Abstract
Prediction of islet yield and posttransplant outcome is essential for clinical porcine islet xenotransplantation. Although several histomorphometric parameters of biopsied porcine pancreases are predictive of islet yield, their role in the prediction of in vivo islet potency is unknown. We investigated which histomorphometrical parameter best predicts islet yield and function, and determined whether it enhanced the predictive value of in vitro islet function tests for the prediction of posttransplant outcome. We analyzed the histomorphometry of pancreases from which 60 adult pig islet isolations were obtained. Islet function was assessed using the β-cell viability index based on flow cytometry analysis, oxygen consumption rate, ADP/ATP ratio, and/or concurrent transplantation into NOD/SCID mice. Receiver operating characteristic (ROC) analysis revealed that only islet equivalent (IEQ)/cm2 and the number of islets >200 μm in diameter significantly predicted an islet yield of >2000 IEQ/g (p < 0.001 for both) and in vivo islet potency (p = 0.024 and p = 0.019, respectively). Although not predictive of islet yield, a high proportion of large islets (>100 μm in diameter) best predicted diabetes reversal (p = 0.001). Multiple regression analysis revealed that the β-cell viability index (p = 0.003) and the proportion of islets >100 μm in diameter (p = 0.048) independently predicted mean posttransplant blood glucose level (BGL). When BGL was estimated using both these parameters [area under the ROC curve (AUC), 0.868; 95% confidence interval (CI), 0.730-1.006], it predicted posttransplant outcome more accurately than the β-cell viability index alone (AUC, 0.742; 95% CI, 0.544-0.939). In conclusion, we identified the best histomorphometric predictors of islet yield and posttransplant outcome. This further enhanced the predictive value of the flow cytometry analysis. These parameters should be useful for predicting islet yield and in vivo potency before clinical adult porcine islet xenotransplantation.
Introduction
Prediction of islet yield and posttransplant outcome in advance of islet transplantation is essential for the clinical application of porcine islet xenotransplantation. Although several histomorphometric parameters of biopsied porcine pancreases have been reported to predict islet yield, variation inherent to the qualitative nature of analysis of islet shape and demarcation has limited the widespread use of these parameters in preclinical trials on islet xenotransplantation (7,18,19). By contrast, a retrospective study suggested that the total number of islets and the proportion of large islets (>100 μm in diameter) may be useful quantitative parameters (4). However, this finding needs to be confirmed because only 18 pancreases were evaluated. In addition, the best cut-off value for the diameter of large islets is controversial because another study (15) demonstrated that pancreases that contain mainly large islets (>200 μm in diameter) yield more islets than pancreases that contain mainly small islets. Mean islet volume density, another quantitative histomorphometric parameter (13), did not predict islet yield in a recent study (12). Therefore, it is unclear which quantitative histomorphometric parameter best predicts islet yield.
More importantly, the relationship between histomorphometric parameters and in vivo islet potency has not been evaluated. Given that small islets function better under rat (17) and human (16) allogeneic islet transplantation conditions than large islets do, the utility of histomorphometric parameters associated with large islets should be reevaluated. Furthermore, most in vitro islet function tests for predicting posttransplant outcomes have only been evaluated for human islets (6,9,21,26) and when porcine islets have been studied, concurrent histomorphometric analysis of the porcine pancreas was not performed (8,14). For this reason, it is unclear whether the predictive value of in vitro tests for porcine islet xenotransplantation would be enhanced by consideration of histomorphometric parameters. Therefore, we identified the quantitative histomorphometric parameters of biopsied porcine pancreases that best predict islet yield and potency and determined whether these parameters could enhance the predictive value of in vitro islet function tests for the prediction of posttransplant outcomes.
