Abstract
To address a number of questions regarding the experimental use of bone marrow (BM) stem cells in hindlimb ischemia, including which is the best cell type (e.g., purified hematopoietic stem cell or monocytes), the best route of delivery [intramuscular (IM) or intravenous (IV)], and the mechanism of action (transdifferentiation or paracrine effects), we have compared the neovascularization capacities of CD133+ stem cells and monocytes (CD11b+) from the BM of Tie2-GFP mice either via IV or IM in a murine severe hindlimb ischemia model. To test the effect of cytokine administration, an extra group received BM conditioned medium. Peripheral blood flow as well as capillary density and GPF-positivity detection in ischemic muscles was evaluated 7, 14, and 21 days postinjection. In addition, CD133+ and CD11b+ cells from transgenic animals were cultured in vitro with angiogenic media for 7, 14, and 21 days to assess GFP expression. In all four cell-treated groups, blood flow and capillary density significantly recovered compared with the mice that received no cells or conditioned medium. There were no differences with respect to cell types or administration routes, with the exception of a faster flow recovery in the CD133+-treated cell group. We did not find GFP+ cells in the ischemic muscles and there was no GFP expression after in vitro proangiogenic culture. Our study shows that both purified CD133+ stem cells and myeloid mononuclear cells, either IM or IV administered, have similar neoangiogenic ability. Nevertheless, transdifferentiation into endothelial cells is not the mechanism responsible for their beneficial effect.
Introduction
Evidence has accumulated over the past few years to suggest that bone marrow (BM) is a reservoir of cells with angiogenic properties, generally designated as endothelial progenitor cells (EPC) (12, 18, 29). In a seminal paper, Asahara and colleagues demonstrated for the first time in humans, the presence of circulating endothelial progenitor cells, based on the expression of CD34 and Flk-1 (3). Later, several groups showed that EPC also expressed the prominin-1 antigen (also called AC133 or CD133) (14, 27). Mouse prominin-1 was initially isolated in 1997 from neuroepitelial stem cells and named for its location on the protrusion of cell membranes (42). Although prominin-1 function is not completely known, is coexpressed by both EPC and hematopoietic stem cells (HSC) and other tissue-specific stem cells, but not by mature endothelial cells (34). Several groups, including ours, have employed isolated human CD133+ cells for the treatment of myocardial (5, 37) or hindlimb ischemia (7).
Because the definition and characterization of EPC is still controversial (16), some authors argue that the use of selected populations may exclude important cell subsets with vasculogenic potential. For this reason, alternative approaches based on cell therapy for ischemic diseases consist of delivering nonselected whole BM cells or BM mononuclear cells (BM-MNC). BM or G-CSF mobilized peripheral blood (PB) MNC administration in experimental preclinical models has shown promising results, which has prompted the launch of clinical trials with nonselected MNC (6, 21). However, a potential disadvantage of nonselected BM-MNC is the coadministration of several cell subsets with proinflammatory activity in an ischemic microenvironment, which could increase the risk of secondary effects (35, 44). Monocytes, the main population contained in BM or PB MNC, have shown “in vitro” and “in vivo” vasculogenic potential (23, 30, 33, 40). To avoid contamination with lymphocytes or other inflammatory cells, the administration of purified monocytes (CD14+ cells in humans or CD11b+ cells in murine models) is therefore an attractive approach. Selected hematopoietic stem cells and monocyte administration has not been directly compared to date. Few data are available from in vivo preclinical experiments (4), and none of them compared CD133+ cells with monocytes.
There are at least two possible routes of administration. Cells can be delivered directly by intramuscular (IM) injection into the ischemic area or can be systemically distributed by intravenous (IV) administration. Both methods have proved to be effective (17, 29), but few studies have directly compared the effects of either approach within the same experiment (15), and none in the hindlimb ischemia setting.
