Abstract
Hepatocyte transplantation is an alternative therapy to orthotopic liver transplantation for the treatment of liver diseases. However, the supply of hepatocytes is limited given the shortage of organs available to isolate good-functioning quality cells. Neonatal livers may be a potential source alternative to adult livers to obtain good-performing hepatic cells for hepatocyte transplantation, which has not yet been explored profoundly. High-yield preparations of viable hepatocytes were isolated from 1- to 23-day-old liver donors, cryopreserved, and banked. Cell integrity and functional quality assessment were performed after thawing. Neonatal hepatocytes showed better postthawing recovery compared with adult hepatocytes, as shown by the viability values that did not differ significantly from freshly isolated cells, a higher expression of adhesion molecules (β1-integrin, β-catenin, and E-cadherin), better attachment efficiency, cell survival, and a lower number of apoptotic cells. The metabolic performance of thawed hepatocytes has been assessed by ureogenesis and drug-metabolizing capability (cytochrome P450 and UDP-glucuronosyltransferase enzymes). CYP2A6, CYP2C9, CYP2E1, and CYP3A4 activities were found in all cell preparations, while CYP1A2, CYP2B6, CYP2C19, and CYP2D6 activities were detected only in hepatocytes from a few neonatal donors. The expression of UGT1A1 and UGT1A9 (transcripts and protein) was detected in all hepatocyte preparations, while activity was measured only in some preparations, probably due to lack of maturity of the enzymes. However, isoforms UGT1A6 and UGT2B7 showed considerable activity in all preparations. Compared to adult liver, the hepatocyte isolation procedure in neonatal livers also provides thawed cell suspensions with a higher proportion of hepatic progenitor cells (EpCAM+ staining), which could also participate in regeneration of liver parenchyma after transplantation. These results could imply important advantages of neonatal hepatocytes as a source of high-quality cells to improve human hepatocyte transplantation applicability.
Keywords
Introduction
Hepatocyte transplantation (HT) has been demonstrated to be a safe therapeutic option to improve the clinical outcome of patients with acute and chronic hepatic liver diseases, especially children with inborn metabolic diseases (14,18,28,30,45). The principle behind hepatic cell therapy is based on the hypothesis that transplanted hepatocytes, when implanted into the host liver, can undertake hepatic metabolic functions, which are lacking or perform poorly in patients (18,30). In fact, patients undergoing HT have shown clinical improvement and/or a partial correction of the underlying metabolic defect (18,30,46). The clinical effectiveness of this process likely relies on many factors, such as the nature of the disease, the functional performance of the transplanted hepatocytes, and their capability to engraft and survive in the host liver (5,14,16,30,46). However, the supply of good quality livers for hepatocyte isolation is a major challenge. HT patients must compete for organs for orthotopic liver transplantation (OLT); hence, current sources of tissue for hepatocyte isolation are mainly adult organs considered unsuitable for whole OLT, often of a marginal quality. This emphasizes the need to explore other sources of human hepatic tissue for hepatocyte isolation. An interesting alternative to adult liver is the use of unsuitable neonatal livers for OLT as a potential source of differentiated hepatic cells which, to our knowledge, has been poorly explored to date (30,39,40). Neonatal liver grafts are not suitable for OLT due to low weight and the technical difficulties to perform vascular sutures and biliary trees. However, these grafts may have very good quality cells. In this type of donor, brain death certification is very difficult and problematic. Despite the brain stem test in newborns, toddlers, and children being similar to that in adults, there are some differences as to the duration of the observation period and also with the instrumental tests performed, which make it much more difficult to diagnose. This inconvenience led us to consider another type of donation, such as non-heart-beating donation (NHBD). According to the Maastrich classification, types III and IV donors are an important source of grafts for transplantation, especially type III, whose asystole happens after the controlled withdrawal of all the therapeutic measures (28). Therefore, we have evaluated the suitability of livers from neonates meeting these criteria and expect to obtain high yields of viable and functional hepatic cells.
Cryopreservation seems the most appropriate form of cell banking as it offers the advantage of immediate availability of cells for both programmed and repeated treatment of liver-based metabolic disorders and emergency treatments for patients with acute liver failure when an organ is not immediately available. Moreover, it allows a thorough functional assessment of each batch of hepatocytes to help make decisions about their quality and suitability for clinical use (5,18,37,40,46,51,56). Meyburg et al. (39) reported that the viability values of neonatal liver cells after thawing did not significantly differ from freshly isolated cells; however, the functional capacity and structure integrity of cryopreserved neonatal hepatic cells, as currently performed in adult hepatocytes, has not yet been assessed in detail (5,16,46).
In general terms, the purpose of this work was to explore livers from neonates as alternative sources to isolate human hepatic cells, which could help overcome the shortage of organ donors. To achieve this goal, we focused on a) exploring the suitability of neonatal livers that do not fulfill the requirements for OLT as a potential source of high-quality hepatocytes for banking; b) assessing cell viability, preservation of membrane integrity, expression of adhesion molecules (E-cadherin, β-catenin, and β1-integrin), and postthawing attachment efficiency as an indication of the potential ability of cryopreserved neonatal hepatocytes to be engrafted and to survive in the host organ; c) investigating the functionality [ureogenesis and cytochrome P450 and uridine 5′-diphospho-(UDP) glucuronosyltransferase (UGT) enzymes] of thawed neonatal hepatocytes to determine their suitability for clinical application; and d) analyzing the percentage of progenitor cells in cryopreserved hepatocyte preparations.
