyl violet (0.5%; Sigma), dehydrated in ethanol (Millipore), bleached in xylene (Panreac, Barcelona, Spain), and mounted in DPX (Sigma) according to standard procedures.
Images and Data Analyses
Analyses and photography of fluorescent or DAB stained samples (Immunocytochemistry and Immunohistochemistry) were carried out in an inverted Leica CTR 6000 microscope equipped with a digital camera Leica DC500 or Leica DM IRB microscope equipped with a digital camera Leica DFC350FX (Nussloch, Germany). In some experiments, digitized images were captured using Leica IM500 Software. Confocal analysis was carried out in Leica TCS SP2 Spectral Confocal Scanner mounted on a Leica DM IRE2 inverted fluorescent microscope. Digitized images were analyzed using Leica confocal software. Colocalization was determined through orthogonal sectioning by comparing signals on the same z planes (DCX = 1.1 μm; GFAP = 2.67 μm; NG2 = 1.97 μm; NF-M = 4.36 μm; GABA = 2.42 μm) to avoid false-positive staining. Descriptive statistics were run using Prophet Software (NIH, Bethesda, MD, USA). Results are shown as the average±SEM of data from three experiments.
Results
Cell Isolation and Characterization of Cultured PDL-Derived Cells
To determine if human PDL (hPDL) cells retain stem cells and neural crest characteristics, tissue obtained from human PDL (hPDL) was examined for stem cell and neural crest (NC) markers by RT-PCR analysis. As a result, both stem cell (nestin and Oct3/4) and NC cells markers (Twist and Slug) were detected in hPDL tissue (Fig. 1A). Based on previous studies (42,46), we isolated cells from hPDL tissue in plastic dishes. Cells were expanded in 10% calf serum-containing culture media (the basal media). We studied the mRNA expression of several markers using RT-PCR analysis. Stem cell markers (Oct3/4, nestin, Sox2, and MLS1) and a subset of NC cell markers (Slug, Twist, and Sox9) were also expressed in hPDL-derived cells (Fig. 1A). Western blot analysis verified the expression of Nestin (Fig. 1B). Additionally, the cells were plated on poly-l-lysine-coated multichambers, fixed, and processed for immunocytochemistry. Almost all cells were positive for surface markers CD44 (99.60±0.69%) and CD90 (99.30±1.05%). Furthermore, 99.11±1.59% hPDL-derived cells were positive for NC cell marker p75NTR [low-affinity nerve growth factor (NGF) receptor] and 29.62±3.43% for neural stem cell marker nestin (Fig. 1C, D). There was no expression of neuronal differentiation markers (MAP2, MAP1b) or glial cell markers (GFAP, NG2, and O4) (data not shown). These results suggest the existence of stem cells with NC molecular properties in hPDL tissue and in hPDL-derived cells.

Analysis of the expression pattern and genetic modification of cultured hPDL-derived cells. (A) mRNA expression of stem cell and neural crest markers in human periodontal ligament (hPDL) tissue and in hPDL-derived cells determined by RT-PCR. –/+, absence or presence of reverse transcriptase, respectively. hPDL tissue mRNA expression are shown at the top and hPDL-derived cells mRNA expression are show at the bottom. (B) Western blot analysis of hPDL-derived cells in basal media stained to detect nestin. Blots were also double-stained for βb-actin, as a loading control. (C) Immunocytochemical images showing the expression of cluster of differentiation 44 (CD44), CD90, low-affinity neurotrophin receptor (p75NTR), and nestin of hPDL-derived cells in basal media. DAPI nuclear staining is also shown. Scale bar: 100 μm. (D) Percentage of CD44+, CD90+, p75NTR +, and nestin+ cells in hPDL-derived cells in basal media as determined by immunocytochemistry. Data represent mean±SEM (n = 3). (E) Stability of enhanced green fluorescent protein (eGFP) overexpression in hPDL-derived cells. Photomicrographs show phase-contrast (PhC) and fluorescence images of eGFP+ transfected cells in basal media (24 h and 21 days after transfection and selection) and neural induction media (10 days). Scale bar: 100 μm. (F) Percentage of eGFP+ cells in basal media (24 h and 21 days after transfection and selection) and in neural induction media (10 days). Data represent mean±SEM (n = 3). (G) Transgenic eGFP overexpression: colony-forming cell population. An example of colony-forming cultures derived from hPDL-transfected cells stained with eGFP and nestin. DAPI nuclear staining is also shown. Scale bar: 100 μm. OCT3/4, octamer binding transcription factor 3/4; MLS1, musashi 1; Sox2, sex-determining region Y box 2.