Materials and Methods
Animals
The donor pigs included adult market pigs from a local slaughterhouse (n = 39), specific pathogen-free (SPF) Seoul National University (SNU) miniature pigs (12,13) bred in a barrier-sustained SPF facility (n = 8), Prestige World Genetics (PWG) miniature pigs (n = 12; Prestige World Genetics Korea Co. Ltd, Kyoungki-Do, Korea), and a transgenic pig with HLA-G genes (MGEN, Seoul, Korea). Donor characteristics are summarized in Table 1. For assessment of in vivo islet potency, we used NOD/SCID mice (Jackson Laboratories, Bar Harbor, ME, USA) of up to 27 weeks of age. All animal procedures were approved by the Institutional Animal Care and Use Committee of the Seoul National University Hospital.
Baseline Donor Pig Characteristics *

PWG, PWG miniature pig; SNU, Seoul National University miniature pig; Farm, adult market pig; IEQ, islet equivalent.
A transgenic pig with HLA-G genes (n = 1) is not included in this table.
t-Test, chi-square test, or one-way analysis of variance with post hoc comparison using Tukey's multiple comparison test, as appropriate.
Virgin female SNU miniature pig (n = 4).
The rate of achieving diabetes reversal among the assessed islet isolation cases.
The same letters indicate nonsignificant difference between groups based on Tukey's multiple comparison test.
Pancreas Procurement and Islet Isolation
For SNU miniature pigs, PWG miniature pigs, and the transgenic pig with HLA-G genes, a total pancreatectomy was performed without warm ischemia time as described previously (12). For adult market pigs, the splenic lobe of the pancreas was harvested at the local slaughterhouse. Before transport, cold SNU solution (24), which is a modified University of Wisconsin solution, was injected intraductally (20). After transportation, islet isolation was performed using the modified Ricordi method (12). Briefly, the harvested pancreas was distended intraductally using a preservation solution containing 3.3 mg/ml Liberase PI (Roche Biochemicals, Basel, Switzerland) at a rate of 1.0 ml of preservation solution per gram of tissue. The distended pancreas was digested in a modified Ricordi chamber at 35–37°C. Liberated islets were purified using a continuous Opti-Prep density gradient (Axis-Shield, Oslo, Norway) and a Cobe 2991 cell separator (Gambro BCT Inc., Lakewood, CO, USA). The islets were then cultured using the free-floating method in Medium 199 (Gibco, BRL, Life Technologies Ltd, Paisley, Scotland, UK) supplemented with 10% porcine serum at 37°C. The medium contained 1 mM calcium nitrate (Sigma Chemicals, St. Louis, MO, USA) and 10 mM nicotinamide (Sigma Chemicals) (8). The medium also contained 1 μM of alkaline hydrolysis-activated (3) simvastatin (kindly donated by Pacific Pharma, Seoul, Korea) because a preliminary experiment showed that inclusion of this reagent in the culture medium reduces apoptosis of porcine islets.
Histomorphometric Analysis
A small piece of tissue was obtained from the junction of the duodenal and splenic lobes of the pancreas (5) (Fig. 1A). Three frozen sections were stained with dithizone. Images of entire nonoverlapping areas 1 cm2 in area were captured for each section using a digital zoom camera (TS100; Nikon, Tokyo, Japan) connected to a microscope (TE2000-U; Nikon). Images were analyzed using i-Solution software (IMT Inc., Daejeon, Korea). The mean diameters of islets were estimated from the largest and smallest widths of all islets in the images (13).

The anatomical location in the pancreas from which the frozen sections were obtained. A small section of tissue at the junction of the duodenal and splenic lobes, indicated by a circle, was cut and biopsied (A). Representative images of dithizone-stained islets in a frozen section of donor pancreas (B, C). Scale bars: 150 μm. Original magnification: 40x.
For each image, dithizone-stained islets (Fig. 1B) with a diameter greater than 50 μm were counted and sorted according to mean diameter. The number and size of each islet were converted to the islet equivalent (IEQ) using the algorithm used to determine the IEQ after digestion and purification of islets (23). The sum of the IEQs was then divided by the total area of the section included in the image analysis to obtain the IEQ per area of donor pancreas (IEQ/cm2). The number and proportion of islets larger than 100 and 200 μm in diameter were estimated from the mean diameter.
