Abstract
Duchenne muscular dystrophy (DMD) is an inherited disease and a main target of myogenic cell transplantation (MT). After the failure of the first clinical trials with DMD patients, the poor migration of transplanted cells has been suspected to be a major problem for a more effective clinical application of MT. Previous investigations suggested that the quantity and dispersion of myofibers containing donor cell nuclei might be improved by increasing the migration of the transplanted cells outside the injection sites. Because the coinjection of motogenic factors with human myoblasts enhanced their intramuscular migration following MT in SCID mice, the present study aimed to investigate whether this approach was appropriate to increase MT success in muscles of nonhuman primates. In vitro studies indicated that IGF-1 or bFGF increased components of proteolytic systems involved in myoblast migration. In vitro and in vivo experiments also demonstrated that coinjection of bFGF or IGF-1 was able to improve monkey myogenic cell migration and invasion. Sixty hours after MT in skeletal muscle tissue, the migration distances reached by monkey myoblasts increased by nearly twofold when one of the growth factors was coinjected with the cells. However, long-term observations in adult monkeys suggest that promigratory treatments are not intrinsically sufficient to improve the success of MT. Even if short-term observations reveal that grafted cells are not always trapped inside the injection site and in spite of the fact that both factors enhanced transplanted cell migration, myofibers including grafted cell nuclei were still restrained to the injection trajectory without notable difference in their amount or their dispersion. The incapacity of transplanted cells to fuse with undamaged myofibers, which are located outside the injection sites, is a priority problem to solve in order to improve transplantation success and reduce the number of injections required for the treatment of DMD patients.
Introduction
Duchenne muscular dystrophy (DMD) is the most frequent hereditary myopathy and the principal target of myogenic cell transplantation (MT). This therapeutic approach consists in injecting normal muscle precursor cells in DMD patient muscles, so they can fuse together and with host fibers to repair them. After incorporation of donor cell nuclei, new hybrid myofibers are able to express the missing dystrophin protein. In addition to its capacity to introduce normal genes in preexisting myofibers, cell transplantation may be the only treatment that could also reconstitute the satellite cell population of the muscle (8, 26, 29, 41). Following the promising results obtained with MT in mice (25, 26), premature clinical trials were conducted with DMD patients (7, 9, 14, 21, 22, 37, 38). Results obtained by these clinical trials were practically negative and three problems were pointed out to explain these poor graft successes: immune-specific reactions against the donor cells, massive mortality of grafted cells, and absence of transplanted cell migration outside the injection sites (6, 12, 13, 35).
Ever since, our group has been working to solve or reduce the impact of these problems. Tacrolimus (FK-506) has been found effective to control acute rejection of the transplanted myoblasts; increasing the number of grafted cells helped to balance the high mortality rate, and transplanting myoblast by injections very close to each other compensated for the absence of cell migration (16, 31, 34). All therapeutic approaches taken into account, the results observed in the most recent clinical trials of MT are the best ever obtained so far in DMD patients (29, 30). However, improvements such as reducing the high number of injections required to treat a muscle are still necessary for a more effective clinical application of MT. In dystrophic patients, like in nonhuman primates, each injection trajectory leaves a thin path of fibers expressing donor proteins (31–33). Consequently, the closer the cell injections are performed the better is the transplantation success. Considering that normal myoblasts migrate to less than 500 μm of the injection site after their intramuscular injection (3–5, 34, 36), it has been suggested that the quantity and dispersion of myofibers containing donor cell nuclei might be improved by increasing the migration of transplanted cells outside the injection sites. If a better spreading of hybrid myofibers is obtained following a single injection, the number of injection sites and the distance between them will consequently be reduced without affecting the success of MT.
A new approach to enhance the migration of human skeletal myoblasts consists in coinjecting motogenic factors (19, 20, 24). In recent studies, insulin like growth factor-1 (IGF-1), basic fibroblast growth factor (bFGF), interleukin-4, and mechanical growth factor (MGF) were found to possess chemokinetic potential and were able to increase the migration distances reached by human myoblasts 48–60 h after their intramuscular transplantation in SCID mouse muscles. Among the different mechanisms by which these treatments were able to enhance myoblast migration processes, results indicate that components of the metalloproteinase and the fibrinolytic systems were involved. Initially identified to be highly implicated in the in vivo migration of mouse myoblasts, these proteolytic systems help the migrating cells to reduce the resistance imposed by the extracellular matrix (4, 5).
