Abstract
Subarachnoidal grafting of monoamine-producing cells has been used with success to treat chronic pain in animal models. In the search for a source of autologous transplantable cells, capable of delivering neuroactive substances to the cerebrospinal fluid (CSF) to treat pain, we have tested adipose tissue-derived stromal cells (ADSCs) transduced to produce levodopa. Intrathecally grafted ADSCs survive for long term adhered to spinal cord and nerve root meninges. Cultured ADSCs were retrovirally transduced with tyrosine hydroxylase (TH) and/or GTP cyclohydroxylase 1 (GCH1) genes and stably expressed them for at least 6 weeks in culture. Singly transduced cultures did not produce measurable levodopa but doubly transduced or a mixture of singly transduced ADSCs were able to efficiently synthesize and release levodopa. When 0.5–1 × 106 TH-and GCH1-expressing ADSCs were intrathecally grafted in rats, elevated levels of levodopa and dopamine metabolites were found in CSF at 3 days, although at lower concentrations than expected. Unexpectedly, no levodopa was measurable in CSF at 6 days. In a rat model of neuropathic pain, intrathecal grafting of doubly transduced cells did not produce antiallodynic effects at 2 or 6 days, even when histological analysis revealed the presence of weak TH-immunoreactive subarachnoidal cell clusters. These results suggested that doubly transduced cells could indeed function as biological minipumps to enhance the dopaminergic neurotransmission at the spinal cord level but transgenes were rapidly silenced after intrathecal grafting. Transgene silencing was mimicked in culture by serum deprivation for 3 days. Serum addition at this point recovered trans-gene expression in just 6 h, as did, to a smaller degree, dbcAMP or histone deacetylase inhibitors. Transgene expression silencing in serum deprivation conditions was prevented by 5′-terminal IRES sequences. The present study does not discard the use of transduced cells as a strategy to treat chronic pain but shows that controlling transgene silencing in implanted cells needs to be achieved first.
Introduction
Neuropathies of varied etiology often cause chronic pain that can be difficult to treat. Allodynias (pain triggered by innocuous stimuli), dysesthesias (abnormal, unpleasant sensations), or hyperalgesia (increased sensitivity to pain) are common symptoms of painful neuropathies, producing great discomfort in patients. Antidepressants, anticonvulsants, opioids, NMDA antagonists, cannabinoids, and topical drugs are used to treat these symptoms with limited success (17,45), so the search for more effective treatments is still intense.
Subarachnoidal grafting of cells has been proposed as an alternative approach to deal with neuropathic pain. More than 20 years ago, Sagen et al. (35) showed that intrathecal implantation of chromaffin cells from the adrenal medulla induced analgesia in rats. Thereafter, chromaffin cells have been extensively tested in animal models of neuropathic pain (8,21,49) obtaining pain relief. Nevertheless, it is not known yet if the analgesic properties of chromaffin cells can be attributed to their secretion of monoamines, opioids, GABA, or even neurotrophic factors (13). Analgesia has been reportedly obtained also by intrathecal implants of opioid-releasing cells (46,47), serotonergic cells (15), and GABAergic cells differentiated from human neural progenitors (31) or from cell lines (14,16). We have shown that intrathecal grafting of monoamine-releasing cells in rats with mononeuropathy produced analgesic effects that were concomitant with increased monoamine metabolite levels in cerebrospinal fluid (CSF) (9). The problem associated with all the cell types that have been tested in the above experiments is that they are either of tumoral origin, so their growth cannot be controlled, or they are allogenic, and thus prone to immune response from the host. Even purified cultures of chromaffin cells, which are known for being negative antigen presenters (30), when placed in the CNS environment will be eventually rejected (43). Immunosuppression or encapsulation of cells inside semipermeable membranes that allow free passage of small molecules but isolate the graft from the host immune system might circumvent these problems (50) but still the risk/benefit ratio might be too high.
