Abstract
The first indication of hepatocyte transplantation is inborn liver-based metabolic disorders. Among these, urea cycle disorders leading to the impairment to detoxify ammonia and Crigler-Najjar Syndrome type I, a deficiency in the hepatic UDP-glucuronosyltransferase 1A1 present the highest incidence. Metabolically qualified human hepatocytes are required for clinical infusion. We proposed fast and sensitive procedures to determine their suitability for transplantation. For this purpose, viability, attachment efficiency, and metabolic functionality (ureogenic capability, cytochrome P450, and phase II activities) are assayed prior to clinical cell infusion to determine the quality of hepatocytes. Moreover, the evaluation of urea synthesis from ammonia and UDP-glucuronosyltransferase 1A1 activity, a newly developed assay using β-estradiol as substrate, allows the possibility of customizing cell preparation for receptors with urea cycle disorders or Crigler-Najjar Syndrome type I. Sources of human liver and factors derived from the procurement of the liver sample (warm and cold ischemia) have also been investigated. The results show that grafts with a cold ischemia time exceeding 15 h and steatosis should not be accepted for hepatocyte transplantation. Finally, livers from non-heart-beating donors are apparently a potential suitable source of hepatocytes, which could enlarge the liver donor pool.
Introduction
Human hepatocyte transplantation (HTx) has been performed as an alternative to orthotopic liver transplantation (OLT) for a variety of indications (4, 19, 22). According to current data, the first indications of HTx are liver-based inborn metabolic disorders (4, 19, 22). Inborn errors of metabolism are rare diseases that affect approximately one in 900 live births (19). These patients require severe diet restrictions and special education, leading to a poor quality of life (22). In principle, despite HTx not being a permanent therapeutic option, it is applicable to any metabolic disease in which the deficient enzyme is primarily expressed in the hepatocyte, where a slight increase in enzyme activity can be expected to have a significant clinical effect on the outcome of the disease. Among inborn liver-based metabolic disorders, urea cycle disorders (UCD) leading to the impairment to detoxify ammonia to urea and Crigler-Najjar Syndrome type I (CNI), a deficiency in the hepatic UDP-glucuronosyltransferase 1A1 (UGT1A1), an isozyme responsible for the conjugation and excretion of bilirubin, present the highest incidence. Therefore, both metabolopathies are most frequently treated with HTx (4, 19, 22). UCD have a poor prognosis, especially if the onset of the disease is within the neonatal period; this is a very promising target disease for HTx. Treatment of CNI remains cumbersome, and the perpetual threat of neurological complications weighs on these children until a transplant can be performed. CNI would seem to be a good candidate for cell therapy because 2–3% of UGT1A1 activity is sufficient for subjects to lead a normal life.
The improvement of the HTx is closely related to the availability of human liver tissue and to the quality of transplanted hepatocytes. In the majority of studies conducted, hepatocytes are obtained from organs of cadaveric donors for OLT that have been discarded. The frequent poor quality of liver sources makes it necessary to develop fast and sensitive procedures, as we recently published (7), to evaluate the functional quality of hepatocytes in order to determine their suitability for HTx. However, stricter criteria for the metabolic characterization of cell preparations are required to optimize cell infusions to patients with metabolic disorders.
In the present work, we summarize the recent effort of the HTx unit at our hospital for the purpose of establishing a cell bank of cryopreserved human hepatocytes for clinical application. To achieve this goal we focused on: a) expanding the liver donor pool by exploring the suitability of livers from non-heart-beating donors (NHBDs) as a potential source of hepatocytes for transplantation; b) analyzing the influence of the length of the cold ischemia time on the outcome of isolated hepatocytes; c) improving the cryopreservation protocol; and, finally, d) assessing the functionality of hepatocyte preparations with a view to promoting customized cell preparation for each receptor.
Materials and Methods
Isolation of Human Hepatocytes From Whole Organs or Splits Discarded for OLT
Nineteen cadaveric livers obtained in agreement with the rules of the hospital's Ethics Committee were used. Hepatocytes were isolated from whole livers (n = 5), livers split into right and left lobes (n = 7), right lobes (n = 2), and left lobes (n = 5). Liver tissue was maintained in cold Celsior solution until cell isolation. Briefly, the major liver vessels were canulated and fixed. Hepatocyte isolation was carried out using a two-step collagenase perfusion technique (12). The perfusion system was run by a peristaltic pump at a flow rate of 200 ml/min. All the buffers were maintained at 37°C. After digestion, the tissue was disrupted and the suspended hepatocytes were filtered, washed three times by centrifugation at 500 × g for 5 min. Cell viability was assessed by the dye exclusion test with 0.4% trypan blue. The complete isolation process was performed in a class A laminar air hood (ISO 14644-1) in a class A clean room. Mandatory environmental monitoring was carried out, including the microbiological testing of personnel, air, surfaces, media, and cellular suspensions.
