Abstract
Currently there are no effective treatments targeting residual anatomical and behavioral deficits resulting from stroke. Evidence suggests that cell transplantation therapy may enhance functional recovery after stroke through multiple mechanisms. We used a syngeneic model of neural transplantation to explore graft–host communications that enhance cellular engraftment. The medial ganglionic eminence (MGE) cells were derived from 15-day-old transgenic rat embryos carrying green fluorescent protein (GFP), a marker, to easily track the transplanted cells. Adult rats were subjected to transient intraluminal occlusion of the medial cerebral artery. Two weeks after stroke, the grafts were deposited into four sites, along the rostro-caudal axis and medially to the stroke in the penumbra zone. Control groups included vehicle and fibroblast transplants. Animals were subjected to motor behavioral tests at 4 week posttransplant survival time. Morphological analysis demonstrated that the grafted MGE cells differentiated into multiple neuronal subtypes, established synaptic contact with host cells, increased the expression of synaptic markers, and enhanced axonal reorganization in the injured area. Initial patch-clamp recording demonstrated that the MGE cells received postsynaptic currents from host cells. Behavioral analysis showed reduced motor deficits in the rotarod and elevated body swing tests. These findings suggest that graft–host interactions influence the fate of grafted neural precursors and that functional recovery could be mediated by neurotrophic support, new synaptic circuit elaboration, and enhancement of the stroke-induced neuroplasticity.
Keywords
Introduction
Stroke is one of the leading causes of death and long-term disability in the world. Each year approximately 750,000 people in the US experience a new or recurrent stroke. Efforts to develop effective therapeutic approaches, such as thrombolytics, have targeted acute stroke with typically short peri-infarction time windows. Although acute thrombolysis has significant impact on the management of stroke, fewer than 5% of acute patients receive this treatment due to the restricted time window of eligibility and other exclusion criteria. The vast majority of stroke patients experience progression of ischemia and neurological deficits.
Cellular therapy offers a promising approach in regenerative medicine to repair diseased or injured brain areas through cell replacement or functional enhancement. Recent studies have demonstrated the efficacy of cell transplants in improving behavioral deficits in rodent models of stroke. Delivery of stem cells derived from bone marrow, cord blood, and from the central nervous system indicate that cellular grafts can improve motor function via mechanisms involving cell replacement, neurotrophic factors, anti-inflammatory-mediated neuroprotection, angiogenesis, and endogenous neurogenesis (3, 27). We and others have previously used xenotransplants of human-derived neural cells in the stroke model to demonstrate engraftment, migration, and functional improvement (5, 9, 11, 13, 15, 16, 24, 33). Xenotransplantation is necessary in preclinical research and development to test the safety and efficacy of therapeutic human cell lines. However, while species cross-interaction of cytokines occurs, there could be limitations in the bidirectional graft–host interactions that are species specific. Syngeneic transplantation could be informative in regards to basic mechanisms of transplant engraftment.
The median ganglionic eminence (MGE) of the embryonic basal forebrain produces multiple subpopulations of migrating neuronal precursors destined to populate the striatum, neocortex, pallidum, and hippocampus (14, 19). MGE cells migrate dorsally from the pallidal primordium through the striatal anlage and into the cortical plate where they integrate and differentiate into local GABAergic interneurons (2, 22). The migratory and engraftability characteristics of the MGE progenitors in the adult brain (1, 29) suggest that these cells could provide useful data in a disease model. In the present study, we used the MGE neural precursors derived from E15 EGFP-transgenic rats in a syngeneic transplant model to analyze graft-host interactions, dispersion, differentiation, and behavioral recovery in the middle cerebral artery occlusion (MCAO) model of stroke.
