Abstract
The objective of this study was to investigate safety and feasibility of autologous bone marrow mononuclear cells (BMMNC) transplantation in ST elevation myocardial infarction (STEMI), comparing anterograde intracoronary artery (ICA) delivery with retrograde intracoronary vein (ICV) approach. An open labeled, randomized controlled trial of 30 patients admitted with STEMI was used. Patients were enrolled if they 1) were successfully reperfused within 24 h from symptoms onset and 2) had infarct size larger than 10% of the left ventricle (LV). One hundred million BMMNC were injected in the infarct-related artery (intra-arterial group) or vein (intravenous group), 1% of which was labeled with Tc99m-hexamethylpropylenamineoxime. Cell distribution was evaluated 4 and 24 h after injection. Baseline MRI was performed in order to evaluate microbstruction pattern. Baseline radionuclide ventriculography was performed before cell transfer and after 3 and 6 months. All the treated patients were submitted to repeat coronary angiography after 3 months. Thirty patients (57 ± 11 years, 70% males) were randomly assigned to ICA (n = 14), ICV (n = 10), or control (n = 6) groups. No serious adverse events related to the procedure were observed. Early and late retention of radiolabeled cells was higher in the ICA than in the ICV group, independently of microcirculation obstruction. An increase of EF was observed in the ICA group (p = 0.02) compared to baseline. Injection procedures through anterograde and retrograde approaches seem to be feasible and safe. BMMNC retention by damaged heart tissue was apparently higher when the anterograde approach was used. Further studies are required to confirm these initial data.
Keywords
Introduction
Growing evidence suggests that cell-based therapies can repair the injured myocardium after an acute infarction. However, comparative data analyses of this new field have been hampered by the great variety of cell phenotypes and protocol design in different clinical trials. Although the largest experience comes from bone marrow-derived cell studies, which mostly point to myocardial regeneration and improvement of ventricular function, the mechanisms of their actions are still poorly understood (15). From the very beginning of these studies, one of the most frequently asked questions has been which is the best way to deliver cells into the heart. Animal studies give us important data but they are not reliably applicable to humans (19). We and others have previously reported the use of transendocardial injections for bone marrow mononuclear cells (BMMNC) delivery in ischemic cardiomiopathy (13,20). Other approaches were described for different clinical settings such as transepicardial and intracoronary injections (20). The latter approach was used in several clinical trials demonstrating safety of BMMNC transplantation in the acute phase of ST elevation myocardial infarction (STEMI). The evidence for efficacy of the therapy has been growing, but the ideal technique for delivery of cells into the myocardium in humans has not been extensively investigated.
There is no clearly established relationship among delivery techniques, cell engraftment, and clinical effects, and ambiguous results regarding infarct-related coronary artery infusion of BMMNC after STEMI have been observed (5). Despite some controversies about the efficacy of BMMNC, the largest reported trial showed a significant improvement for patients who received cells after the fifth day of large infarcts. On the other hand, microvascular obstruction may play an important role in this case, as proposed by Janssen et al. (10), who reported that microvascular obstruction could impair cells' uptake by heart tissue and consequently heart function improvement. We hypothesized that coronary intravenous approach may overcame this issue, because diapedesis of circulating cells into the adjacent cardiac tissue occurs in the venous side of microcirculation (11). Little is known regarding retrograde percutaneous approach through the coronary vein, even though this route has been shown as an effective way to deliver cells in animal and human models (6,9,18).
The primary objective of this study is to compare two different delivery techniques for the infusion of BMMNC in STEMI: the anterograde intracoronary artery (ICA) delivery with the retrograde intracoronary vein (ICV) approach. Furthermore, we used radiolabeled cells in order to evaluate their distribution pattern in the heart once injected and their relationship with left ventricle function improvement.