Thirty-eight small surgical human liver biopsies (<5 g) obtained in agreement with the rules of the hospital's Ethics Committee were used to isolate hepatocytes by microperfusion of the tissue with collagenase as previously described in detail (12).
Hepatocyte Cryopreservation and Thawing
Hepatocytes isolated from cadaveric livers were cryopreserved in ice-cold University of Wisconsin solution with 10% DMSO (standard medium) in 50-ml freezing bags (2 × 107 cells/ml) using a controlled-rate freezer, and were long-term stored in liquid nitrogen at −196°C. Only freshly isolated hepatocytes with a viability >80% were cryopreserved. For cell thawing, bags were placed in a water bath at 37°C. The thawed contents were washed with 10 volumes of thawing media at 4°C and centrifuged at 500 × g for 5 min (10). Finally, cells were resuspended in the appropriate medium in accordance with their use.
To improve the freezing conditions, cryopreservation medium supplemented with 300 mM glucose and 10–6 M dexamethasone (improved medium) was compared with the standard medium in hepatocyte preparations obtained from five small surgical liver biopsies. Hepatocytes were frozen in cryovials (107 cells/vial) and freezing and thawing procedures were performed as described above.
Hepatocyte Attachment and Functional Assessment
Hepatocyte attachment capability and urea formation from NH4Cl were measured as previously described in detail (10). Drug-metabolizing activities were assessed by incubating hepatocytes in suspension (1.25 × 106 cells/ml) with HEPES-saline solution (10 mM HEPES, 2 mM Cl2Ca, 137 mM NaCl, 5 mM KCl, 1 mM Na2HPO4, 0.5 mM MgCl2, 10 mM glucose; pH 7.4) containing a cocktail of appropriate substrates for measuring five cytochrome P450 (CYP) enzymes as well as two phase II activities, UDP-glucuronyltransferase (UGT) and sulfotransferase (SULT). Cocktail composition (10 μM 7-ethoxyresorufin, 5 μM coumarin, 90 μM diclofenac, 90 μM chlorzoxazone, and 5 μM midazolam) was selected on the basis of a previously published procedure for measuring the activity in human cultured hepatocytes (16). Midazolam 1′-hydroxylation (CYP3A4), diclofenac 4′-hydroxylation (CYP2C9), chlorzoxazone 6-hydroxylation (CYP2E1), coumarin 7-hydroxylation (CYP2A6), 7-ethoxyresorufin O-deethylation (CYP1A2), and 7-hydroxycoumarin conjugation (UGT and SULT) were quantified by HPLC/MS/MS as described previously (16).
Activity of UGT1A1 enzyme was assessed by incubating hepatocytes in suspension (1.25 × 106 cells/ml) in HEPES-saline solution containing β-estradiol as selective substrate and 1 mM UDP-glucuronic acid as endogenous cofactor at 37°C. To set up assay conditions, different β-estradiol concentrations (up to 15 μM) and incubation times (up to 60 min) were tested. At regular intervals, aliquots of incubation medium (250 μl) were taken and reaction was stopped with cold acetonitrile (1:1). Then, samples were centrifuged at 10,000 × g for 10 min. The metabolite formed during the activity assay was quantified by using a Waters Micromass Quattro Micro with a 2795 Alliance HPLC system (Waters, Milford). An aliquot (20 μl) was injected into a Teknokroma Mediterranea C18 column (100 mm × 2.1 mm, 3 μ particle size). The flow rate was 0.4 ml/min. The mobile phase was A, 0.1% formic acid in water, and B, 0.1% formic acid in acetonitrile. The proportion of acetonitrile was increased linearly from 0% to 90% in 6 min. Then the injection column was allowed to reequilibrate at initial conditions. The complete run lasted 15 min. The column eluent was directed to an atmospheric pressure ionization interface in negative mode at 320°C without splitting, where nitrogen was used as desolvation gas (500 L/h). The mass spectrometer (MS) was operated in the selected multiple reaction monitoring mode. The MS conditions were set up as follows: MS/MS transition was 447 > 113 m/z, cone voltage and collision energy were 40 V and 25 eV, respectively. The limit of quantification for estradiol 3-glucuronide defined as the concentration at which the MS/MS screening peak has a signal-to-noise response of 10 was 2 ng/ml.