Materials and Methods
Materials
Culture media and complements were purchased from Gibco BRL (Invitrogen, Barcelona, Spain). The primary antibodies for this study are summarized in Table 1, and the secondary antibodies were purchased from Amersham Biosciences/GE Healthcare (Buckinghamshire, England). Trizol reagent was purchased from Invitrogen; SYBR Green I Master was acquired from Roche Applied Sciences (Barcelona, Spain). Fluorescent probes were purchased from Molecular Probes (Invitrogen). Other chemicals were obtained from Sigma Aldrich (Madrid, Spain) unless otherwise noted.
Antibodies Used for Western Blot and Flow Cytometry

Sigma-Aldrich (Madrid, Spain); Santa Cruz Biotec (Heidelberg, Germany); Epitomics, Inc. (Burlingame, CA, USA); Miltenyi Biotec (Madrid, Spain); Cell Signaling Technology (Leiden, Netherlands); Abcam (Cambridge, UK). WB, Western blot; FC, flow cytometry; EpCAM-PE, epithelial cell adhesion molecule-phycoerythrin; UGT1A1; uridine 5′-diphospho-(UDP) glucuronosyltransferase 1 family, polypeptide A1.
Neonatal Tissue Procurement for Perfusion Via the Portal Vein
Human hepatocytes were isolated from the procured neonatal livers of NHBDs under the supervision of the hospital's Transplantation Coordination and Ethics Committee, and with the parents' informed consent. Potential donors included all the newborns admitted to the Neonatal Intensive Care Unit (ICU), who met the criteria for either brain stem death or NHBD in the Maastricht III and IV categories. In NHBD, diagnosis is made based on cardiac criteria, whereas it is made based on brain stem criteria in a heart-beating donor diagnosis. Controlled donation takes place when death occurs within an ICU/hospital setting with nonsurvivable injuries following treatment withdrawal; the transplant team can pay special attention at the time of treatment withdrawal and can retrieve organs after cardiac arrest has occurred. Our patients are Maastricht category III, and the NHBD diagnosis of death was made based on cardiac criteria (32).
The newborn period is considered to be the first 28 days of life (7). Eligible donors were identified based on the criteria developed with our transplantation surgeons and our local organ procurement organization. Our inclusion criteria for organ donation were patients whose pathologies did not respond to medical and/or surgical treatment and with life incompatibility, congenital anomalies, intracranial hemorrhage grades III–IV intraventricular, congenital disorders of the central nervous system with no activity in the electroencephalography (EEG), surgical pathology not amenable to treatment and incompatible with life, anencephalic, cranioencephalic trauma, premature newborns with pathologies that do not respond to therapeutic maneuvers, surgery patients with postoperative cerebral anoxia, and NHBD.
Short warm and cold ischemia times were considered a major issue. Potential donors included all the newborns admitted to the neonatal ICU who met the criteria for either brain stem death or NHBD in the Maastricht III and IV categories (32). Inclusion criteria were as listed above. Patients with HIV1 and 2, HBV, HCV, and sepsis with multiorgan dysfunction were excluded.
A super-rapid technique was used for retrieval. Abdominal and thoracic cavities were accessed through a sternotomy and a midline laparotomy. The intrathoracic and infrahepatic inferior vena cava were sectioned to avoid congestion, and the hepatic hilus was also sectioned. Special attention was paid to liver cooling with ice. The portal vein or umbilical vein within the umbilical cord was identified and was therefore cannulated according to vessel size and was fixed with a 4-0 silk tie. This cannula was then connected to the tissue perfusion liquid (Celsior solution; Genzyme, Madrid, Spain), and liver perfusion started. At this stage, the gallbladder was also removed. The liver, with the catheters in place, was placed into a sterile plastic bag and transported to the lab while being maintained in ice-cold Celsior solution until cell isolation (5).
Hepatocyte Isolation
Cold ischemia never exceeded 2.5 h (Table 2). Liver tissue was enzymatically dissociated using a two-step collagenase perfusion technique (22) adapted for neonatal liver. The perfusion system was run by a peristaltic pump (Ref PD 5201; Heidolph Instruments GmbH & Co. KG, Schwabach, Germany) at a flow rate of 100 ml/min. All of the buffers were maintained at 37°C. After collagenase (200 U/ml) digestion by perfusing for 10–15 min, the tissue was disrupted, the suspended cells were filtered, and hepatocytes purified by selective centrifugation at 300 × g for 5 min. Then the pellet was washed twice by centrifugation at 100 × g for 5 min. Cell viability was assessed by the dye exclusion test with 0.4% trypan blue.
Adult human hepatocyte samples (three females, five males with mean age 50) were obtained from a Biobank setup from previously reported cryopreserved hepatocytes (16).