Materials and Methods
Patients
Between January 3, 2005 and January 6, 2006, patients admitted in Pró-Cardíaco Hospital or Miguel Couto Municipal Hospital, Rio de Janeiro, were selected to be included into the study if they were between 18 and 80 years old and had a STEMI. Patients selected at Miguel Couto Hospital were transferred to Pró-Cardíaco Hospital where all procedures related to the study were performed. Patients were enrolled if they had: 1) a STEMI successfully reperfused with either thrombolytic therapy or primary angioplasty up to 24 h after the symptoms onset, and 2) fixed perfusion defect larger than 10% of the LV mass after 72 h on technetium 99m methoxy-isobutyl-isonitrile (99mTc-MIBI) single-photon emission computed tomography (SPECT) with sublingual nitrate. Exclusion criteria were: 1) indication to undergo coronary artery bypass graft; 2) creatinine level >2.0 mg/dl or hemodialysis; 3) infarct-related coronary artery with thrombolysis in myocardial infarction (TIMI) flow <3 by the time of cells injection; 4) sepsis; 5) persistent cardiogenic shock after 72 h; 6) significant valvar disease; 7) mechanical complications of STEMI; 8) liver failure; 9) severe pulmonary disease; 10) left bundle branch block; 11) implanted pacemaker; 12) hematological illnesses; 13) neoplasia; 14) other disorders of hemostasis and other pathologies that could have impact on life expectancy.
The Ethics Review Board of Pró-Cardíaco Hospital, Rio de Janeiro, and the Brazilian National Ethics Council for Human Research (CONEP, Brasilia) approved the protocol. Written informed consent was obtained from all patients.
Study Design and Randomization
This is an open-labeled, randomized controlled trial of a consecutive patient series. Between the third and sixth day after successful reperfusion of the infarct-related artery, included patients were randomly assigned into one of the three groups: ICA approach, retrograde ICV approach, and control. Random allocation was stratified according to infarct size (≥ 25% or <25%) in three blocks of different size (7, 5, and 3, respectively) for each stratum, with the use of sealed envelopes.
Bone Marrow Cells Harvesting and Isolation of Mononuclear Cells
In subjects assigned to either of the two treatment arms of this study, about 80 ml of autologous bone marrow was aspirated under local anesthesia from the posterior iliac crest in the morning of the procedure, which was anticipated to occur 6 h later. BMMNC were isolated by density gradient centrifugation on Ficoll-Paque Plus (Amersham Biosciences, São Paulo, Brazil) and manipulated under aseptic conditions. They were washed with saline containing 5% human serum albumin. The cells were resuspended in saline with 5% human serum albumin and filtered for injection through 100-μm nylon mesh to remove cell aggregates. A small fraction of the cell suspension was used for cell counting and viability control using the trypan blue exclusion assay. Cell viability was >90% in all subjects (93.26 ± 2.9%). Characterization of leukocyte differentiation markers by flow cytometry and functional assays were done on another fraction of cells following study intervention. The clonogenic capacity of hematopoietic progenitors was evaluated by colony-forming assays [granulocyte-macrophage colony-forming units (CFU-GM)], as previously described (4). Fibroblast colony-forming assay (CFU-F) was done to estimate the presence of putative progenitors of the mesenchymal cell lineage, as previously described (3).
Antibodies and Staining Procedure for Fluorescence-Activated Cell Sorter/FACS Analysis
The following antibodies were either biotinylated or conjugated with fluorescein isothiocyanate (FITC), phycoerythrin (PE), PE-TR (PE-Texas red), or PerCP. Anti-CD45 as a pan-leukocyte marker (clone HI30), anti-CD34 as a hematopoietic progenitor marker (clone HPCA-II), anti-CD3 as a pan-T-cell marker (clone SK7), anti-CD4 and anti-CD8 as T-cell subpopulation markers (clones SK3 and SK1, respectively), all from Becton Dickinson (B&D, São Paulo, Brazil); anti-CD105 as a mesenchymal cell marker (clone 166707), anti-CD14 as a monocyte marker (clone TUK4), anti-CD19 as a pan-B-cell marker (clone SJ25-C1), and anti-CD56 as a NK-cell marker (clone NKI nbl-1), all from Caltag Laboratories (Burlingame, CA); and anti-HLA-DR (MHC-II, clone B8.12.2) from Beckman-Coulter (Fullerton, CA). The biotinylated antibodies were revealed with streptavidin PE-TR (Caltag Laboratories). Three-color immunofluorescence analysis was used for identification of leukocyte populations within the total nucleated bone marrow cell suspensions. After staining, erythrocytes were lysed with the B&D lysis buffer solution according to the manufacturer's instructions, and CD45 antibody was used to assess the percentages of leukocytes in each sample. Data acquisitions were performed on a FACS Calibur cytometer, and analyses were performed using CellQuest software (B&D).
