Abstract
Autologous brain cell transplantation might be useful for repairing lesions and restoring function of the central nervous system. We have demonstrated that adult monkey brain cells, obtained from cortical biopsy and kept in culture for a few weeks, exhibit neural progenitor characteristics that make them useful for brain repair. Following MPTP treatment, primates were dopamine depleted but asymptomatic. Autologous cultured cells were reimplanted into the right caudate nucleus of the donor monkey. Four months after reimplantation, histological analysis by stereology and TH immunolabeling showed that the reimplanted cells successfully survived, bilaterally migrated in the whole striatum, and seemed to have a neuroprotection effect over time. These results may add a new strategy to the field of brain neuroprotection or regeneration and could possibly lead to future clinical applications.
Introduction
The use of neural grafts to restore function after lesions or degeneration of the central nervous system has been widely reported. Most of the transplantation experience acquired during the last two decades was focused on fetal neuronal grafts (2, 20, 24, 36, 39). The objective was to replace degenerated neurons, which were restricted to a limited brain area, such as the known pathologies of Parkinson's and Huntington's disease (3, 5, 14, 29, 32, 35, 37). However, despite the great enthusiasm generated by this approach, ethical controversies, immune rejection (38), poor tissue survival, and lack of fetal donors remain major problems.
For many years, the debate has evolved to include the relative merits of using embryonic and adult stem cells for cell therapy (28). Embryonic and fetal stem cells are also controversial, more likely to be rejected when they mature, and fail to produce large numbers of DA neurons after implantation (11, 26, 27, 40, 46). Recent methods for gene reprogramming of somatic cells to pluripotent states would likely avoid immune rejection (11, 31, 42, 49). A recent report demonstrated, in a rat PD model, the potential of such neurons derived from reprogrammed cells to integrate in the brain (50). If it is possible to minimize the risk of tumor formation by separating contaminating pluripotent cells and committed neural cells (16, 50), the risks could be reduced for cell sources that present a high rate of proliferation in vitro. Therefore, autotransplantation of adult brain cells that were shown to have a limited proliferation rate (9) represents an attractive alternative to bypass many of these caveats and to elucidate mechanisms of neural repair.
Efforts have been made over the past few years to develop appropriate methods to prepare long-term primary cultures from primate cortical biopsies (22, 23, 25, 30, 47). After optimization of the procedure for brain cell culture preparation, using a medium with preselected fetal calf serum (pFCS), we succeeded in producing long-term primary cultures of adult human brain cells obtained from fresh or cryopreserved tissues collected during epilepsy or trauma neurosurgeries (7, 8). Furthermore, previous studies suggested that the reconstruction of the cortical circuitry responsible for sensory, motor, or cognitive function may be possible by allogeneic stem cell transplantation in mature mouse neocortex (18).
In a preliminary study in a motor cortex lesion model, we have demonstrated that the autotransplanted cells could survive, migrate, and acquire mature neuronal characteristics over time after reimplantation in the donor cortex of mice (9). Here, we demonstrate for the first time the feasibility of adult brain cell autotransplantation in the brain of asymptomatic MPTP-treated monkeys. We show that the adult cortical brain cell autografts into the striatum can positively influence the number of dopaminergic neurons in the substantia nigra in mildly dopamine-depleted but asymptomatic parkinsonian monkeys. For these initial studies, we decided to choose an asymptomatic low-dose MPTP model in order to determine biological effects prior to subjecting monkeys to the full effects of parkinsonian disabilities and the more extensive care required to maintain them.
Materials and Methods
Animals
Eight adult male St. Kitts green monkeys (Chlorocebus sabaeus) were used in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals and with approval of the Institutional Animal Care and Use Committee. Animals were trapped and maintained in individual cages at the St. Kitts Biomedical Research Foundation (St. Kitts, West Indies); they were fed regularly on a diet of fruit and monkey chow (Teklad) and had free access to water. The eight animals were separated into three groups: two normal controls without any intervention, two MPTP controls that only received MPTP without surgical intervention, and four reimplanted animals that were first biopsied, then treated with MPTP and reimplanted with their autologous brain cells (Table 1).
Animal and Experimental Timetable
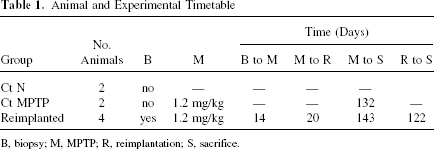
B, biopsy; M, MPTP; R, reimplantation; S, sacrifice.