Hepatocyte Cryopreservation and Thawing
Freshly isolated hepatocytes were pelleted and suspended at a density of 10 × 106 viable cells/ml in cryopreservation medium [90% University of Wisconsin solution (DuPunt Merck Pharmaceutical, Wilmington, DE, USA) and 10% dimethyl sulfoxide (DMSO), v/v maintained at 4°C] (16). Cryopreservation and thawing processes were performed as described in detail elsewhere (16,59).
Assessment of Hepatocyte Viability and Attachment Efficiency After Thawing
After thawing, cellular viability was assessed by the trypan blue test. To assess attachment efficiency, hepatocytes were seeded on fibronectin/collagen type I-coated dishes (Sigma-Aldrich) at a density of 8 × 104 viable cells/ cm2 in the culture medium Ham's F-12/Williams (1:1) supplemented as formerly described (21). Cell attachment and monolayer formation were monitored by observation under a phase contrast microscope. The protein content of the attached cells was determined by the Bradford method (Sigma-Aldrich) 1 h after cell plating.
Functional Assessment of Banked Hepatocytes
The S9 fractions from thawed hepatocytes were prepared as previously described (15). Briefly, samples were homogenized in 50 mM Tris HCl pH 7.4 containing 150 mM KCl and 1 mM ethylenediaminetetraacetic acid (EDTA). Homogenates were centrifuged at 10,000 × g for 20 min at 4°C, and the supernatant (S9 fraction) was collected. Cytochrome P450 (P450) activities were assayed by incubating the S9 fractions (1 mg protein/ml) with anicotinamide adenine dinucleotide phosphate(NADPH)-regenerating system (15) and a cocktail mixture of selective substrates for eight individual P450 enzymes (20): 10 μM phenacetin (cytochrome P450, family 1, subfamily A, polypeptide 2; CYP1A2), 5 μM coumarin (CYP2A6), 10 μM bupropion (CYP2B6), 10 μM diclofenac (CYP2C9), 50 μM mephenytoin (CYP2C19), 10 μM bufuralol (CYP2D6), 50 μM chlorzoxazone (CYP2E1), and 5 μM midazolam (CYP3A4). After 20 min of incubation at 37°C, reactions were quenched by adding two volumes of acetonitrile. Formation of the corresponding metabolites was quantified by high-performance liquid chromatography tandem mass spectrometry (HPLC/MS; Waters, Milford, MA, USA), as formerly described (20).
UDP-glucuronosyltransferase (UGT) activities were assayed by incubating the S9 fractions (1 mg protein/ml) in 50 mM Tris-HCl buffer, pH 7.4, containing 2 mM UDP-glucuronic acid, 8 mM Cl2Mg, alamethacin (25 μg/ml), and the corresponding selective substrates: 15 μM β-estradiol (UDP glucuronosyltransferase 1 family, polypeptide A1; UGT1A1), 25 μM 1-naphtol (UGT1A6), 15 μM propofol (UGT1A9), and 250 μM naloxone (UGT2B7). After incubation at 37°C, the assays were stopped by adding two volumes of cold acetonitrile–1% formic acid. The formation of the corresponding metabolite was measured by HPLC/ MS, as previously described in detail (17). Ureogenesis was assessed in the thawed cells by measuring the formation of urea from NH4+, according to Donata et al. (16).
Details of Donors and Organs

F, female; M, male.
Western Blotting
After cryopreservation and thawing, neonatal cell preparations were centrifuged, and pellets were lysed in mammalian protein extraction reagent (Thermo Scientific, Madrid, Spain). Protein content was quantified by the Bradford assay. Protein equivalents from each sample were resolved in sodium dodecyl sulfate-polyacrylamide electrophoresis (BioRad, Madrid, Spain), and were electrotransferred to polyvinylidene difluoride (PVDF) membranes (Immobilon, Millipore, Madrid, Spain) using a Bio-Rad Trans-Blot system, according to the manufacturer's instructions. Membranes were probed with adequate primary antibodies, as summarized in Table 1. β-Actin was used as a loading control. Membranes were washed and incubated with the appropriate peroxidase-conjugated secondary antibodies and were visualized as previously detailed (23).
RNA Extraction and Real-Time Quantitative PCR
Total RNA was extracted from hepatocytes using Trizol Reagent following the manufacturer's recommendations. The amount of isolated RNA was estimated by ribogreen fluorescence, and its purity was assessed by the absorbance ratio 260/280 nm. Total RNA (1 μg) was reverse transcribed and real-time quantified using SYBR Green I Master and the appropriate primers for UGT1A1 and UGT1A9 (47) in a Lightcycler 480 instrument (Roche Applied Sciences, Barcelona, Spain). In parallel, the mRNA concentration of human housekeeping β-actin (FP5′ CGT ACC ACT GGC ATC GTG AT3′; RP5′ GTG TTG GCG TAC AGG TCT TTG3′) was always analyzed as an internal normalization control. The real-time monitoring of the PCR reaction and the precise quantification of the products in the exponential phase of the amplification were performed with the LightCycler Relative Quantification Analysis quantification software in accordance with the manufacturer's recommendations. Moreover, a positive sample with a stable ratio of target and reference cDNAs (a calibrator) was included in each PCR run to normalize all the samples within one run and to provide a constant calibration point among several amplification runs.