Materials and Methods
Cell Preparation and Transplantation Surgery
All animal experiments were conducted according to the National Institute of Health (NIH) guidelines and approved by the IACUC. Sprague-Dawley (SD) adult rats (275–310 g, Charles River Laboratories, Wilmington, MA) were subjected to 1.5 h of transient MCAO by intraluminal suture, as previously described (9). The MGE cells were dissected out from E15 transgenic SD rat fetuses carrying GFP transgene (Green Rat CZ-004; kindly provided by Dr. Masaru Okabe, Osaka University, Osaka, Japan). Briefly, the E15 rat fetuses were collected into sterile phosphate-buffered saline (PBS) and the brains were extracted. The floor of the forebrain was then exposed by making an incision across the dorsomedial surface of the cerebral hemispheres. The dorsal-most aspect of the MGE was dissected and mechanically dissociated with a fire-polished Pasteur pipette in sterile PBS. Two weeks after the ischemic lesion, 2 μl of the MGE-derived primary cell suspension, at a concentration of 50,000 cell/μl, were stereotaxically transplanted into four sites within the lesioned striatum and cortex (n = 11) at the following coordinates in mm: AP/ ML/DV: +1.0/+3.2/–5.0, −2.5; +0.5/+3.0/ −5.0, −2.5; −0.5/+3.0/ −5.0, −2.5; −1.0/+3.5/ −5.0, −2.5. For controls, we used rats subjected to ischemia and transplanted either with fibroblasts derived from E15 GFP-Tg embryos (n = 5) or with the vehicle (n = 8). All animals underwent baseline motor behavioral assessment before and 2 weeks after the ischemic lesion, and 4 weeks after cell transplantation.
Behavioral Tests
An accelerating rotarod test was performed to measure motor function in the rats. The rats were first trained to stay on the rotating spindle of the rotarod in three sessions with three trials per session at the beginning and a single trial after the rat could stay more then 60 s on the spindle. The acceleration of the rotarod was automatically set to increase from 4 to 40 rpm within 5 min. Trials automatically ended when the animals fell off the spindle. The elevated body swing test (EBST) was used to assess body asymmetry after MCAO and evaluated quantitatively, as previously described (4). Animals were suspended by tail and the frequency of initial head swings contralateral to the ischemic side was counted in 20 trials and represented as percent of total. Ischemic rats with more than 75% biased swing were used in the study.
BDA Injections
Three weeks after cell transplantation, three randomly selected animals from each of transplanted and vehicle-treated groups were anesthetized and placed in the strereotaxic apparatus. After craniotomy, 0.5 μl of biotinylated dextran amine [BDA, 10,000 molecular weight (MW), Molecular Probes, Eugene OR; 10% w/v solution in sterile PBS] was injected stereotaxically into the sensorimotor cortex opposite to the stroke lesion site at the stereotaxic coordinates: AP: +0.5 m, ML: 2.5 mm, and DV: −1.5 mm. The scalp was then closed and the animal returned to its cage. Animals were sacrificed 1 week after BDA injection.
Electrophysiology
Coronal brain slices (250 μm) were prepared from stroked rats implanted with MGE cells, 4 weeks after transplantation. Slices were allowed to recover in a submerged holding chamber containing artificial cerebrospinal fluid (ACSF). Whole-cell voltage-clamp and current-clamp recordings were obtained from visually identified GFP-positive MGE cells (n = 30). Events were analyzed using Minianalysis software (Synaptosoft, Decatur, GA) with detection parameters set at greater than 5 pA amplitude and less than 3 ms rise time.
Immunocytochemistry
Animals were killed 4 weeks after transplantation by transcardiac perfusion with PBS followed by 4% paraformaldehyde. The brains were cryoprotected in an increasing gradient of sucrose solution (10%, 20%, and 30%) and cryostat sectioned at 40 μm. Sections were processed for immunocytochemistry with the following antibodies: anti-GFP (1:1000; AVES); anti-β-tubulin class III (1:100; Covance); anti-GAD (GAD67; GAD65-67; 1:1000), anti-glutamate (1:5000); anti-Pva (1:1000); anti-Clb28K (1:1000) (all from Sigma); anti-NeuN (1:100); anti-GFAP (1:1000); anti-GC (1:250) (all from Chemicon); anti-synaptophysin (1:100, Zymed); anti-doublecortin (DCX, 1:250, SantaCruz Biotech). Indirect immunofluorescence microscopy was carried out. Sections were rinsed in PBS for 3 × 10 min, then incubated overnight with the appropriate primary antibodies for multiple labeling. Secondary antibodies raised in the appropriate hosts and conjugated to FITC, RITC, or AMCA chromogenes (Jackson ImmunoResearch) were used. Sections were counterstained with the DNA marker 4′,6-diamidine-2′-phenylindole dihydrochloride (DAPI). Positive and negative controls were included in each run. Immunostained sections were coverslipped using fluorsave (Calbiochem). Fluorescence was detected, analyzed, and photographed with a Zeiss LSM550 laser scanning confocal photomicroscope. For GFP immunohistochemistry, sections were rinsed in PBS, quenched in 3% H2O2 for 20 min, blocked in serum solution with 0.1% Triton-X 100 in PBS for 30 min, and incubated with primary antibody and subsequently with biotinylated secondaries (1:300, Vector Labs). The antibody binding was visualized with streptavidin horseradish peroxidase and DAB chromogen with nickel. Sections were then coverslipped and examined under a light microscope. Visualization of the immunohistochemical staining and the stereological analysis were carried out using a Nikon Eclipse TE 300 microscope.