Cell Delivery Techniques
Cell delivery was performed 8.5 ± 1.44 h after bone marrow cells harvesting. Arterial accesses were performed using femoral or radial approach. All patients received 10,000 IU of nonfractionated heparin after sheet insertion. Non-infarct-related vessel intervention was performed before cell transfer in three patients of the ICA group and in one patient of the ICV group. Electrocardiography, pulse oximetry, vital signs, and any clinical symptoms were monitored throughout the procedure.
In both the ICA and ICV delivery groups, a coronary angiography was performed (GE Medical System, Advantx LCV plus, WI). A 6-F guiding catheter was then placed at the ostium of the infarct-related coronary artery in order to confirm target vessel patency and to assess coronary blood flow (TIMI 3) before cell injection.
In the ICA group an over-the-wire (OTW) balloon catheter (Maverick® Over-The-Wire balloon, Boston Scientific, Natick, MA), with a diameter 0.5 mm greater then the implanted stent, was positioned inside the previously implanted stent in the culprit vessel to transiently interrupt anterograde blood flow during infusions through a stop-flow technique. Ten milliliters containing 100 million autologous BMMNC were infused through the central lumen of the balloon catheter during three coronary occlusions, each lasting 2–3 min, followed by 2 min of balloon deflation. One milliliter of labeled cells solution (see below) was diluted in 3 ml of saline and infused into the ICA as a final solution, using the same technique described above.
In the ICV group, right internal jugular vein access was used to position 5 or 6 Fr JR or multipurpose guiding catheters in the coronary sinus, in addition to arterial access. The same type of OTW balloon catheter (Maverick® Over-The-Wire balloon), varying from 3.5 to 4.0 × 9.0 mm in size, was then advanced through the cardiac vein corresponding to the culprit vessel and positioned side by side with the balloon in the coronary artery, at the previously implanted stent. Total occlusion of the cardiac vein was then performed and maintained for at least 12 min. Four intermittent coronary artery occlusions were performed simultaneously, aiming to reduce cells washing by anterograde flow pressure. One milliliter of labeled cells (see below) was diluted in the cell solution containing 100 million mononuclear cells. During the first anterograde stop-flow inflation the final 11 ml of cells solution was infused through the central lumen of the venous balloon catheter during 1 min, at one shot.
A final coronary angiography was performed at the end of all procedures as control in order to ascertain TIMI frame count pre- and post-PCI and cell transfer. EKG and cardiac enzymes were done before and timely after cell transfer.
Mononuclear Cells Labeling
In 12 patients of the ICA group and 6 of the ICV group, about 40 min before injection, a fraction of cells (1% of 108 cells) were incubated under sterile conditions in a 10-ml tube with 150 MBq of 99mTc-hexamethylpro-pylenamineoxime (HMPAO) (Amersham Biosciences, Piscataway, NJ) per 107 cells for 30 min in a saline solution containing 2.5% human albumin.
Labeling efficiency was estimated after removing the excess of unbound radioactivity by washing the cells with the saline solution with 2.5% human albumin. Radioactivity was measured with a dose calibrator (PTW Curiementor 2, Freiburg, Germany). Viability of the cells was assessed by trypan blue exclusion.
Cells labeled with 99mTc-HMPAO were injected as described above. Tissue distribution was observed with early and late total body images at anterior and posterior projections, with 1024 × 256 pixels and 12 cm/min speed. For topographical location of cells in the heart, thorax images tomography were acquired with a protocol similar to that used for myocardial perfusion images, 64 projections of 20 s each, with resolution of 64 × 64 pixels. Images were reconstructed with Butterworth filter using the software e-Soft 3.0 n (Cedars. Sinai, QGS and Emory Cardiac Toolbox) and compared to the acquired perfusion images tomography. All images were acquired in a dual head gamma camera (Ecam-Duet, Siemens Medical Systems Inc, IL).