Flow Cytometric Analysis
To determine the proportion of progenitor cells copelleted with hepatocytes in the final suspension after the selective centrifugation in the hepatocyte isolation procedure, postthawing cells were fixed with 2% paraformaldehyde in PBS for 10 min at room temperature and were then incubated in permeabilization/blocking buffer (2% Triton X-100, 10% goat serum in PBS) for another 60-min period before incubation with monoclonal antibody anti-epithelial cell adhesion molecule-phycoerythrin (EpCAM-PE) (Table 1) and washing. The analysis was performed in an EPICS XL flow cytometer (Beckman Coulter, Madrid, Spain) using appropriate regions of interest in the compensated fluorescence dot plot diagrams.
For the determination of the proportion of apoptosis after thawing, a total of 1 × 106 cells were suspended in 1 ml PBS and incubated with 0.1 μM YO-PRO-1 and 1.5 μM propidium iodide (PI) for 30 min at 4°C. After incubation, stained cells were analyzed by flow cytometry using 488 nm excitation wavelengths with green fluorescence emission for YO-PRO-1 and red fluorescence for PI. Single-color stained cells were used to perform standard compensations. Apoptotic cells showed a higher level of green fluorescence, whereas dead cells displayed both red and green fluorescence.
Data Analysis
All the data were expressed as mean ± SD (or ±SEM in the figures) values. A Student's t test was used for the statistical evaluations. The level of significance was considered p ≤ 0.05.
Results
Influence of the Source of Human Liver Tissue in the Hepatocyte Isolation Procedure
In order to assess the outcome of the cell isolation procedure from neonatal livers, cell viability, cell yield, and donor characteristics were analyzed for each hepatocyte preparation (Table 2). The mean donor gestational age was 35 weeks (range 26–40 weeks). The mean cell viability of fresh hepatocytes was 87 ± 4%, greater than previous values reported in adult hepatocytes (74 ± 27%) (5), with a mean yield of 21.2 ± 14.4 × 106 cells per gram of tissue, also superior to adult cell suspensions (10.1 ± 6.4×106) (5). Donor sex did not significantly affect the isolation outcome (viability and cell yields). The cell yield analysis revealed that gestational age could influence cell yield (4.5 ± 0.7 × 106 cells per gram of tissue; samples H4/10, H6/11, and H3/12). No influence of cold ischemia time, which never exceeded 2.5 h, was observed on the hepatocyte isolation procedure outcome (Table 2). Warm ischemia was always less than 30 min.
Cell Attachment and Expression of Adhesion Molecules After Thawing
Cell attachment and monolayer formation of thawed hepatocytes were also investigated. Adhesion to the culture substrate of attachable hepatocytes was completed after 40 min of cell seeding. The 1-h plating efficiency after hepatocyte seeding was high and ranged from 46% to 90% (Table 3). Thawed neonatal hepatocytes exhibited greater plating efficiency (82 ± 5) than cryopreserved adult cells (43 ± 13) after 1 h of cell seeding (Table 3). Monolayer formation was followed under the microscope up to 48 h of culture. The representative appearances of the cryopreserved and thawed hepatocytes from a neonatal donor are shown in Figure 1A. The expression of the adhesion molecules was also examined (Fig. 1B). The important role of adhesion molecules has been described in both cell engraftment and the prevention of cryopreservation-induced cell death. To determine whether there was a differential expression of the adhesion molecules in neonatal cells compared with adult hepatocytes, the expressions of E-cadherin, β-catenin, and β1-integrin were determined after cryopreservation and thawing. The Western blot experiments demonstrate a significantly higher level of expression of the adhesion molecules in all the neonatal hepatocyte preparations when compared with adult ones (Fig. 1B, C).
Quality Assessment of Cryopreserved Neonatal Hepatocytes


Increased attachment efficiency and expression of adhesion molecules in cryopreserved and thawed neonatal hepatocytes. (A) Representative photographs of neonatal hepatocytes cultured for 1, 6, 24, and 48 h after thawing. Scale bar: 100 applies to all images. (B) The protein expressions of E-cadherin, β-catenin, and β1-integrin were determined by immunoblotting using β-actin as the loading control. Representative immunoblottings are shown. (C) Western blot bands were quantified by a densitometric analysis (integrated optical density of E-cadherin, β-catenin, or β1-integrin). The immunoreactivity for each protein was normalized for the amount of β-actin for each condition and was expressed as a value in relation to the value in adult hepatocytes. The mean ± SEM is provided. *p ≤ 0.01 compared to adult hepatocytes (Student's t test).
Functional Quality Assessment of Human Hepatocyte Preparations After Thawing
Eleven human hepatocyte suspensions were obtained from neonatal donor livers. Cell viability, attachment efficiency, urea synthesis, P450-dependent, and UGT activities were determined as the criteria to assess the functional quality of hepatocyte suspensions after cryopreservation (Tables 3 and 4). Thawed hepatocyte preparations showed a mean viability of 82 ± 5% (range 72–88%).