The cell survival was estimated for each animal by counting GFP+ cells using an unbiased stereological method aided by Stereo-Investigator software (Micro-BrightField, Inc.) as previously described (9). Double labeling of the GFP+ grafted MGE cells with all phenotypic markers was determined by random sampling of more than 100 cells per marker for each animal and scoring first for GFP, followed by DAPI nuclei and then the phenotypic marker. The double labeling was always confirmed in x–z and y–z cross-sections produced by the orthogonal projections of z-series. Two experimenters blinded to condition were used for quantitative analysis.
BDA-labeled axon terminals were detected using Cy3-conjugated streptavidin (1:200, Jackson ImmunoResearch laboratories) or histochemically using Vectastain Elite Kit (Vector Labs) for peroxidase reaction with diaminobenzidine. Sections were counterstained with toluidine blue (0.1% in 1N HCl, LabChem Inc.), rinsed with water and then dried and coversliped with DPX mounting medium (Electron Microscopy Sciences).
For the quantitative analysis of synaptophysin and BDA-labeled axon terminals, three sections approximately 500 μm apart were selected from each experimental case. The lateral ventricle, corpus callosum, striatum, and anterior commissure were used as landmarks for selecting sections, according to the rat stereotaxic atlas (23). The fiber terminals were measured in each scanned field (100 μm2 surface area for synaptophysin, 400 μm2 for BDA) using NIH ImageJ Analysis Software (version 1.39 software, http://rsbweb.nih.gov/ij/). The background from each image was adjusted by thresholding the image so that only legitimate fibers and puncta remained above threshold. ImageJ's “Particle Analyzer” plug-in was used to identify and measure the number of BDA fiber terminals. The number, mean area, mean minimum, and maximum pixel intensities for each image were recorded. The accuracy of the ImageJ's counts was confirmed by manual counting. The results from the computerized method validated the manual counts. The total number of BDA-labeled cell bodies at the injection site is critical for a comparative measurement of patterns of connections between cases. In the same sections used for axon terminal quantification, the total number of BDA+ cells at the injection site was determined and used to normalize the total number of BDA-labeled axon terminals for each experimental case. Two experimenters blinded to conditions performed the quantitative analysis.
Ultrastructural Analysis of Engrafted Cells
Preembedding immunohistochemistry was performed on 80-μm vibratome sections. Immunohistochemistry for GFP with control runs was carried out as described above. Sections were then fixed in 4% PFA+ 0.02% glutaraldehyde for 15 min at RT. Samples were washed three times with PBS and incubated in 1% osmium tetroxide for 60 min at 4°C. After further washing with water the sections were incubated overnight in an aqueous solution of uranyl acetate (1%), dehydrated through graded alcohols and the propylene oxide for 15 min, and embedded in EMbed 812 resin (Electron Microscopy Sciences). Ultrathin sections were analyzed with Jeol JEM-1230 transmission electron microscope.
Statistical Analysis
Outcome measurement for each experiment was reported as mean ± SEM. All data were analyzed using SPSS 11 for Mac OS X (SPSS Inc.). Significance of intergroup differences was performed by applying Student's t-test where appropriate. The one-way ANOVA analysis was used to compare groups. Differences between the groups were determined using Bonferroni's post hoc test. A value of p < 0.05 was considered to be statistically significant.