Islet Potency Test
An in vivo islet potency test was performed using islets isolated from 30 pancreases. After overnight culture, 2500 IEQs of isolated islets per recipient were transplanted under the renal capsules of two to four streptozotocin-induced diabetic NOD/SCID mice. To estimate the 2500 IEQ, 30,000 islet equivalent (IEQ) were suspended in 30 ml of Medium 199, and then four 2.5ml aliquots of the suspended islets were taken. Islets in each aliquot were allowed to settle for 10 min and the islet pellet was then transplanted to the mice. We chose this method so that the islet size distribution of the islet grafts would coincide with that of the isolated islets. In parallel, we subjected a 500-IEQ aliquot, estimated using the same method, to DNA quantification using Pico Green dsDNA probe reagent (Molecular Probes Eugene, OR, USA) to confirm the same islet equivalent had constant cell number. Mice were regarded as diabetic when two consecutive blood glucose levels were higher than 300 mg/dl.
In this study, in vivo islet potency was expressed in terms of diabetes reversal for the categorical analysis and in terms of mean posttransplant blood glucose level for the quantitative analysis. We defined diabetes reversal as the presence of all of the following conditions in at least one mouse per group: (A) two consecutive blood glucose levels of less than 200 mg/dl within 10 days of islet transplantation, (B) normoglycemia for longer than 8 weeks, (C) an area under the curve (AUC) for an intraperitoneal glucose tolerance test (28) conducted 8 weeks after islet transplantation of less than 1000 mmol/min, and (D) reversion to hyperglycemia (>300 mg/dl) when the graft-bearing kidney was removed. In this study, mean posttransplant blood glucose level was defined as the mean of random blood glucose levels (mg/dl) obtained on days 1 and 56 after islet transplantation.
In vitro islet potency tests were performed concurrently with the in vivo islet potency tests. We screened three tests that have been reported to be predictive of posttransplant outcome and can be performed in <4 h: measurement of the oxygen consumption rate by the rdO2 index obtained with a BD oxygen biosensor system (BD Biosciences, San Jose, CA, USA), which uses an oxygen-sensitive fluorophore (6), ADP/ATP ratio using the ApoGlow kit (Lonza Rockland Inc., Rockland, ME, USA) (8), and fractional β-cell viability based on flow cytometry (9). All in vitro islet potency tests were performed as previously described (6,8,9), except that FluoZin-3 (Invitrogen, Carlsbad, CA, USA) was used as a β-cell indicator and the gating strategy was modified as described in a previous study on human islets (11). Because the porcine islets did not exhibit a bimodal distribution after exposure to the zinc-binding fluorochrome, Newport Green, we gated FluoZin-3-positive cells and then determined the proportion of tetramethylrhodamine ethyl ester (TMRE; Invitrogen)-positive cells. The cutoff value for positive staining with FluoZin-3 was determined at a fluorescence intensity corresponding to the 99.5% percentile of unstained samples because this percentile correlated best with concomitant insulin staining results in our preliminary experiments. The β-cell viability index was defined as the percentage of FluoZin-3-positive TMRE-positive cells/100.
Statistical Analysis
All values are expressed as the mean ± SD. The predictive values of each indicator and each in vitro islet potency test were compared using receiver operating characteristic (ROC) analysis. Means were compared using t-tests or one-way analysis of variance followed by Tukey's multiple comparison test. Categorical variables were compared using the chi-square test or Fisher's exact test as appropriate. Linear regression analysis was performed to identify correlations between continuous variables. Multiple regression analysis was performed to identify independent predictors of mean posttransplant glucose level and to estimate mean blood glucose level from the predictors. A probability of p < 0.05 was considered statistically significant. All analyses were performed using SPSS version 12.0K software (SPSS Inc., Chicago, IL, USA).
Results
IEQ/cm2 and Number of Very Large Islets (>200 μm in Diameter) Were the Best Predictors of Islet Yield
Of the four histomorphometric parameters evaluated, only IEQ/cm2 and the number of very large islets (>200 μm in diameter) significantly predicted both islet yield and in vivo potency in terms of diabetes reversal (Table 2, Figs. 2 and 3).
