Abstract
Successful pancreas preservation during storage in oxygenated perfluorodecalin (PFD) is mainly related to oxidative ATP generation during storage. Increasing the storage temperature would accelerate this process essential for resuscitation of ischemically damaged pancreatic tissue. The present study aimed at comparing islet isolation outcome from adult pig pancreata preserved in oxygenated PFD by means of a one-layer method during storage on ice or at 20°C. Resected pancreata were intraductally flushed with cold UW solution and promptly processed (n = 6) or stored for 3 h in continuously oxygenated PFD at 4°C (n = 5) or 20°C (n = 7). Prior to digestion-filtration pancreata were intraductally injected with UW supplemented with Serva collagenase NB8 and neutral protease. Islet quality assessment determined viability, glucose stimulation index, mitochondrial activity, intracellular ATP content, and transplant function in diabetic nude mice. Pancreata oxygenated for 3 h at 20°C yielded islet numbers similar to organs oxygenated at 4°C. Compared to a storage temperature of 20°C, preservation at 4°C reduced islet ATP content (p < 0.05) as well as islet viability (p < 0.05). Nevertheless, PFD storage at 20°C decreased insulin response to glucose compared to unstored pancreata (p < 0.05). In contrast to unstored pancreata or cold-stored organs, transplantation of islets isolated after oxygenation at 20°C was characterized by an early loss of transplant function in 50% of recipients (p < 0.05). The present study demonstrates that PFD storage at 20°C enhances islet ATP synthesis within a short period of oxygenation but deteriorates islet function. We conclude that the present data reflect an equilibration between reduced depression of metabolic activity resulting in damage of islets and temperature-stimulated acceleration of ATP synthesis. Future studies are required to adjust the optimum storage temperature for pancreas oxygenation in different species.
Introduction
The broad application of islet allotransplantation for the treatment of type 1 diabetic patients is mainly restricted by the shortage of suitable human donors (35, 38). The disproportion between demand on islets and availability of human donor pancreata will further increase in the near future (1). The use of a xenogeneic islet source such as the pig would offer an attractive solution for the upcoming problems (12, 15) as shown by long-term survival of adult and neonatal pig islets transplanted into immunosuppressed primates (11, 16). However, before clinical islet xenotransplantation becomes reality, stringent regulatory guidelines have to be fulfilled aiming on biosecurity and minimization of the risk for public health (40). The costs for breeding, maintenance, and monitoring of pathogen-free and/or genetically modified pigs in biosecure, fully enclosed facilities run by educated and trained staff has not been calculated yet but will easily reach the level that is known for human pancreas procurement (32). Therefore, the limited availability of cost-intensive pathogen-free pigs requires the processing in centralized isolation facilities experienced in producing high-quality pig islets obtained from juvenile or adult donors.
Compared to pancreata of other species, such as dog or human, the pig pancreas is characterized by a high sensitivity against hypoxic conditions (17, 24, 44). Therefore, strategies are required to minimize ischemically induced damage during shipment of explanted pig pancreata to distant isolation laboratories. In previous studies we demonstrated that pancreas storage in oxygenated perfluorodecalin (PFD) is superior for subsequent islet isolation compared to simple immersion in University of Wisconsin solution (UWS). The higher efficiency of oxygenated PFD, to protect cellular integrity during cold storage, can be attributed to the oxygen-stimulated synthesis of ATP in the Krebs cycle (22). The time that is needed to reach a critical cellular concentration of ATP is determined by the extent of ATP consumption on the one hand and the magnitude of ATP generation on the other (21, 23). The Krebs cycle contributes to the highest extent to the oxidative ATP synthesis but is more susceptible toward hypothermia than the cytosolic pathways of nonoxidative glucose metabolization (13). It could be demonstrated in canine pancreas autotransplantation that an increase of the storage temperature to 20°C allows significant reduction of the oxygenation time required for successful pancreas resuscitation after ischemically induced damage (27). An acceleration of this process would enhance the flexibility in the logistics for pancreas procurement and subsequent islet isolation. Because data about the effect of pancreas storage in oxygenated PFD at room temperature on subsequent islet isolation are still lacking, the present study aimed at comparing the outcome of islet isolation from PFD-preserved pancreata subjected to storage at 4°C or 20°C.
