Abstract
Hepatocyte transplantation (HCT) is a potential bridging therapy or an alternative to liver transplantation. Conventionally, single-cell hepatocytes are injected via the portal vein. This strategy, however, has yet to overcome poor cell engraftment and function. Therefore, we developed an orthotopic HCT method using a liver-derived extracellular matrix (L-ECM) gel. PXB cells (flesh mature human hepatocytes) were dispersed into the hydrogel solution in vitro, and the gel solution was immediately gelated in 37°C incubators to investigate the affinity between mature human hepatocyte and the L-ECM gel. During the 3-day cultivation in hepatocyte medium, PXB cells formed cell aggregates via cell–cell interactions. Quantitative analysis revealed human albumin production in culture supernatants. For the in vivo assay, PXB cells were encapsulated in the L-ECM gel and transplanted between the liver lobes of normal rats. Pathologically, the L-ECM gel was localized at the transplant site and retained PXB cells. Cell survival and hepatic function marker expression were verified in another rat model wherein thioacetamide was administered to induce liver fibrosis. Moreover, cell–cell interactions and angiogenesis were enhanced in the L-ECM gel compared with that in the collagen gel. Our results indicate that L-ECM gels can help engraft transplanted hepatocytes and express hepatic function as a scaffold for cell transplantation.
Introduction
Hepatocyte transplantation (HCT) has been explored as a less invasive therapy for patients with acute liver failure or metabolic liver diseases, with the potential to be a bridging therapy or alternative to liver transplantation. HCT was first performed in 1976 in a rat model of Crigler-Najjar syndrome 1 . Since the first attempt at HCT in a human patient with cirrhosis in 1992 2 , HCT has been performed in over 100 patients with liver disease globally, with intraportal single-cell injection being the standard procedure 3 . Nevertheless, the therapeutic impact of intraportal HCT is limited because of inadequate cell engraftment caused by cell emboli and instant blood-mediated inflammatory reactions4,5. Moreover, in patients with cirrhosis, an end-stage liver disease, cell engraftment was even poorer due to the negative effect of portal hypertension 6 . These impediments continue to hinder the use of HCT in clinical settings.
To circumvent the difficulty of intraportal HCT, ectopic HCT of the spleen, 7 subcutaneous spaces, 8 lymph nodes, 9 and intraperitoneal cavity 10 has been reported. Furthermore, scaffold biomaterials, including cell sheets and hydrogels, have gained prominence for ectopic HCT owing to their ability to retain transplanted hepatocytes in space11–14. These ectopic transplantations successfully maintain hepatic function 15 . However, the confinement of transplanted hepatocytes to the ectopic space limits cell–cell and cell–extracellular matrix (ECM) interactions inherent in the tissue-specific microenvironment of the liver. These organ-specific environments are essential for hepatocyte proliferation and expression of inherent functions16,17.
Many studies have demonstrated the use of orthotopic HCT coupled with biomaterials, such as cell sheets and patch grafts, as another approach to HCT18,19. These studies reported contact between transplanted hepatocytes and the liver parenchyma of the recipient, as well as substantial cell engraftment. Recently, biological scaffolds have attracted the interest of researchers because of their biocompatibility and bioactivity 20 . ECM hydrogels prepared by solubilizing decellularized tissues and removing cells and nucleic acid components are beneficial for cell culture 21 . They have been reported to promote the inherent functions of hepatocytes both in vitro and in vivo22,23. Angiogenesis is another important factor in hepatocyte engraftment 24 , and is related to the long-term engraftment of transplanted hepatocytes 25 . Kidney-derived decellularized ECM hydrogels have also been reported to enhance vascularization26,27.
In this study, we hypothesized that a decellularized liver hydrogel contributes to the inherent function of transplanted hepatocytes via self-organization and vascularization and human donor-derived human primary hepatocytes vary with donor age, condition, method of cell isolation, and genetic background 4 . In this study, we used PXB cells that are pure human hepatocytes freshly isolated from PXB mice, which have been used in some in vitro and in vivo studies28,29. Here, we developed a liver-derived extracellular matrix (L-ECM) gel that functions as a scaffold and investigated the orthotopic transplantation of mature hepatocytes in a fibrotic liver model. Our findings will help establish the therapeutic potential of L-ECM hydrogels as scaffolds for metabolic diseases, acute liver failure, and end-stage liver cirrhosis.
Materials and Methods
Animals
Adult female Sprague–Dawley (SD) rats between 8 and 16 weeks of age (weighing approximately 180–250 g) were used for all gel implantation experiments. All the rats were bred ad libitum in a room at 24–25°C, 40–70% humidity, and 12 h of light and dark. Göttingen minipigs (Oriental Yeast Co., Ltd, Tokyo, Japan) weighing approximately 13–15 kg were used as the source of L-ECM gel. All experiments were approved by the Institutional Animal Care Committee. The experimental procedures and protocols were approved by the Animal Ethics Committee of Keio University Tokyo, Japan (approval number: A2022-229), and were performed in accordance with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health, Bethesda, MD, USA). Animal experiments were performed according to the ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines 2.0, as a guideline for animal experiments, and all necessary checklist criteria were met.
Human Hepatocytes
HepG2 and PXB cells were obtained from Cellular Engineering Technologies, Inc, and Phoenix Bio, Japan, respectively. Thus, human donor-derived human primary hepatocytes are not good candidates for proof-of-principle HCT. Moreover, human-induced pluripotent stem (iPS) cell-derived hepatocytes are generally immature, and hepatocyte isolation suitable for clinical application has not yet been developed. Therefore, in this study, we used PXB cells, which are mouse-derived primary human hepatocytes. For HepG2 culture, the HepG2 Hepatocellular Carcinoma Expansion Media was used. PXB cells were cultured in six-well plates according to the manufacturer’s instructions.
Decellularization of Porcine Liver
L-ECM was prepared according as previously described, with some modifications27,30. Briefly, livers were removed from the disinfected peritoneal cavity of pigs, frozen at −80°C, and thawed at 4°C for over 24 h for the freeze-thaw process, a common initial decellularization step. Porcine liver was cut into small pieces using the food dicer DC-203 (Emura Food Machine Co, LTD, Japan) for uniform tissue decellularization. The fragments were then rinsed three times a day for 2 days at 4°C with phosphate-buffered saline (PBS; Fujifilm Wako Pure Chemical Inc, Osaka, Japan) under constant stirring. Thereafter, the fragments were decellularized with 1% TritonX-100 (Sigma-Aldrich Co., Tokyo, Japan) and ammonium hydroxide under continuous stirring for 5 days. To remove the remaining detergent, pieces of decellularized liver were rinsed with PBS, and the solution was changed three times per day for 2 days. The pieces were then mixed with PBS containing an antibiotic, antimycotic (Thermo Fisher Scientific K.K., Tokyo, Japan), and 300 μg/ml sodium colistin methanesulfonate (Fujifilm Wako Chemicals, Osaka, Japan) for 1 h, followed by the addition of PBS containing 1× antibiotic–antimycotic (Nacalai Tesque Co., Ltd, Tokyo, Japan) and 200 μg/ml gentamicin sulfate (Nacalai Tesque Co., Ltd, Tokyo, Japan) for 1 h. Decellularized liver ECM (L-dECM) powder was produced after 3 days of lyophilization and subsequent milling.
Hepatocyte Cultivation in Hydrogel
Cultured PXB and HepG2 cells were individually dissociated using Accutase (Thermo Fisher Scientific, USA) and mixed with the gel solution. In total, 8.0 × 105 human hepatocyte cells (PXB or HepG2) were encapsulated in a solution of 40 and 200 μl of either the L-ECM gel or collagen gel. Following immediate gelation in an incubator for 10 min, three-dimensional (3D) culture was initiated in the medium. After 3–6 days, we conducted enzyme-linked immunosorbent assay (ELISA) and quantitative real-time polymerase chain reaction (qRT-PCR) on the supernatant to assess human albumin production and hepatocyte-related gene expression, respectively. In addition, histological examinations of the cells and hydrogel were performed along with immunostaining to evaluate protein expression. PXB cells were encapsulated in the hydrogel and grown in Corning’s ultra-low attachment six-well plates.