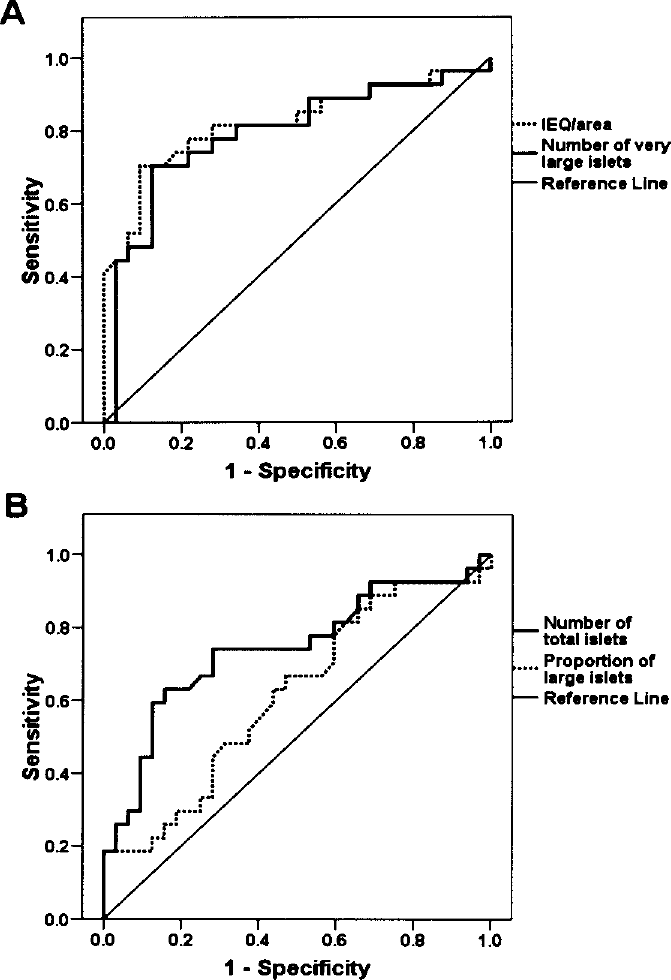
Receiver operating characteristic curves for each of the histomorphometric parameters for prediction of islet yield >2000 islet equivalents (IEQs)/g. (A) IEQ/area and the number of very large islets (>200 μm in diameter). (B) The total number of islets and the proportion of large islets (>100 μm in diameter). The area under the curve is specified in Table 2.

Receiver operating characteristic curves for each histomorphometric parameter used to predict diabetes reversal in NOD/SCID mice transplanted with porcine islets. (A) The number of very large islets (>200 μm in diameter) and islet equivalent (IEQ)/area. (B) The proportion of large islets (>100 μm) and the total number of islets. The area under the curve is specified in Table 2.
Comparison of the Predictive Value of Each Histomorphometric Parameter for Islet Yield >2000 IEQ/g and In Vivo Islet Potency in Terms of Diabetes Reversal

IEQ, islet equivalent; ROC, receiver operating characteristic; CI, confidence interval.
An area under the ROC curve of 1 indicates 100% correct classification and an area under the ROC curve of 0.5 indicates that there is no predictive relationship.
>200 μm in diameter.
>100 mm in diameter.
Porcine pancreases with ≥ 100 IEQ/cm2 yielded a higher total IEQ (normalized by donor pancreas weight) and islet recovery rate after purification and exhibited no impairment of islet survival rate after overnight culture. Moreover, the diabetes reversal rate in NOD/SCID mice transplanted with the islets was higher when the islets were from pancreases for which the IEQ/cm2 was ≥ 100 (p = 0.028) (Table 3). A similar result was obtained with the number of very large (>200 μm in diameter) islets (data not shown). When analyzed in the adult market pig subgroup (n = 39), IEQ/cm2 (AUC 0.868, 95% CI 0.756–0. 979, p < 0.001) and the number of very large (>200 μm in diameter) islets (AUC 0.831, 95% CI 0.699-0.962, p < 0.001) still remained a significant predictor of islet yield higher than 2000 IEQ/g.