Statistical Analysis
ANOVA followed by a Bonferroni test were used for statistical evaluations. The level of significance was p < 0.05.
Results
Influence of the Source of Human Liver Tissue in the Hepatocyte Isolation Procedure
In order to assess the outcome of the cell isolation procedure, cell viability, cell yield, the cold and/or warm ischemia time prior to hepatocyte isolation, and donor characteristics were analyzed for each hepatocyte preparation (Table 1). The mean donor age was 45 years (range, 4 days to 83 years). The mean cell viability of fresh hepatocytes was 74 ± 27, with a mean yield of 10.1 ± 6.4 × 106 cells/g of tissue. Donor sex and age did not significantly affect the isolation outcome (viability and cell yields). Severely steatosic organs (steatosis >40%, samples 2, 4, and 10) gave rise to a significantly lower viability than normal livers (15 ± 4% vs. 88 ± 9%, p < 0.001). The influence of cold ischemia time in the outcome of the hepatocyte isolation procedure was analyzed in 38 small surgical human liver biopsies (<5 g). Analysis of cell viability revealed a dependence on cold ischemia time (Table 2), indicating that this time should be kept as short as possible (<15 h) prior to hepatocyte isolation. Livers from the three NHBDs (samples 8, 9, and 15; Table 1) were processed and the effect of warm ischemia time (40, 50, and 10 min, respectively) on the outcome of cell isolation process was also investigated. Cell viability and cell yield values were similar to those obtained in heart-beating donors (excluding severe and mild steatosic samples) (89 ± 6% vs. 87 ± 8%, p > 0.05 and 9.7 ± 4.8 vs. 10.1 ± 6.4 × 106 cell/g, p > 0.05, respectively).
Details of Donors and Organs

W, whole liver; LL, left lobe; RL, right lobe; NA, noradrenaline.
Correlation Between Ischemia Time and Viability
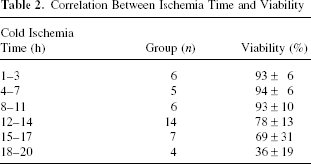
Functional Assessment of Cryopreserved Hepatocyte Preparations
Viability, attachment capability, and metabolic functionality were quantified to estimate the quality of cryopreserved hepatocyte preparations (Table 3). Viability of thawed hepatocytes significantly differed from that of freshly isolated hepatocytes (63 ± 8% vs. 87 ± 8%, n = 13, p < 0.001) (Tables 1 and 3). The attachment efficiency of hepatocytes after thawing ranged from 20% to 63% (Table 3). Preliminary experiments to improve the cryopreservation protocol were performed using hepatocyte preparations obtained from surgical biopsies with a cold ischemia time <15 h. The results showed no significant differences in cell viability between hepatocytes cryopreserved in standard and improved medium. Meanwhile, a significant increase of attachment efficiency (48 ± 12% vs. 63 ± 7%, n = 5, p < 0.05) and an improvement of monolayer formation by polygonal-shaped cells after overnight culturing (data not shown) were observed in the glucose- and dexamethasone-supplemented medium.
Functional Characterization of Cryopreserved Hepatocytes
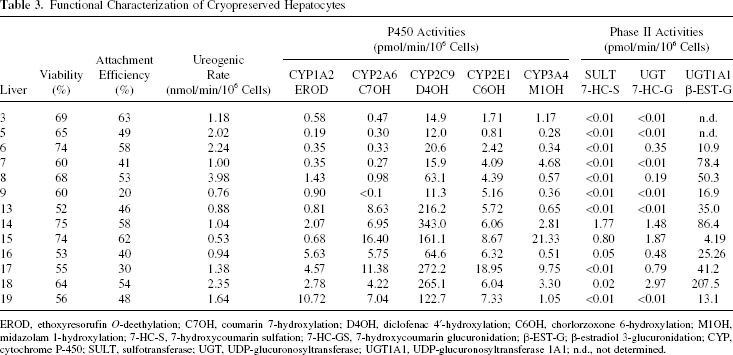
EROD, ethoxyresorufin O-deethylation; C7OH, coumarin 7-hydroxylation; D4OH, diclofenac 4′-hydroxylation; C6OH, chorlorzoxone 6-hydroxylation; M1OH, midazolam 1-hydroxylation; 7-HC-S, 7-hydroxycoumarin sulfation; 7-HC-GS, 7-hydroxycoumarin glucuronidation; β-EST-G; β-estradiol 3-glucuronidation; CYP, cytochrome P-450; SULT, sulfotransferase; UGT, UDP-glucuronosyltransferase; UGT1A1, UDP-glucuronosyltransferase 1A1; n.d., not determined.