Biopsies
The four adult St. Kitts African green donor monkeys were deeply anesthetized with pentobarbital to achieve and maintain a stage III surgical anesthesia under sterile surgical conditions; the doses varied from 25 to 50 mg/kg depending on duration. The animals were carefully monitored for vital signs, EKG, and pO2 and given supplemental oxygen and room air by endotracheal tube if needed. Biopsies (3 mm2) were surgically obtained from the dorsolateral prefrontal right cortex (Fig. 1A–D), placed into tubes that were completely filled with Hibernate A medium (Brain Bits, Springfield, IL, USA), and shipped from St. Kitts to Lausanne, Switzerland.

General observations of re-implanted monkey brains. (A–D) Macroscopic pictures of the monkey brains W802, W808, W818, and W825, respectively; arrowheads and gray spots correspond to the biopsy, the white arrows represent the injection tracts, the dark arrow shows the hemorrhage that occurred in the W802 case (A). (E) The mosaic reconstruction of GFAP immunohistostaining (in dark) is a part of cortex at the level of the hemorrhage showed in (A) and revealed the gliosis that occurred under the hemorrhage area. (F) Higher magnification of dark frame box (E) that shows hypertrophied GFAP-positive astrocytes in the gliosis. Scale bar: (E) 500 μm, (F) 100 μm.
MPTP Treatment
Two weeks after biopsy, the four donor monkeys were intramuscularly injected with MPTP 0.4 mg/kg/12 h times three doses to reach a final dose of 1.2 mg/kg. The two MPTP control monkeys received the same doses and were studied over the identical time periods. A parkinsonian factor and healthy behavior scores were derived from a prior study using a principal component factor analysis on 15,000 individual behavioral observations from 77 MPTP or control monkeys (36, 43, 44), and the score was averaged per week of observations. Individual behavioral observations of the MPTP-treated monkeys confirmed their asymptomatic status because they had normal healthy behavior and did not show parkinsonian behavior.
Preparation of Cultured Primate Brain Cells From Adult Tissue
The cortical biopsies were dissected with a razor blade to obtain enriched fractions of gray matter. Primary cultures were generated by mincing and mechanically triturating the tissue with fire-polished glass pipettes of decreasing diameters. Cells were resuspended at 50,000 cells/ml in RPMI-1640 medium (1×) without L-glutamine (31870-025, Invitrogen AG, Basel, Switzerland) supplemented with NaHCO3 44 mM, 20% preselected fetal bovine serum (pFBS), and an antibiotic/anti-mycotic cocktail (A7292 Sigma-Aldrich, St. Louis, MO, USA) directly into 25-ml glass Erlenmeyer at 37°C in a water-saturated atmosphere containing 6.5% CO2/93.5% air under horizontal agitation at 70 rpm. A cell suspension was plated on glass coverslips into a 24-well plate under the same incubator conditions. Fifteen days later, the concentration of serum was reduced to 10%. After 20 days, when cells became confluent on the coverslip, the cells were cultured in medium without serum and were maintained in these conditions until reimplantation 10 days later.
Labeling and Preparing Cells for Transplantation
Cell aggregates from one flask were pooled by centrifugation at 800 rpm. Supernatant was recovered for resuspension. Aggregates were resuspended in 500 μl diluent C with 5 μM green fluorescent viable dye PKH67 (MINI67, Sigma-Aldrich) for 3 min. FCS (500 μl) was added for 1 min and aggregates washed three times with RPMI medium. After the last wash, aggregates were resuspended with the recovered medium previously centrifuged at 4000 rpm to eliminate cellular wastes. Tubes containing fluorescent dye-stained aggregates were completely filled for transportation at room temperature from Lausanne to St. Kitts. For reimplantation, aggregates were settled and supernatant was partially removed until a volume of 100 μl remained, in which the aggregates were resuspended at a cell concentration of 3,000/μl.