Because the investigations carried out previously with human myoblasts were done in SCID mice (19, 20), the present report aimed to evaluate whether motogenic factor coinjection is appropriate to enhance myoblast migration and improve the transplantation success in a nonhuman primate model. In opposition to the great transplantation successes observed in mice (10, 16, 18), myoblast transplantations in monkeys are less successful and can be extrapolated more easily to humans (15, 31, 34). Our results demonstrate that both IGF-1 and bFGF improved the overall migration of monkey myoblasts in a serum-free environment. Treatments with these factors also stimulated components of proteolytic systems and enhanced cell invasion in vitro. As observed with human myoblasts, growth factor coinjection with monkey myoblasts also increased their intramuscular migration in SCID mice. However, observations in adult monkeys suggest that the migration of the transplanted cells is not the principal factor that restricts the presence of hybrid myofibers along the injection trajectories. The incapacity of myogenic cells to fuse with undamaged myofibers, which are located outside the injection sites, is also a priority problem to solve to improve transplantation success and reduce the number of injections required for the treatment of DMD patients.
Materials and Methods
Cell Culture
Monkey myoblasts were obtained from an adult monkey (Macaca mulata) biopsy. Myogenic cells were released from minced fragments by enzymatic dissociation. After 1-h incubation in collagenase (600 UI/ml; Sigma-Aldrich, St. Louis, MO, USA), cells were incubated for 30 min in Hank's buffer salt solution (HBSS) including 0.1% trypsin and 0.02% EDTA w/v (Gibco product, Invitrogen, Ontario, Canada). Dissociated cells were grown in a modified MCDB 120 medium containing 10% fetal bovine serum, 10 ng/ml of epidermal growth factor, 100 U/ml of penicillin, and 100 μg/ml of streptomycin. All cells were cultured below 80% confluence.
In Vitro Studies of Secreted Proteins
At the desired confluence, cells were rinsed with HBSS and incubated for 24 h in serum-free media with or without different concentrations of recombinant IGF-1 and/or bFGF (IGF-1: Sigma-Aldrich, USA; bFGF: Strathmann Biotec AG, Germany). After the incubation, conditioned media were collected and concentrated using a Centricon YM-30 (Millipore Corp., Ontario, Canada). Bio-Rad protein assay (Bio-Rad Laboratories, Hercules, CA, USA) was used to measure protein concentrations. Secreted proteins/lane (12 μg) were loaded for zymography analysis. Coomassie blue-stained gels were always performed as loading controls to verify that the same amounts of proteins were loaded for each protein extraction used in our experiments.
Zymography
To assess the activity of gelatinases, proteins were electrophorized on 10% polyacrylamide gel containing 1.5 mg/ml gelatin (Laboratoire Mat Inc., Quebec, Canada). The gel was subsequently washed twice with 50 mM Tris-HCl, 2.5% Triton X-100 for 30 min, and the gelatinase activity was promoted by incubating the gel overnight at 37°C in the activation buffer (50 mM Tris-HCl, pH 7.6, with 150 mM NaCl and 10 mM CaCl2). After staining with 0.5% Coomassie brilliant blue, gelatinase activity was visualized as a clear band at a specific molecular weight (62–72 kDa for MMP-2) and was submitted to NIH Image densitometric analysis (software version 1.62).
Transwell Migration Assay
Twenty-four-well transwell assay dishes (Corning Life Sciences, Acton, MA, USA) were used to examine myogenic cell migration. Briefly, this modified Boyden chamber consists in a well (lower compartment) containing an insert (upper compartment). The upper and lower compartments are separated by a 8-μm pore size polycarbonate membrane. Prior to cell seeding, transwell permeable supports were preincubated for 2 h in the appropriate media to improve cell attachment and spreading. Myoblasts were harvested and suspended in serum-free medium at a concentration of 3 × 105 cells/ml. One hundred microliters of the cell suspension was then loaded into the upper chamber of a transwell containing 0.6 ml of serum-free medium in the lower chamber. To assess chemokinesis, 50 ng/ml of IGF-1 or bFGF was added to the cell suspension in the upper chamber. Following a 14-h incubation period at 37°C, the inserts were removed and washed three times in phosphate-buffered saline (PBS). Cells remaining on the upper surface of the insert (nonmigrated cells) were removed gently using a cotton swab, and cells on the lower surface were stained for 20 min in a solution containing 0.4% crystal violet, 2% ethanol, and 0.1% ammonium oxalate. After staining, inserts were rinsed three times by dipping into PBS and left to air dry. Colored cells that had migrated through the pores of the polycarbonate membrane were visualized using a stereomicroscope. Results were digitized using a Nikon COOLPIX-4500 camera (Nikon Canada Inc., Ontario, Canada), and colored cells were analyzed using the NIH Image software.