Protein Analysis
To analyze the samples, each sample was first digested and peptized in a solution containing 0.001% trypsin, 10 μM Tris-HCl, 0.005% n-octyl glucopyranoside, and 0.7 M guanidine hydrochloride (pH 8.8) for 12 h. Subsequently, the resulting peptides were reduced with 5 mM Tris(2-carboxyethyl) phosphine for 30 min at 65°C and alkylated with 10 mM iodoacetamide for 30 min at 25°C. The resulting digested peptides were then analyzed using an Evosep One LC system (EVOSEP) connected to a Q-Exactive HF-X mass spectrometer (Thermo Fisher Scientific). The mobile phase consisted of 0.1% formic acid (solution A) and 0.1% formic acid/99.9% acetonitrile (solution B). The analysis was performed in the data-dependent acquisition mode, and the top 25 recorded mass spectrometry spectra between 380 and 1,500 m/z were selected for further analysis. All MS/MS spectra were then searched against the protein sequences of the Sus scrofa (NCBI: txid9823) protein database using Proteome Discoverer 2.2 with the SEQUEST search engine. The peptide spectrum match was subjected to a false discovery rate (FDR) set at 1% for reliability. GraphBio was used to generate a heat map of the matrisomes identified by LC-MS.
Electron Microscopy
The resulting organ-derived hydrogel solution was placed onto a glass slide and was transferred to the gel in the same conditions described above. The L-dECM gel was fixed on a glass slide in freshly prepared 2.5% glutaraldehyde solution for scanning electron microscopy. Samples were fixed with 1.0% osmium tetroxide (TAAB Laboratories) for 2 h at 4°C, rinsed in 0.1 M phosphate buffer (pH 7.4, Muto Pure Chemicals), and then treated with a series of escalating ethanol concentrations. The samples were coated with osmium to a thickness of approximately 2 nm using a conductive osmium coater after drying using a critical point drier (CPD300, Leica Biosystems; Neoc-ST, Meiwafosis). Using an SU6600 (Hitachi High Tech) apparatus, scanning electron microscopy images were captured at 5 keV.
Rheological Characterization
Collagen concentration in the matrix gel solution was measured for each L-dECM concentration (8, 12, 16, and 20 mg/kg).
The rheological characteristics of the L-dECM and rat tail collagen I hydrogels (Corning) were assessed using a longitudinal vibration viscoelastic meter (PZ-Rheo NDS-1000, Taisei Co., Ltd, Japan). The samples were tested at 25°C with a fixed frequency of 3 Hz and amplitude of 250 m to determine the dynamic viscosity. Compression tests were conducted to determine the storage modulus. Samples were measured using a 10-mm diameter parallel plate at 25°C with a fixed frequency of 3 Hz and an amplitude of 10 µm.
The rheological characteristics of the solubilized L-ECM gel were investigated under the same conditions. In the linear regime, the total resistance of the hydrogel to deformation was assessed by the complex viscoelastic modulus (E* = E′ + iE′′), where E′ represents the storage modulus and E′′ represents the loss modulus. Moreover, tan delta (δ = E′′/E′) represents the frictional dissipations of the hydrogel.
Hepatocyte Transplantation
SD rats were used for in vivo experiments. Figure 4A depicts the surgical procedures. Splenectomy was performed after the incision. The left lateral lobe was mobilized into the median lobe. Suturing of the lateral left, right, and internal left lobes with 3-0 vicryl was performed and a pocket was made. Peeling of the bottom of the pocket on the lateral left lobe with a large 12 mm diameter biopsy trepan (Kai Corporation, Japan). Subsequently, the PXB or HepG2 grafts were transplanted into the pocket. In the rat model of liver fibrosis, two sets of 8.0 × 105 cells of PXB embedded in 40 μl of L-ECM gel were transplanted. PXB cells were cultured in the L-ECM gel for 3 days before transplantation. HepG2 cells were not pre-cultured in L-ECM gel before transplantation. The procedure was finalized by suturing the lobes and securely packing the gel into the liver lobes.
Postoperative Management
Immunosuppressive agents were administered daily after transplantation. According to the protocol 31 , 20 mg/kg of PSL was administered intramuscularly, and 1 µg/kg of tacrolimus was administered intrasubcutaneously. Ceftriaxone (20 mg/kg), an antibiotic, was administered daily for 1 or 2 weeks. The rats were sampled 1 week after transplantation. For long-term experiments, rats were sampled 2 weeks after transplantation. Whole livers were resected, and major organs were preserved, including the heart, lungs, and kidneys. The postoperative schedule is shown in Fig. 4B.
Biochemical Blood Examination
Blood (500 µl) was collected from the subclavian vein of the rats. Of this volume, 100 µl was used for biochemical analysis with the Piccolo Xpress (Central Scientific Commerce, Inc, Japan) to assess liver enzyme levels and complete blood count in the rat. The remaining blood was used for ELISA to evaluate the ability of transplanted PXBs to produce human albumin.
Gene Expression Analysis
Total RNA from HepG2 and PXB cells was extracted using the RNeasy Plus Mini Kit (Qiagen, USA). QuantiTect Rev was used to generate cDNAs from 500 ng of total RNA for RT-PCR. A transcription kit (Qiagen, USA) was used according to the manufacturer’s instructions. SYBR Green PCR Master Mix was used for PCR (Thermo Fisher Scientific, USA). Supplemental Table 1 lists the sequences of the used primers. Rhelixa, Inc carried out RNA sequencing (RNA-seq) for a whole-transcriptome study (Tokyo, Japan). Briefly, library construction was performed using the NEBNext® Ultra™ II Directional RNA Library Prep Kit (Illumina) according to the manufacturer’s instructions. Sequencing was performed on the Illumina NovaSeq 6000 platform. Expression profiles were derived as read counts and fragments per kilobase of transcripts per million mapped reads for each sample, transcript, and gene. The Benjamini–Hochberg approach was also used to identify differentially expressed genes (DEGs) using the criteria of|log2 (Fold Change, FC)| > 1 and P value 0.05. Using ggVolcanoR, a volcano plot was produced using the FC and P value of the DEGs. Using GraphBio, heat maps were produced using Z-scores of normalized numbers. GOATOOLS (version 1.1.6) was used for GO analysis of DEGs, whereas ClusterProfiler was used for GSEA (Version 3.12.0).
Protein ELISA
Human albumin was quantified in the culture media and rat blood samples using the Human Albumin ELISA Kit (Bethyl Laboratories, Inc) on day 3. Rat blood samples were collected and assayed on days 3, 5, 7, and 14 after transplantation. Rat blood was collected via the portal vein on day 7 in the 1-week transplantation experiment and on day 14 in the 2-week transplantation experiment.
Histological Analysis
The harvested livers were fixed using 4% paraformaldehyde for 48 h. Sections were immersed in paraffin blocks, cut to a thickness of 0.3 µm, and stained with either azan or hematoxylin and eosin (H&E) (Sigma-Aldrich Co.) according to established procedures. The slides were imaged using a BZ-X800 microscope (KEYENCE Co., Osaka, Japan).
Immunohistochemical Analysis
The paraffin-embedded sections were deparaffinized and rehydrated. Citrate buffer (Dako, Agilent Technologies Japan, Ltd) was used to extract the antigens from each segment for 15 min at 120°C. The sections were blocked with 1% bovine serum albumin (Sigma-Aldrich Co.) and treated with a primary antibody solution at 4°C overnight. The primary antibodies and dilutions were as follows: anti-CK8/18 (P05786, Progen Bio), anti-human albumin (ab10241, Abcam), anti-claudin-1 (Cell Signaling Technology, 13255), anti-E-cadherin (ab76055, Abcam), anti-collagen IV (ab6586, Abcam), anti-HNF 4α (3113S, CST), ani-Pcam1 (ab182931, Abcam), anti-human mitochondria (Merck, MAB1273), anti-DPP IV (Cell Signaling Technology, 40134S), anti-ZO-1 (Invitrogen, 33-9100), anti-integrin (Abcam, ab52971), and laminin (ab11575, Abcam).
After rinsing, the sections were mounted with DAPI (4′,6-diamidino-2-phenylindole) and ProLong Diamond Antifade Mountant (Invitrogen Inc, Carlsbad, CA, USA). The slides were imaged using a BZ-X810 microscope (KEYENCE Co., Osaka, Japan). Claudin-1- or Pcam1-positive areas were counted using ImageJ. Three samples were randomly chosen and claudin-1- or Pcam1-positive area were counted under the same conditions (range, threshold).
Statistical Analysis
Paired continuous data were evaluated using Student’s t test. Statistical significance was defined as P < 0.05. SPSS (SPSS 28.0, Chicago, IL, USA) was used for statistical analysis.