Cell Reimplantation
The four MPTP donor adult St. Kitts African green monkeys were deeply anesthetized under sterile surgical conditions as described for biopsy. The resuspended aggregates were administered into the target site via a 22-gauge 100-μl Hamilton syringe attached to a microinfusion device (Stoelting, Wood Dale, IL, USA). Insertion of the needle was performed slowly and allowed to remain at the target sites for a 2-min period before injection, and the cannula tip was permitted to remain in the implantation site for at least 2 min before removal. Suspension was extruded at a maximum rate of 2 μl/min. Needle withdrawal occurred at a rate of 1 mm/min for 5 min, followed by a more rapid withdrawal for the remaining distance. Two sites in the right caudate nucleus (AP +23.1 mm and 19.1 mm, lateral 4 mm, +19mm above the ear bar zero and AP +19.1 mm, lateral right 4 mm, +19 mm above) were injected with 50 μl per site of PKH67-labeled aggregates corresponding to 280,000 ± 18,000 cells per animal (mean ±SD, four animals). Culture medium was injected into the left caudate nucleus in the same animals. The control monkeys were not injected.
Necropsy
Four months after reimplantation, and nearly 5 months after MPTP treatment, the monkeys were sacrificed using ketamine (8–10 mg/kg, IM) and sodium pentobarbital (30 mg/kg, IV, or more until loss of deep corneal reflex). The two normal controls were sacrificed using the same necropsy procedure and the two MPTP control monkeys were sacrificed at the same period of post MPTP treatment. Monkeys were then perfused with heparinized saline and fixed with 4% paraformaldehyde in phosphate buffer; brains were removed, postfixed for 12 h with 4% paraformaldehyde in phosphate buffer, and cryopreserved in 30% sucrose solution before freezing. Sections (50 μm thick) were prepared with a cryotome (HM560, MICROM, Volketswil, Switzerland) and were stored at −80°C until used in the cryoprotection 50 mM phosphate buffer pH 7.4, solution that contains 25% glycerol (G7893, Sigma-Aldrich), 30% ethylene glycol (33068, Ridel-de-Haën, Seelze, Germany).
Immunocytochemistry and Immunohistochemistry
Brain cryosections were incubated in PBS with 0.1% casein (C8694, Sigma-Aldrich) and then incubated in PBS 0.3% bovine albumin with antibodies against the following antigens: glial fibrillary acidic protein (GFAP) (monoclonal G3893, 1/1000, Sigma-Aldrich; polyclonal rabbit Z0334 1/500, Dako, Glostrup, Denmark), nestin (AB 22 polyclonal rabbit, Chemicon, Millipore, Billerica, MA, USA), doublecortin (guinea pig polyclonal DCX AB5910, 1/1000, Chemicon, Millipore), tyrosine hydroxylase (TH) (monoclonal MAB318, 1/1000, and polyclonal MAB 152, 1/500, Chemicon, Millipore), glial-derived neutrophic factor (GDNF) (MAB212, 1/1000, R&D Systems Europe Ltd, Abingdon, GB). They were then washed and incubated with fluorescent or horseradish peroxidase- conjugated secondary antibodies. After the last washes, they were revealed for peroxidase activity with diaminobenzidine or mounted in Vectashield® with DAPI to counterstain nuclei before fluorescent microscopic analysis or infrared fluorescence scanning (Odyssey LI-COR, Lincoln, NE, USA).
Histological Observation and Quantification
Drawings and cell counting of immunostained sections were made with an Olympus BX40 epifluorescence microscope equipped with a motorized X–Y-sensitive stage and a video CDD camera connected to a computerized image analysis system (Explora Nova, La Rochelle, France). Images were acquired using three software programs (Mosaic, Morpho Expert, and Fluo3D, Explora Nova, La Rochelle, France). Series of slices on whole brain were observed to define the brain section where PKH67 cells were detected. For stereological analysis, representative 50-μm slices separated by 750 μm in the defined brain section were used. The edges of the section and the structures such as caudate nucleus, putamen, and ventricles were outlined for each slice. The threshold of green fluorescence intensity and the limit of cell surface from 100 to 600 μm2 were applied to automatically identify all PKH67 cells on 11 to 13 slices depending on the cell migration (Mercator, Explora Nova, La Rochelle, France). A visual control for this PKH67 cell identification was done on 20 of 2500–3500 fields per slices. The obtained models of the slices with identified cells allowed 3D reconstruction and 3D cell counting through computer-based stereological analysis (Map3D, Explora Nova, La Rochelle, France). The total cell counts were compared with the number of implanted cells to obtain a percentage of survival and in parallel the percentage of cells per anatomical structures could be estimated.