Effect of Islet Equivalent (IEQ) per Area of Biopsied Donor Pancreas on Islet Yield and Potency

IEQ/g, islet equivalent per gram of pancreas.
Islet mass expressed as IEQ after purification divided by islet mass before purification.
IEQ after overnight culture divided by IEQ immediately after purification.
Glucose-stimulated fold increase in oxygen consumption rate measured using a microplate containing an oxygen-sensitive fluorophore.
The rate of achieving diabetes reversal among the assessed islet isolation cases.
A High Proportion of Large Islets (>100 μm in Diameter) Was the Best Histomorphometric Predictor of Diabetes Reversal
Although not significantly predictive of islet yield, a high proportion of large islets (>100 μm in diameter) was the best predictor of diabetes reversal of the four histomorphometric parameters evaluated (Table 2, Fig. 3). The predictive value of this parameter remained significant (AUC, 0.790; 95% CI, 0.602–0.977, p = 0.021) when analyzed in the adult market pig subgroup (n = 23).
To exclude confounding effects, we compared islets transplanted from pancreases with a large islet content greater than or less than 50.55% (Table 4). The DNA contents of 500 IEQs were quantified in parallel for every transplantation; mean DNA contents were associated with very low standard deviations and did not differ between groups (Table 4). The islet size distribution of transplanted islets did not differ between groups. There was a difference of borderline significance (p = 0.063) in isolation index (islet mass expressed as IEQ divided by islet mass expressed as the actual number of islets) (Table 4). However, despite the presence of correlations between mean posttransplant blood glucose level and the proportion of large islets (>100 μm diameter) (p = 0.012) and the isolation index for donor pancreases (p = 0.012) (Fig. 4A, B), the isolation index of isolated islets that were cultured overnight was not correlated with mean posttransplant blood glucose level (p = 0.920) (Fig. 4C). Therefore, posttransplant in vivo islet potency was predicted by the islet size distribution of biopsied pancreases, but not by that of isolated islets.
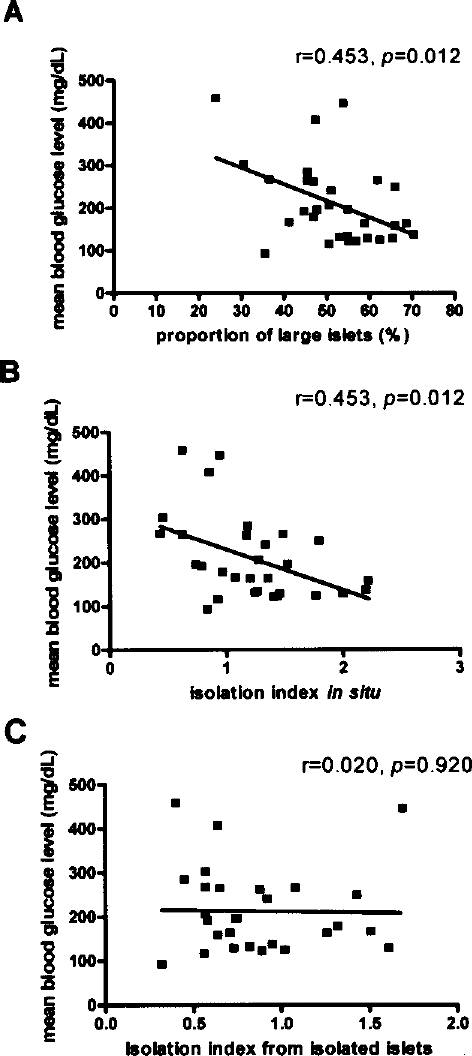
The proportion of large islets (>100 μm in diameter) (A) and the isolation index (B) from biopsied donor pancreases are correlated with mean posttransplant blood glucose level, whereas the isolation index of overnight cultures of isolated islets is not (C). The isolation index was calculated as islet mass expressed as an islet equivalent divided by islet mass expressed as the actual count of islets.