Results
Characterization of the L-ECM Gel
Figure 1A depicts the protocol for preparing the L-ECM gel via decellularization, lyophilization, pulverization, acidification, solubilization, neutralization, lyophilization, dilution, and gelation. Proteomic analysis revealed the components of porcine liver and L-ECM gel (Fig. 1B), indicating that the L-ECM gel contained liver-specific heterogeneous matrisomes and non-matrisomes. Matrisomes detected in the liver and L-ECM gel were the most common, and liver-specific ECM components were retained (Supplemental Figure 1). Figure 1C shows the ratio of the ECM characteristics of the matrisome detected in the L-ECM gel via proteomic analysis. Type IV collagen and laminin, which are the components of the basement membrane, were detected. Moreover, ANXA2 32 , which is associated with angiogenesis; FBN1, a promoter of ECM integrity 33 ; BGN, a key factor in scaffolding the collagen fibrils that mediates cell signaling 34 ; and FLNA 35 , a crosslinking dynamic 3D structure, were detected. A heat map of all matrisomes detected via proteomic analysis showed that several components, such as type IV and type I collagen, FLNA, and FBN1, were more concentrated in the L-ECM gel than in the liver (Fig. 1D).
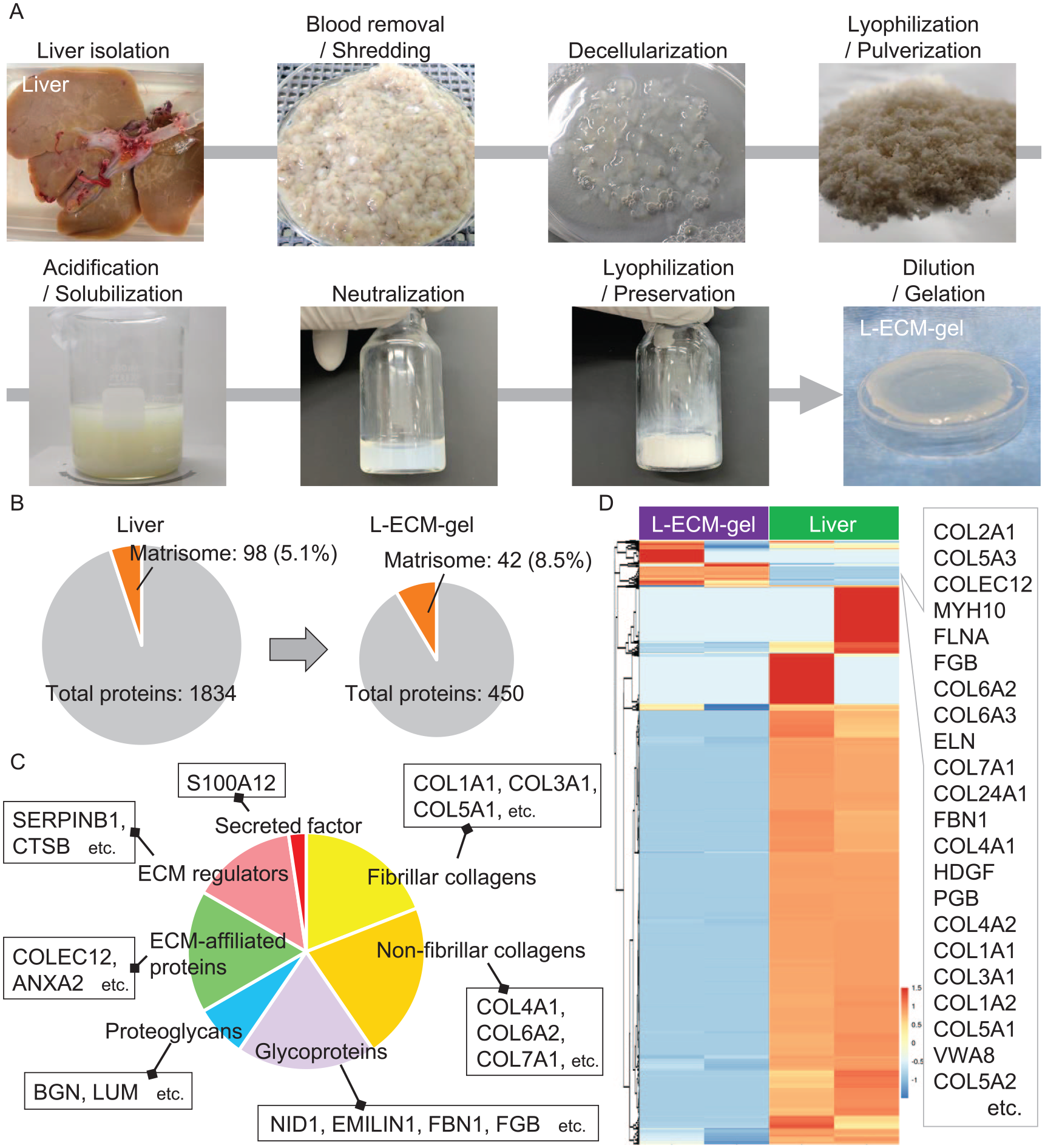
(A) Protocol for preparing decellularized liver hydrogel and the characteristics of liver-derived extracellular matrix (L-ECM) gel. The porcine liver is minced using a food dicer. This decellularization takes 5 days. Subsequently, freeze-drying and solubilization were performed. The appearance of pre-decellularized tissues, post-decellularized tissues, and hydrogel is shown. (B) Pie graph from proteomic analysis of the liver and L-ECM gel. (C) The ratio of matrisome components in L-ECM gel detected by proteomic analysis. (D) Hierarchical clustering analysis of matrisomes in the liver and the L-ECM gel.
Evaluation of the Proper Viscoelasticity of the L-ECM Gel for Hepatocytes In Vitro and In Vivo
The L-ECM gel was classified according to its viscoelasticity, n = 3, Fig. 2A. Electron microscopy revealed that the L-ECM gel had a structure similar to that of the collagen fibers in vitro (Fig. 2B). HepG2 cells are widely used as a model for evaluating a new hepatocyte culture system. Rajalekshmi et al. 36 reported that a fibrin-incorporated alginate dialdehyde–gelatin hydrogel has the potential to be used as a scaffold for liver tissue engineering using HepG2 cells. Moreover, Mirdamadi et al. 37 reported that a 3D-printed poly-lactic acid (PLA)/Gel hybrid has architectural effects on biological functions in HepG2 cells. Thus, we used HepG2 cells as a model when initially evaluating the correlation between gel stiffness and hepatocyte functionality and establishing a 3D culture system. For both functional analysis and handling evaluation, HepG2 cells were cultured for 6 days in the L-ECM gel according to viscoelasticity. Physical stability was achieved in a stiff L-ECM gel. In contrast, HepG2 cells did not survive in the stiff L-ECM gel (Fig. 2C). In soft and intermediate L-ECM gels, HepG2 cells survive and form a spheroid-like structure. However, soft gels are extremely fragile. Figure 2D depicts human albumin production in HepG2 cells as measured by ELISA. This was similar for the soft and intermediate L-ECM gels. Relative gene expression of hepatocyte markers and hepatic function, including ALB, AFP, EPCAM, and HNF4α, was similar between soft and medium L-ECM gels (n = 3) (Fig. 2E). Based on these results, we determined that the actual viscoelasticity of the L-ECM gel for the in vivo experiments was intermediate. Immunohistochemistry analysis of HepG2 cells within the intermediate L-ECM gel demonstrated that HepG2 cells expressed the hepatocyte marker (CK8) and hepatic function (HNF4α and albumin) within the L-ECM gel, which is rich in collagen IV (Fig. 2F).