TH immunolabeling were quantified on 10 cryosections covering the caudate and the putamen by infrared scanning (Odyssey LI-COR, Lincoln, NE, USA). Circles were drawn in different structures to obtain the integral intensity values, expressed in arbitrary units that reflect the number of emitted photons per surface pixel unit; for each animal, an integral intensity value was obtained for each structure and substructure for right and left hemispheres.
GDNF immunolabeling was quantified as TH immunolabeling with infrared intensity measurement done on four cryosections covering the area where PKH67 reimplanted cells were observed.
For the stereology of the dopaminergic neurons in the substantia nigra, TH neurons were counted on six sections covering anteroposterior extent of the whole substantia nigra. The sections were matched anatomically, verifying that the cross sections of the substantia nigra were similar in all animals. The criteria of DAB immunostaining intensity, cell surface limits from 100 to 600 μm2, and a form coefficient between 0.4 and 0.8 were defined by measuring cell surface and form coefficient on 20 TH positive neurons to detect TH neurons for the automatic computer based on each section and to perform the 3D cell counting through computer-based stereological analysis (Mercator, Explora Nova, La Rochelle, France).
All the immunolabeling were identically done for all the animals for cell counting or intensity quantification in order to compare the results despite the limited number of animals. To blindly collect the data, the slices were randomly labeled for the eight animals by a person other than the observer.
The values or ratios obtained with normal and MPTP control animals were compared with expected values from previous studies, such as the number of TH neurons in substantia nigra in normal and in asymptomatic MPTP controls or the decrease of TH in striatum after MPTP; we compared these control groups to the group of four reimplanted monkeys. Statistical differences were determined using one-way analysis of variance and Bonferroni Multiple Comparisons Test, using GraphPad Instat® software.
Results
The locations of the prefrontal biopsies, performed in four monkeys (Chlorocebus sabaeus), are illustrated (Fig. 1A–D). There were no signs of hemorrhage or other complications at the site of the biopsies and they were well tolerated by the animals. The dorsolateral part of the caudate nucleus as the target of reimplantion sites was precisely identified for the four reimplanted animals. The only complication related to the needle tract was a cortical hemorrhage, which was not considered clinically significant, followed by a gliosis observed histologically at the time of necropsy in case W802 (Fig. 1A, E, F). Individual behavioral observations of the MPTP-treated monkeys (reimplanted or not) confirmed their asymptomatic status because they had normal healthy behavior and did not show parkinsonian behaviors.
In vitro, the monkey adult brain cells proliferated by showing different mitotic states in culture over time (Fig. 2A–E) and expressed neuroectodermal and progenitor markers such as GFAP (Fig. 2F), neurofilament, vimentin, or nestin as described in previous studies (7, 8). In order to facilitate reimplantation, cells were cultured in suspension under slow agitation (50–75 rpm with an ES-W shaker-Kuhner). Cells obtained in suspension expressed the same markers (GFAP, vimentin, nestin, and doublecortin), as observed in monolayer cultures (Fig. 2F). They formed aggregates that contained few cells (5–10) with GFAP-positive long processes that surrounded small nestin-positive and doublecortin-positive cells (300–1,000) (Fig. 2G).
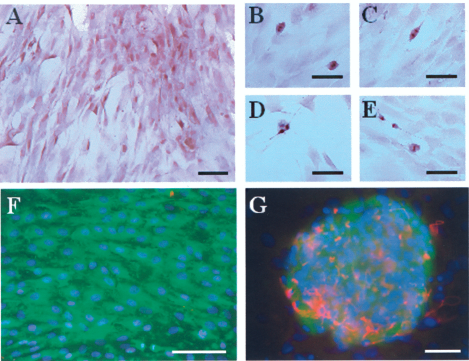
In vitro adult brain cell culture. (A–E) Hematoxylin staining of gray matter-derived cells. (A) Classical monolayer coverslip culture after 3 weeks. (B–E) Example of four mitosis stages observed in a proportion of less than 0.1%. (F) Nestin (green) and GFAP (red) immunolabeling in coverslip culture where only very few GFAP cells were found. (G) Doublecortin (DCX) (green) and GFAP (red) immunolabeling in suspension culture where aggregates were constituted with a few GFAP cells with long processes that surround a large group (300–1,000) of DCX cells. Scale bar: 100 μm.