In Vitro Invasion Assay
The evaluation of the myoblast invasion was performed with the Chemicon collagen-based Cell Invasion Assay Kit (Chemicon international, Temecula, CA). The invasion chamber is a well from a 24-well plate (lower compartment) containing an insert (upper compartment). The bottom of the insert is a 8-μm pore size polycarbonate membrane covered by a layer of collagen matrix. In all experiments, 300 μl of a serum-free medium suspension containing 1 × 106 myoblasts/ml was added to the upper compartment, whereas the lower chamber was filled with 500 μl of medium containing 15% fetal bovine serum. The promigratory potentials were evaluated by coinjecting the factor to be tested with the cells in the upper chamber (50 ng/ml of IGF-1 or bFGF). Invasion assays were performed at 37°C for 72 h. Cells remaining on the upper surface of the insert were removed gently using a cotton swab. Cells trapped in the filter pores and cells that migrated on the lower surface of the membrane were visualized by dipping the inserts in the staining solution (0.4% crystal violet, 2% ethanol, and 0.1% ammonium oxalate). Colored cells were visualized under a stereomicroscope and results were digitized using a Nikon COOLPIX-4500 camera (Nikon Canada Inc, Ontario, Canada).
In Vivo Quantification of Myoblast Proliferation by Real-Time PCR
Monkey male myoblasts (5 × 105) suspended in 10 μl of HBSS with or without 50 ng of IGF-1 or bFGF were transplanted into female SCID mouse TA muscles. At various time points, transplanted muscles were removed and their DNA was extracted. The amount of monkey Y chromosome was quantified in each muscle by qPCR using a LightCycler 480 (Roche Diagnostics). Primers used to amplify monkey Y chromosome were: 5′-GCT CTT CCT TCC TTT GCA CTG A-3′ and 5′-GCT GCT TGC TGA TCT CTG AGT TTC 3′. A mouse internal control was also used. The primer sequences used to amplify HSD3B1 intron were: 5′-CAC CCC TTA AGA GAC CCA TGT T-3′ and 5′-CCC TGC AGA GAC CTT AGA AA AC-3′. The PCR profiles for the monkey Y chromosome and the mouse internal control were 10 s at 95°C, 10 s at 62°C, 14 s at 72°C, and reading at 80°C during 5 s, and were repeated for 50 cycles.
In Vivo Cell Death Assay
To quantify the in vivo cell mortality, monkey myoblasts were radiolabeled by culturing them 48 h in growth medium containing 0.25 μCi/ml [methyl-14C] thymidine (50 mCi/mmol). Radiolabeled cells (5 × 105) were injected into 12 sites of the TA muscle of 12-week-old SCID mice using a glass micropipette (Drummond Scientific Co., Broomall, PA, USA). The right TA was injected with myoblasts suspended in 10 μl of HBSS containing 50 ng of IGF-1 or bFGF while the left TA muscle was used as control (transplanted with myoblasts suspended in HBSS). Five muscles were used for each group. Muscles were then removed at the indicated time points, snap-frozen in liquid nitrogen, and stored at −80°C. The amount of radiolabel within each TA muscle was measured on DNA extracts using liquid scintillation counter (Mod. Wallac 1409, Woodbridge, Ontario, Canada).
Analysis of Intramuscular Migration with the Microtube Technique
This work was authorized and supervised by the Laval University Animal Care Committee and conducted according to the guidelines set by the Canadian Council of Animal Care. Approximately 2-month-old SCID mice (Charles River, Toronto, Canada) were used as hosts for these experiments. Perforated microtubes (Intramedic Polyethylene Tubing, Clay Adams-Becton Dickinson, NJ, USA) were implanted in both tibialis anterior (TA) muscles following the technique previously described (5). To visualize the transplanted cells in vivo, dissociated cells were labeled with PKH-67 (green fluorescence) using the supplier's protocol (Sigma Immuno Chemicals) and the labeled myoblasts (1 × 106 cells/Ependorf) were kept on ice until transplantation. Before myoblast injection, cells were pelleted and suspended in 7 μl of HBSS with or without 50 ng of IFG-1 or bFGF. With a 50-μm-tip glass micropipette (Drummond Scientific Co.), cells were slowly injected from the proximal extremity of the polyethylene microtube, which had been placed intramuscularly. Treated or untreated cells were injected in the contralateral TA muscle. After 60 h, TA muscles were taken out and entirely cut in transversal cross sections (18 μm thick). Only the cross section showing the maximum migration distance in each injected muscle was used to measure the migration distance from the injection site depicted by the microtube. After images were digitized using a Nikon COOLPIX 4500 camera, concentric equidistant circles were superimposed on the photograph of the selected muscle cross section. Distance between each bold circle was 165 μm, and migration distance was measured from the external surface of the microtube up to the farthest located group of PKH-labeled fluorescent cells.