Transgenic eGFP Overexpression
As noted in the introduction, this work aims to evaluate the potential of hPDL-derived cells for their use in gene therapy and cell replacement studies. The development of optimal gene marking procedures for hPDL-derived cells is crucial for analyze cellular behavior and improve the success of possible therapeutic strategies. We tested whether hPDL-derived cells could be conveniently modified in culture for the expression of marker gene eGFP. hPDL-derived cells were transfected by electroporation using the p(eGFP)IRES1hyg plasmid vector (as described in Materials and Methods). Twenty-four hour after transfection, fluorescence was visible in 27.08±1.37% cells (Fig. 1E, F). eGFP transfected cells, after 1 month of continuous drug selection, rendered vigorously growing cultures in which 81.75±1.65% cells were fluorescent under the microscope (Fig. 1E, F). Importantly, no morphological or proliferative changes were observed in the fluorescent hPDL-derived cells for prolonged (months) periods of time. Then, it was required that hPDL-derived cells retain their fluorescent properties after differentiation. For it, stably transfected eGFP-expressing hPDL-derived cells were exposed to serum-free neural induction media. The number of fluorescence cells after 10 days in neural induction media cultures was 74.86+1.59% (Fig. 1E, F). Also, we decided to test if cellular subclones could be isolated from eGFP+ hPDL cultures (in basal media) by limiting dilution. Of 58 clones isolated, 51 were fluorescent under the microscope 26 days following isolation. These subclones expressed Nestin (Fig. 1G). The results indicate that DNA transfection by electroporation in hPDL-derived cells allows for stable expression of marker gene eGFP. In addition, hPDL-derived cells are suitable for gene therapy carriers since isolation of clonal cells with homogenous properties is feasible.
Generation of PDL-Derived Spheres From PDL Cell Culture
Neurosphere derivation has been used as an in vitro test to define the persistence of NSCs in neural tissues (24,39). Free-floating spheres were generated from eGFP+ hPDL-derived cells cultured by applying serum-free neural induction media (as described in Materials and Methods). Most of the cells were observed floating in the medium after 1 day in culture. The single cells formed small spheres within a few days (3–4 days), which continued to grow after 1 week in culture (Fig. 2A). Free-floating spheres were collected and seeded onto poly-l-lysine-coated multi chambers (Fig. 2B). As a result, the spheres attached and formed a monolayer culture (Fig. 2C, D). It is important to mention that the cells retained their fluorescent properties after forming the spheres. The results indicate that it is possible to generate neurospheres from hPDL-derived cells by applying serum-free culture medium in the presence of bFGF and EGF.

Morphological changes during neural induction in hPDL-derived cells. Adding neural induction media to eGFP+ hPDL cell cultures resulted in the cells detaching from the culture dish and form free floating spheres (A). The hPDL-derived spheres were later collected and seeded into poly-l-lysinecoated multichambers (B), where they attached and proliferated forming a monolayer (C, D). DAPI nuclear staining is also shown. Scale bar: 100 μm. (E) Microphotographs of eGFP+ hPDL-derived cells before and after neural induction. hPDL-derived cells presented a flat and polygonal morphology in basal media. After exposure to neural induction media, hPDL-derived cells developed a spherical cell body with bipolar or multipolar processes, forming part of the dense and complex network of processes. Scale bar: 100 μm.