Four months after reimplantation, the four monkeys were sacrificed and the brains processed. The 3D reconstruction done with 11 to 13 sections that cover the area where PKH67-labelled cells were targeted showed that reimplanted cells were viable and present not only in the reimplanted striatum (Fig. 3) but also in the contralateral striatum and in the corpus callosum (Fig. 3). Unbiased stereological counting analysis obtained from the 3D computerized reconstructions (Fig. 3) confirmed that 50.4 ± 14.6% (n = 4) of the autotransplanted cortical gray matter cells survived (Table 2). The majority (59.83 ± 3.95%, n = 4) of these cells were located in the implanted caudate nucleus. The remaining cells were distributed primarily in the striatum (ipsilateral putamen 14.17 ± 0.95%, contralateral caudate nucleus 11.77 ± 2.66%, contralateral putamen 0.32 ± 0.07%; n = 4). There were 13.91 ± 6.06% (n = 4) of the reimplanted cells located outside the striatum in the corpus callosum (Fig. 3, Table 2). No reimplanted cells were detectable in other structures except for one monkey. For this particular case (Fig. 3B), autotransplanted cell survival was lower than for the other three monkeys (30.65% compare to 49% to 65%) and 22.94% of the reimplanted cells were located in the needle tract as well as in the cortex where a small hemorrhage took place and a significant gliosis was observed (Fig. 1A, E, F). In the four reimplanted animals, gliosis was also observed in the striatum (Fig. 4K, L).

Localization of PKH67-labeled cells 4 months after reimplantation. (A–E) All four cases are represented by 3D reconstruction (Map3D Explora Nova, La Rochelle) to illustrate the distribution of PKH67 reimplanted cells in the striatum and the corpus callosum. For each slice used for stereology, a model was established by drawing the edges of the section (in red), the ventricles (in blue), the caudate nucleus (in brown), and the putamen (in dark brown) and by pointing the PKH67 cells (in green points). Each 3D reconstruction was done with 11 to 13 models that corresponded to the slices 50 μm thick, separated by 750 μm, and represented the complete slice of the brain where PKH67 cells were found. This brain section is illustrated for the case W808 (A, C). The dark arrows in (A) correspond to the entry of needle tracts. Note that in case W802 (B), some PKH67 cells were found near the needle tract and leading into the cortex (white arrow) where a small hemorrhage took place. Scale bar: 10 mm.

Phenotype of PKH67-labeled cells four months after reimplantation. The first column corresponds to the pictures of the PKH67 fluorescent cells. The second column corresponds to the DAB immunostaining for nestin (B, E), TH (H), and GFAP (K). The third column corresponds to the merged images of the first two first, respectively. In detail, as in all cases, in case W818, PKH67 cells (in green) were observed near the implantation site (in rows A–C, G–I), in the whole striatum (row J–L), and in the corpus callosum (row D–F). In (A–F), PKH67 cells (A, D) still express nestin revealed in brown (B, E) as shown by the merged images (C, F). Note that all nestin-positive cells were PKH67 positive. In (G–L), no green PKH67-positive cells (G, J) express TH (H), as shown in the merged image (I), or GFAP (K), as shown in the merged image (L). Scale bar: 100 μm.
Distribution of Reimplanted Cells in the Brain Structures of the Four Monkeys

To evaluate the phenotype of the autologous transplanted cells, immunohistochemistry revealed that PKH67 cells still expressed neural stem cell markers such as nestin (Fig. 2A–F), but did not express TH (Fig. 4G–I) or GFAP, even in the reimplanted caudate nucleus where a gliosis took place (Fig. 4J–L).
TH immunohistochemistry was quantified by gray level analysis after peroxidase revelation (data not shown), and also following infrared scanning with the LICOR Odyssey system (Fig. 5). Both analyses gave the same results. In the two control low-dose MPTP-treated monkeys, a significant depletion of the TH immunolabeling (48.74± 1.68% in the right caudate and 51.79 ± 2.11% in the left caudate) was observed only in the dorsolateral caudate nucleus compared to normal control (p < 0.001), but not in the rest of the striatum. This decreased TH intensity was less significant in the four reimplanted monkeys, and was limited to 30.89 ± 4.41% in the reimplanted side (p < 0.05) and to 41.37 ± 3.26% in the contralateral side (p < 0.01) (n = 4) (Fig. 5).