Effect of the Percentage of Large Islets (>100 μm in Diameter) in Biopsied Donor Pancreases on Islet Yield, Islet Potency, and the Characteristics of Transplanted Islets

IEQ/g, islet equivalent per gram of pancreas.
Estimated from the characteristics of overnight cultures of isolated islets from which the transplanted islets were derived.
IEQ after overnight culture divided by IEQ immediately after purification.
IEQ divided by actual total number of islets.
The Proportion of Large Islets (>100 μm in Diameter) Enhanced the Accuracy of an In Vitro Islet Function Test for Predicting Posttransplant In Vivo Islet Potency
To identify the most appropriate in vitro islet function test for porcine islets, we screened the following tests for predictive value: the β-cell viability index based on flow cytometry, the rdO2 index for estimating the glucose-stimulated increment in oxygen consumption ratio, and the ADP/ATP ratio. Of these tests, only the β-cell viability index was significantly associated with in vivo islet potency in terms of mean posttransplant blood glucose level (p = 0.001) (Fig. 5).
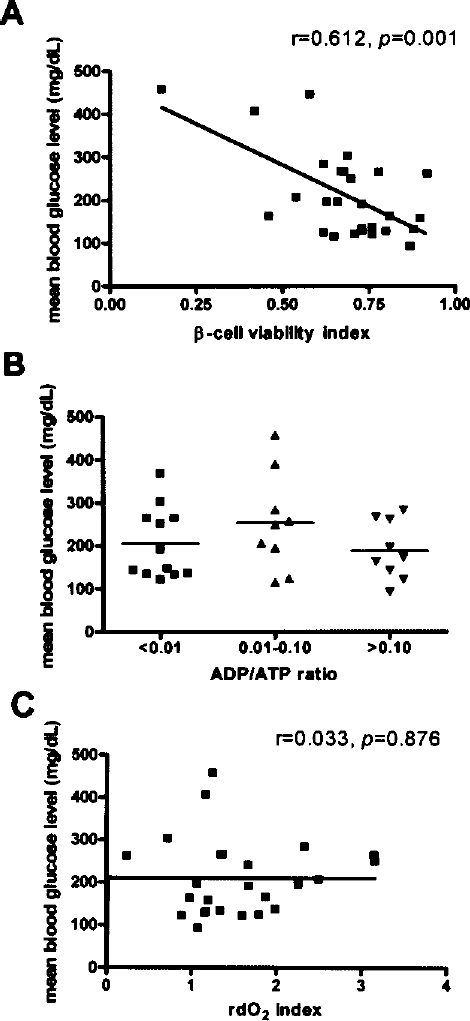
The β-cell viability index based on flow cytometry was correlated with mean posttransplant blood glucose level (A), whereas ADP/ATP ratio (B) and the rdO2 index (C) were not. The β-cell viability index is expressed as the proportion of FluoZin-3-positive tetramethylrhodamine ethyl ester-positive cells. The rdO2 index is expressed as the glucose-stimulated fold increase in DNA-normalized oxygen consumption rate estimated using an oxygen-sensitive fluorophore.
Because the β-cell viability index (p = 0.003) and the proportion of large islets (>100 μm in diameter) (p = 0.048) were both independent predictors of mean posttransplant blood glucose level during the first 8 weeks after islet transplantation, mean blood glucose level estimated from these two parameters was a more appropriate predictor of posttransplant outcome than the β-cell viability index alone. The following equation for predicting mean posttransplant blood glucose level (mg/dl) was derived from multiple regression analysis: 578.3 - 319.6 × β-cell viability index - 2.837 × proportion of large islets (%) (R2, 0.471; p < 0.001). The AUC of this parameter (0.868; 95% CI, 0.730–1.006; p = 0.001; 78.6% sensitivity and 84.6% specificity at the cutoff value of 200.6 mg/dl) was higher than that of the β-cell viability index alone (0.742; 95% CI, 0.544–0.939; p = 0.033; 78.6% sensitivity and 76.9% specificity at the cut-off value of 0.70) (Fig. 6A).