Evaluation of Damaged Myofibers
Evans blue dye uptake, indication of sarcolemmal damage, was used to visualize damaged myofibers after cell transplantation through the microtube. The low-molecular-weight dye solution (200 μg/ml) was prepared in PBS and sterilized by 0.2-μm filtration. The Evans blue solution was injected intraperitoneally (100 μl/25 g) 16 h before SCID mice euthanasia.
Dystrophin Detection
TA muscles of SCID mice were removed 2 weeks after MT through the microtube. Frozen muscle cross sections were blocked in PBS with 10% FBS for 30 min and then incubated 1 h with the mouse monoclonal antibody for human dystrophin (NLC-DYS3, diluted 1:20, Novocastra, Newcastle, UK). Finally, muscle sections were incubated 1 h with a goat anti-mouse biotinylated antibody (diluted 1:150, DAKO Diagnostics, Mississauga, ON, Canada), and labeled with streptavidin-Cy3 (diluted 1:500, Sigma-Aldrich). Muscle cross sections were washed with PBS before and after incubation with both antibodies and all incubations were performed at room temperature.
Long-Term Analysis of Graft Success in Adult Monkey
Myoblasts were infected three times in vitro with a retroviral vector (LNPOZ) encoding the LacZ gene (gift from Dr. C. Cepko, Harvard University, Boston, MA, USA). Before intramuscular implantation, myoblasts were suspended in HBSS supplemented or not with IGF-1 or bFGF (50 ng/106 cells) and injected in a portion of the biceps brachii using a 50-μl Hamilton syringe with a 27-gauge needle. Interinjection distance was 2 mm and 6 to 10 injections were performed parallel to each other. For each trajectory, 1 × 106 cells were delivered during the needle withdrawal, these trajectories being roughly 1 cm (the needle length). Monkey was immunosuppressed with FK506 as described previously (31). Biopsies of the transplanted area were performed 1 month after MT and entirely cut in serial cross sections of 30 μm thick. Finally, cross sections were stained for β-galactosidase (β-Gal) to visualize the myofibers that had incorporated transplanted cell nuclei.
Statistical Analysis
For migration, proliferation, and survival assays, the significance of results was evaluated using an analysis of variance (ANOVA) on Stat View 512 software (Brain Power, Calabasas, CA, USA) followed by Fisher's PLSD test.
Results
Growth Factors Improved the Overall Rate of Movement in a Serum-Free Environment
To determine whether IGF-1 or bFGF improves the overall rate of cell movement in a serum-free environment, monkey myogenic cells were loaded into the upper compartments of transwell migration assays. As revealed by colorimetric analysis (Fig. 1), both cytokines enhanced cell migration compared with control condition. When IGF-1 or bFGF was added to the serum-free medium, the quantity of myoblasts that migrated through the 8-μm pore size polycarbonate membrane to reach the lower chamber of the assay increased by 2.7- and 2.5-fold, respectively (p < 0.0034 and p < 0.0054).

IGF-1 and bFGF improve the overall migration rate of monkey myoblasts in serum-free medium. Cells (3 × 104/100 μl were loaded into the upper chamber of a transwell migration assay. Media in the upper chamber were complemented or not with 50 ng/ml of IGF-1 or bFGF; while the lower chamber contained only serum-free medium. Following 14 h of incubation, migrating cells were stained and colored cells were analyzed using the NIH Image software. Representative pictures of the colored inserts are shown above the graph. νp < 0.0054 relative to the control. φp < 0.0034 relative to the control.