Materials and Methods
Donor Animals and Organ Procurement
All donor sows (≤24 months) were obtained from a central breeding station and slaughtered as retired breeders in a local abattoir after mechanical induction of cerebral death. The splenic lobe of the glands was dissected ex situ after 12–15 min of warm ischemia time. Subsequently, the pancreata were intraductally flushed with 2 ml/g of cold UWS (33), soaked into cold saline (Braun, Melsungen, Germany) and transported within 30 min to the isolation laboratory.
Organ Storage
During storage the organs were preserved by the one-layer method (OLM) as previously described (6). Briefly, trimmed and intraductally flushed pancreata were placed into a modified plastic container (30) and completely immersed in 600 ml of PFD (Novaliq GmbH, Heidelberg, Germany) initially precharged for 10 min with 100% oxygen at a flow rate of 2000 ml/min. Thereafter, the oxygen flow rate was adjusted to 300 ml/min. During 3 h of OLM storage the temperature was adjusted to 4°C (n = 5) or 20°C (n = 7). Storage time started after onset of pancreas oxygenation and did not include transportation from the abattoir to the isolation facility.
Islet Isolation and Purification
Subsequent to procurement or organ storage, pancreata were intraductally distended with 4.4 PZ-U of collagenase NB8 (Serva Electrophoresis, Uetersen, Germany) dissolved in 1 ml of cold UWS per gram trimmed pancreas weight. After preliminary experiments activity of neutral protease NB (Serva Electrophoresis) was adjusted to respectively 1.1 or 0.9 DMC-U/g for pancreata either immediately processed (n = 6) or subjected to storage at 4°C (n = 5) or 20°C (n = 7).
Isolation and purification were performed as previously described (8) with minimal modifications regarding the digestion temperature adjusted between 30°C and 37°C. Purified islet fractions were pooled for subsequent characterization in supplemented CMRL 1066 (Biochrom, Berlin, Germany).
Islet Characterization
Islet yield of dithizone-stained samples was evaluated in quadruplicate before and after purification. Islets were converted to islet equivalents (IEQ) with an average diameter of 150 μm (37). After purification total tissue volume was precisely measured in a conical glass tube with a fine-graded scale, and related to the calculated volume of counted islets assuming that porcine islets are ellipsoids (14, 45). Additionally, purity was conventionally determined in dithizone-stained samples by visual estimation of the surface of stained particles (islets) in relation to nonstained particles. Morphological integrity of islets was estimated using a fragmentation index (FI) that was calculated as the ratio between islet number (IN) and IEQ. After quantification, aliquots of 500 IEQ were assessed for intracellular insulin content, ATP content, and mitochondrial activity as described previously in detail (6, 7). Mitochondrial activity was determined by conversion of the tetrazolium compound 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS, Promega, Mannheim, Germany) into formazan, which was measured at 490 nm and expressed as optical density (OD) per 1000 IEQ. The luminometric quantification of the intracellular ATP content of freshly isolated and overnight-cultured islets was performed utilizing a commercially available assay (Roche, Mannheim, Germany).
After overnight culture at 37°C, in vitro function of quantified islet aliquots was determined after 120 min of static incubation in CMRL 1066 containing either 2.8 or 20 mM glucose. Insulin release was measured utilizing an enzyme immunoassay specific for human insulin (DRG, Marburg, Germany) and cross-reactive to porcine insulin. Glucose-stimulated insulin release was expressed as stimulation index (SI) and calculated as the ratio of stimulated to basal insulin release (20). Islet viability was simultaneously examined by the trypan blue exclusion assay (18).