Neural Differentiation Potential of hPDL-Derived Cells
After exposure to neural induction media, hPDL-derived cells dramatically changed their morphology, giving rise to a variety of morphologies, including unipolar, bipolar, and multipolar cells of variable branch complexity (Fig. 2E). To study the neural differentiation potential of the hPDL-derived cells, cultures were maintained for 21 days in neural induction media. Immunocytochemical studies were performed to characterize in the hPDL-derived cells different neural phenotypes (Fig. 3). A percentage of the cells (39.66+3.22%) expressed neuronal marker β-III tubulin (TUJ1 antibody) (Fig. 3A, C). In addition, we also detected cells positive for microtubule-associated protein 1B (MAP1B), microtubule-associated protein 2 (MAP2), neuronal specific nuclear protein (NeuN), γ-aminobutyric acid (GABA), glutamic acid decarboxylase (GAD65-67), high-affinity GABA plasma membrane transporter (GAT1), choline acetyltransferase (ChAT), and synaptophysin (Fig. 3A). Furthermore, approximately 25% showed glial markers (13.85±1.59% of hPDL-derived cells) were immunopositive for GFAP (Fig. 3B, C), while a similar percentage (13.05±2.15%) were NG2 positive (Fig. 3B, C). Moreover, hPDL-derived cells were immunopositive for another glial marker, the surface antigen O4 (Fig. 3B). The results described above show that hPDL-derived cells have the ability to differentiate in vitro into neural-like cells: 40% neurons, 25% glial cells, and the remaining cells are nestin-positive undifferentiated cells (Fig. 3D).

Neural markers gene expression of hPDL-derived cells in neural induction media. Immunocytochemical images depicting neuronal (A) and glial (B) markers in eGFP+-overexpressing hPDL-derived cells after 21 days in neural induction media. DAPI nuclear staining is also shown. Scale bar: 100 μm. (C) Percentage of β-III tubulin (TUJ1)+, glial fibrillary acidic protein (GFAP)+, and chondroitin sulfate proteoglycan (NG2)+ cells in hPDL-derived cells after 21 days in neural induction media as determined by immunocytochemistry. Data represent mean+SEM (n = 3). (D) Immunocytochemical images showing the expression of nestin in eGFP+-overexpressing hPDL-derived cells after 21 days in neural induction media. DAPI nuclear staining is also shown. Scale bar: 100 μm. Abbreviations: MAP1b, microtubule-associated protein 1b; NeuN, neuronal nuclei; GABA, γ-aminobutyric acid; GAD 65/67, glutamate decarboxylase; GAT1, high-affinity GABA plasma membrane transporter; ChAT, choline acetyltransferase; O4, oligodendrocyte marker 4.
In Vivo Experiments
We assessed the in vivo potential of hPDL-derived cells after transplantation into the adult mouse brain. Stably eGFP transfected hPDL-derived cells cultured for 21 days in neural induction media were grafted in the hippocampus of seven 8-week-old immune-suppressed mice, and the animals were histologically analyzed 1 month later (Fig. 4). All of the seven animals showed surviving grafts at the time of the analysis, as determined by the presence of the large mass of eGFP-positive cells in the dorsal lamina of the dentate gyrus (DG) in the hippocampus (Fig. 4A, K, LL, O), as well as several dispersed fluorescent