Results
Engraftment of the MGE Cells
Four weeks after transplantation, 16.5 ± 3.7% of the grafted cells survived, expressed the GFP reporter gene, migrated toward the stroke-lesioned regions (Fig. 1A–F) and dispersed in 3.6 ± 1.3 mm3 volume that represented 16.4 ± 6.2% of the host striatum and cortex. In the cortex, the transplanted MGE cells displayed bipolar morphologies and were radially oriented. Others acquired morphologies similar to native cortical neurons and were frequently arranged in colinear orientations reminiscent of the columnar cytoarchitecture of the cortex (Fig. 1B). In the striatum, many MGE cells acquired neuronal morphologies, with complex dendritic arborization and axon-like single thin processes emanating from the cell body (Fig. 1C). Others showed a typical medium aspiny interneuron-like with perykarya ranging in size from 10 to 15 μm in diameter. The neuronal population expressed β-tubulin class-III (13 ± 4%; n = 732 cells analyzed), NeuN (19 ± 6%; n = 822 cells analyzed) (Fig. 2), and doublecortin (DCX) (5 ± 1%; n = 647 cells analyzed) (Fig. 2B). Less than 1% expressed the astrocytic marker glial fibrillary acidic protein (GFAP; n = 674 cells analyzed), and the oligodendroglial markers galactocerebroside (GC; n = 692 cells analyzed) and NG2 (n = 771 cells analyzed) (Fig. 2C–E).

Transplanted MGE cells disperse and acquire various morphologies. Schematic drawing of a frontal section through the striatum (A) illustrating the distribution and dispersion of GFP+ grafted cells. Shaded area in (A) and (D) represent the extent of focal ischemia. (B, C; insets in A) Photos of grafted MGE cells taken from the cortex (Ctx) and the striatum (Str), respectively. (E, F; insets in D) Dispersion of the MGE cells (E) and a fascicle of MGE cell projection fibers in the striatum (F). Scale bars (B, C) 20 μm; (E) 100 μm; (F) 10 μm.
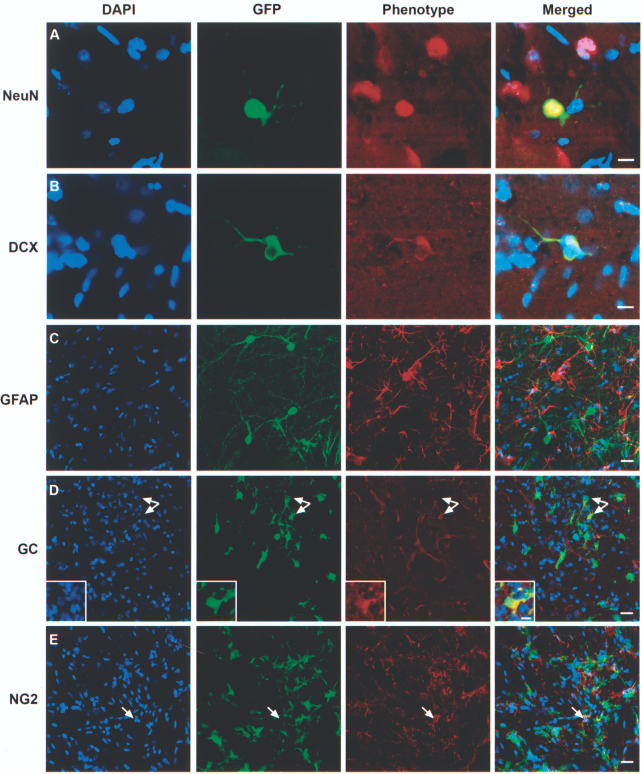
Differentiation of the grafted cells. (A, B) Grafted MGE cells expressed NeuN, doublecortin, and rarely did they express GFAP (C) and GC (D). Arrows in (D) show examples of grafted MGE cells expressing GC; inset shows high magnification of a GFP/GC+ cell. (E) An example of endogenous NG2 cell negative for GFP. Scale bars: (D, E) 20 μm; (A–C, inset in E) 10 μm.
Molecular Properties of Grafted MGE Cells
Immunostaining for glutamic acid decarboxylase (GAD67), a marker for GABAergic neurons, demonstrated that less than 2% (n = 867 cells analyzed) of grafted MGE cells expressed this isoform (Fig. 3C), while 68.1 ± 9.2% (n = 791 cells analyzed) expressed the GAD65 isoform. The later population were located in the transplant zone (Fig. 3A, B) while the GAD67+ cells were located outside the graft zone. Immunostaining for glutamate revealed that 15 ± 6% (n = 651 cells analyzed) of the grafted cells in the striatum (Fig. 3D, E) and 40 ± 8% (n = 637 cells analyzed) in the cortex (Fig. 3F, G) were immunoreactive.

Neurotransmitter phenotype of the grafted MGE cells. Grafted MGE cells expressed the GABAergic markers GAD65/67 in the transplant site (A). Arrows show example of double-labeled cells and arrowheads show MGE cells negative for GAD65/67. Cells outside the transplant site expressed GAD67 (C). The grafted cells also expressed glutamate (red in D–G). (E, G) Higher magnification of the inset in (D) and (F), respectively. Scale bars: (A, B, D, F) 20 μm; (C, E, G) 10 μm.