Urea is formed within the urea cycle and represents the major end product of ammonia detoxification in the liver. It is a good indicator of the degree of hepatocyte mitochondria preservation. As shown in Table 3, the hepatocytes from all of the analyzed livers were able to synthesize urea from ammonia at high rates (0.77 to 2.19 nmol/min/106 viable cells), which is slightly lower than the mean in adult hepatocytes (3.38 nmol/min/106 viable cells).
The drug detoxification competence of thawed hepatocytes was assessed by measuring the activities of the major P450 enzymes responsible for the oxidative metabolism of drugs in the human liver (CYP1A2, CYP2A6, CYP2B6, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP3A4) and UGT enzymes (UGT1A1, UGT1A6, UGT1A9, and UGT2B7). In general, CYP3A4 and CYP2C9 (the major P450s involved in the metabolism of immunosuppressant and many drugs) exhibited the highest activity in all the hepatocyte preparations, followed by CYP2A6 and CYP2E1. However, CYP1A2, CYP2B6, CYPC19, and CYP2D6 activities were not detected in all the hepatocyte preparations, and they showed very low or undetectable levels (Table 4). It is noteworthy that the hepatocyte preparations from the oldest donors (H14/11 and H5/12, 9-day- and 23-day-old donors, respectively) were the only ones that presented measurable levels of all the P450 activities. No apparent influence of gestational age on activity levels was observed.
UGT1A6 and UGT2B7 activities were found in hepatocytes from all neonatal donors, while measurable UGT1A1 and UGT1A9 activities were observed only in hepatocytes from the oldest donors (Table 4). In view of the low activity of UGT1A1 and UGT1A9, their expression was studied at both the mRNA and protein levels. The analysis revealed that all the hepatocyte preparations from neonatal donors showed measurable levels of UGT1A1 and UGT1A9 enzymes, and of their corresponding transcripts, although the level of expression was lower than in adult human hepatocytes (at least p ≤ 0.05) (Fig. 2).
P450 and UGT Activities in Neonatal Hepatocytes After Cryopreservation and Thawing
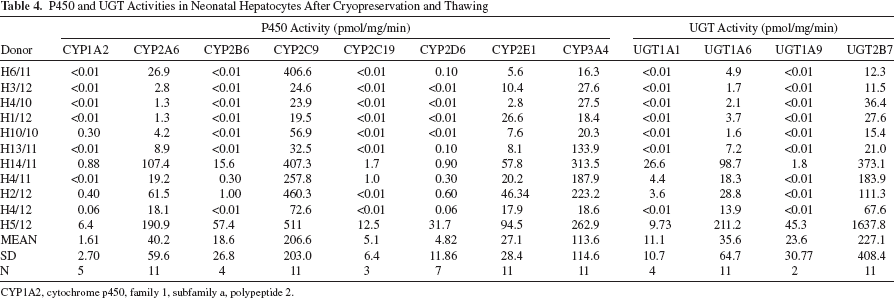
CYP1A2, cytochrome p450, family 1, subfamily a, polypeptide 2.
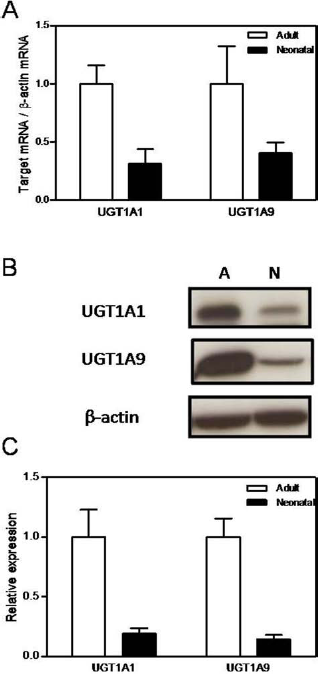
UGT1A1 and UGT1A9 expression in cryopreserved and thawed neonatal hepatocytes versus adult cells. (A) Uridine 5′-diphospho-(UDP) glucuronosyltransferase 1 family, polypeptide A1 (UGT1A1), and UGT1A9 mRNAs were quantified by real-time RT-PCR and normalized to the expression of the housekeeping β-actin mRNA. Data represent the mean of 7 adult and 11 neonatal thawed samples. (B) Protein expression of UGT1A1 and UGT1A9 determined by immunoblotting, using β-actin as the loading control. A representative immunoblot is shown. A, adult; N, neonatal. (C) Western blotting bands were quantified by densitometric analysis (integrated optical density of UGT1A1 and UGT1A9). Immunoreactivity was normalized for the amount of β-actin for each condition and was expressed as a value in relation to the value in adult hepatocytes. The mean ± SEM is provided.
Apoptotic and Necrotic Cell Death
The proportion of apoptotic hepatocytes in cell suspensions after thawing was determined by staining with YO-PRO-1 and PI. Apoptotic cells were detected in both adult and neonatal suspensions, although the percentage in adult cells (7.2%) was significantly higher (p = 0.023) than in those isolated from newborn donors (3.6%) (Fig. 3A). Dead cells were identified by both red and green fluorescence in both hepatocyte suspensions. However, the proportion of dead hepatocytes was lower in the hepatocytes isolated from neonatal donors (10.8%) than in adult cells (19.4%) (p = 0.031) (Fig. 3B).