cells were detected through the needle trajectory in the dorsal cortex and CA1 (Fig. 4A, E, Q). The cells were also detected in other areas, including the corpus collosum, fimbria (Fig. 4G), neighboring epithelium of lateral ventricles (Fig. 4A–D, F, J, L, M, N, R), and the caudal pole of subventricular zone (Fig. 4A, H, P). A detailed histological analysis revealed no signs of transplant-induced tissue damage or uncontrolled cell growth, ectopic or abnormal cellular masses, in any of the transplanted animals. The expression of eGFP in hPDL-derived cells allowed, for the first time, the evaluation of the integration, migration, and differentiation of the cells, with a cellular morphological resolution (Fig. 5). Grafted cells that populated the ventricular epithelium presented epithelial cell morphology (Fig. 4J, L, M), in some cases even presenting radial-oriented extensions into the subventricular zone (Fig. 4D, L, R). hPDL-derived cells leaving the grafts showed bipolar morphology, suggesting a migration from the graft, integrated close to the subventricular zone, towards distal regions of host DG lamina, throughout hippocampus cellular and molecular layers (Fig. 5A). Some of the grafted hPDL-derived cells also appeared to persist in stem cell niches, the subventricular zone (SVZ), and the subgranular zone (SGZ) of the dentate gyrus (Figs. 4H, P and 5A). eGFP-hPDL-grafted cells showed typical morphology of neural stem cells in the subgranular zone of the DG (Figs. 4LL and 5A), described in Yang et al. (45). This might indicate that those cells could respond to either molecular signals for migration and differentiation and local survival cues. Immunohistochemistry studies revealed expression of neural markers in eGFP+ hPDL-grafted cells (Fig. 5B) Dcx, NF-M, and GABA as well as glial markers GFAP and NG2. These observations demonstrate that hPDL-derived cells survive, integrate, migrate, and generate neural-like cells in the hippocampus of adult mouse brain. Since Fgfr1 is a good marker of subgranular neuronal progenitors during development and adult brain (11) (Fig. 6), being expressed in migrating neural crest cells during development (Fig. 6B). Interestingly, this receptor was also detected in hPDL-derived cells (Fig. 6A), which could represent the target receptor for the potential FGF-mediated attractive signal from the DG cells that could drive the active migration of SGZ precursors in physiological and experimental conditions (Fig. 6B–E). Thus, Fgf1 expressed in DG neurons could represent an attractive signal in subventricular precursors to migrated into the subgranular layer and differentiate as granular stem cells.
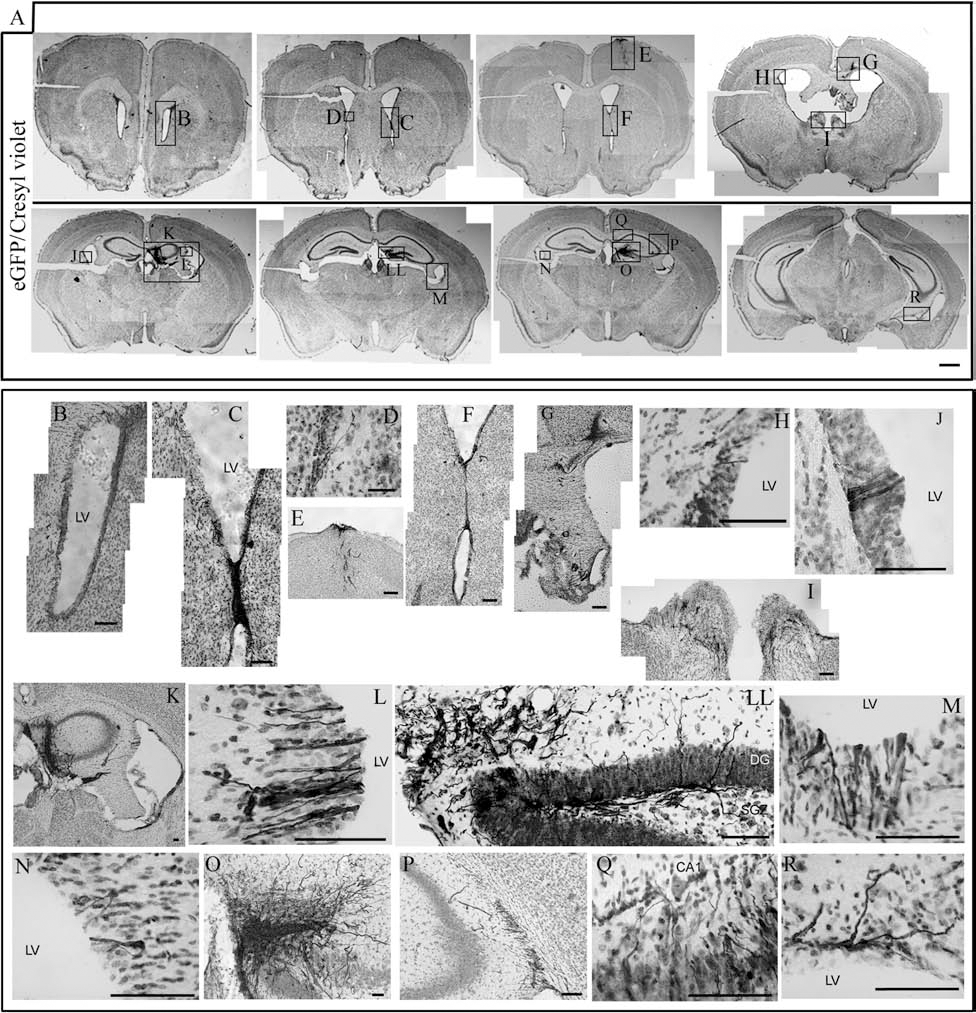
Transplantation of neural-differentiated eGFP+ hPDL-derived cells into the adult mouse hippocampus. (A) Integration and migration of hPDL-grafted cells into the adult mouse brain after 1 month of transplantation. Photomicrographs of coronal sections showing representative transplants stained for eGFP and cresyl violet. Scale bar: 1,000 μm. (B–R) Magnified views of graft region from (A). Scale bar: 100 μm. LV, lateral ventricle; DG, dentate gyrus; SGZ, subgranular zone; CA1, CA1 area of the host's hippocampus.