Ureogenic capability and phase I (CYPs) and phase II (UGT, SULT) activities were used to assess the metabolic capacity of cryopreserved cells (Table 3). Hepatocytes from all the livers analyzed were able to synthesize urea from ammonia at high rates (0.53–3.98 nmol/min/106 viable cells). The lowest value corresponded to sample 15 from the youngest donor (4 days old).
Biotransformation competence was assessed by measuring the activity of five human CYPs responsible for the oxidative metabolism of drugs (CYP3A4, CY2C9, CY2E1, CY2A6, CY1A2) and the phase II activities UGT and SULT using a previously described cocktail of specific substrate probes (16). All the samples analyzed displayed measurable levels of CYP activities (Table 3), indicative of their metabolic competence. As expected, a noticeable variability was observed among the individual donors. Hepatocytes from liver 5 showed, in general, lower activities than those from the other donors, whereas the highest values were obtained from livers 14, 17, and 18. Phase II activities were determined through the formation of glucuronide and sulfate conjugates of 7-hydroxycoumarin, a substrate for several UGT and SULT enzymes. In contrast to CYPs, low UGT and SULT activities were found in most hepatocyte preparations. Reduced levels of endogenous conjugating cofactors (UDP-glucuronic acid and 3′-phosphoadenosine 5′-phosphosulfate) were reported as a potential reason for the low UGT and SULT activities, respectively, in cryopreserved hepatocytes (30).
In view of the particular importance of UGT1A1 in HTx, a new specific assay for measuring UGT1A1 activity in hepatocyte suspensions was developed in the present study. The assay consists in incubating cells with β-estradiol and UDP-glucuronic acid. The formation of estradiol-3-glucuronide, the metabolite formed by UGT1A1, proceeded linearly for a period of about 30–60 min (Fig. 1). To ensure a linear metabolite production for both the low and high activity cells, a β-estradiol concentration of 7.5 μM and a 30-min incubation time were standardized for the activity assays. Using this procedure, UGT1A1 activity was determined in cryopreserved hepatocytes from 11 donors (Table 3). All cell preparations showed measurable activity levels and, as expected, high interindividual differences (50-fold variability) were found, showing hepatocytes from donor 15 (4 days old) the lowest UGT1A1 activity.

Formation of estradiol-3-glucuronide in human hepatocyte suspension. Cryopreserved hepatocytes from three different donors were incubated at a density of 1.25 × 106 viable cells/ml in the presence of 7.5 or 15 μM β-estradiol as indicated. Glucuronide metabolite released into the incubation medium was quantified at the indicated times. Data correspond to activity assays in duplicate.
Discussion
Individuals with inborn errors of metabolism who have undergone HTx show clinical improvement and a partial correction of the underlying metabolic defect (4, 19, 22). In most cases, sustained benefits are not observed. This may be partly related to an inadequate cell dose and/or to variations in the quality of the hepatocyte preparations. To date, no general consensus exists on protocols for the use of freshly isolated or cryopreserved hepatocytes for HTx. However, cryopreservation remains the most appropriate way of cell banking, allowing a programmed cell administration to patients. Cryopreservation can adversely lead to both the structural and functional impairment of hepatocytes (29). Nevertheless, it has been reported that cell membrane stabilization by dexamethasone (15, 25) and a high concentration of glucose as an energy source (28) are apparently key factors contributing to the better preservation of hepatocytes, likely contributing to help engraftment of transplanted cells in the liver, which is a critical step for the success of hepatocyte transplantation (18, 20, 21, 27). In agreement with these previous observations, our results comparing standard with improved cryopreservation medium showed an improvement of attachment efficiency and monolayer formation with glucose- and dexamethasone-supplemented medium.