Most importantly, irrespective of the cell source or route of administration, a further topic of interest is the understanding of the mechanisms involved in the therapeutic effect of the administered cells. In this regard, some of the studies from animal models have shown that donor BM cells may “transdifferentiate” into host tissue cells and remain in the corresponding damaged organ (the contribution of donor-derived EPC to neovascularization may range from 5% to 25%) (10, 25). However, other studies have shown that only a very small proportion of cells, if any, transdifferentiate into endothelial cells, suggesting that most of the beneficial effects are mediated by paracrine-secreted factors (26, 36, 39), supporting that cytokine or growth factor administration could be an alternative or an adjuvant to cellular therapy (22).
The availability of cell-tracking systems may shed some light on the discussion of the mechanism of action of the transplanted cells (19). In this regard, the Tie2-GFP transgenic model initially described by Motoike et al. (24) is valuable. In this model, GFP expression in transgenic mice is controlled by the Tie2 promoter, a well-known endothelial marker activated during angiogenesis (32). If several stem cell subsets from the BM of transgenic mice are transplanted into ischemic areas of wild-type animals, they will only become GFP positive if they express Tie2. If this is accompanied by their location within the vascular endothelial surface and the coexpression of other endothelial-specific molecules [e.g., endothelial nitric oxide synthase (eNOS)], it may be evidence that transdifferentiation is the main mechanism of action controlling the vasculogenic effects of the transplanted BM cells.
In the present work, we have employed the Tie2-GFP murine model in order to address the aforementioned questions concerning the preferable stem cell source, route of administration, and mechanism of action.
Materials and Methods
An outline of all the experiments performed in this study is depicted in Figure 1.

Outline of the experimental design. (A) In vivo studies. (B) In vitro studies.
Mice
Animal care and procedures were conducted in compliance with European and Spanish policies and all experiments were approved by the Local Ethics Committee. Wild-type FVB mice were obtained from Charles River Laboratories (Barcelona, Spain) and FVB-TgN (Tie2-GFP)287 Sato (24) mice were purchased from Jackson Laboratories (Bar Harbor, ME). Animals were housed in a conventional clean facility for at least a week before experimental use, and given ad libitum access to mouse chow and tap water. Donor and recipient mice were between 6 and 8 weeks of age (16–22 g) at the time of study.
Bone Marrow Harvest and Cell Selection
Donor Tie2-GFP mice were anesthetized and sacrificed by cervical dislocation. Tibiae, femurs, iliac bones, and vertebrae were removed and cleaned. BM cells were obtained using the optimized method described by Colvin et al. (9), crushing the bones in a mortar with a pestle using cold PBS. Marrow cells were then filtered through a 40-μm strainer and washed once. MNC were obtained after gradient density centrifugation with Ficoll-Paque (GE Healthcare Bio-Sciences, AB, Uppsala, Sweden). For prominin-1 and CD11b selection, we employed the respective selection kits from Miltenyi Biotec (Bergisch Gladbach, Germany), strictly following the manufacturer's instructions. Briefly, MNC were incubated for 15 min at 4°C with FcR blocking reagent and anti-mouse CD133 monoclonal antibody. Labeled cells were loaded on a manual magnetic separation column device (VarioMACS, Miltenyi Biotec). Purity of the selected populations (both prominin-1 and CD11b) was assessed by flow cytometry by anti-mouse CD133 conjugated with phycoerythrin (PE) and anti-CD11b conjugated with allophycocyanin (APC), respectively, both from Miltenyi Biotec. Purity was >95% in both cases.
Induction of Hindlimb Ischemia
Hindlimb ischemia was induced by ligation of the left iliac artery as described by Peñuelas et al. (28). Wild-type FVB mice were anesthetized with IP administration of ketamin (75 mg/kg) and xylazin (10 mg/kg) and subjected to surgery under sterile conditions. A laparotomy was performed and the iliac arteries were localized at the bifurcation of the abdominal aorta. The left iliac artery was then ligated proximally to the bifurcation and distally close to the inguinal ligament with 7/0 nylon ligatures. All animals received an anti-inflammatory agent (ketoprofen, 5 mg/kg subcutaneously) daily for 3 days and an antibiotic (enrofloxacin, 25 mg/kg, in drinking water) for 5 days after surgery. Between 7 and 10 ischemic mice were employed for each experimental group described in Figure 1 (CD133 IV, CD133 IM, CD11b IV, CD11b IM, vehicle-injected IV, vehicle-injected IM, conditioned medium-injected IV, and conditioned medium-injected IM) and for each time point evaluated (7, 14, and 21 days after the induction of ischemia).