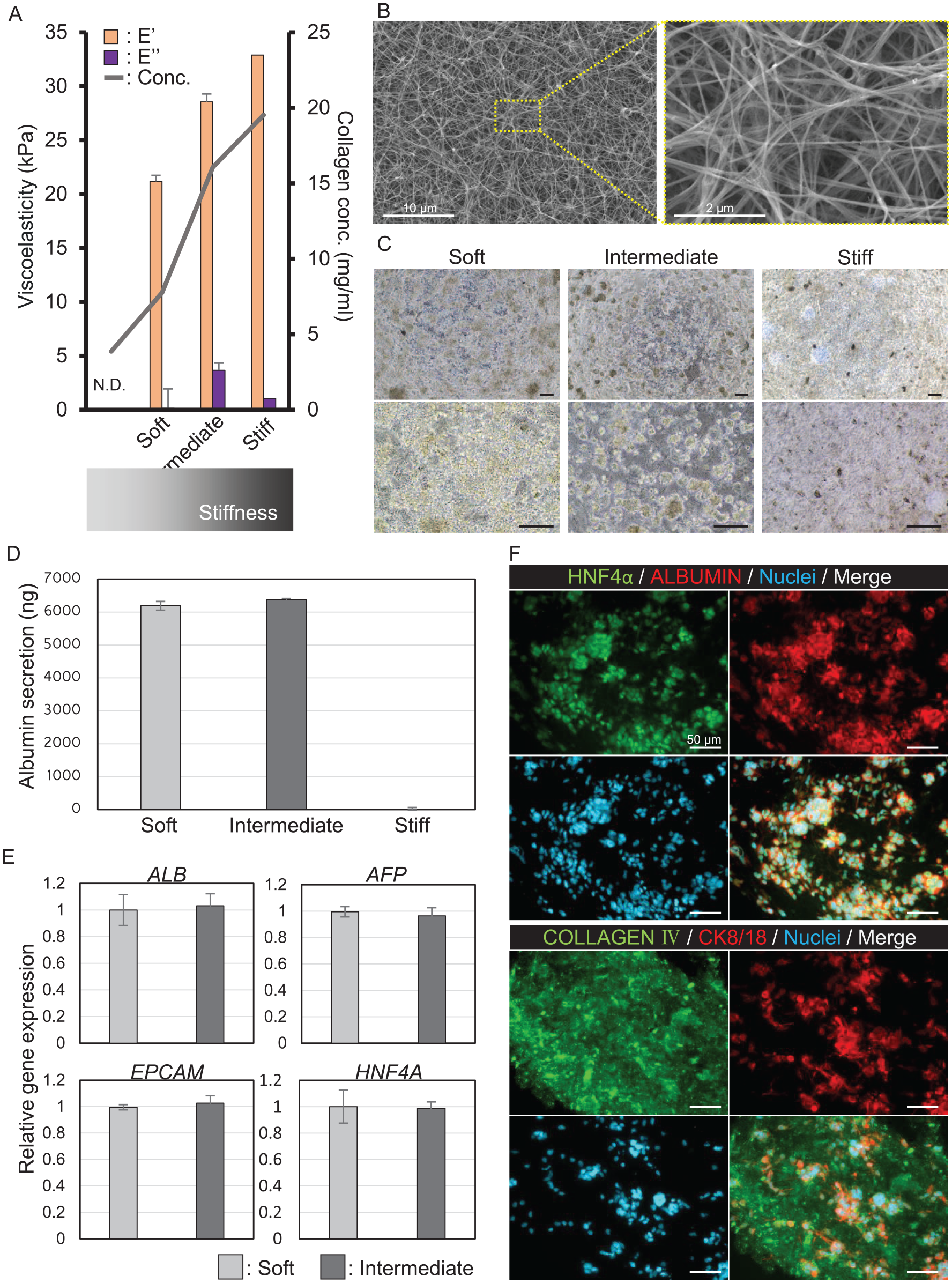
Characterization of HepG2 in L-ECM gel according to the viscoelasticity of the L-ECM gel. (A) Viscoelasticity of the L-ECM gel. Ultrasoft, soft, medium, and hard characteristics were determined by complex viscoelastic modulus (E′E′′ [kPa]). The collagen concentration is described as a line graph. (B) Electron microscopy of the L-ECM gel. (C) The appearance of hepatocytes (HepG2) in the L-ECM gel in vitro according to the viscoelasticity of the gel. Scale bar, 50 μm. (D) Human albumin production by hepatocytes (HepG2) in culture medium according to the gel viscoelasticity. (E) Relative gene expression of hepatocytes (HepG2) according to gel viscoelasticity. (F) Immunostaining analysis of HepG2 cells encapsulated in the L-ECM gel. Scale bar, 50 μm.
Functional and Histological Analyses of the L-ECM and Collagen Gels as Scaffold for Hepatocytes In Vitro
The functional and histological findings of PXB cells encapsulated in the L-ECM gel were compared with those in the collagen gel in vitro (Fig. 3A). For the PXB cells, a dimpled parafilm substrate was prepared to generate L-ECM gel droplets by layering a square of parafilm over an empty tip tray for 200 µl tips 28 . The intermediate viscoelasticity of the L-ECM gel was employed for in vitro analysis. PXB cells were cultured for 3 days before being characterized on the last day. Figure 3B shows macroscopic changes in hepatocytes cultivated in the L-ECM gel between days 0 and 1, indicating cell aggregation in the L-ECM gel. Human albumin production was similar between the two hydrogels in quantitative ELISA (L-ECM gel, n = 7; collagen gel, n = 5), as shown in Fig. 3C. Immunohistochemistry analysis of PXB cells within L-ECM gel demonstrated that PXB cells expressed hepatocyte marker (CK8), hepatic function (HNF4α and albumin), and intercellular adhesion marker (E-cadherin) within the L-ECM gel, which is rich in collagen IV (Fig. 3D). Relative gene expression analysis revealed that hepatocyte markers and PXB-cell hepatic function were not significantly different between the L-ECM and collagen gels; however, CYP3A4 expression was reduced in the L-ECM gel without a significant difference (P < 0.10). The expression of cell–cell interaction markers, such as CLDN1 and CLDN4, and Yes-associated protein (YAP) pathways, such as CCN2 and CCN1, was significantly increased in the L-ECM gel (Fig. 3E). RNA sequencing was performed to investigate the gene behavior of PXB-cell in two hydrogels: L-ECM and collagen gels. The volcano plot revealed that the expression of albumin, HNF4α, and EPCAM in PXB cells was not different in the L-ECM and collagen gels, as well as in the PCR data. In addition, the expression of hepatic function markers, such as CYP3A4 and UGT1A, was downregulated in the L-ECM gel compared with that in the collagen gel. In contrast, the expression of markers related to endothelial-to-mesenchymal transition (EMT) such as TWIST2, LOX, and SNAIL, was upregulated in the L-ECM gel (Supplemental Figure 2A). Moreover, gene set enrichment analysis (GSEA) demonstrated that the expression of SMAD2 and SMAD4, downstream genes of the YAP pathway, and EZH2, an EMT-related gene, was significantly enriched in L-ECM gel, whereas the expression of liver-specific genes was significantly reduced (Supplemental Figure 2B). Figure 3F shows the gene sets of mature hepatocytes, EMT, partial EMT, and related angiogenesis of PXB cells encapsulated in hydrogels (L-ECM gel; n = 3, collagen gel; n = 1) using RNA sequencing. A gene set of mature hepatocytes, including CYP3A4, was downregulated in the L-ECM gel. EMT genes, such as SNAI2 and TGFBI, were partially upregulated in the L-ECM gel. Moreover, a gene set of partial EMT- and angiogenesis-related genes was upregulated in the L-ECM gel. Gene ontology (GO) enrichment analysis revealed that cell–cell signaling, growth factor activity, angiogenesis, and integrin complexes were enhanced in the L-ECM gel (Fig. 3G).