Morphology and neural marker expression of hPDL-grafted cells. (A) Immunohistochemical analysis of different regions of the brain where hPDL-derived cells were detected. Scale bar: 100 μm. (B) Brain slices were stained for eGFP and doublecortin (DCX), GFAP, NG2, neurofilament-medium (NF-M), and GABA. DAPI nuclear staining is also shown. Orthogonal projections confirm the colocalization of eGFP and neural markers. Scale bar: 20 μm. LV, lateral ventricle; OR, oriens layer of the hippocampus; Py, pyramidal cell layer of the hippocampus; DG, dentate gyrus; SGZ, subgranular zone; CA2, CA2 area of the host's hippocampus.

Expression of Fgfr1 in PDL developing cells and SGZ in adult mouse brain. (A) mRNA expression of fibroblast growth factor receptor 1 (Fgfr1) in hPDL-derived cells cultured in basal media determined by RT-PCR. –/+, absence or presence of reverse transcriptase, respectively. (B) Tooth development at bell stage (E14.5), sagittal section showing Fgfr1 expression in the dental mesenchyme cells (dm) (from Eurexpress database: www.eurexpress.org; T31230; section 14). (C) In the developing medial pallium at E14.5, the dentate gyrus region (DG) in the hippocampal mantle layer presents a strong expression of Fgfr1 (from Eurexpress database: www.eurexpress.org; T31230; section 11). (D, E) Sagittal sections of P28 mouse hippocampus showing the complementary localization of Fgfr1 expression in subgranular zone (SGZ) cells (D) and Fgf1 in the DG cells (E) (Allen Developing Mouse Brain Atlas [Internet]. Seattle, WA: Allen Institute for Brain Science. ®2009. Available from: http://developingmouse.brain-map.org. Image Series 100040785; section 14 for D, and Image Series 100056893; section 10 for E). de, dental epithelium; DG, dentate gyrus; dl, dental lamina; dm, dental mesenchyme; GCL, granular cell layer; Hi, hippocampus; ML, molecular layer; Th, thalamus.
Discussion
The use of adult stem cells in regenerative medicine is promising since certain types are easy to obtain and isolate and can be expanded in vitro (5,15,28). In addition, immunosuppressive treatments are not required in autologous transplantations, making autologous cells ideal candidates for clinical trials (21,33). Several recent studies have shown that neural crest-derived stem cell can be isolated from different adult tissues (19,40,43,44). The isolation and manipulation of autologous neural crest-derived stem cell from human periodontal ligament (hPDL) might represent an accessible source of adult neural stem cells for their use in cell replacement therapies and gene transfer to the diseased central nervous system.