Cell Administration
Twenty-four hours after left iliac artery ligation, 1 × 106 CD133+ selected cells or 1 × 106 CD11b selected cells, depending on the experimental group (Fig. 1), were administered either IV or IM. For the IV injection, cells were diluted in a final volume of 200 μl and injected in the tail vein. For IM administration, cells were diluted in 30 μl and injected in the left quadriceps. The groups that received no cells (vehicle-treated mice) received the aforementioned volumes of PBS, either IV or IM.
BM Conditioned Medium Administration
In order to compare the potential angiogenic effects of cell injection with the effects of in vivo cytokine and growth factor administration, BM-MNC conditioned medium was also administered to a subgroup of ischemic mice, and the IV and IM routes were compared. For this purpose, MNC were obtained as indicated before, and seeded at a density of 1 × 106 cells/cm2 in fibronectin-coated six-well culture plates containing Dulbecco's modified Eagle's medium, low glucose (DMEM; Gibco, Invitrogen, Paisely, UK), 10% fetal calf serum (FCS; BioWhittaker, Lonza, Verviers, Belgium), and 1% penicillin/streptomycin (Gibco, Invitrogen). BM-MNC were maintained at 37°C in a humidified atmosphere of 5% CO2 for 48 h. Supernatants were collected, filtered with 0.22-μm-pore nylon filters, and administered to ischemic wild-type FVB mice in the same volumes, using the same procedures as for the cell-treated groups.
Blood Flow Assessment
On days 7, 14, and 21 after cell administration, blood flow (in μl/s) was measured by laser-Doppler (Moor Instruments, Axminster, Devon, UK) in the left femoral artery surface area. A median number of 15 measures were performed at each time point per animal. Basal blood flow was determined in the same way in 25 recipient mice before surgery.
Capillary Density in Ischemic Muscles and GFP+ Cell Detection
On days 7, 14, and 21 after cell administration, depending on the experimental group, mice were deeply anesthetized with ketamin and xylazin and perfused intra-aortically with 0.9% saline followed by a fixative solution containing 4% paraformaldehyde and 0.2% picric acid in phosphate buffer. The ischemic muscles (adductors) were dissected and postfixed in the same solution for 4 h, then washed and cryoprotected in 30% sucrose buffer and finally embedded in optimal cutting temperature (OCT) compound (Sakura Finetek Europe, Zoeterwoude, The Netherlands) and frozen at −20°C. For all staining and analysis, 6-μm-thick sections were obtained in a Leica CM2000 cryostat (Leica Microsystems, Nussloch, Germany). Slides were incubated with a primary rabbit polyclonal anti-eNOS antibody (Santa Cruz Biotechnology, Santa Cruz, CA) and with a mouse monoclonal anti-GFP antibody (Abcam, Cambridge, UK) to amplify the GFP signal, diluted in PBS containing 5% goat serum, and 0.2% Triton X-100. After several rinses in PBS, sections were further incubated with a mixture of Cy3-conjugated goat anti-rabbit IgG and Cy2-conjugated goat anti-mouse IgG (Jackson Immunoresearch, West Grove, PA), diluted in PBS with 0.2% of Triton X-100. Sections were then counterstained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma, St. Louis, MO). Finally, sections were washed, mounted, and cover-slipped with the antifading mounting medium Fluoromount (Sigma). Slides were analyzed on an Olympus Provix AX70 photomicroscope (Olympus, Tokio, Japan) equipped with epifluorescence and appropriate filter sets. Images were digitalized by a DP-70 digital camera (Olympus) adapted to the microscope and connected to a computer with the DP manager software (Olympus). To accurately estimate the capillary density, the Image J software (freely available at http://rsb.info.nih.gov/ij/) was used. The capillary density was quantified as the percentage of the muscular area formed by blood capillaries. Capillaries were identified by the eNOS immunofluorescence displayed by endothelial cells. Five eNOS-stained muscular images were randomly taken from each slide using the 20× objective. Between 5 and 10 slides were analyzed per animal. Those digital images were treated to balance the signal-to-noise ration so that positive elements were clearly distinguishable from the background. They were then manually transformed into binary images where only immunostained elements appeared as white pixels. Then, the capillary density was calculated as the white/black pixel ratio in the entire image. Within the same analysis, number of GFP+ endothelial-compatible cells was determined by direct quantification of elements displaying green fluorescence in each analyzed field. The number of GFP+ cells was calculated as the percentage of total endothelial cells (eNOS+) per slide (at least 500 endothelial cells were counted per slide).