TH immunohistology in the striatum 4 months after adult brain cell reimplantation. Infrared intensity of TH immunolabeling was measured on 12 sections covering the striatum. The sections were matched anatomically in each animal, verifying that the cross sections of the substantia nigra were similar in all animals. (A) Example of representative scanned slices from each group (normal, MPTP, and reimplanted). Immunolabeling infrared intensity was measured by drawing a circle (white in part A) on each different substructure of the striatum after infrared scanning with Odyssey software (LI-COR). Only the dorsolateral head of caudate nucleus showed a significant decrease in all six MPTP-treated monkeys. (B) The graph represents the means with standard deviation that were determined from values of three consecutive sections per animal per group. Note that the decrease is more significant in the control MPTP animals than in four reimplanted ones. Scale bar: 1 cm. A one-way ANOVA was performed with Bonferroni multiple comparison post hoc test. Only the right side was reimplanted.
Stereological counts of TH-positive cells in substantia nigra (SN) showed a significant TH neuron depletion in the MPTP control monkey SN (p < 0.01, ANOVA Bonferroni) in both sides; the remaining TH neurons represented 31.49 ± 11.93% compared to the control (n = 2). In the four reimplanted monkeys, TH neurons in the SN almost attained normal values: 106.33 ± 20.26% in the right implanted side and 76.31 ± 11.79% in the left contralateral side (n = 4) (Fig. 6).

TH-ir neurons in substantia nigra, 4 months after adult brain cell reimplantation. (A) Two pictures per group showed the representative TH immunostaining obtained in the rostral and caudal part of the subtantia nigra in the three groups (normal, MPTP, and reimplanted ones). The sections were matched anatomically in each animal, verifying that the cross sections of the substantia nigra were similar in all animals. (B) Three examples of models obtained by pointing the TH-positive cells (as described in Materials and Methods) showed representative TH cell counting for the three groups. Neuronal cells expressing TH were counted in whole substantia nigra on six sections per animal covering anteroposterior extent of substantia nigra. (C) Graphs done with the means that were determined from the values per animal per group. Scale bar: 1 cm. A one-way ANOVA was performed with Bonferroni multiple comparison post hoc test. Only the right side was reimplanted.
GDNF immunohistochemistry were quantified in the area where PKH67 cells were detected in the reimplanted monkeys or in the corresponding area in controls. The average intensity values obtained revealed an important increase of GDNF in reimplanted monkeys with a mean value of 98.9 ± 11.2 arbitrary infrared intensity units compared to normal and MPTP controls with mean values of 52.5 ± 7.3 and 54.8 ± 13.8, respectively (Fig. 7).

GDNF immunohistology 4 months after adult brain cell reimplantation. Infrared intensity of GDNF immunolabeling was measured on four sections covering the area where PKH67 reimplanted cells were observed or in the corresponding area in controls after infrared scanning with Odyssey software (LI-COR). The average values were measured for each animal and the mean was determined from the values per animal per group. Note the increase of GDNF intensity in four reimplanted monkeys compared to normal and MPTP controls.
Discussion
As with human and macaque brain cell culture, we succeeded in producing long-term primary cultures of St. Kitts green monkey brain cells from the gray matter-enriched fraction. In vitro, we demonstrated that these monkey adult brain cells proliferate and present a progenitor/stem cell phenotype in culture over time. The present study aimed to determine in vivo survival, migration, or other effects of these cells, derived from cortical gray matter cells that were cultured for 1 month when reimplanted in the caudate nucleus of MPTP-treated dopamine-depleted monkeys from which the cells were collected (an autograft).
After the biopsy, MPTP treatment induced a partial dopamine depletion. This level of depletion was not sufficient to induce grossly observable parkinsonian motor signs (12).
In a previous study, we demonstrated that cells survive in vivo over time in ibotenic acid-lesioned brains but not in normal ones (9). Therefore, the asymptomatic MPTP model allowed us to investigate the biology of cortical cells in dopamine-depleted striatum without significant morbidity of the animals. A previous study showed a loss of 70% of TH-immunolabeled cells in the substantia nigra in a group of six monkeys 20 days after an identical MPTP treatment (19). The depletion of dopamine [29.6 ± 4.6% (19–44) of control in caudate and 42.0 ± 4.9% (28–79) in the putamen] was also observed following long-term treatment (1 year after MPTP treatment) in the striatum of a group of nine monkeys classified asymptomatic after MPTP treatment (12). These results showed the significant impact of MPTP on the striatal level of dopamine even if there are no parkinsonian signs (12, 19), and many investigators have agreed that there is a threshold of 75–85% dopamine depletion that is necessary for motor signs to appear (1, 21).