There are multiple subclasses of the MGE-derived interneurons that could be identified based on other cellular markers, such as the calcium binding proteins parvalbumin (Pva) and calbindin (Clb). MGE cells grafted in the cortex expressed Clb (Fig. 4A, B) and Pva (Fig. 4C, D) in 6 ± 1.7% (n = 751 cell analyzed) and 3 ± 0.8% (n = 686 cell analyzed), respectively. These Ca2+ binding proteins are markers for cortical interneurons. In the striatum, the grafted MGE cells did not express the cAMP-regulated phosphoprotein 32 kDa (DARP-32), a marker for striatal projection neurons (Fig. 4E).
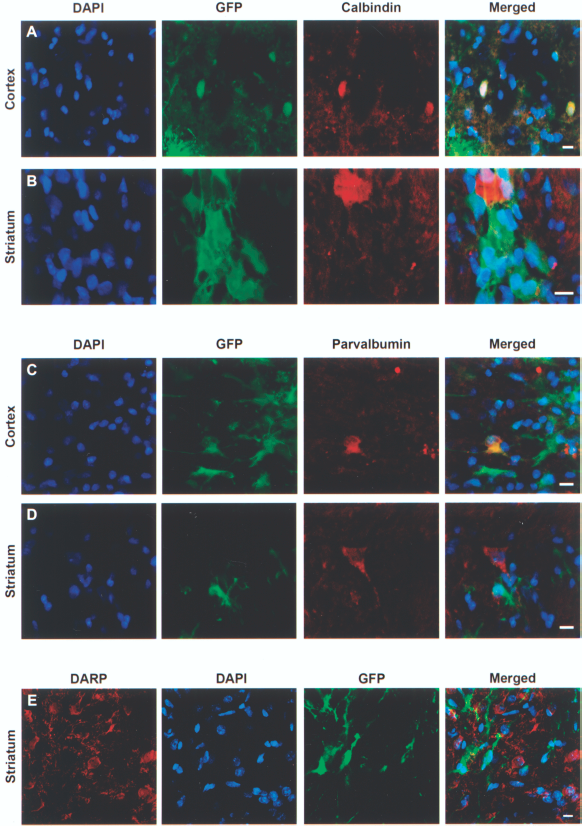
Molecular characterization of MGE grafts in the cortex and striatum. (A, C) Calbindin- and parvalbumin-immunoreactive MGE cells grafted in the cortex. (B, D) Examples of calbindin and parvalbumin immunonegative MGE cells in the striatum. (E) MGE cells grafted in the striatum did not express DARP-32. Scale bars: (A–E) 10 μm.
Grafted MGE Cells Enhance Local Synaptic Formations and Make Connections with Host Neurons
Synaptophysins are glycoproteins localized to synaptic vesicles in presynaptic axonal terminals. Immunostaining for synaptophysin revealed a significant decrease in the density of synaptophysin+ puncta in the stroke hemisphere (Fig. 5A–C). Notably, the MGE grafted stroked area displayed an increased expression of synaptophysin (Fig. 5D). Quantitative analysis demonstrated that in comparison to vehicle and fibroblast groups, synaptophysin expression in transplant/stroke zone was enhanced in the MGE grafted animals (Fig. 5E). To determine whether MGE cells made connections with host cells, we performed preliminary in vitro electrophysiological recording from brain slices (Fig. 5G–I). Transplanted GFP+ MGE cells were identified under epifluorescence in the region of the striatum for patch-clamp recording. Brain slices were prepared 1 month after transplantation. Some MGE cells close to the injection site were electrophysiologically silent 4 weeks after transplantation. Injection of a depolarizing current triggered a single action potential in one of seven GFP+ cells recorded in the dorsal part of the striatum of a stroked rat (Fig. 5G). Spontaneous excitatory postsynaptic currents (sEPSCs) were observed from this same cell (Fig. 5H), as well as from others (n = 10) with frequency of 10.9 Hz and average amplitude of 12.9 pA when holding the membrane potential at −70 mV. The native resting membrane potential for this cell was −56 mV. Immunoelectron microscopy analysis confirmed that the MGE cells established synaptic contacts with host cells (Fig. 5J). Together, these data suggest that grafted MGE cells are starting to integrate into the local neural circuit.