In vivo function of overnight-cultured islets was assessed in NMRI nude mice (Harlan, Hanover, Germany) rendered diabetic by a single IV injection of 240 mg/kg streptozotocin (Sigma, Deisenhofen, Germany) 4 days prior to transplantation beneath the kidney capsule. Blood samples were taken from the tail vein of recipients and analyzed utilizing a Beckman glucose analyzer (Beckman Instruments, Fullerton, CA, USA). Pretransplant, the nonfasting serum glucose levels of all recipients exceeded 350 mg/dl. After preliminary experiments islet mass was adjusted to 3000 and 4000 IEQ for immediately processed and OLM-stored pancreata, respectively. After transplantation nonfasting serum glucose levels <200 mg/dl were defined as normoglycemic and considered as graft function. Twenty-six days posttransplant nephrectomy of graft-bearing kidneys was performed to demonstrate immediate return of hyperglycemia.
Data Analysis
All values were expressed as median (range). Comparisons of data were carried out by Mann-Whitney test following Kruskal-Wallis test. Multiple comparisons were corrected utilizing the Bonferroni method. Paired samples were analysed utilizing the Wilcoxon test. Graft function (time of normoglycemia) was analyzed utilizing the Log-rank test. Significance was expressed as p-value and considered for p < 0.05. Values of p > 0.05 are termed nonsignificant (NS).
Results
Islet Isolation Outcome
The donor characteristics of 18 processed retired breeders were not significantly different between the experimental groups with regard to the donor age, carcass, and pancreas weight (data not shown). The digestion parameters were almost similar in all experimental groups as reflected by the recirculation time of 23 (21–28), 26 (22–32), and 21 min (18–29) (NS) or the total dissociation time of 55 (48–64), 50 (48–53), and 47 min (35–56) (NS) as determined for pancreata either immediately processed or stored at 4°C or 20°C, respectively.
The isolation outcome is presented in Table 1. No significant differences were found prepurification with regard to islet yield (IEQ/g) and morphological integrity determined as fragmentation index (FI). Compared to unstored pancreata [3899 IEQ (1227–6200)], purified islet yield was slightly decreased after OLM storage at 4°C [2640 IEQ (2212–3974), NS] and 20°C [2448 IEQ (1936–6717)]. The FI and recovery of purified islets were not influenced by the storage temperature in relation to unstored organs. A minor reduction in islet volume-related purity was observed after storage at 4°C [59% (48–80%)] and 20°C [64% (43–77%)] in comparison to islets isolated from unstored pancreata [82% (42–111), NS]. Because visually estimated purity was higher than 95% in all experimental groups, we assumed that single cells or fragments of islets rather than exocrine particles contribute to the relative increase of puried tissue volume observed after OLM storage.
Isolation Variables
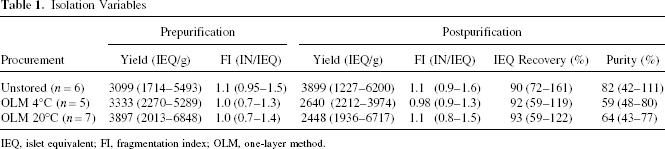
IEQ, islet equivalent; FI, fragmentation index; OLM, one-layer method.
Islet Characterization
The data of islet characterization are presented in Table 2. Assessment of islet viability revealed a small but significant difference between islets from pancreata subjected to OLM storage at 4°C [96.5% (95.0–97.5%), p < 0.05] and organs either unstored [100% (99.5–100%)] or preserved at 20°C [99.7% (97.3–100%)]. With respect to mitochondrial activity the storage temperature did not affect islet metabolism when compared to unstored pancreata. As expected, a strong effect of storage temperature was found with regard to ATP synthesis. Compared to storage at 4°C [67 ng/1000 IEQ (36–433), NS vs. unstored] a significant increase of the intraislet ATP content was measured after storage at 20°C [964 ng/1000 IEQ (211–2326), p < 0.05 vs. 4°C, NS vs. unstored]. After overnight culture, pancreata preserved at 4°C were characterized by a significant increase in islet ATP content that was equivalent to 154% (128–219, p < 0.05) compared to preculture. No significant increase in ATP content was measured after overnight culture in islets isolated from either unstored organs [76% (36–227) or pancreata stored at 20°C (152% (58–321)].