Factors directly derived from the differences in the procurement of the liver sample notably influence the quality of hepatocyte preparations. Livers from organ donors are perfused in situ with a cold preservation solution to avoid warm ischemia, and the tissue is usually maintained under these conditions for several hours until hepatocyte isolation. According to our experience (Table 2), a short cold ischemia time (<15 h) is recommended to prevent harmful effects on cells. In addition, low levels of steatosis (<40%) yield a higher viability and number of hepatocytes (6, 9, 17). Because one of the limitations of HTx is the restricted availability of donor liver tissue, we evaluated the suitability of livers exposed to warm ischemia, such as those from the NHBDs, as a source of hepatocytes. Cell viability of >60% is required for HTx (2, 5, 13, 14, 23, 26). Both fresh and cryopreserved hepatocytes from the three NHBD livers processed meet this criterion (Tables 1 and 3). In hepatocytes isolated from NHBD tissue, not only cold ischemia but also warm ischemia can be a major concern. A previous study found a negative correlation between hepatocyte viability and the length of warm ischemia (14). The high viability and good functionality shown by hepatocytes isolated from the three NHBD livers can be due to the relatively short warm and cold ischemia times (<50 min and <6 h, respectively).
Recently, we reported a reliable assay to assess the functionality of fresh hepatocytes or recently thawed hepatocytes (7). However, a more careful and complete analysis of cell quality based on the function/activity of interest would allow the customization of hepatocyte preparation for each receptor. UCD and CNI are the metabolopathies most frequently treated with HTx (4, 19, 22). Urea is the main form of ammonium removal, and a deficiency in any of the five enzymes of the urea cycle causes UCD. HTx is one of the techniques applied to treat disorders due to the deficient expression of urea cycle enzymes, carbamyl phosphate synthetase I or ornithine transcarbamylase. We previously described a rapid assay to determine the capacity of hepatocytes to synthesize urea from ammonia, which indicates that this pathway is fully active (7). This is of great help to choose the appropriate hepatocyte batches to be infused in patients with UCDs.
Biotransformation capacity is of great importance for HTx, and this function has been proposed as the functional criteria of human hepatocyte preparations (1, 8, 21). Drug-metabolizing enzymes are among the most sensitive liver functions for the isolation process, and high preparation-to-preparation differences are usually found in hepatocytes obtained from different donors (5) (Table 3). To date, most studies on drug metabolism focus on oxidations catalyzed by CYPs, and little attention has been paid to conjugating enzymes. However, the relevance of phase II reactions is increasingly recognized. Recently, we developed a new cocktail strategy for the simultaneous determination of five individual CYP activities, and also of UGT and SULT enzymes in hepatocytes (16). Glucuronidation and sulfation of 7-hydroxycoumarin are often used as indicators of the phase II function in human hepatocyte preparations (20, 21, 30).
Our results are in good agreement with previous studies showing that glucuronidation by UGTs is the major route of phase II metabolism of 7-hydroxycoumarin, whereas sulfation activity is low, even undetectable, in most preparations, particularly for those of cryopreserved hepatocytes (Table 3) (20, 21). Some authors found significant alterations of phase II drug-metabolizing activities during hepatocyte cryopreservation (1, 24), whereas most studies reported slight or no appreciable losses of functional competence (3, 11, 21). Therefore, we consider that the evaluation of conjugating activities, particularly UGT1A1, in the panel of assays to assess functionality of hepatocytes is essential. In the present article, we describe a procedure for the first time that not only estimates the general glucuronidation capacity of hepatocytes for HTx purposes, but also a specific assay for examining UGT1A1. In view of the great variability in UGT1A1 (estradiol 3-glucuronidation) activity levels observed for cryopreserved hepatocytes from different donors (Table 3), this specific assay could be used as a key criterion in the selection of hepatocyte preparations for HTx in CNI patients. By way of example, thawed hepatocytes from liver 15 (with a high viability, attachability and CYP function, but low ureogenic and UGT1A1 activities) did not seem to be the most appropriate for the treatment of UCD or CNI disorders. Cell preparations from other donors should be recommended as the first choice for HTx of patients with UCD (i.e., livers 8 or 18) or CNI (i.e., livers 14 or 18).
In short, we found that a) good quality hepatocytes can be obtained from NHBD livers, which can extend the pool of available organs; b) grafts with cold ischemia >15 h and >40% steatosis should not be accepted for HTx; and c) the assessment of specific functional parameters can help to make decisions about the suitability of each hepatocyte batch for a given recipient, thus contributing to improve the probability of a successful clinical outcome.
Footnotes
Acknowledgments
The authors thank the financial support of the ALIVE Foundation, the European Commission (LSSB-CT-2004-037499), and the Regional Valencian Government (Generalitat Valenciana) (Ev-Tec 003/200-2005). The technical assistance of S. López and A. Negro is also acknowledged. A.L. gratefully acknowledges Ministerio Ciencia e Innovación/Instituto de Salud Carlos III for a Miguel Server contract.