Planar and SPECT images were obtained at 4 h (3.67 ± 1.03 h) and 24 h (22.8 ± 4.46 h) after injection in all patients. Retention was defined as the percentage of cardiac-originated number of counts in the anterior and posterior projections (cardiac counts), compared to the total number of body counts of radiolabeled cells (body counts) in both early and late images. Washout rate was calculated by using the equation: [early (cardiac counts/body counts) – late (cardiac counts/body counts)] × 100/early (cardiac counts/body counts). Washout rate per hour was calculated by dividing washout rate per the interval between the early and late images retention.
Radionuclide ventriculography (MUGA) was performed 3–5 days after reperfusion and at least 24 h after 99mTc-MIBI SPECT. Injected 99mTc-pertechnetate activity was 925 MBq (25 mCi), after injection of 5 mg of pyrophosphate. Conventional planar gated blood pool (GBP) imaging was performed in the appropriate left anterior oblique (LAO) projection angle. Data were acquired with low-energy, high-resolution (LEHR) collimators, by use of a dual-detector gamma camera with the detectors set at 90° relative to one another. No caudal tilt was used in setting up patients for optimal planar GBP imaging. Data were acquired as 64 × 64 matrices, with LEHR collimators for 24 frames per R-R interval for 10 min. Processing was performed with commercially available software, involving two-dimensional spatial smoothing and one-dimensional time filtering of LAO data sets. Software supplied by the manufacturer generated automated outlines for LV identification and background correction regions, but the observer was free to alter these as necessary to conform to the visual impression of true LV boundaries and appropriate background locations. Observer drew LV outlines for end-diastolic frames, primarily guided by the visual impression of the LV shape and aided by Fourier amplitude and phase maps. Counts within end-diastolic and end-systolic regions were corrected for average background counts derived from a region drawn beside the LV region. Background-corrected counts were used to compute LVEF. From the time–activity curve we were able to obtain the LVEF as a systolic parameter and the diastolic parameter named time to peak filling (TPF) rate. TPF rate is measured from end-systole to the time of the peak LV filling rate. It is calculated from the time–activity curve and its first derivative. The maximal slope of this derivative of the filling portion, normalized for the number of counts at end-diastole, gives rise to the peak filling rate.
Cardiac magnetic resonance imaging (MRI) was performed 3–5 days after reperfusion. All studies were done with commercially available cardiac MRI software (GE Healthcare Milwaukee, WI, USA) as previously described (16). Briefly, microvascular obstruction (MO, or MVO) was defined on late enhanced images taken early (within 2–5 min) after injection of 0.20 mmol/kg of gadopentetate dimeglumine (Gd-DTPA), in the LV short and long axis at the same locations used for cine-MRI as a dark, subendocardial zone within the infarct area. We defined infarct area as the zone of bright signal on late-enhanced images (10–20 min after contrast injection) by inversion-recovery gradient-echo technique. All MRI studies were analyzed on an off-line workstation.
Follow-up
All patients were standard treated. Follow-up to assess clinical status, review of current medication, and to reevaluate cardiac function through MUGA occurred at 3 and 6 months after the procedure. In addition, at 3 month follow-up all patients were scheduled to repeat coronary angiography.
Statistical Analysis
All image exams were independently analyzed by an experienced observer unaware of patients' group allocation throughout the study.
Continuous variables are presented as mean ± SE (unless stated otherwise). Categorical variables were compared with chi-square or Fisher's exact test, as appropriate. Continuous variables were compared with t-test or ANOVA, as appropriate. Comparisons of the changes from baseline to 3 and 6 months among the groups were made with repeated-measures ANOVA. Kendall's tau correlation coefficient was used to correlate continuous data.
Statistical significance was assumed at a value of p < 0.05. All reported p-values are two-sided. All statistical analysis was performed with SPSS (Version 13.0, SPSS Inc.).
Results
Thirty patients were included in the study, 14 in the ICA group, 10 in the ICV group, and 6 in control group. There was no significant difference in demographic parameters among the studied groups (Table 1), including infarct size and cell phenotypes and functionality (Table 2).
Demographic Data

CAD, coronary artery disease; RD, rest defect; AMI, acute myocardial infarction; PTCA, percutaneous transluminal coronary angioplasty; NA, not applicable.
Characteristics of Injected Bone Marrow Mononuclear Cells

Number of cells represents the total number of cells injected according to viability and specific phenotype.