The cells were stereotactically reimplanted into the right caudate nucleus at the dorsolateral level that was previously described to be the only affected area in asymptomatic MPTP monkeys for TH immunolabeling (12). Just prior to reimplantation, cells were stained for tracking with PKH67 green fluorescent viable dye that irreversibly binds to viable cell membranes and does not label resident host cells as already described for intracerebrally transplanted cells (17).
In the reimplanted brain, the stereological counting revealed that autologous reimplanted cells survived at an impressively high rate (50%) 4 months after reimplantation. In the other approaches, such as fetal progenitor cell grafting, it has been shown that a large number of transplanted cells (up to 70–90%) die after transplantation (6, 51), even if antideath strategies were used to improve this survival (10, 33, 34, 45, 48). The impressive rate of the autologous brain cell survival could be related to the fact that acute or chronic rejection seems less extensive for brain cell autotransplantation than for the typical allografts that constitute fetal tissue or grafts from stem cell lines (6, 41, 48, 51).
Furthermore, these cells migrated antero-posteriorly in the reimplanted striatum but also to the contralateral striatum through the corpus callosum. The large majority of the reimplanted cells were found in the dorsolateral part of the caudate nucleus, which is the most TH-depleted area of the striatum, as previously described (12) and as we demonstrated in the two control MPTP monkeys. The migration observed in the four reimplanted monkeys is consistent with the migratory ability of neural stem cells demonstrated in adult rat brain (13, 15) and in MPTP monkey brain transplanted with human neural stem cells (4). The important migration and the presence of cells in the corpus callosum could also be due to the injection target at the superior part of the caudate nucleus. Our previous studies also showed that autologous monkey brain cells migrate into the lesioned motor cortex (9). We hypothesize that this migratory ability is related to the fact that adult cortical brain cells express DCX (Fig. 2G), the migrant neuroblast marker, and probably respond to chemoattractant agents promoting migration (4).
The fact that no PKH67 reimplanted cells expressed TH indicates that they do not participate in cell replacement of dopamine neurons (which are not normally found in this target region anyway, but project there from the SN); however, they seem to have a neuroprotective effect, and may produce some growth factors. Indeed, by using Raybio™ human cytokine array I (RayBiotech, Inc.), the presence of neurotrophic factors such as GDNF or BDNF was shown in medium conditioned by adult cortical brain cells (unpublished data), which may contribute to a bystander effect. This hypothesis is supported by the fact that the number of TH neurons in the substantia nigra is close to normal values and the decrease of TH intensity is less important in the dorsolateral caudate nucleus when autotransplantation has been performed. It is also suggested by the immunodetection of a higher level of GDNF in the area where reimplanted cells were found compared to controls. Moreover, the absence of PKH67 cells in the substantia nigra suggests that this neuroprotective effect is provided by a distal retrograde effect.
This positive impact on nigral dopaminergic neurons suggests two interpretations for future investigations: either the cells present a neuroprotective effect at the level of the striatal dopaminergic processes that influence the nigral dopaminergic neurons, or the cells have the ability to stimulate endogenous dopaminergic neurogenesis.
Taken together with our previous studies (7–9), these results confirm that adult brain cells can be obtained, cryopreserved, and kept in culture before being reimplanted in the donor, and survive in vivo at least 4 months. In a mild dopamine-depleted MPTP monkey model, these cells have been demonstrated to have a neurotrophic effect on the TH neurons in the substantia nigra and their processes in the dorsolateral caudate nucleus over time.
These encouraging results would support studies of the therapeutic role of these cells in a symptomatic model of Parkinson's disease and to further explore their potential mechanisms. These results and their potential clinical impact should encourage the further development of the field of autotransplantation in the central nervous system.
Footnotes
Acknowledgments
This work was supported by grants from Foundation Swiss Parkinson, Swiss National Funds (No. 3100AO-103924), Axion Research Foundation, St. Kitts Biomedical Research Foundation. and Lausanne University Hospital Neurosurgery Department. We express our gratitude to Laurence Grollimund, Marie-France Hamou, and the staff of the St. Kitts Biomedical Research Foundation for their expert technical assistance. We thank John Elsworth for advice and data regarding dopamine depletion after MPTP treatment in St. Kitts monkeys.