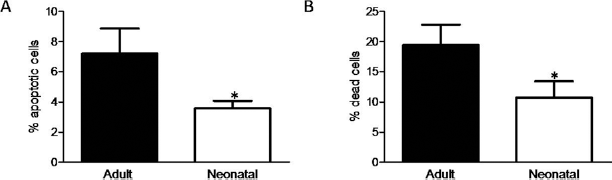
Percentage of apoptotic and dead cells in cryopreserved and thawed adult and neonatal hepatocytes. (A) The percentage of apoptotic cells in thawed hepatocytes was identified by YO-PRO1 staining. (B) The percentage of necrotic hepatocytes was identified by propidium iodide staining. The mean ± SEM of at least six separate cell preparations is provided. *p ≥ 0.05 (Student's t test).
Analysis of Progenitor Cells
The presence of progenitor cells in postthawing cell preparations was investigated by determining the expression of hepatic progenitor cell marker EpCAM. The Western blotting analysis reveals a significantly higher level of EpCAM expression (p = 0.014) in all the neonatal hepatocyte preparations when compared with thawed adult ones (Fig. 4A, B). This correlates with the results obtained by the flow cytometric analysis of thawed neonatal hepatocyte preparations, which showed a significant number of EpCAM+ cells (5.8-fold increase) compared to the adult hepatocyte preparations which were copelleted in the isolation process (p = 0.016) (Fig. 4C).

Increased EpCAM expression and cell number in neonatal livers. (A) Protein expression of EpCAM determined by immunoblotting using β-actin as the loading control. A representative immunoblot is shown. A, adult; N, neonatal. (B) Western blot bands were quantified by a densitometric analysis (integrated optical density of EpCAM). Immunoreactivity was normalized for the amount of β-actin for each condition and was expressed as a value in relation to the value in adult hepatocytes. The mean ± SEM is provided. (C) Relative number of EpCAM-positive cells detected in thawed adult and neonatal cell suspensions. Data are expressed as the relative number of cells from neonatal livers compared with cells from control adult livers. EpCAM, epithelial cell adhesion molecule. *p < 0.05 compared with adult cell suspensions (Student's t test).
Discussion
The current source of isolated hepatocytes is donor organs, which are unused or deemed unsuitable for OLT. Hence, the major challenge faced while developing cell-based therapies for liver diseases is the limited availability of the human liver donors that can provide high-quality human hepatocytes (28,30,45). Livers from neonatal donors are not usually valid for OLT because of their small size. Therefore, we explored neonatal tissue as an alternative source to isolate high-performing cells for HT.
It is known that both warm and cold ischemia times of the donor liver affect the viability and functionality of cells after isolation. Short warm (<30 min) and cold (<2.5 h) ischemia times, and flushing the tissue with preservation solutions, are recommended (5,57). The neonatal livers included in our study accomplished these requirements (Table 2). The outcome of the isolation process in neonatal livers was assessed by each hepatocyte preparation's cell yield and viability. A high yield of viable hepatocytes was isolated from neonatal liver tissue, consistent with the data reported from the hepatocytes isolated from both a 9-day-old neonate (39,40) and a newborn calf (65).
One of the main advantages of HT compared with OLT for human application is the possibility of cryopreserving and storing isolated hepatocytes until they are required for emergency treatments and prescheduled clinical use. Banked hepatocytes are constantly available, and extensive quality testing can be done to determine their suitability for transplantation (16). However, it is known that cryopreservation can cause detrimental effects on hepatocytes; thus, upon thawing they are often not suitable for clinical use (28,30,51,57). Apoptosis has been identified as an important cause of freshly isolated and banked hepatocyte death, with mitochondria as the key players in the initiation of apoptosis and in cryopreservation-induced cell damage (50). In fact, the thawing process damages mitochondria. Alterations are found in the mitochondrial respiratory chain, and ATP production diminishes (50). In agreement with the previous nonpublished observations of Jorns et al. (30), no significant loss of cell viability or in function was found when comparing neonatal thawed hepatocytes with fresh ones (data not shown). In contrast, a decrease in both cell viability and attachment has been previously described in adult hepatocytes after thawing (16). Our results not only indicate that neonatal hepatocytes can be cryopreserved without significant loss of viability but also show smaller apoptotic and necrotic cell numbers if compared with adult ones (Fig. 3). These findings are very likely related to the fact that the mitochondrial function is not substantially altered by cryopreservation in neonatal cells (39).