In the present study, we found the expression of several stem cell and neural crest cell markers in hPDL tissue and hPDL-derived cells. Under proliferation conditions, hPDL-derived cells express the surface proteins CD44 and CD90 [stem cells derived from human dental pulp also express these proteins (1)], the neural stem cell marker nestin, and the neural crest cell marker p75NTR. Under neural induction culture conditions, hPDL-derived cells can also proliferate as neurosphere-like clusters and differentiate mainly into neuron-like cells (approximately 40% of cells were TUJ1+, as well as other neuronal markers, like MAP2, MAP1b, GAD65/67, GABA, NeuN, ChAT, GAT, and synaptophysin at heterogeneous proportions), as well as astrocyte-like cells (GFAP; 14% approximately) and oligodendrocyte-like cells (NG2; 13% approximately) and O4. It is important to mention that hPDL-derived cells not only expressed neural markers but also dramatically changed their morphology, mainly neuronal-like cells, developing spherical-shaped cell bodies with bipolar or multipolar processes forming part of the dense and complex network of processes. In vivo, grafted hPDL-derived cells survive after transplantation, integrate, migrate, and differentiate into DCX+, NF-M+, GABA+, GFAP+, and NG2+ cells in the adult mouse brain. The grafted hPDL-derived cells were mainly detected in the dentate granular cell layer, as well as in the hippocampal CA1, CA2, and CA3 cell layers, 1 month after transplantation. Moreover, some of the grafted hPDL-derived cells also appeared to persist in stem cell niches such as the ventricular epithelium and the SVZ of the anterolateral ventricle wall as well as the SGZ of the hippocampal dentate gyrus. Previous works suggest that neural stem cells in the SVZ correspond to a population of astrocytes that contain only one or two primary processes (12,34) while hippocampus neuronal precursors have their cell bodies in the SGZ, with a radial process that goes through the granular cell layer and sort tangential processes extending along the SGL under the blades of the dentate gyrus (2). Interestingly, the hPDL-derived cells located in the stem cell niches showed neural stem morphology, supporting that the hPDL contain stem cells that originate from the neural crest (9,19). Although previous data have already indicated a ventricular origin of the SGZ, originating in hippocampal ventricular epithelium during embryonic and early postnatal stages (30), migration guidance mechanisms have not been suggested for this cellular stream. The expression patterns of Fgf1 and Fgfr1 strongly suggests that FGF could represent the signal that acting from the DG neurons attracts SGZ migrating precursors by binding Fgfr1 receptors. These receptors have been also evidenced in hPDL-derived cells, strongly suggesting that engrafted cells move to the SGZ recapitulating physiological developmental mechanisms.
Previous studies suggest that survival and neural differentiation of grafted mesenchymal stem cells may be dependent upon microenvironmental conditions according to the site of implant in the brain (23). hPDL-derived cells could have also niche dependent survival and differentitation, but since we have grafted hPDL-derived cells always in the same brain area we have not observed significant differences in cell survival and cellular differentiation patterns. Transplants of hPDL-derived cells cultured before and after neural induction media in neurogenic and nonneurogenic adult mouse brain regions will be performed in the future to determine whether or not they develop different behavior.
We have also tested if PDL-derived cells could be conveniently modified in culture with nonviral vectors for the expression of foreign gene product. Downregulation of the gene expression in vivo has been a general problem for ex vivo gene therapy applications, particularly with the use of engineered stem cells (18). In the present study, we demonstrate that DNA transfection by electroporation in hPDL-derived cells allows stable expression of eGFP. Downregulation both in vitro and in vivo was not observed. Stably expressing eGFP+ hPDL-derived cells subclones could be isolate thus allowing the isolation of clonal cells with homogenous properties, considered to be valuable properties for a potential clinical product (25,35). The results described above indicate that hPDL-derived cells also seem to be an attractive tool for the stable transfer of foreign genes or application of growth factor-expressing cells within the central nervous system.
In summary, we have identified a population of multipotent stem cells in the hPDL originating from the neural crest. The engraftment and differentiation properties of hPDL-derived cells in the adult brain indicate that hPDL-derived cells are a potential alternative for the treatment of neurodegenerative diseases, although future research is required to confirm this possibility.
Footnotes
Acknowledgments
This work was supported by the grants EUCOMMTOOLS (Contract 261492), Spanish MICINN BFU-2008-00588, CONSOLIDER (CSD2007-00023), Institute of Health Carlos III, TERCEL and CIBERSAM, Generalitat Valenciana (PROMETEO 2009/028 and 11/2011/042), and Alicia Koplowitz Foundation. The authors declare no conflicts of interest.