Evaluation of Growth Factor Effects on Myoblast Proteolytic Components and Invasion Capacity In Vitro
As previously demonstrated with human and mouse myoblasts, the metalloproteinases (MMPs) are essential for the intramuscular migration of muscle precursor cells. Before doing in vivo experimentation, we had to confirm that IGF-1 and bFGF treatments could also improve the proteolytic activities and invasion capacity of monkey myoblasts. Among the proteolytic components implicated in human skeletal myoblast migration, growth factor effects on monkey cells were evaluated on one metalloproteinase (MMP-2). Zymography analysis revealed that monkey myoblasts treated with both factors exhibited a significant effect on gelatinase-A (MMP-2) (Fig. 2a), while showing no effect on MMP-9 (data not shown).
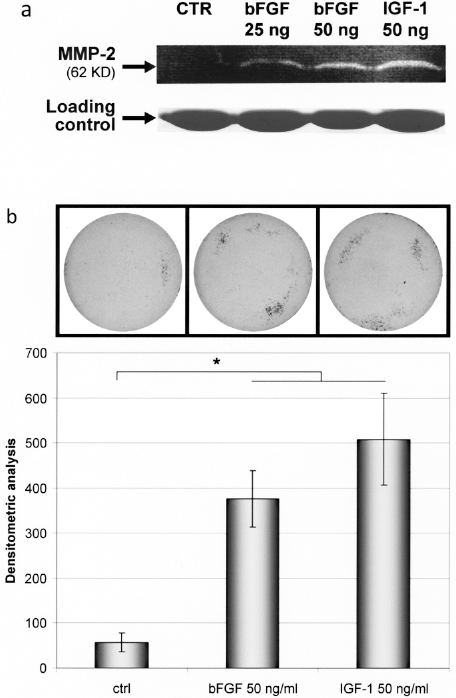
IGF-1 and bFGF improve proteolytic activities and invasion capacity of monkey myoblasts. (a) After 24 h of incubation in serum-free media with or without the GF to be tested, secreted proteins in the media were collected and concentrated. Gelatin zymography was used to assess the activity of gelatinases. Gelatin degradation is visualized as a clear band at a specific molecular weight (62–72 kDa for MMP-2). (b) Myoblasts (3 × 105) in a serum-free medium were placed in the upper chamber of an invasion assay well with or without IGF-1 or bFGF (50 ng/ml). The lower chamber of the well was filled with complete medium. Following 72-h incubation, cells that reached the lower chamber and cells trapped in the filter pores were stained and were analyzed using the NIH Image software. Representative pictures of the colored inserts are shown above the graph for each group. *p < 0.0065 relative to the control.
Invasion assays were used to determine whether the presence of the growth factors with the cells improves their migration rate toward a gradient of serum. Cells were placed in the upper chamber of the assay with or without 50 ng/ml of IGF-1 or bFGF. The lower chamber of the assay was filled with medium containing 15% serum to serve as chemoattractant. In order for the cells to migrate through the porous membrane, they must digest away enough of the collagen layer that covers it. As revealed by colorimetric analysis (Fig. 2b), both cytokines enhanced cell invasion compared with control condition. The amount of invading cells that migrated through the collagen layer and through the 8-μm pore size membrane increased by 6.6-fold in the presence of bFGF and by 8.9-fold when IGF-1 was added (p < 0.002).
Improvement of Intramuscular Migration Following Growth Factor Coinjection with Transplanted Myoblasts
To assess the chemokinetic potential of bFGF or IGF-1 in an in vivo model, 1 × 106 labeled myoblasts were transplanted in the tibialis anterior of SCID mice using the microtube technique. Monkey myogenic cells were coinjected with or without growth factors (50 ng/106 cells) and the highest migration distance was measured as described above. Sixty hours posttransplantation, the radial migration reached by untreated myoblasts was 503 ± 112 μm from the external surface of the microtube (Fig. 3a). When bFGF was coinjected with the myogenic cells, the maximal migration distance increased significantly and reached 802 ± 93 μm (p < 0.003). For IGF-1, the maximal migration distance increased by 2.2-fold to reach 1089 ± 148 μm (p < 0.001). These results confirm the promigratory effects observed in vitro and suggest that growth factor coinjection with monkey myoblasts might promote their migration and potentially their fusion outside the injection site following MT in skeletal muscles.