The mean blood glucose level estimated from the proportion of large islets and the β-cell viability index together was a better predictor of diabetes reversal (AUC, 0.868; p = 0.001) than the β-cell viability index alone (AUC, 0.742; p = 0.033) (A). Categorization with the proportion of large islets improved the predictive value of the β-cell viability index (B). The p-value was obtained using a one-way analysis of variance. Diabetes reversal was defined as a blood glucose level <200 mg/dl within 10 days that was sustained for more than 8 weeks.
The correlations between mean blood glucose level and the proportion of large islets (>100 μm in diameter) and between mean blood glucose level and the β-cell viability index (Figs. 4A and 5A) were strengthened by categorization of both parameters (Fig. 6B). The diabetes reversal rate was 9 out of 10 when β-cell viability index was >0.70 and the proportion of large islets (>100 μm in diameter) was ≥ 50.55%. However, the diabetes reversal rate was 0 out of 7 when the β-cell viability index was <0.70 and proportion of large islets (>100 μm in diameter) was <50.55% (Fig. 6B).
Discussion
Previous studies have indicated that a porcine pancreas composed mainly of large islets is a better donor pancreas because it yields more islets (4,15). Recently, this concept has been challenged by the observation that smaller islets in humans (16) and rats (17) are better in terms of islet potency. The conflict between the yield, function, and size of islets needs to be resolved. In this study, we showed that some quantitative histomorphometric parameters are predictive of islet yield and function. Moreover, the proportion of large islets was correlated with in vivo islet potency and inclusion of this variable enhanced the predictive value of flow cytometry analysis for posttransplant in vivo potency of islets.
The IEQ/cm2 and the actual count of islets larger than 200 μm in diameter had a higher predictive value for islet yield than that of previously proposed quantitative parameters such as total islet number and the proportion of islets of more than 100 μm in diameter (4). We initially speculated that histomorphometric variables associated with large islets might be inversely associated with in vivo islet potency because it has been proposed that small islets have better in vivo potency than large islets (16,17). However, the results of this study indicate that IEQ/cm2 and the number of very large islets (>200 μm in diameter) indeed predicted in vivo potency, in contrast to the previously suggested quantitative histomorphometrical parameter of total islet number (4). Another previously suggested quantitative parameter (4), the proportion of large islets (>100 μm in diameter), was the most precise predictor of in vivo islet potency, rather than a predictor of islet yield. Taken together, histomorphometric analysis of donor pancreases before islet isolation has much stronger predictive power for in vivo islet potency than other methods because the size distribution of overnight cultures of isolated islets alone is insufficient to predict in vivo islet potency (Fig. 4).
The results of this study contrast with those of previous studies that have demonstrated that small islets are superior to large islets in terms of in vivo potency (16,17). The main reason for this discrepancy may be that we did not compare small and large islets from the same pancreas but islets from different pancreases composed mainly of large or small islets. Another reason for this discrepancy may involve species differences in the fragility of islets. Because porcine islets are more fragile than human or rat islets (27), the degree of damage during the islet isolation process is a more important issue with porcine islets than with human or rat islets. Given that the islets of adult pigs are generally larger than those of young pigs (13,22), the results of this study are consistent with a previous study that showed that islets from adult pigs have better yield and function than those from young pigs because of less fragmentation during the islet isolation procedure (1). This finding is in contrast to the observation that the function of isolated human islets decreases with donor age (10).
Because female retired breeders have been suggested to be one of the most favorable factors in selecting islet donor (15), it is important to know whether this factor is related to certain histomorphometrical characteristics. However, the effect of this factor is not conclusive in this study because only four pigs were confirmed female retired breeders. Although some of the adult market pigs included in this study might have been retired female breeders, the probability of this occurring is very low considering the age of pigs that are slaughtered for meat production. Although the number of pigs was insufficient to make a definitive conclusion, the four virgin female SNU miniature pigs, which had a favorable pancreatic histomorphometry, had high islet yields. This indicates that pancreatic histomorphometry has stronger predictive power for islet yield than sex or age of the donor. This finding was unexpected because virgin female SNU miniature pigs generally have poor islet yields (12), as do most other pig strains. Experiments with large numbers of such pigs are required to confirm previous results (15) showing that histomorphometry is more predictive of islet yield than simple donor characteristics.