One major problem commonly found after HT is the small number of transplanted cells found in the graft. However, if HT is to be done as a therapeutic approach, cells need to adhere to the hepatic sinusoidal endothelium as part of the engraftment process (31,45). The success of engraftment into the host liver requires both the membrane integrity and adhesion molecules involved in cell-cell and cell-extracellular matrix adhesion mechanisms being preserved (43,44). However, it has been described that cryopreservation of adult hepatocytes significantly diminishes cell attachment efficiency (58,59) and leads to a downregulation of the key adhesion molecules at the gene and protein levels (44,58,59). Our results reveal that cryopreserved neonatal cells exhibit greater plating efficiency and survival than adult hepatocytes (5), which correlates with a significantly higher expression of E-cadherin, β1-integrin, and β-catenin than adult hepatocytes (Fig. 1). There have been previous indications of a strong correlation between loss of β1-integrin and degradation of E-cadherin and a low attachment of cryopreserved adult cells (44,58). E-Cadherin has also been reported to be responsible for protecting hepatocytes from a novel form of caspase-independent cell death (36). These findings suggest a role of adhesion molecules in the prevention of cryopreservation-induced cell damage (24). In addition, other reports have revealed that transplanted newborn (6) and fetal (12) rat hepatocytes and neonatal mouse hepatocytes (54) can integrate, expand, and differentiate by producing more colonies in the host liver than adult liver cells, irrespective of the recipient's age. This suggests an improved engraftment and survival advantage of transplanted fetal or neonatal hepatocytes in the host liver as opposed to adult hepatocytes.
The functional competence (urea synthesis and drug-metabolizing capability) of hepatocytes was assessed after cryopreservation and thawing. Detoxification, one of the liver's vital functions, involves both waste removal and xenobiotic biotransformation. Urea is formed within the urea cycle. This hepatic function is essential for the elimination of ammonia generated from protein catabolism in order to prevent or relieve hyperammonemia in recipients and also for the utilization of most amino acids for gluconeogenesis. It is also a good indicator of the degree of hepatocyte mitochondria preservation (38). Our results indicate that this pathway is fully active in cryopreserved neonatal hepatocytes, with ureogenic rates comparable to those of cryopreserved adult hepatocytes (5). In fact, Meyburg et al. (40) transplanted thawed neonatal hepatocytes from the same donor to four different pediatric patients to treat urea cycle disorders with considerable beneficial therapeutic effects.
The drug-metabolizing function is of special interest for the clinical HT setting in which recipients receive extensive medication. Based on current knowledge, the largest number of drug-metabolizing enzymes is expressed at negligible or low levels in the fetus. A number of reports into ontogeny development of the phase I and II drug metabolism enzymes provide information on their abundance/ expression levels (mRNA and protein) in neonatal livers, but very few contribute enzyme activity data (3,11,25-27,42). To our knowledge, no systematic evaluation of the catalytic activity of the P450 and conjugating enzymes in neonatal hepatocytes, either fresh or cryopreserved, has been reported.
We found that the functional P450s in neonatal hepatocytes after cryopreservation showed a similar balance to that in the adult liver (19): CYP3A4 and CYP2C9 gave the highest activity values in all cell preparations, followed by CYP2A6 and CYP2E1 with lower levels, and finally by CYP2C19, CYP2B6, and CYP2D6 with the lowest activity values (Table 4). The relatively high CYP3A4 activity noted in neonatal hepatocytes after thawing is a good indication of their metabolic competence because the drugs commonly used in standard immunosuppression for HT (e.g., tacrolimus, cyclosporine, or prednisone) are metabolized by this enzyme. Individual P450 enzymes mature at variable age rates to reach 30% of adult levels with great interindividual variability, and independently of gestational age, during the postnatal period (3,11,25-27,39). Although some P450 isoforms are expressed in the fetal liver and others surge within hours of birth, significant increases in expression to mature levels occur within weeks of birth for most enzymes (25). In fact, there is considerable variation in the fetal expression patterns of P450s. The two hepatocyte suspensions obtained from livers collected at early term (26 and 27 weeks, H6/11 and H3/12, respectively) are comparable to the data from fetal livers (11,27,42) and showed low, but detectable, levels of CYP2A6, CYP2E1, and CYP3A4, while CYP2C9 showed great variability (406.6 and 24.6 pmol/mg/min, respectively) in the two cell preparations analyzed. This probably indicates the importance of postnatal age for the maturation of this isoform (33,60). Nevertheless, early neonatal isozymes have been considered CYP2D6 (4,52) and CYP2E1 (29,61), which rose during the first week of birth (10,26). A second group includes neonatal P450 isoforms, which develop later and gradually increase during the postnatal period, including CYP2A6 (26), CYP2C9 and CYP2C19 (33,60), and CYP3A4 (35,52). Finally, CYP2B6 is expressed later on, during the postnatal period (55), while CYP1A2, the last isoform expressed in the human liver, shows a slow, moderate increase in activity during the first months of life (up to 5% of adult levels are reached after 2-3 months of birth) (3,11,26). In agreement with the literature, our results suggest a certain donor age-enzyme activity relationship of neonatal isoforms, which develop after birth (CYP2A6, 2C9, 2C19, 3A4), as shown by the lower activity values in the hepatocytes of ≥3-day-old donors, while no clear age relation was observed in early neonatal isoforms (CYP2E1, 2D6). Finally, as our hepatocytes were obtained from ≥23-day-old donors, CYP1A2 and CYP2B6 activity was very poor or undetectable, which was expected.