Our aim was to find a source of easy and innocuous to obtain autologous cells that could be used to treat patients suffering unbearable chronic pain. Additionally, we consider that the ideal cell type for this goal should: a) produce and release analgesic products, b) survive for a long time, integrated in the nervous environment without producing adverse reactions, and c) its activity—or presence—be terminated if required. Cells fulfilling all these requisites are extremely difficult to find, but we are looking for ways to address the lack of any of these properties in cells that accomplish most of them. In that respect, we have considered the use of adipose tissue-derived stromal cells (commonly referred to as ADSCs). These cells have several characteristics that might be interesting for clinical application. First, they are very easy to obtain from patients by minimally invasive surgery (lipoaspirate) and they are also easy to process by culture methods. Second, they show stem cell characteristics like pluripotentiallity and integrate to different tissue environments, where they will stop proliferating once culture mitogens are not present. Third, although they do not produce pharmacological amounts of any of the proposed endo-analgesics (monoamines, endorphins, GABA, endocannabinoids, galanin, etc.), gene transfer strategies can be devised so that ADSCs synthesize and release them in a controlled manner (20) and, additionally, are programmed to undergo apoptosis if their action is not longer required or if cells produce undesired effects. Due to their high proliferation rate in culture, ADSCs are an easy material for retroviral transduction.
For the present study, we have chosen to introduce the genes of tyrosine hydroxylase (TH), an enzyme that catalyses the conversion of phenylalanine into levodopa (also known as l-DOPA), and GTP cyclohydrolase 1 (GCH1), the limiting enzyme in the synthesis of tetrahydrobiopterin (BH4), which is a necessary cofactor for the function of TH. The genes were cloned in retroviral plasmids under the control of cytomegalovirus (CMV) promoter to drive their constitutive expression. As we show here, double transduction of ADSC cultures induces the production of levodopa by these cells, while single transduction by either of the above genes does not. Levodopa is the precursor of catecholamines in dopaminergic and noradrenergic terminals so addition of exogenous levodopa increases the synthesis and synaptic release of dopamine (DA) and noradrenaline (NA). The spinal dopaminergic system has been shown to modulate the transmission of nociceptive signals, especially through D2-type receptors (1,18,19,23–25,28,32,41). When levodopa is applied locally to the spinal cord, the increased dopaminergic neurotransmission produces analgesia (38). We have also shown (Cobacho et al., submitted) that both systemically and intrathecally injected levodopa has antiallodynic effects on a rat mononeuropathy model. The present work is a further step of those studies aiming to test if levodopa can be delivered by autologous cells to alleviate neuropathic pain.
Materials and Methods
Experimental Outline
For the present study, the following experiments were performed: 1) Testing of the long-term survival and distribution of intrathecally implanted ADSCs. 2) Transduction of ADSCs in culture with retroviral vectors carrying the genes for TH and/or GCH1. 3) Measuring levodopa production and release in vitro. 4) Intrathecal grafting of transduced cells in rats with a mononeuropathy; pain tests as well as HPLC-ED analyses of CSF were performed. 5) Given that, as we describe below, no pain relief was obtained and levodopa/dopamine metabolites in CSF disappeared by 6 days postimplantation, in vitro experiments to test downregulation of transgene expression were performed. Using GCH1 expression as a tool, immunofluorescence and Western blot experiments showed that serum deprivation decreased trans-gene expression in less than 3 days. Both transductional and translational changes appeared to be implicated. 6) Protection by IRES from translation silencing of trans-genes in serum deprivation.
Animals
Male Sprague-Dawley rats weighing 250–300 g at the beginning of the experiments were used for the present study. The experimental procedures using animals followed IASP's ethical guidelines for investigations of experimental pain in conscious animals (51) and were approved by the Ramon y Cajal Hospital Animal Welfare Ethic Committee.