Although some quantitative histomorphometric parameters predicted islet function in this study, postisolation confirmation of islet function was warranted to evaluate damage induced by the isolation process. Therefore, we screened the predictive value of β-cell viability based on flow cytometry (9), ADP/ATP ratio (8), and the glucose-stimulated increment in oxygen consumption rate (OCR) measured using oxygen-sensitive fluorophores (6).
The ADP/ATP ratio and the OCR did not have significant predictive value in our screening test. For the ADP/ATP ratio, a negative value was frequently observed with the ApoGlow kit (Lonza), although we adhered strictly to the methods of a previous study (8). Previous authors have also reported that negative ADP/ATP ratios were frequently obtained using this kit (25). Although the manufacturer's insert states that a negative ratio is indicative of a low ADP content, we did not compare results obtained using this kit with high-performance liquid chromatography estimates of ADP/ATP ratio. For this reason, we could not determine whether our finding that the ADP/ATP ratio has no predictive value is an anomaly caused by erroneous estimates of ADP and ATP content. In addition, we failed to reproduce the predictive value of glucose-stimulated increases in OCR in human islets using oxygen-sensitive fluorophores (6). However, we are unable to determine whether this problem stems from species differences or technical error because we were unable to compare our estimated OCR values with estimates obtained using instruments that measure OCR directly (21,26). Despite these limitations, it is noteworthy that not all of the in vitro islet function tests predictive of the vivo potency of human islets are predictive of the in vivo potency of porcine islets.
We confirmed that β-cell viability based on flow cytometry (9), which has been evaluated only for human islets, also predicts posttransplant outcomes in porcine islets. In addition, we demonstrated that the modified gating strategy proposed in a study involving an in vitro model (11) also predicted posttransplant outcomes with our in vivo model. Because the non-β-cell fraction of porcine islets is less prominent than that of human islets (2), it was not possible to conduct gating using a bimodal distribution with zinc-binding dye staining (9). Therefore, we gated FluoZin-3-positive cells using a fixed cut-off percentile derived from an unstained sample to avoid subjectivity.
Our study showed that the predictive value of the β-cell viability index is high when it is concordant with predictions derived using histomorphometric analysis (17 of 27 cases). Therefore, prediction of in vivo islet potency using histomorphometric criteria in advance of islet isolation and its confirmation using an in vitro method such as the β-cell viability index (10,12) just before islet transplantation would ensure adequate in vivo islet potency in advance of islet transplantation. This would be of value for clinical islet xenotransplantation because the current gold standard, a bioassay using immune-deficient mice transplanted with the islets, cannot be used prospectively.
This study has several limitations. First, Liberase PI (Roche) is not appropriate for good manufacturing practice (GMP)-grade islet isolation. The results of this study could have been different if a GMP-grade collagenase blend had been used because isolation-related islet damage may have been different. Second, we used pancreases from four strains of pig. Although the results of the subgroup analysis using only adult market pigs remained similar, caution should be exercised in applying the results of this study to porcine pancreases obtained from a single pig strain.
In conclusion, we identified the best histomorphometric predictors of islet yield and posttransplant in vivo potency. IEQ/cm2 and the number of very large islets (>200 μm in diameter) predicted islet yield and in vivo potency. The proportion of large islets (>100 μm in diameter) best predicted posttransplant outcome and its inclusion enhanced the predictive value of flow cytometry analysis. These parameters can be used to predict islet yield and in vivo islet potency in advance of clinical adult porcine islet xenotransplantation.
Footnotes
Acknowledgments
This work was supported by a grant from the Korea Health 21 R & D Project, Ministry of Health, Welfare and Family Affairs, Republic of Korea (Project A040004).