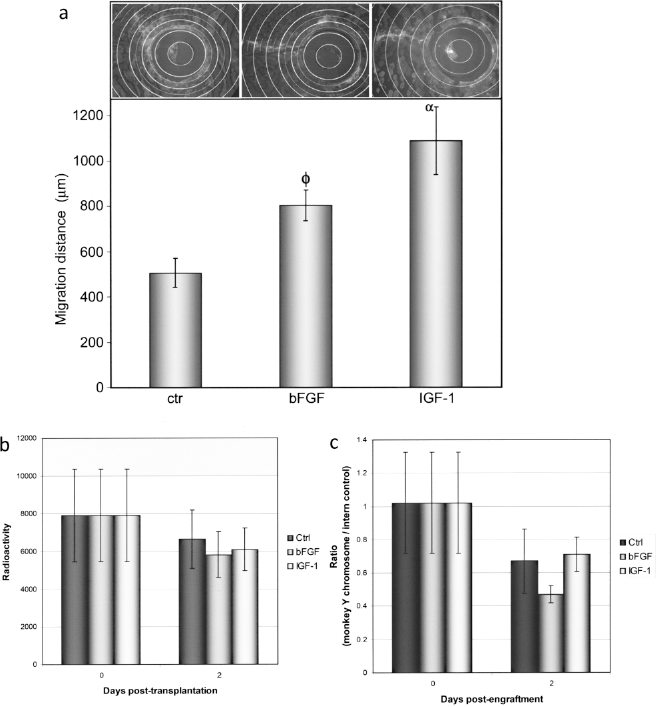
Short-term evaluation of the in vivo migration, survival, and proliferation of monkey myoblasts. (a) Labeled myoblasts (1 × 106) were transplanted in the tibialis anterior of SCID mice using the microtube technique. Cells were coinjected with or without cytokine and their migration distance was measured 60 h posttransplantation. Representative patterns of migration are shown above the graph for each group (bold circles are 165 μm equidistant). Graph presents the radial migration reached by control myoblasts (503 μm) versus myoblasts coinjected with bFGF (802 μm) or IGF-1 (1089 μm). The values represented in the graph are the means ± SEM (n = 5 for each group). (φ and α indicate significant differences relative to the control with values of p < 0.05. (b, c) Male myoblasts (5 × 105 coinjected with or without cytokine were transplanted in the tibialis anterior of female SCID mice. At different time points (30 min and 2 days after the engraftment) muscles were sampled and DNA was extracted. The radiolabeled DNA extracts were measured using liquid scintillation counter (b) and the amount of monkey Y chromosome was quantified in each muscle by quantitative PCR (c). No significant differences were observed between control and treated group DNA samples.
IGF-1 and bFGF Fail to Improve the Overall Survival Rate and Proliferation of Monkey Myoblasts In Vivo
To verify if the increased migratory capacity of the cells was related to potential effects of the growth factors on cell proliferation or survival, radiolabeled male myoblasts were transplanted in the TA muscles of female SCID mice. As for the microtube experiments, cells were injected with or without growth factors (50 ng/106 cells). Grafted muscles were collected immediately after cell transplantation (T0) or after 48 h. The amount of radioactivity measured from DNA extracts revealed that neither IGF-1 nor bFGF had effect on myoblast survival (Fig. 3b). Moreover, real-time PCR amplification of the Y chromosome indicated that both factors failed to improve the in vivo proliferation of monkey myoblasts 2 days after their transplantation (Fig. 3c).
Long-Term Graft Success Following Transplantation of Monkey Myoblasts Coinjected with IGF-1 or bFGF
After being infected with a retroviral vector containing the LacZ gene, monkey myoblasts were transplanted in the biceps of an adult monkey. For the transplantation, myoblasts were coinjected or not with IGF-1 or bFGF (50 ng/1 × 106 cells/injection) and the cell injections were performed parallel to each other. To evaluate graft success 1 month posttransplantation, transplanted muscle cross sections were stained for β-Gal. As observed in Figure 4, muscle fibers that have incorporated transplanted cell nuclei are identified by their β-Gal staining. Unfortunately, bFGF or IGF-1 coinjection with the cells did not improve long-term graft success in the primate. For all the conditions tested, no β-Gal-stained myofibers were observed in the interinjection areas. The hybrid myofibers expressing this donor protein were still restricted to the injection trajectories without notable difference in their amount or their dispersion.

Long-term graft success following transplantation of monkey myoblast coinjected with growth factor. Myogenic cells infected with a LacZ retroviral vector were transplanted in the biceps of an adult monkey with or without IGF-1 or bFGF (50 ng/1 × 106 cells/injection). Transversal injections were performed parallel to each other with a 2-mm interinjection distance and muscle biopsies were performed 1 month posttransplantation. Biopsy cross sections were stained for β-Gal to visualize myofibers that incorporated transplanted cell nuclei. β-Gal myofibers are seen as different degrees of a dark precipitate. Scale bars: 1 mm; area shown in the insets: 500 μm width by 900 μm length.