In Vitro Analysis of Endothelial Differentiation
In a different set of experiments, selected CD133+ cells and CD11b+ cells (obtained as indicated before) from Tie2-GFP mice were cultured in two different standard proangiogenic media (38, 45). The first one was the endothelial growth medium-2 (EGM-2) Bullet kit (endothelial cell basic medium, hydrocortisone, FGF-b, VEGF, R3-IGF-1, ascorbic acid, EGF, GA-1000, and heparin) supplemented with 10% fetal bovine serum (Cambrex, Walkersville, MD, USA). The second media consisted of DMEM (Gibco), 20% FCS (BioWhittaker), 1% penicillin/streptomycin (Gibco), and 100 μg/ml of endothelial cell growth supplement (Sigma). CD133+ cells and CD11b cells were seeded in fibronectin-coated six-well culture plates at a density of 5 × 105 cells/cm2 and at 37°C in a humidified atmosphere of 5% CO2. Cells were maintained for 7, 14, and 21 days (depending on the experiment). As controls, CD133+ and CD11b+ cells from wild-type FVB mice were equally selected and cultured. At the aforementioned times, cells were harvested after trypsinization and then washed. Cells were then processed for flow cytometric evaluation of GFP expression and were also incubated with anti-mouse CD31 conjugated with PE (Becton Dickinson Biosciences-BDB, San Jose, CA). At least 5 × 104 events were acquired in a dual-laser FACSCalibur flow cytometer using the CellQuest software (BDB). The latter, together with Paint-A-Gate PRO software (BDB), were employed for data analysis, as previously described (31).
Statistical Analysis
All statistical analyses were performed with SPSS 15.0 software (SPSS Inc., Chicago, IL). For each variable under study, percentages or means, SEMs, medians, and ranges were calculated. For continuous variables, normality was assessed by the Kolmogorov-Smirnov test. When the assumption of normality was met, differences between groups were tested by one-way ANOVA with Bonferroni, Scheffe, and Tukey post hoc corrections. Otherwise, the nonparametric Kruskal-Wallis test was performed. Differences between groups were considered to be statistically significant for values of p < 0.05.
Results
Blood Flow Recovery
As it is depicted in Figure 2, blood flow in the ischemic muscles progressively recovered after iliac artery ligation, even in the control group, although this improvement reached a plateau around day 7. Nevertheless, in all the cell-treated groups (CD133+ IM, CD133+ IV, CD11b+ IM, and CD11b+ IV) blood flow was significantly higher than in controls at every time evaluated (p < 0.001 in all cases). At day 21, blood flow in all treated groups was close to the normal values. When comparing CD133+ with CD11b+ cell administration, blood flow recovery was significantly faster at day 7 in animals treated with CD133+ cells (183 ± 4 vs. 162 ± 5 μl/s; p < 0.001), and also at day 14 (210 ± 3 vs. 197 ± 6 μ/s; p = 0.001), but these differences disappeared at day 21 after transplantation (Fig. 2A). Regarding the administration route, there were no differences in blood flow recovery between those mice that received either CD133+ or CD11b+ cells with the IM route and those who were treated with the IV route (Fig. 2B).

Blood flow recovery evaluated by the laser Doppler technique. (A) Comparison of cell types. (B) Comparison of administration routes.