Islet Characterization

IEQ, islet equivalent; OD, optical density; OLM, one-layer method; SI, stimulation index.
p < 0.05 versus OLM 4°C.
p < 0.05 versus OLM 20°C.
p < 0.01 versus OLM 20°C.
In contrast to OLM storage at 4°C [640 mU/1000 IEQ (314–991), NS], a preservation temperature of 20°C decreased the intracellular insulin content from initially 715 mU/1000 IEQ (615–1339) in islets from unstored organs to 448 mU/1000 IEQ (158–744, p < 0.05). In correspondence with this observation, insulin release during static glucose incubation was significantly reduced after OLM storage at 20°C in terms of both basal [8.1 mU/1000 IEQ/2 h (6.7–29.0), p < 0.05] and stimulated insulin release [12.5 mU/1000 IEQ/2 h (9.3–35.0), p < 0.01] compared to islets from unstored pancreata. As a consequence, the stimulation index was significantly reduced after OLM storage at 20°C as well [1.4 (0.9–1.9), p < 0.05 vs. unstored]. When 4°C was selected as storage temperature the reduction of neither basal [22.0 mU/1000 IEQ/2 h (7.7–118.0)] and stimulated insulin release [35 mU/1000 IEQ/2 h (11–197)] nor stimulation index [1.6 (1.5–1.9)] reached significance in comparison to islets from unstored pancreata.
The detrimental effect of storage at 20°C on islet function was finally confirmed in diabetic nude mice. As shown in Figure 1, these islets were characterized by a final transplant function in 50% of recipients until nephrectomy of graft-bearing kidneys was performed. In contrast, implantation of islets from cold-stored pancreata resulted in immediate and sustained reversal of hyperglycemia in 92% of recipients (p < 0.05 vs. OLM 20°C).

Cumulative graft function of pig islets isolated from pancreata (n = 3) either immediately procured (unstored, open circles, n = 12) or subjected to prior oxygenation utilizing the one-layer method (OLM) at 20°C (OLM 20°C, filled squares, n = 10) or 4°C (OLM 4°C, filled triangles, n = 12). Islets were transplanted beneath the kidney capsule of STZ-treated NMRI nude mice. Graft removal through nephrectomy (Nx) was performed as indicated at day 26 posttransplant.
Discussion
The established protocols for pancreas preservation aim at maximum reduction of cell metabolism and decrease of enzyme activity during ischemia by means of hypothermic organ perfusion with UWS followed by storage at 0–4°C (46). Although UWS contains precursors for ATP synthesis, this preservation solution does not prevent the intracellular depletion of ATP during long-term storage of pancreatic tissue on ice (22). This observation correlates with the increased susceptibility of the oxidative ATP production toward hypothermia compared to nonoxidative pathways of glucose metabolization in islets (13).
Only very few studies exist so far comparing different temperatures for organ storage prior to pancreas transplantation or islet isolation. Among these, a study in rats revealed that a moderate increase of the storage temperature from 4°C to 7–10°C significantly enhances islet yield isolated after 24 h of UWS incubation performed without oxygenation. A further increase to 15°C resulted in the lowest islet recovery observed among all experimental groups (25). In contrast, initial experiments in dogs demonstrated that 5 h of oxygenation at 20°C resuscitates ischemically damaged pancreata prior to autotransplantation in opposite to storage at 4°C (27).