Follow-Up of Myogenic Cell Transplantation Through the Microtube
To verify if new hybrid myofibers could be produced by the migrating myogenic cells, 1 × 106 labeled myoblasts were transplanted through a polyethylene microtube inserted in the TA of SCID mice. Two weeks after monkey myogenic cell transplantation, dystrophin staining revealed that practically no hybrid myofibers could be observed at more than 200 μm of the microtube. The same results were obtained with bFGF or IGF-1 coinjection (i.e., only rare dystrophin expressing myofibers were found and these were located close to the microtube) (Fig. 5).

Short- versus long-term observations following MT through the polyethylene microtube. PKH-67 labeled monkey myoblasts (1 × 106) were suspended in HBSS containing 50 ng of bFGF or IGF-1 and transplanted in SCID mice using the microtube technique. (a) Systemic injection of Evans blue revealed that only rare damaged myofibers (red florescence following Evans blue incorporation) could be observed next to microtube an hour after its insertion and after cells injection (green). (b) Sixty hours posttransplantation, green labeled myogenic cells (PKH-67) could be visualized at more than 700 μm of the microtube. (c) Dystrophin staining revealed that hardly any hybrid myofibers could be observed 14 days posttransplantation. Only occasional dystrophin expressing fibers were found close to the microtube insertion site. Scale bars: 165 μm.
Evans blue was administered systemically to elucidate whether the scarce fusion with host myofibers was spontaneous or could be correlated with myofibers that are damaged by cell implantation through the microtube. Results revealed that only rare damaged myofibers could be detected next to the microtube 1 h after its insertion and after cell injection (Fig. 5a). Even if myoblasts were detected at more than 700 μm of the microtube when bFGF or IGF-1 was coinjected with the cells (Fig. 5b), the long-term observations seem to indicate that migrating cells have not fused with the myofibers they reached (Fig. 5c). As expected, the frequency and localization of hybrid myofibers observed 14 days after cell transplantation were in accordance with those of damaged muscle fibers observed shortly after transplantation. Taken together, these results suggest that transplanted myogenic cells might only fuse with damaged host muscle fibers.
Discussion
Cell transplantation remains to date the only treatment that could both reconstitute the satellite cell population of the dystrophic muscle and restore dystrophin expression in preexisting myofibers. Even if MT produced the best results observed until now in DMD patients (29, 30, 33), a poor dispersion of hybrid myofibers outside the injection sites is a major problem that remains for a more effective clinical application.
Because the restriction of hybrid myofibers to the injection sites has been attributed to a limited migration potential of myoblasts (4, 5, 36), many promigratory treatments were attempted to increase the intramuscular migration of transplanted myoblasts. The goal of these treatments was to obtain migrating cells fusion with myofibers located outside the injection trajectories. By increasing the number and dispersion of hybrid myofibers produced by a single cell injection, the final objective was to reduce the high quantity of injections required to obtain an optimal transplantation success for the treatment of DMD patients. Unfortunately, our results suggest that promigratory treatments are not intrinsically sufficient to improve MT success in the primate.
Fundamentally, MT is an approach that rests principally on the basic mechanisms of the muscular regeneration. After muscle injury, the major function of myoblasts produced by activated satellite cells is to repair the damaged myofibers. Once muscular damage is repaired, these myogenic precursors are not found any more between myofibers. Myogenic cells that do not proceed to the terminal differentiation and that are unable to return to the state of quiescence next to myofibers probably die by apoptosis (27, 39) and are phagocyted by macrophages. Following MT, it is likely that the grafted cells are only able to fuse with the myofibers damaged by the injection needle. If this is indeed what occurs, no improvement of the transplantation success would be observed even if an approach allowing transplanted cells to migrate across an entire muscle is developed.
In an article by Watt et al. (40), which had particularly encouraged us to improve the migratory potential of the transplanted myoblasts, it was reported that C2C12 injected into a muscle could form hybrid myofibers in the adjacent muscle. However, it should be noted that hybrid myofibers were observed only when the adjacent muscle tissue was injured. Even in the case of myogenic cells with strong migratory potential, it is likely that muscular damage is also necessary to permit fusion of the transplanted cells. In SCID and dystrophic mice, the highest percentages of dystrophin-positive fibers were observed only after damaging the irradiated host muscles or by performing successive transplantations to stimulate regeneration (10, 17). In primates, we have also demonstrated that successful myoblast transplantation depends on appropriate cell delivery and induction of host muscle regeneration (34). In this article, authors demonstrated that hybrid myofibers produced by monkey myoblasts injected without notexin are restricted to the injection sites. In contrast, hybrid myofibers were homogeneously distributed into the muscle when notexin was coinjected with the cells.