The time period between AMI and cell injection was 5.5 ± 1.3 days and 6.1 ± 1.4 days in the ICA and ICV groups (p = 0.14), respectively.
ICA and ICV injections were successfully performed in all but one case in the ICV group due to anterior interventricular vein tortuosity. CK-MB elevation (3× normal) occurred in one patient in the ICA group and in two patients of the ICV group. One subacute thrombosis in the ICA group occurred 10 days after the procedure. One sudden death in the ICV group occurred 1 month after the therapy. All treated patients were asymptomatic by the time they were submitted to the invasive follow up 3 months after injection. Four binary restenoses were observed in the target vessel (one in the ICA group and three in the ICV group), and two in the nontarget vessel (one in the ICA group, one in the ICV group). One symptomatic new lesion in the nontarget vessel was observed in the control group. Controls were not submitted to invasive follow-up.MUGA results are shown in Table 3. Data regarding cell retention are shown in Table 4. In most cases, cell retention was precisely in the infarcted area, as demonstrated in Figures 1 and 2.

Nitrate-enhanced Tc-99m sestamibi scan (first, third, and fifth rows) and BMMN Tc-99m HMPAO–labeled cell images (second, fourth, and sixth rows). Perfusion images demonstrated a severe defect in the anterior, septal, and apical walls. A scan of labeled cells revealed intense regional accumulation of radioactivity in the septal and anterior walls.

Nitrate-enhanced Tc-99m sestamibi short-axis slices (first row) and BMMN Tc-99m HMPAO–labeled cell slices (second row). An absence of radioactivity in the lateral and inferior walls and intense accumulation of labeled cells in the anterior and septal walls in the fused images (third row), revealing the precise localization of the BMMN Tc-99m HMPAO–labeled cells in the infarcted area.
Radionuclide Ventriculography Data

Microvascular obstruction (MVO) was observed in 50% of the ICA group and in 60% of the ICV group. There was no difference in cell retention regarding presence of MVO within the groups, as shown in Figure 3. However, a higher percentage of cell retention was observed in patients submitted to anterograde compared to retrograde approach even in the presence of MVO.
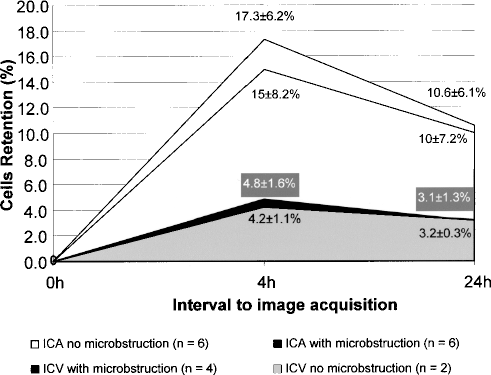
There was no difference regarding radiolabeled cells retention comparing the subgroups with and without microbstruction within the intracoronary artery (ICA) (p = 0.6) or intracoronary vein (ICV) (p = 0.6) groups; early (4 h) and late (24 h) cells retention were higher in the ICA group than in the ICV group, independently of microbstruction presence (p = 0.02 and 0.03, respectively) or absence (p = 0.05 and 0.05, respectively).
A significant correlation among early and late labeled cell retention and the percentage of EF variation between baseline and 6-month follow-up was observed (Fig. 4).

Kendall's tau correlations among early and late radiolabeled cells retention and relative EF improvement at 6 months.
Discussion
The present study has given three sets of information: 1) even though anterograde and retrograde approaches were shown to be feasible and safe, cell delivery into the infarcted myocardium was more efficient in the former than in the latter one, in terms of number of retained cells; (2) microcirculation obstruction did not interfere with cell access and retention within the infarcted myocardium of each studied group; (3) the higher the cell retention, the better the change observed in ejection fraction from baseline to 6-month follow-up.
In most studies of stem cell therapy for the treatment of STEMI, autologous BMMNC fraction was injected through infarct-related artery after reperfusion, aiming to reach the area of myocardium that has been directly affected by interruption of blood circulation. This anterograde approach has been shown to be feasible and safe. However, blood circulation in the infarcted area can be severely affected, hampering potential access of the injected cells into the area where they are required for tissue repair. Thus, alternative retrograde intravenous approach has been proposed; it is feasible and safe, but it has been used less often for stem cells delivery. A comparison between these two techniques was necessary in order to assess their efficiency in improving heart function and clinical outcome of a recent acute myocardium infarction.