Compared with P450s, the data on UGTs, which catalyze the conjugation of hydrophobic compounds into glucuronides, in neonates are limited. Glucuronidation activity in neonates has been described to develop from minimal to almost adult levels within months of birth, showing extensive interindividual variability (1,2,37,53). Particularly, the expression in humans of UGT1A1, the major enzyme responsible for bilirubin glucuronidation, is modulated in a developmental fashion, to the extent that its activity is absent or very poor in the fetal liver. It develops after birth to reach adult levels by 14 weeks of postnatal life (2,26,37,53,62). This developmentally determined pattern of UGT1A1 activity accounts for, to a large extent, the genesis of neonatal hyperbilirubinemia. Moreover, type I Crigler-Najjar syndrome, a genetic deficiency in hepatic UGT1A1, is a metabolopathy that is frequently treated with HT (46). Therefore, we consider that the assessment of UGT1A1 and other UGT enzymes should be considered key criteria in the selection of hepatocyte preparations for HT in type I Crigler-Najjar syndrome patients. UGT1A6, UGT1A9, and UGT2B7 have already been seen to be expressed in fetal liver, but they develop slowly during the postnatal period. UGT1A9 (41) and UGT2B7 (53) activity correlates with expression and age, and reached the adult activity levels by 2-12 months of age (9,63), while UGT1A6 maturation takes years (until puberty) to reach adult levels (13). In fact, early neonates (H6/11 and H3/12) have shown one of the lowest values for both UGT1A6 and UGT2B7, which was expected (9,53,63).
Our results show that despite UGT1A1 and UGT1A9 expression being found at the mRNA and protein levels in all donors, the catalytic activity of UGT1A1 can be detected only in 4 of the 11 hepatocyte preparations, while the same may be said of UGT1A9 activity in 2 of the 11 cell preparations. It has been reported that enzyme maturation rates vary between UGTs, but that they may well extend beyond the age of 2 years (34). Thus, consistent with the literature, lack of enzyme maturation is very likely responsible for the absence of the catalytic activity of both UGTs in some cell preparations. However, UGT1A6 and UGT2B7 activity was present in the hepatocytes from all donors, thus providing them detoxification capability by the conjugation and elimination of hydrophobic compounds.
Both fetal and postnatal human livers have been shown to contain pluripotent hepatic progenitors, which express the EpCAM transmembrane glycoprotein (48,49,64). EpCAM expression has been evidenced in human livers in all developmental stages (i.e., fetal, neonatal, pediatric, and adult livers) (49,64). Nevertheless, a larger number of EpCAM+ cells has been described in fetal livers, whereas after birth, relatively smaller and constant numbers have been found in livers at all donor ages (49). Our results indicate that after the hepatocyte isolation procedure, a mixed population of hepatocytes and EpCAM+ cells is obtained. A significantly higher percentage of EpCAM+ cells were found in neonatal cell preparations compared to hepatocyte preparations from adult livers (Fig. 4). The higher proportion of progenitor cells may be due to the fact that we used very early neonatal livers (1- to 23-day-old donors), with a wide range of gestational age (26-40 weeks), containing progenitor cells, probably at a similar proportion to fetal livers. Thus, it can be hypothesized that the cotransplantation of neonatal hepatocytes with a small percentage of progenitor cells could augment the post-cell transplantation in vivo expansion, as recently suggested by cotransplanting liver-derived mesenchymal stromal cells with hepatocytes (31).
Another important issue for neonatal human hepatocytes is that they are diploid and can pass through the usual mitotic cycles several times by maintaining their initial ploidy, while the liver cell polyploidization process is generally considered to indicate terminal differentiation and senescence, leading to diminished replication capacity (8). Therefore, the larger number of progenitor cells and diploidy maintenance through several mitotic cycles confer important advantages to the hepatic cells isolated from early neonatal livers over adult ones to improve engraftment, proliferation, and long-term survival in the host liver.
In summary, we found that high-quality hepatocytes can be obtained from neonatal livers, which can extend the pool of available organs for HT. It is important to note that neonatal hepatocytes can be cryopreserved with no significant loss of viability after thawing, suggesting that the cells isolated from neonatal livers seem less vulnerable to cryopreservation effects than adult hepatocytes. This was observed by greater viability, cell attachment efficiency, adhesion molecule expression, and by less apoptotic cell death if compared with adult hepatocytes. Moreover, a larger number of progenitor cells in thawed hepatocyte suspensions suggests their advantage of being engrafted into the host liver and their better long-term survival. In addition, the assessment of the specific functional parameters analyzed after thawing reveals that neonatal cells have functional capabilities, such as P450 and conjugating enzymes activity, as well as ureogenesis. Thus, all the characteristics of thawed neonatal hepatic cells likely confer important advantages to neonatal hepatocytes for HT compared with adult cells.
Footnotes
Acknowledgment
The authors thank financial support from the Lubasa Foundation and the ALIVE Foundation. L.T. was a recipient of a Sara Borrell Contract from the “Instituto de Salud Carlos III” from the Spanish Ministry of Economy and Competitiveness. The authors declare no conflicts of interest.