In the present study, nearly no hybrid myofibers were observed when myoblasts were injected through a perforated microtube placed in a muscle, and were allowed to migrate from this microtube for 2 weeks or more (similar results were obtained with monkey and human myoblasts). This absence of dystrophin-expressing myofibers was initially explained by the fibrosis that develops around the microtube. However, it is also possible that migrating cells were simply unable to fuse with undamaged myofibers. This hypothesis is supported by the systemic injection of Evans blue, which revealed that only rare damaged myofibers could be found next to the microtube after its insertion and after cell injection (Fig. 5). Because the migrating cells observed 60 h after MT in SCID mice had no muscle fibers to repair, they possibly died because of their incapacity to differentiate.
Short-term observations performed after MT in adult monkeys also supported the new hypothesis. Contrary to our beliefs, short-term observations revealed that myogenic cells are not rigorously contained within the needle trajectory after their injection in skeletal muscle tissue. In opposition to the distribution of hybrid myofibers that follow the original injection trajectories 1 month posttransplantation, the distribution of injected cells turned out to be highly variable following a single intramuscular injection (unpublished observations). The grafted cells were mostly located in layers of connective tissue ripped open by the needle and/or by the pressure of the injection (ruptures are generally found at the level of large connective tissue bands such as the perimysium or the epimysium). Unpredictable cell distribution and cell pockets inside the connective tissues were also observed following single transversal injections performed in mouse muscles. During the first 3 days following transplantation, no fusion or dystrophin detection could be observed even if myoblasts were found around host myofibers. The important point that comes from these observations is that even if transplanted cells scatter differently from one intramuscular injection to another, the long-term results remain the same (i.e., the majority of hybrid myofibers remain located along the injection trajectories).
In previous results from our group, myoblast treatment with bFGF improved the transplantation success in a dystrophic muscle (mdx mouse) (17, 18). One hypothesis that could explain an improved transplantation success in dystrophic mice is the presence of more degenerating muscle fibers. In the presence of fibers to repair, the increased migratory capacity given by the growth factor could have led to the formation of hybrid myofibers at sites further from the injection site. Because dystrophic primates do not exist, we had to perform the present experiments in nondystrophic animals and this may be one reason that explains why a similar improvement of myoblast transplantation has not been observed in the monkey.
In conclusion, our results suggested that limited cell migration is not the principal limiting factor restricting the presence of hybrid myofibers only to the injection trajectories. If we want the promigratory treatments to be efficient, the migrating cells must also have the ability to fuse with the myofibers they reach. The difficulty of transplanted cells to fuse with host myofibers not damaged by the injection is a priority problem to solve to improve the transplantation success and reduce the number of injection sites required for the treatment of DMD patients (Fig. 6). For a more effective clinical application of MT, future strategies should be based on the two phenomena normally responsible for myoblast fusion with existing muscle fibers. The first strategy consists simply in harming the muscle fibers so that they can be repaired by the grafted cells (2, 11, 28, 30, 34), while the second consists in stimulating host muscle fibers hypertrophy via the recruitment of new transplanted mononucleated cells. As already shown by our group, induction of a muscular hypertrophy and improvement of MT in mice was achieved by blocking the myostatin signal (1) and by systemic administration of an IGF-1-derived peptide (MGF-Ct24E peptide) (23).

Reevaluation of the migration problem. The restriction of the hybrid muscle fibers, which is observed after a single myoblast injection, is due to the muscular damage produced by the injection (a) rather than limited cell dispersion (b). Without the ability of migrating cells to fuse with the myofibers they reached, the development of protocols that increase the intramuscular migration of transplanted cells will not improve the success of MT (c). The most important methods to obtain a higher dispersion and number of hybrid myofibers (d) would be either to damage more fibers around the injection trajectory or to improve transplanted cell recruitment by undamaged host fibers following stimulation of a muscular hypertrophy.
Footnotes
Acknowledgments
This work was supported by grants from the FRSQ (Fonds de la Recherche en Santé du Québec), the AFM (Association Française contre les Myopathies), the MDC (Muscular Dystrophy Canada), ALS (Amyotrophic Lateral Sclerosis), and CIHR (Canadian Institute of Health Research).