The administration either via IV or IM of BM conditioned medium did not induce any improvement in the blood flow, as can be observed also in Figure 2.
Capillary Density and Detection of GFP+ Cells
In accordance to what was observed in the blood flow analysis, capillary density decreased promptly after iliac artery ligation, but progressively recovered to a maximum value around 3% of total muscle area (Fig. 3). Capillary density was significantly higher in all cell-treated groups than in controls (p < 0.01 in all cases). Upon comparing different cell sources, capillary density was similar in mice treated with CD133+ cells and those receiving CD11b+ cells. Concerning the administration route, we found similar values in the intramuscular and intravenous groups.
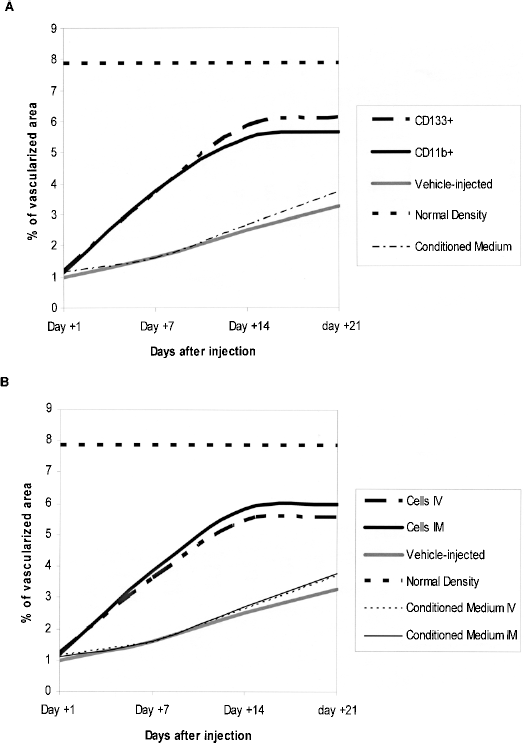
Capillary density. (A) Comparison of cell types. (B) Comparison of administration routes.
Finally, and in accordance to what was observed in the blood flow analysis, the administration of BM conditioned medium did not improve the capillary density compared to the vehicle-injected animals, as also shown in Figure 3.
Together with the eNOs staining that was used for capillary density quantification (see Materials and Methods section), we also investigated the presence of GFP+ cells in the ischemic muscles analyzed. We could not find any significant number of GFP+ eNOS+ endothelial-like cells. Therefore, we can estimate that the number of Tie2+ cells is lower than 1 in 500 eNOS+ cells.
In Vitro Proangiogenic Differentiation
In vitro differentiation of both cell types (CD133+ and CD11b+) from Tie2-GFP mice was tested by cell culture with two different proangiogenic media during 7, 14, and 21 days. We found that, irrespective of the differentiation medium employed, cultures of neither cell type gained CD31 expression, another classic endothelial marker, throughout the culture. Most importantly, neither CD133+ nor CD11b+ activated Tie2 gene expression, because fluorescence was absent in the FITC channel (where GFP expression would be detected) (Fig. 4).
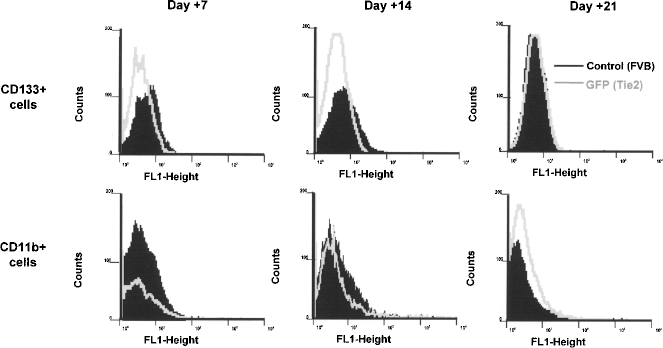
GFP expression in vitro after proangiogenic differentiation of CD133+ and CD11b+ cells. GFP expression was evaluated in CD133+ and CD11b+ cells from Tie2 transgenic mice after 7, 14, and 21 days of culture in proangiogenic media. The same cells from wild-type FVB animals experienced the same experimental conditions and were used as a control.