MGE grafted cells enhanced synaptophysin expression and exhibited synaptic connectivity. Fluorescence micrographs show synaptophysin expression (red) in the contralateral (A) and ipsilateral (B–D) sides to the stroke lesion. (C) GFP+ grafted fibroblasts, MGE cells (D) stained with DAPI. Fiber outgrowths and soma of grafted MGE cells appear decorated with synaptic puncta (D). Scale bars: 10 μm. (E) Synaptophysin expression was quantified in the contralateral and ipsilateral sides (insets in F). Data are expressed as mean ± SEM. *p < 0.05 versus vehicle and fibroblast transplanted groups. (G) Representative current clamp trace of an action potential from MGE neuron implanted into the dorsal striatum of stroked rat elicited in response to a 400 pA current injection. (H) Sample trace of spontaneous excitatory postsynaptic currents (sEPSCs) in the MGE neuron held at −70 mV (native resting membrane potential for this cell was −56 mV). (I) Scaled average sEPSC. (J) Electron micrograph taken from transplant zone showing an axodendritic synapse. The DAB-immunostained GFP is visible in the dendrite. Arrow points to synaptic vesicles at the axon terminal. Scale bar: 500 nm.
Transplanted MGE Cells Stimulate Axonal Reorganization in the Sensorimotor Cortex
Axonal sprouting and reorganization occurs after stroke injury. This anatomical reorganization occurs in the cortex immediately adjacent to stroke-induced injury (8) as well as corticocortical and cross-callosal projections (10, 20, 31). To investigate whether the MGE transplant influenced rewiring of the stroke-damaged side, axons originating from the intact hemisphere were labeled by injecting BDA into the right sensorimotor cortex 3 weeks after cell transplantation (Fig. 6A, B). The quantitative analysis of BDA-labeled terminals, normalized to the total number of labeled somas at the injections site (see Materials and Methods for details) (Fig. 6D, E), revealed an increase in the transplanted side (Fig. 6C).

MGE grafted cells enhanced axonal sprouting and improved motor behavior. (A) Schematic drawing of a frontal section through the striatum illustrating the extent of focal ischemia (shaded area) and the BDA injection site. (B) Low magnification photo of a frontal section taken showing the BDA injection site. Sections were counterstained with toluidine blue; arrowheads delineate the stroke zone in the striatum. (C) Quantitation of axon terminals in the sensorimotor cortex symmetric to the BDA injection site. (D, E) Photos showing examples of anterogradely labeled BDA axon terminals. LV: lateral ventricle, Str: striatum, cc: corpus callosum. *p < 0.05 versus vehicle group. Scale bars: (D, E) 100 μm. (F) The MGE grafted group showed significant improvement on the rotarod test compared to pretransplantation and to groups that received vehicle or fibroblast. (G) The MGE transplanted group showed a significant reduction in body asymmetry. Data are expressed as mean ± SEM. *p < 0.05 versus pretransplant, fibroblast-, and vehicle-transplanted groups.
To determine if MGE grafts had an effect on motor function following stroke, the animals were subjected to motor behavioral tests. The ischemia-lesioned rats were first tested for their baseline rotarod and EBST after stroke. Our results showed that the MGE transplanted rats improved in their locomotion and motor coordination with a significant improvement in both tests (p < 0.05) (Fig. 6F, G).
Discussion
Cell transplantation is a promising therapeutic strategy for brain repair. Mechanisms of stem cell-mediated functional recovery in diseased or injured brain tissues are still not completely understood. After stroke the brain undergoes limited self-repair to compensate for the lost structures. These changes in reorganization or neuroplasticity have long been suspected to underlie the partial spontaneous functional recovery that often occurs over time following stroke. Our study demonstrated that grafted cells differentiate and establish connections with the host, enhance axonal sprouting and synaptogenesis, and help restore motor function after stroke.