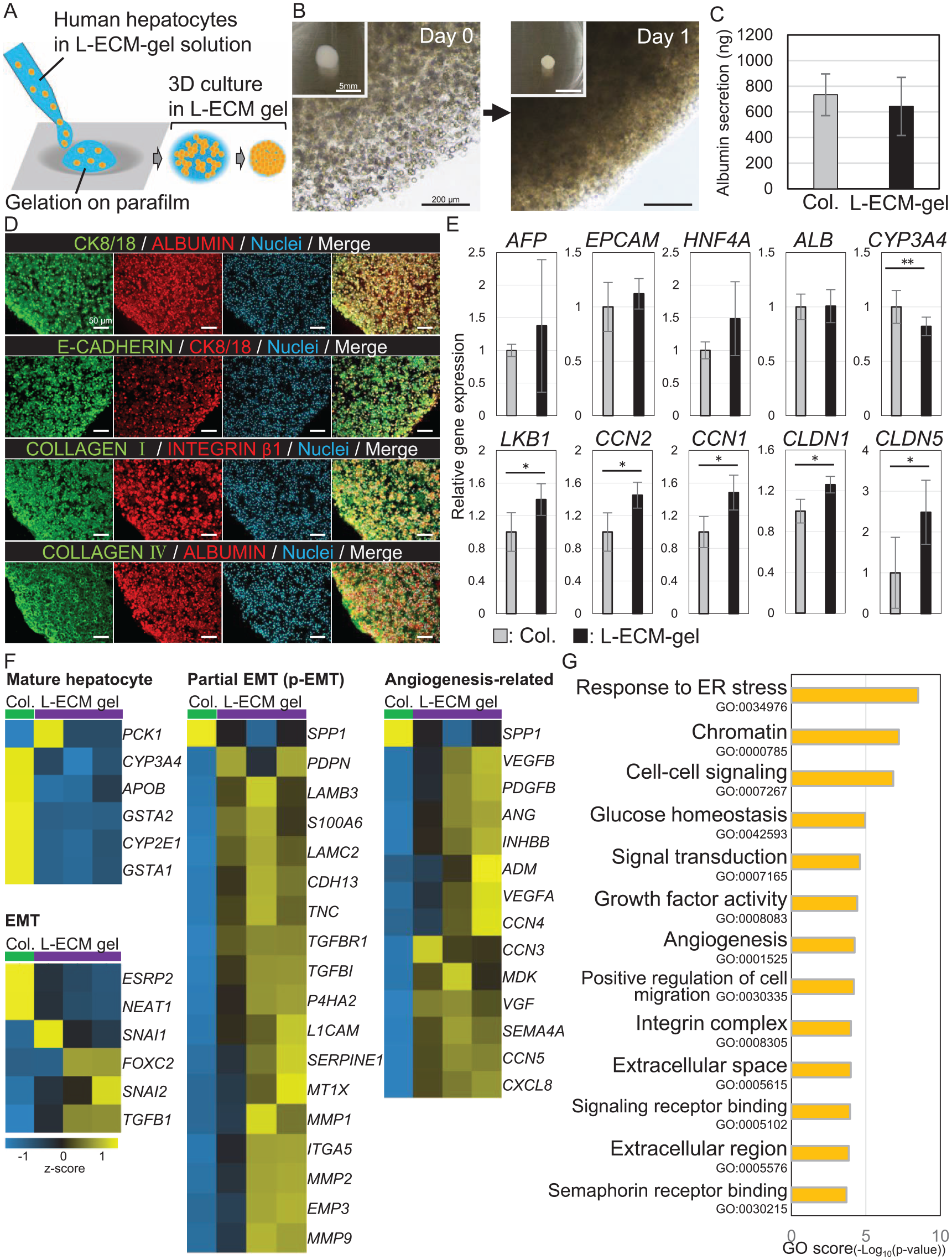
In vitro analysis of hepatocyte (PXB) function in the L-ECM gel vs collagen gel. (A) Scheme of the in vitro analysis of hepatocytes (PXB) in the L-ECM gel. (B) The appearance of hepatocytes in vitro in the L-ECM gel on days 0 and 1. Scale bar, 200 μm. (C) Human albumin production by hepatocytes in the culture medium. There is no statistically significant difference between PXB in the L-ECM and collagen gels. (D) Immunostaining of hepatocytes in the L-ECM gel. Scale bar, 50 μm. (E) Gene expression in the hepatocytes. The expression of AFP, ALB, and EPCAM is not significantly different between hepatocytes in the L-ECM gel and those in the collagen gel. LKB1, CLDN1, and CLDN4 expression is significantly higher in the L-ECM gel than in the collagen gel. (F) Hierarchical clustering profiles of hepatocytes encapsulated in the L-ECM (n = 3) and collagen gels (n = 1). Higher expression relative to the median expression value (black) is indicated in yellow, and lower expression is indicated in blue. (G) Gene ontology (GO) analysis of hepatocytes encapsulated in the L-ECM gel vs collagen gel. The y-axis refers to GO categories. The x-axis shows the GO scores. * and ** indicate P < 0.05, P < 0.10, respectively.
HCT With the L-ECM Gel as a Scaffold
PXB cells were embedded in the L-ECM gel and transplanted into the liver lobes of SD rats. The surgical procedure is illustrated in Fig. 4A. These procedures enabled the PXB cells embedded in the L-ECM gel to remain on the liver surface. The perioperative management is shown in Fig. 4B. Splenectomy was performed during transplantation as an immunosuppressive control, and dual immunosuppressive agents FK506 and methylprednisolone were administered daily. One week after transplantation, the native livers were sampled (n = 5). In the quantitative analysis, human albumin was detected in rat blood by ELISA and increased linearly after transplantation (Fig. 4C). Macroscopic findings demonstrated that the L-ECM gel was sandwiched between the liver lobes 1 week after transplantation (Fig. 4D). H&E staining revealed that the L-ECM gel contained cells (Fig. 4E). The L-ECM gel remained at the transplanted site and contained PXB (human albumin-positive) cells (Fig. 4F). Supplemental Figure 3 shows the results of immunostaining of PXB cells transplantation with collagen gel in normal rats for a week. Immunostaining of engrafted hepatocytes (CK8/18, human albumin-positive) was performed. Moreover, claudin-1 expression was detected in PXB, indicating that cell–cell interactions exist in PXB encapsulated in L-ECM gel. These results suggested that PXB survived in the L-ECM gel and expressed inherent hepatic function.
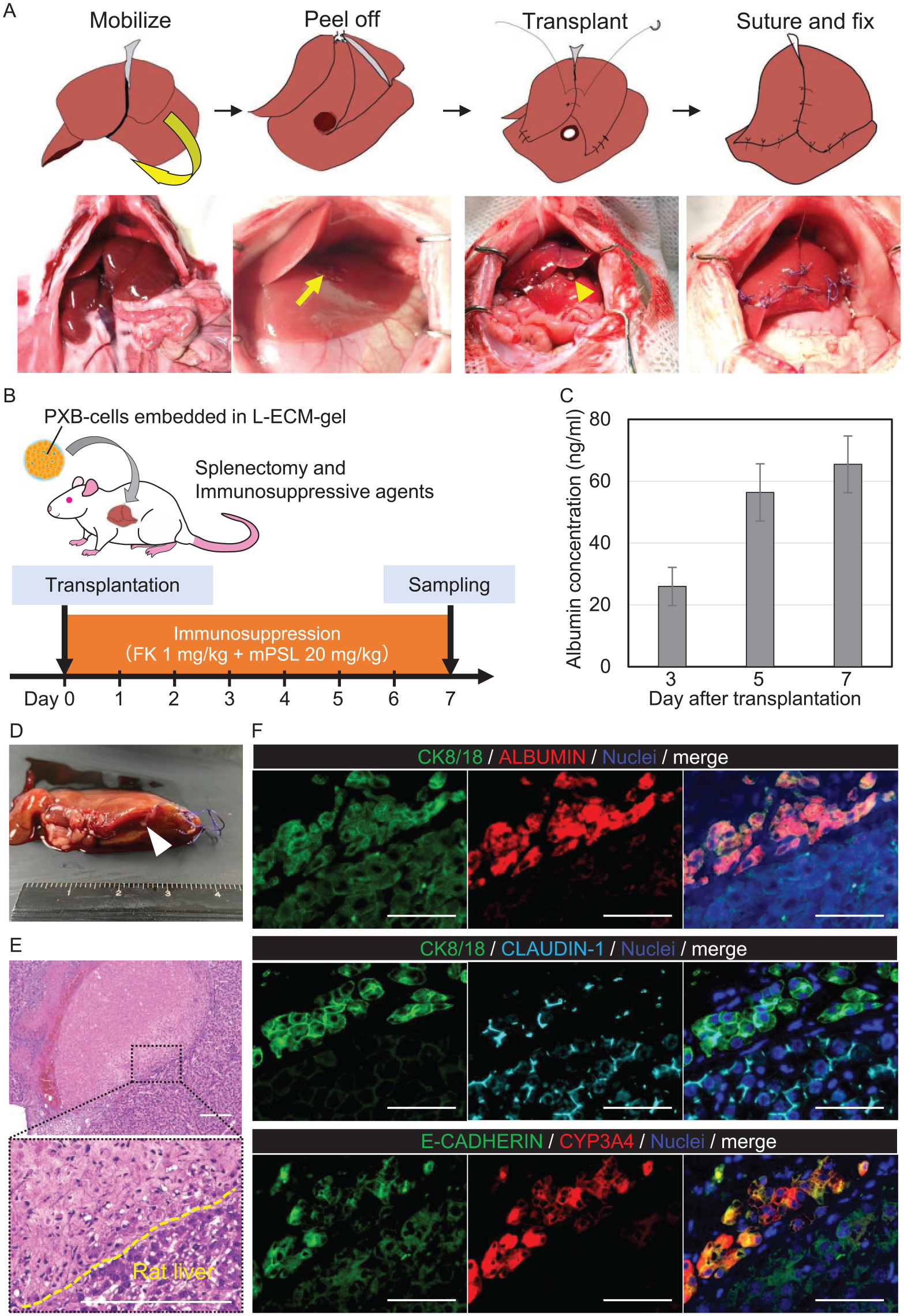
Hepatocyte (PXB) transplantation with the L-ECM gel in normal rats. (A) Scheme of the surgical procedure for hepatocyte transplantation. The peeled-off surface is indicated by arrows. (B) Immunosuppressive agent protocol. FK, tacrolimus, mPSL, and hydrocortisone sodium succinate. (C) Albumin concentration in rat blood samples on days 3, 5, and 7 after transplantation (n = 3). (D) Macroscopic, (E) hematoxylin and eosin (H&E), and immunostaining results. The L-ECM gel was placed between the liver lobes (white arrow). Cells were detected in the L-ECM gel by H&E staining. (F) Immunostaining of hepatocytes in the L-ECM gel; PXB cells (CK8/18, human albumin-positive) expressing CYP3A4. Claudin-1 was expressed in PXB cells. Scale bar, 50 μm.