Discussion
The present article is, to the best of our knowledge, the first to evaluate in the same experiment the angiogenic ability of two cell populations that have already been employed in clinical trials of hindlimb ischemia and that have been claimed as endothelial progenitor cells (CD133+ and monocytes). In addition, we have also assessed the two routes of cell delivery most widely employed (IV and IM) in this setting (2). Moreover, by using the Tie2-GFP transgenic model, we have also tested whether transdifferentiation of these two BM cell subsets into Tie2+ eNOS+ endothelial cells is the main mechanism of action of their angiogenic effect. Unlike cell administration, we have also demonstrated that the IV or IM delivery of BM-MNC conditioned medium has no angiogenic effect.
We have shown that both CD133+ cells and monocytes (CD11b+ cells) have a similar ability to improve the blood flow and the capillary density of the ischemic muscles in our experimental model, with no differences between the two cell sources with the exception of a faster flow recovery when CD133+ selected cells were administered. This has also been shown after CD34+ cell administration (a cell subset that, as opposed to CD133+ cells, contains mature endothelial cells) (34) when compared with CD14+ monocytic cells in a different animal model (4). The clinical implications of a faster improvement when selected hematopoietic stem cell populations (either CD34+ or CD133+ cells) are administered for hindlimb ischemia remain to be determined. It has been shown that MNC administration (mostly but not exclusively composed of monocytes) may be associated with an increase of inflammatory cytokines in the ischemic area as well as inflammatory infiltration that may lead to unexpected events (1, 35, 44), as has also been described after CD34+ cell administration (4) but not when CD133+ are injected (43). The fact that we did not observe a significant inflammatory infiltrate in the ischemic muscles in the CD11b+ group (data not shown) may be justified by the absence of lymphocytes and neutrophils within this administered cell subset, as others have also noted (2, 4).
With respect to the administration route, it is well known that after IV administration, a number of BM cells get trapped in the spleen, liver, or lung microcirculation (11, 13). This may explain why most of the clinical trials of cell therapy for chronic limb ischemia cells use the IM route (2). Nevertheless, in our work the IV administration gave similar results to those with IM administration. Because the engraftment of the administered cells in the damaged tissue is constantly low in most of the published experiments (2), the number of cells that home to the ischemic muscles may be similar in both administration routes, although, unfortunately, the transplant model used in our study, Tie2-GPF FVB donor mice into wild-type FVB, did not allow us to measure the percentage engraftment.
Finally, our study shows that neither CD133+ cells nor CD11b+ cells from Tie2-GFP FVB mice are able to activate the Tie2 promoter either in vitro, after culture in established proangiogenic media, or in vivo, after administration into severely ischemic mice. This strongly supports that their angiogenic potential (that we have demonstrated in vivo) is not mediated by transdifferentiation into endothelial cells. This is consistent with most of the published reports, which have shown that the beneficial effects may be mediated by secreted cytokines and growth factors delivered by the transplanted cells (1, 20).
A result of particular interest is the absence of any angiogenic effect of the administration of BM conditioned medium. This would require a sustained production of those factors (such as can be released into the ischemic microenvironment by administered cells) (20), whereas their local administration undoubtedly has a merely transient effect. Alternatively, cellular contact would be required to induce vasculogenesis. This may explain why the results of clinical trials of angiogenic growth factors for chronic limb ischemic have so far been unsatisfactory (8, 41).
Conclusion
In summary, our study shows that both CD133+ and CD11b+ administered either via IM or IV in a murine model of severe hindlimb ischemia have a similar but significant angiogenic effect compared with vehicle-injected animals. In addition, neither cell type is able to activate, either in vitro or in vivo, the Tie2 promoter suggesting, in the experimental model employed, that the mechanism of action of the transplanted cell is not transdifferentiation into endothelium.
Footnotes
Acknowledgment
This work was partially supported by a research grant from the Consejería de Sanidad de la Junta de Castilla y León (GRS 71/A/06).