The grafted MGE cells dispersed in the striatum and cortex predominantly toward the stroke area. Previous in vitro and in vivo reports showed that the MGE cells possess an inherent ability to migrate when transplanted, cultured in a three-dimensional matrix, or cocultured on coronal sections of adult forebrain (1, 12, 29). The grafted MGE cells exhibited different morphologies and molecular identities depending on their final location within the stroke-damaged parenchyma. They exhibited spontaneous electrophysiological properties and were able to fire action potentials. Although the electrophysiological recording demonstrated connectivity with host tissue, further comprehensive studies are necessary to understand the neurophysiological properties of grafted MGE cells over time in the stroke environment. The electrophysiological properties of grafted neural progeny in the stroke-damaged tissue have been recently investigated using mouse embryonic stem cells (mESCs) that ubiquitously express eGFP (6). Patch-clamp recordings demonstrated that 27.7% of grafted cells expressed action potentials and voltage gated Na+ and K+ currents typical of the neuronal phenotype. Graft-derived neurons showed spontaneous postsynaptic current between 3 and 7 weeks posttransplant, demonstrating that they receive synaptic inputs. Similarly, electrophysiological recording from neural progeny derived from mESCs grafted into the MCAO stroke model demonstrated synaptic connections with the host and behavioral improvement in the fear-conditioning test (17). Together these data suggest that cell replacement could support functional recovery in animal model of stroke.
The mechanisms controlling cellular engraftment and differentiation in a stroke-lesioned environment are still largely unknown. In naive animals, approximately 98% of grafted MGE cells differentiated into neurons (1, 29). While in the ischemic environment, 13% and 20% of grafted MGE cells differentiated into the neuronal lineages, as demonstrated by the expression of β-tubulin-III and NeuN, respectively. We noted differential expression of GAD isoforms associated with their proximity to the graft. Cells within the core of the graft were GAD65+, whereas cells outside the graft expressed GAD67. In agreement with developmental studies, immunostaining for the calcium binding proteins Pva and Clb further confirmed the interneuronal GABAergic phenotype of MGE cells that migrated to the cortex while few migrated in the striatum (18). In addition, the MGE cells that migrated in the striatum did not express DARP-32, marker for stritatal projection neurons. This concords with the developmental origin of the DARP-32+ neurons from the lateral ganglionic eminences (LGE) and not from the MGE (21). We also showed that 15 ± 6% and 40 ± 8% of the grafted cells were immunopositive for glutamate in the striatum and the cortex, respectively. Transplantation studies have demonstrated that in developing cortex approximately 15% and 25% of transplanted MGE cells differentiated into glutamatergic neurons at embryonic ages E12.5 and E16.5, respectively (32). Transplantation of embryonic stem cell-derived neural progeny into developing normal rats demonstrated that 44% exhibit a glutamatergic phenotype (28), while in ischemic brains 13.3 ± 5.8% of the grafted cells expressed glutamate (26). Together these data suggest that stroke environment, proximity to the graft, and the final region of engraftment affect the fate of the grafted MGE cells.
We showed a robust increase in synaptogenesis and axonal terminals from intact cortex to the stroke-damaged and transplanted side. The number of BDA-labeled axon terminals may vary with the size of the injection site and the number of BDA+ cell bodies. We injected 0.5 μl of BDA into specific stereotaxic coordinates in randomly chosen animals from each group. The BDA infusion led to small (approximately 4 mm2) and circumscribed injection sites in the sensorimotor cortex. There was no significant difference in the size of the injection site and the total number of labeled cells between vehicle and transplanted groups. In addition, the data were normalized to the total number of labeled cells. Thus, it is unlikely that the increase of axonal sprouting we observed is due to differences in the size or location of the injection site. Injections of stem or stromal cells derived from cord blood and bone marrow-stimulated brain reorganization and enhanced functional recovery in stroke model (25, 30, 31). Neurosphere-derived cells reduced functional deficits and neuronal damage after focal ischemia by enhancing the expression of various growth factors, including BDNF and VEGF-A, known to be involved in brain plasticity, neuronal survival, and synaptic function (7, 15). Together, these data suggest that grafted MGE cells could exert neurotrophic, humoral effects on stroke-damaged local network and enhance axonal sprouting and synaptogenesis.
Our study suggests that MGE neural precursors promote functional recovery in an experimental stroke model through mechanisms involving cell replacement and local reinnervation. Nonetheless, additional work will be required to determine the time course and to what extent each of these modalities contributes to functional improvement.
Footnotes
Acknowledgments
We thank David Kunis and Guohua Sun for laboratory and technical support, and Beth Hoyte for preparation of the figures. This work was supported in part by Russell and Elizabeth Siegelman, Bernard and Ronni Lacroute, the William Randolph Hearst Foundation, the Edward E. Hills Fund, and NIH NINDS grants RO1 NS27292, P01 NS37520, and R01 NS058784.