Another study indicated the significance of maintaining engraftment and functionality for approximately 2 weeks in HCT experiments as a noteworthy short-term outcome 38 . Supplemental Figure 4 presents the results of a 2-week HCT study using the L-ECM gel and collagen gel in normal rats. Histological analysis revealed the presence of human albumin-positive hepatocytes in the L-ECM gel during this period. However, numerous cells formed cyst-like structures and expressed CK8/18 and ZO-1 but not albumin, suggesting a shift in cholangiocyte characteristics due to prolonged in vivo observation (Supplemental Figure 4A). The concentration of human albumin in rat blood decreased by 30% in the L-ECM gel and 20% in collagen gel within 2 weeks post-transplantation (Supplemental Figure 4B). These results suggested that hepatocytes in the L-ECM gel retained better hepatic function than those in the collagen gel. Similar results were obtained with HCT in HepG2 cells.
Protocol for Constructing a Thioacetamide-Induced Liver Fibrosis Model of Wild Rat
A rat model of liver fibrosis was developed by administering thioacetamide (TAA) to evaluate cell engraftment in damaged livers. TAA (100 mg/kg) was administered subcutaneously three times per week. This administration was scheduled for 8 weeks (Fig. 5A). None of the rats died during the study (TAA: n = 5; control: n = 6). The serum levels of aspartate aminotransferase (AST) and alanine transaminase (ALT) were higher in the TAA group than in the control group, but the difference was not significant. Serum albumin levels were lower in the TAA group than in the control group, but the difference was not significant (Fig. 5B). After the 8-week administration, the rat livers were analyzed. Histological analysis of the liver revealed that the TAA group presented a cross-linked structure, as observed by H&E and azan staining (Fig. 5C). According to the New Inuyama classification of liver fibrosis 39 , the TAA group was matched for F2 (63%) and F1 (13%). qRT-PCR analysis of the liver revealed that the expression of inflammation-associated genes, including IL1 and IL2, MMP, Col, liver regeneration-associated genes, including Fgf2 and Pdgfa, and ECM-associated genes, including Mmp1 and Col1a1, were significantly elevated in the TAA group (Fig. 5D).
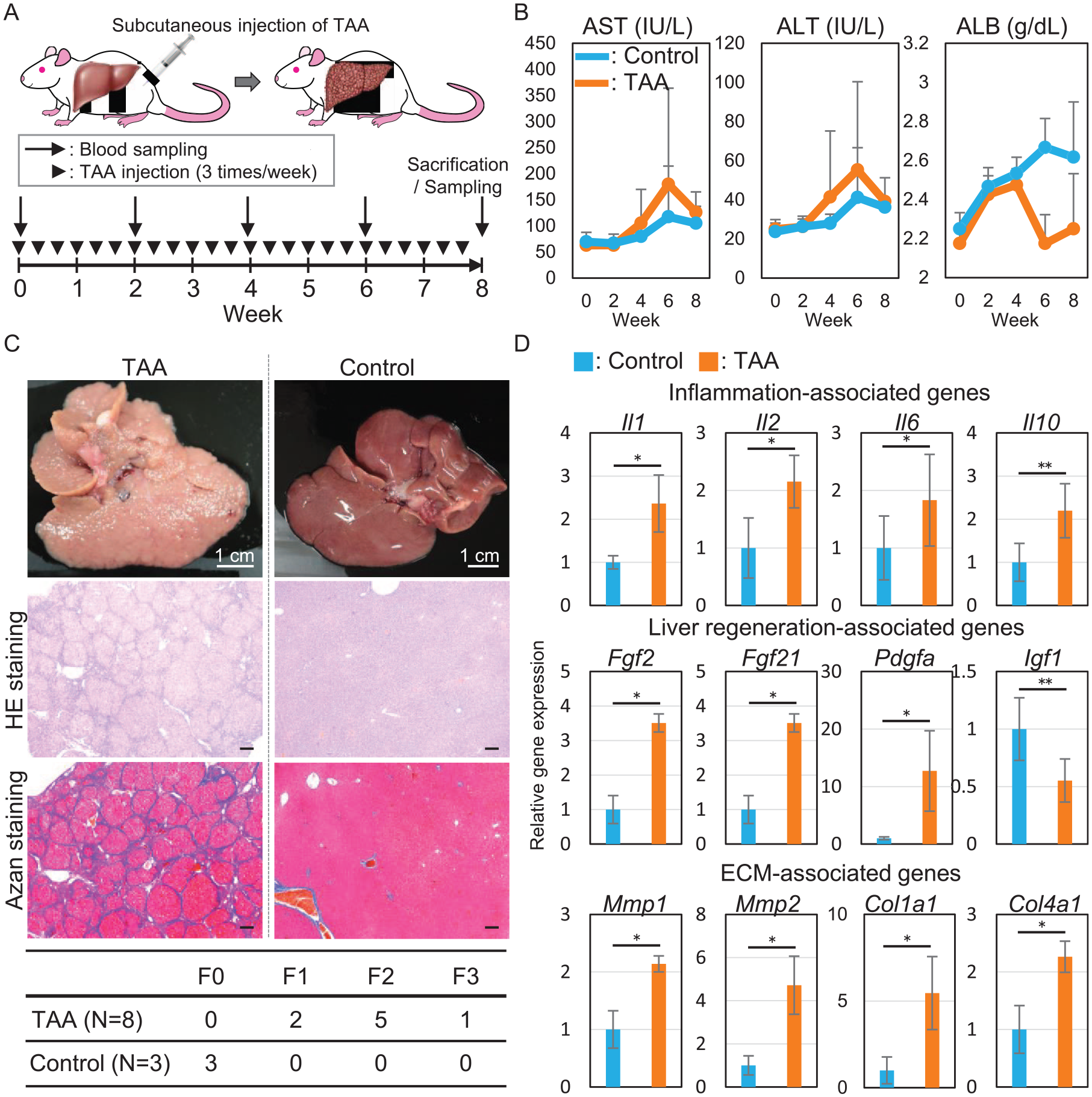
Rat model of drug-induced liver fibrosis using thioacetamide (TAA). (A) Protocol for TAA administration and blood examination. (B) Changes in serum levels of ALT, AST, and albumin in rats. There were no statistically significant differences. (C) Macroscopic, H&E, and azan staining of resected liver after TAA administration. The table below shows the degree of liver fibrosis according to the Inuyama classification. Scale bar, 1 cm or 50 μm. (D) qRT-PCR of the resected liver after TAA administration. The expression of MMP, Col, and Vegf was significantly elevated upon TAA administration compared with that observed in the controls. * and ** indicate P < 0.05 and P < 0.10, respectively.