The present study demonstrates that oxygenated PFD enhances significantly islet ATP production during 3-h storage of porcine pancreas if the storage temperature is enhanced from 4°C to 20°C. In spite the enormously enhanced ATP synthesis, islets isolated after 20°C storage were characterized by a reduced in vitro function when compared to islets from cold-stored pancreata. In agreement with previous studies in humans, islet culture resulted in an enhancement of the initial ATP content in islets partially depleted from ATP during cold storage (5). Although other determinants of a successful islet isolation, such as yield, viability, and morphological integrity, were excellent after oxygenation at 20°C, the cure rate in diabetic nude mice was reduced by nearly 50% in this experimental group. The observation that the ATP content in pig islets does not correlate with posttransplant islet function may be related to the specific sensitivity of the pig pancreas toward ischemia, as observed in other studies (3, 17).
The major attribute of pig islets is their structural integration in the exocrine pancreas, which almost exclusively depends on protease-sensitive cell-to-cell adhesion and on marginal islet encapsulation by collagen (43). Temperature-activated proteases, released from autolytic acinar cells, can therefore easily initiate extensive damage on endocrine tissue (34). Due to the specific structure of pig islets formed of subunits that are composed of a β-cell core and α-cells localized to the periphery (10), the damage of glucagon-containing cells during autolysis has implications for the insulin secretory capacity of islets isolated from pigs (4). We hypothesize that the data of the present study reflect partially an equilibration between reduced depression of endogenous enzyme activity resulting in islet damage, and temperature-stimulated acceleration of ATP synthesis, which has protective effects on islet integrity. This may explain the excellent islet yields isolated after 20°C storage on the one hand, and the reduced in vitro and posttransplant function on the other.
Another explanation for our conflicting results in terms of yield and posttransplant function of islets is the finding in dogs that pancreas oxygenation at 20°C has detrimental effects on the viability of endothelial cells resulting in failure of autotransplanted pancreata after 8 h of storage (28, 29). Because donor-derived intraislet endothelial cells seem to contribute significantly to islet revascularization in allogeneic and xenogeneic models, a temperature-induced deterioration of islet engraftment can not be excluded for pig-to-nude mouse transplantation (26, 31).
Our data may additionally contribute to the actual discussion whether pancreas oxygenation by means of precharged PFD has a beneficial effect at all in comparison to simple storage in UWS (9, 19). According to calculations made by Pappas et al.'s (36) and Avgoustiniatos et al.'s (2) utilizing the pig as a model, the penetration of oxygen into pancreatic tissue is limited to a distance of only 1 mm, suggesting that only a marginal fraction of the pancreatic volume is efficiently oxygenated. However, our observation that the ATP content in islets isolated after pancreas storage at 20°C exceeds the ATP level in islets retrieved from cold-stored or unstored pancreata is incompatible with the van't Hoff equation and rather indicates that an ATP production of this magnitude must be additionally stimulated by oxygen. Assuming that the previous experiments (2, 36) were performed with native, untreated pancreata, we hypothesize that the significant edematous distension of the soft porcine pancreatic tissue by means of intraductal UWS perfusion, as performed in the present study, facilitates the penetration of oxygen into the pancreatic core. Furthermore, other studies indicate that the application of precharged PFD is less effective than continuous oxygen bubbling performed for 3–9 h subsequent to arrival in the laboratory as demonstrated in human pancreata by increased ATP levels and improved function of isolated islets (39, 41, 42).
In summary, the present study revealed that a storage temperature of 20°C enhances the ATP production in islets isolated from PFD-stored pig pancreata during 3 h of oxygenation but seems to be detrimental for islet function in vitro and after transplantation. Considering that a narrow species-specific temperature range exists that allows preservation of pancreas viability during organ storage, a further refinement is required for future experiments in order to adjust the optimum storage temperature for pancreas oxygenation in different species.
Footnotes
Acknowledgment
This work was supported in part by the Juvenile Diabetes Research Foundation International (Grant 1-2007-198).