Radiolabeled Cells Retention and Washout in the Heart Tissue

The decision to use 99mTc was based on the previous reports of lower toxicity of this radiolabeling method over the others. Other contrast agents such as ferumoxides–protamine sulfate complexes, used in MRI, would offer specific advantages, because they are not associated with cell toxicity (1). However, false-positive findings might be provided by macrophage uptake of the particles from dead cells. The 6-h half-life of 99mTc allowed us to evaluate cell distribution for about 24 h, an advantage over 18F-fluorodeoxyglycose, another possible labeling agent, with half-life of about 2 h (8). 111In-oxine has been proven to cause impairment of the in vitro CD34 cell proliferation and differentiation (2). A much smaller effect was observed with the use of 99mTc-HMPAO (7). In addition, in our study the small fraction of cells used for radiolabeling (1%) may prevent any potential induced harm. We have also performed an in vitro evaluation that showed no impairment of 150 MBq 99mTc-HMPAO on human mononuclear cells viability. Based on the above, we consider the toxicity of our radiolabeling technique acceptable.
We have published a description of the first patient of this series (12). Our data revealed that the level of BM progenitors homing to the irreversibly ischemic myocardial area through ICA and ICV approaches were about 10% and 3% of the infused activity, which corresponded to 10 × 106 and 3 × 106 autologous BMMNC, respectively. Our study provided clear evidence that adhesion and retention of BMMNC via ICA injection to infarcted myocardium is feasible and safe and corroborates previous findings of stem cell mobilization and homing signals from injured tissue in the period of acute ischemic injury (17). It also showed that ICA delivery was more efficient in terms of cell retention in the myocardium than the ICV approach. Such information is valuable because a premise for this therapy benefit is the engraftment of the cells in the damaged tissue. Accordingly, the improvement of the heart function reflected by the EF was consistent with the higher cell retention in the infarcted area after anterograde injection.
This study was undertaken to detect significant difference among the groups regarding LV global function, but some differences were observed in MUGA. EF improved in the arterial group at 3 and 6 months after cells injection. An improvement in the TPF rate was also observed in treated patients compared to controls, probably meaning an effect on diastolic function. These data point towards a causal relationship between the total number of cells that participate in infarct repair and the final enhancement of cardiac function. Up to now, there is no human evidence supporting this hypothesis. New improvements of cell therapies should consider possible means to deliver more cells into the region attained by ischemia.
This study was planned to answer the questions of delivery technique and the consequent distribution of cells. Its small sample size and open label characteristic prevent us from conclusions regarding efficacy of the treatment. We decided to label only a small fraction of cells (1%) in order to avoid unpredicted negative effects on cells due to higher total radiation. This small fraction may not reflect the global cell distribution. Although 99mTc-HMPAO has been widely accepted for safe radiolabeling of leukocytes to localize areas of inflammation (14), the long-term effects of 150 MBq of 99mTc-HMPAO on cells motility and function are still unknown. Finally, the observed radiation in the myocardial area due to labeled cell retention does not necessarily reflect cells engraftment. Under our conditions of cell therapy for AMI with autologous BMMNC, anterograde cell delivery appears to have advantages compared to the retrograde one, but further studies may be required introducing new parameters, such as selected cells types or association of growth factors and/or molecules that modulate the inflammatory reaction of the vascular tree.
Footnotes
Acknowledgments
We gratefully acknowledge the participation of Ana Cristina Reis for the outstanding organization of this project; to Christine Rutherford for the psychological assistance; to Dr. Maria Claudia Rodrigues and Dr. Roberto Magalhães for the bone marrow procedures; and to Pró-Cardíaco team for the medical assistance. We also would like to thank Dr. Warren Sherman for reviewing this manuscript. This study was supported by Pró-Cardíaco Hospital—in charge of patients' care—and by Excellion Biomedical Services S/A—in charge of cell preparation and characterization. Stents Taxus® were donated by Boston Scientific Corp. The authors declare that they have no competing interests.