HCT With the L-ECM Gel as a Scaffold in the Rat Model of Liver Fibrosis
A cirrhotic liver induces portal hypertension, which negatively affects hepatocyte injection via the portal vein. Thus, indications for intraportal HCT are limited. This study aimed to investigate engraftment of transplanted hepatocytes in damaged livers. Given the clinical impracticality of intraportal HCT for cirrhotic livers, we shifted our attention to orthotopic transplantation rather than an intravascular approach. In this context, this study employed HCT with collagen gel as a control. TAA (100 mg/kg) was administered subcutaneously three times per week to SD rats. Figure 6A shows the fibrotic livers of SD rats induced by TAA administration during HCT. One week after transplantation, the rats were sampled (n = 3). The L-ECM gel was observed on the surface of the liver lobes by H&E and azan staining (Fig. 6B). H& E staining revealed the presence of red blood cells in the L-ECM gel. Azan staining demonstrated that the L-ECM gel had migrated to the native liver. We investigated the function of transplanted hepatocytes in the L-ECM gel and hydrogel. Cell function was used as an indicator because single-cell isolation from inside a transplanted hydrogel is technically challenging and impractical. Human albumin production in blood samples was quantified using ELISA. The results indicated no significant difference in albumin production by PXBs in the L-ECM gel compared with the collagen gel (Fig. 6C). Relative gene expression was compared between the L-ECM and collagen gels. Hepatic function markers, such as CYP3A4, and hepatocyte markers, such as EPCA, KRT7, and LKB1, were not significantly different between the two hydrogels. EMT markers, such as CCN1 and CCN2, were similar between the two hydrogels. CLDN1 and CLDN3 expression levels did not differ significantly between the two hydrogels. Moreover, the expression of ABCB11, a cholangiocyte marker, did not differ between the groups (Fig. 6D). Immunostaining analyses revealed that PXB cells (CK8/18 and human albumin-positive) expressed hepatic function markers, including CYP3A4 (Fig. 6E). Moreover, ZO-1, a tight junction marker, was detected in the L-ECM gel. Claudin-1 expression was detected in these cells. The rate of claudin-1 positive area in the hydrogel was significantly higher in the L-ECM gel than in the type I collagen gel using ImageJ software (Fig. 6F). Further evaluation of angiogenesis in the hydrogels was also performed. Gene expression of Pecam1 was significantly elevated in the L-ECM gel (Fig. 6G). Figure 6H shows the appearance of a vascular structure in the L-ECM gel. Pecam1-positive areas were compared between the L-ECM and type I collagen gels using ImageJ software. The rate of Pecam1 positive area in the hydrogel was significantly higher in the L-ECM gel than in the type I collagen gel (Fig. 6H). These findings suggested that cell–cell interaction and angiogenesis play important roles in cell engraftment and are promoted in the L-ECM gel compared with the collagen gel in vivo.
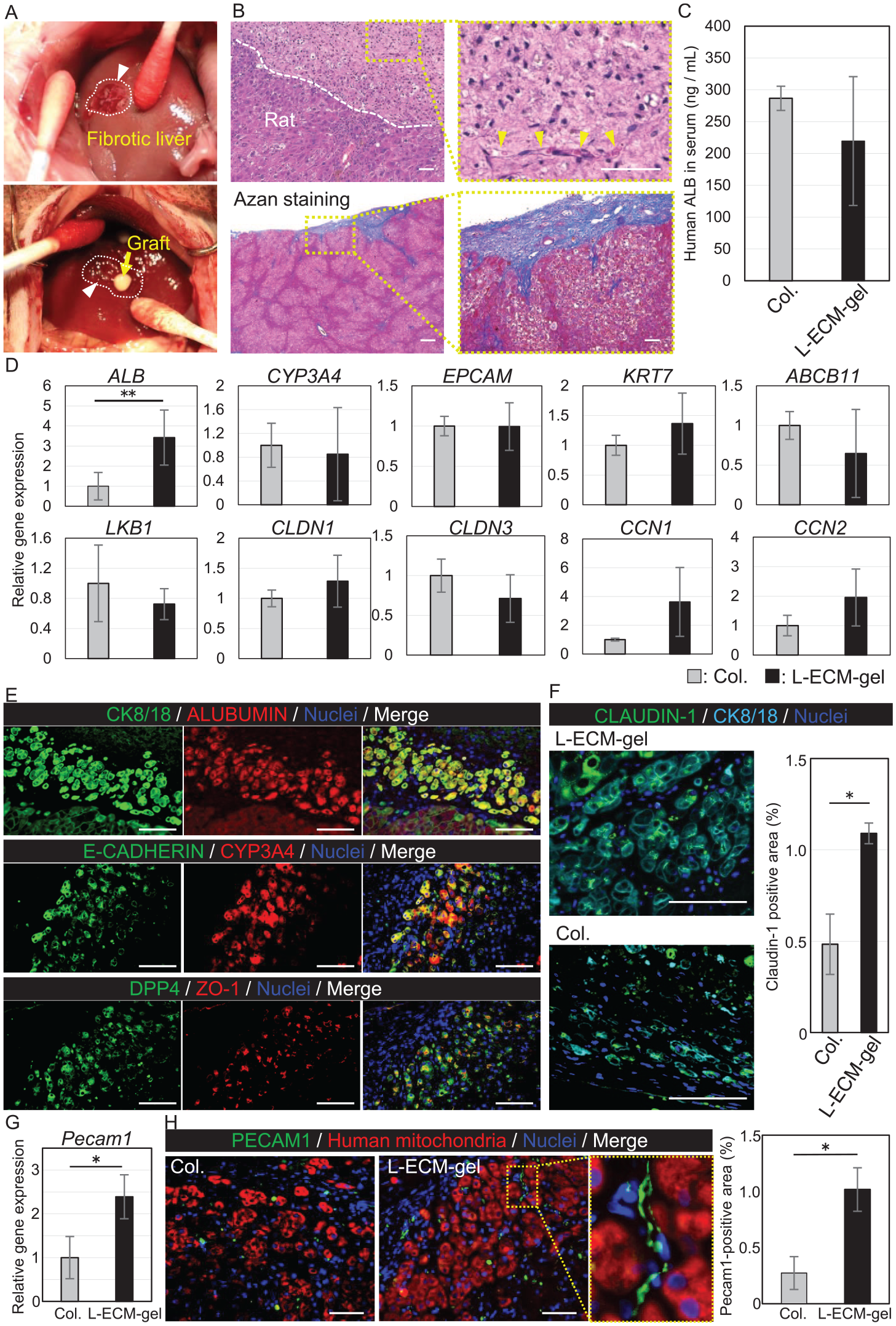
Hepatocyte (PXB) transplantation with L-ECM gel and collagen gel (control) in a drug-induced liver fibrosis rat model. (A) Photographs of the transplantation. The white arrow indicates the peeled-off surface. (B) H&E and azan staining of the resected specimen. L-ECM gel was observed on the liver surface. Red blood cells (yellow arrows) and nuclei cell were observed in the L-ECM gel. Scale bar, 50 μm. (C) Human albumin production in the rat blood samples (n = 3). There was no significant difference between the L-ECM and collagen gels. (D) Gene expression analysis by PCR. ALB was significantly higher in L-ECM gel than in the collagen gel (P < 0.05). Expression of other genes, including CYP3A4, EPCAM, KRT7, ABCB2, LKB1, CLDN1, CLDN2, CCN1, and CCN2 was similar in the two hydrogels. (E) Immunostaining of engrafted hepatocytes (CK8/18, human albumin-positive). The hepatic function marker, CYP3A4, and the tight junction marker ZO-1 were expressed in hepatocytes. Scale bar, 50 μm. (F) The rate of claudin-1 positive area per same square measurement was significantly higher in the L-ECM gel than in the collagen gel (P = 0.024). (G) Pecam1 expression analysis of hepatocytes encapsulated in the L-ECM and collagen gels. A significant difference was observed between the two hydrogels. (H) Angiogenesis in the hydrogel. The rate of CD-31 positive area per graft area was significantly higher in the L-ECM gel than that in the collagen gel (P = 0.014). A vascular structure was observed in the L-ECM gel. Scale bar, 50 μm. * and ** indicate P < 0.05 and P < 0.10, respectively.
Discussion
This study demonstrated short-term cell engraftment of orthotopic HCT in combination with an L-ECM gel as a scaffold. Our orthotopic HCT method revealed the following: (i) survival of translated hepatocytes and expression of their inherent function in a rat model of drug-induced liver fibrosis, (ii) enhancement of the cell–cell interaction of transplanted hepatocytes, and (iii) induction of angiogenesis in the L-ECM gel. These results suggest that the L-ECM gel can promote cell–cell interactions and angiogenesis, and this novel approach to orthotopic HCT engineered using liver hydrogel can be useful even in fibrotic livers.
ECM hydrogels derived from decellularized tissues are promising materials for in vitro and in vivo bioengineering applications 19 . Various studies have reported that organ-specific ECM, including hepatocytes, affect cell function and proliferation20,40–43. Proteomic analysis revealed that matrisome, an essential component of ECM41,44, is retained after decellularization. The L-ECM gel contains various important hepatocyte components, including Type IV collagen, laminin, and glycoproteins45,46. Moreover, ANXA, FBN1, BGN, and FLNA, which are key factors in angiogenesis and cell activity, were detected. Type I collagen consisted of only one component. The maintenance of these matrisomes, which play crucial roles in angiogenesis and cell activity, could promote cell–cell interactions and angiogenesis in in vivo and in vitro experiments using the L-ECM gel. Because the viscoelasticity of hydrogels is closely related to tissue homeostasis and regeneration47,48, we investigated the proper viscoelasticity of the L-ECM gel for cell function and applicable staff for surgical situations. Viscoelasticity can be adjusted in a concentration-dependent manner. In vitro quantitative analyses demonstrated that the production of albumin by mature hepatocytes was similar in the L-ECM and collagen gels. In vivo, the L-ECM gel can provide a physically stable scaffold for cell engraftment and expression of its inherent function.
To investigate cell engraftment in our orthotopic HCT in a fibrotic liver, we generated a rat model of liver fibrosis by the subcutaneous administration of TAA. While immunostaining analysis revealed a fibrotic liver, TAA administration did not cause a significant increase in ALT and AST levels. No animal deaths occurred during the study. These results suggest that our method can be safely performed using the duplicate clinical features of cirrhotic livers. We used HCT in the TAA-induced liver fibrosis rat model as well as in normal rats. Angiogenesis is not only strongly associated with liver regeneration but also with fibrosis 49 . In this model, angiogenesis was promoted in the L-ECM gel with cell–cell interactions of hepatocytes, showing that it was activated in the L-ECM gel in liver regeneration and hepatocyte survival, instead of fibrosis. Currently, liver transplantation is the only life-saving treatment available for patients with cirrhosis. The shortage of donor livers has not yet been solved, resulting in many patients placed on transplant waiting lists, with a significant proportion dying while waiting50,51. In the conventional method, injected hepatocytes are unable to reach the cirrhotic liver owing to the negative effect of portal hypertension 52 . This limits the indication for HCT in non-cirrhotic livers, such as those with metabolic disease 4 . Thus, our HCT method has the potential serve as be a bridging therapy for liver transplantation in patients with decompensated cirrhosis.
Gene expression analysis showed that ALB and EPCAM were retained in hepatocytes encapsulated in the L-ECM gel compared with those encapsulated in the collagen gel in vitro. In contrast, in the L-ECM gel, the cluster gram of mature hepatocytes was downregulated and that of EMT was upregulated compared with that in the collagen gel. Therefore, we performed a further subgroup analysis of partial EMT, which was upregulated. EMT is integral to development, and is associated with wound healing, fibrosis, cancer progression, and stem cell differentiation 53 . In particular, partial EMT is necessary for hepatocyte regeneration54,55. These gene behaviors of hepatocytes in L-ECM gel in vitro indicated that mature hepatocytes do not show a mesenchymal shift to fibroblasts but present the process of regeneration in such a liver-specific environment. In the analysis of transplanted hepatocytes in L-ECM gel in vivo, the expression of mature hepatocyte markers, such as CYP3A4 was similar to that in the collagen gel, and ALB was significantly promoted. In addition, the expression of CCN1 and CCN2 associated with the YAP pathway in the L-ECM gel was not significantly different from that in the collagen gel. These results demonstrated that transplanted hepatocytes are regulated by the epithelial phenotype in vivo 56 . In this study, we observed a decline in hepatocyte function in PXB cells during the cell isolation process of gel embedding. In particular, a transient process of partial pEMT was noted in hepatocytes cultured in the liver gel, potentially leading to reduced hepatocyte function.
Albumin production by PXBs in L-ECM gel and collagen gel (Fig. 3C) did not differ significantly. This finding suggests that L-ECM gel maintains hepatocyte function comparable with the traditionally used collagen gel for hepatocyte embedding in vivo.
As reported in previous studies13,14, hepatocytes require cell–cell and cell–ECM interaction to survive for long time and express their inherent function. CLDN-1 and ZO-1 are essential cell surface proteins involved in cell–cell interactions and tight junctions57,58. Immunostaining demonstrated that the CLDN-1 was strongly expressed in intercellular space and the area of CLDN-1 positives was significantly larger in L-ECM gel than in collagen gel. This suggests that transplanted hepatocytes show an epithelial transition to regenerate in vivo and inherent hepatic function with promoted cell–cell interactions.
Angiogenesis was also observed in the L-ECM gel. Cells and tissues require an oxygen supply and metabolite excretion pathways. Mature hepatocytes have high oxygen demand for survival59,60. Thus, angiogenesis is a crucial issue in tissue engineering 61 . In this method, transplanted hepatocytes do not receive direct blood supply, unlike in intraportal HCT. RNA sequencing of hepatocytes demonstrated that the cluster gram of angiogenesis-related genes had a higher expression in the L-ECM gel than in the collagen gel. In vivo histological analysis demonstrated the presence of red blood cells in the CD31-positive vascular structure of the L-ECM gel from the peel-off surface. These results indicate that the L-ECM gel has the potential to achieve long-term survival of transplanted hepatocytes by promoting angiogenesis. The results obtained two weeks after transplantation (Supplemental Figure 4) support these considerations.
In this study, we aimed to investigate the engraftment of mature hepatocytes in rats with normal livers and in a rat model of liver fibrosis using a new orthotopic HCT approach. The primary objective was to demonstrate the methodology of this new transplantation technique and its applicability in damaged liver conditions, rather than to investigate the quantity of grafts and cells required for therapeutic effects. The current graft size was small (8 × 105 cells/drop), especially compared with the surface area of the rat liver. Histological evidence indicates that the liver surface has ample space for transplantation, potentially up to 10–20 times larger than the current graft size. Although the technical limitation is the liver surface area available for transplantation, it appears feasible to transplant a sufficient number of cells. Further exploration is needed to understand the therapeutic effects and long-term outcomes of the HCT method. Although we did not investigate whether cell–cell interactions and angiogenesis enhanced cell engraftment in our experiments, previous experiments have shown that cell–cell interactions and angiogenesis are important for cell engraftment. Therefore, we believe that L-ECM gel containing various components, instead of collagen gel, is a useful scaffold for new HCT methods and could be applicable to cirrhotic livers. We conclude that orthotopic HCT using L-ECM hydrogels as scaffolds holds clinical therapeutic potential for treating metabolic diseases, acute liver failure, and decompensated cirrhosis.
Supplemental Material
sj-pptx-1-cll-10.1177_09636897241253700 – Supplemental material for A Novel Approach to Orthotopic Hepatocyte Transplantation Engineered With Liver Hydrogel for Fibrotic Livers, Enhancing Cell–Cell Interaction and Angiogenesis
Supplemental material, sj-pptx-1-cll-10.1177_09636897241253700 for A Novel Approach to Orthotopic Hepatocyte Transplantation Engineered With Liver Hydrogel for Fibrotic Livers, Enhancing Cell–Cell Interaction and Angiogenesis by Daisuke Udagawa, Shogo Nagata, Hiroshi Yagi, Kotaro Nishi, Toshinori Morisaku, Shungo Adachi, Yutaka Nakano, Masayuki Tanaka, Shutaro Hori, Yasushi Hasegawa, Yuta Abe, Minoru Kitago and Yuko Kitagawa in Cell Transplantation
Footnotes
Acknowledgements
The authors thank S. Mizutani, K. Zhao, M. Ohtomo, and M. Takahashi for technical laboratory support. They would also thank Editage for English language editing.
Author Contributions
Conceptualization, H.Y.; methodology, D.U., S.N., K.N., and H.Y.; investigation, D.U., S.N., and K.N.; proteomic analysis, D.U., S.N., and S.A.; writing—original draft preparation, D.U.; writing—review and editing, H.Y., S.N., T.M., S.A., M.T., S.H., Y.H., Y.A., M.K., and Y.K.; supervision, H.Y. and Y.K.; project administration, H.Y.; funding acquisition, H.Y. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Material
All raw sequencing data have been deposited in the DDBJ Sequence Read Archive (DRA) database that is publicly accessible with the bioproject accession number PRJDB15729.
Ethical Approval
All experiments were reviewed and approved by the Animal Ethics Committee of Keio University School of Medicine, Tokyo, Japan (approval number: A2022-229).
Statement of Human and Animal Rights
The experiments in this study were conducted by the Guide for the Care and Use of Laboratory Animals (National Institutes of Health, Bethesda, MD, USA).
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Japan Agency for Medical Research and Development (AMED; 22bm1004003h0003), Grant-in-Aid for Challenging Research (Pioneering) (22K18395) awarded to H.Y., and JSPS KAKENHI Grant-in-Aid for Scientific Research(C) (23K11833) awarded to S.N.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