ADSC Cultures
Adipose tissue was obtained from inguinal pads of adult rats in aseptic conditions. The tissue was placed in Leibowitz L-15; major fasciae and blood vessels were tore off under a dissecting microscope and the pad was cut up into small pieces. Then the tissue was digested with collagenase A (Sigma-Aldrich), 1 mg/ml in α20 (see below) for 45 min at 37°C. The pieces were then transferred to a tube with fresh α20 and mechanically dispersed by repeated pipetting using a P1000 automatic pipette with a fire-smoothened sterile tip. The cell suspension was seeded on a 75-cm2 culture flask (Falcon) with α20 medium. After 24 h, the medium was withdrawn and the flask was rinsed twice with Hank's balanced salt solution (HBSS without Ca2+ and Mg2+, Sigma-Aldrich), and new α20 was added. The medium was changed twice between passages, which were performed every 7 days. Passages were done by digesting near-confluent cultures with trypsin 0.05% + EDTA 0.02% (Sigma-Aldrich) in HBSS for 10 min at 37°C and seeding 1:20 in new flasks.
The above referred α20 medium was made of 78% of minimal essential medium, alpha modification (αMEM, Sigma-Aldrich), 1% nonessential amino acids (NEAA, Sigma-Aldrich), 1% antibiotic/antimycotic (Gibco), and 20% fetal bovine serum (FBS, Gibco) from batches selected for undifferentiated growth of rat ADSCs.
Retroviral Constructs
Replication-deficient Moloney murine leukaemia virus-based retroviral vector was used for transduction of ADSCs. The genes to be transduced (human TH and rat GCH1) were cloned into the multiple cloning site of the retroviral plasmid, placed between the CMV promoter and an IRES sequence, that was followed by the cope-pod GFP gene, as shown in Figure 1a. The plasmids were amplified in DH5α bacteria and the clones were selected by their ampicillin resistance.
Retroviral constructs to transduce ADSCs. (A) Schematic representation of the retroviral plasmid construct that was used to transduce ADSCs with either the human TH or the rat GCH1 genes. The genes were cloned in the multiple cloning site (MCS). Downstream, an IRES allowed the expression of the sequence encoding for the green fluorescence protein (GFP), which was used as reporter gene in the initial experiments and was removed in the second series of transplantation experiments to prevent host immune reactions. pCMV, cytomegalovirus promoter; AmpR, ampicillin resistance gene; RU5, Ψ+ and 3′-LTR are retroviral sequences. (B) Different examples of retrovirally transduced cells: ADSC-GFP (GFP autofluorescence in green), ADSC-GCH1 (red immunofluorescence to GCH1), ADSC-TH (green immunofluorescence to TH), and doubly transduced ADSC-GCH1 + TH (showing various degrees of red and green immunofluorescence to GCH1 and TH, respectively). Nuclei were counterstained by bis-benzimide (Hoechst 33342, in blue) in the mounting medium. Scale bar: for all images 100 μm.
Human TH cDNA was generously provided by Karen O'Malley (Washington University of St. Louis, MO, USA) and was cloned into the EcoRI site of the retroviral plasmid. The correct directionality of the cloned hTH was checked by measuring the size of EcoRI and XhoI (Roche Diagnostics) digestion fragments and the expression of the transgene was immunocytochemically tested in C6 glioma cells after transient transfection using Lipofectamine 2000 (Life Technologies).
Rat GCH1 cDNA with added adaptor sequences for restriction enzymes was synthesized by PCR from a viral plasmid generously provided by Un Jung Kang (University of Chicago, IL, USA), using the primers5′-AGT GTCGACATGGAGAAGCCGCGGGG-3′ and 5′-AGT GTCGACGGAAGTTCAGCTCCTGATG-3′ (Tm 66°C, 40 cycles). The PCR product was cleaved with Sal I (New England Biolabs) and cloned into a pGEM plasmid for amplification in DH5α bacteria. Sal I restriction fragments containing the rGCH1 cDNA were then cloned in the XhoI site (which is complementary to the Sal I protruding ends) of the retroviral vector. Directionality of the cloned gene was checked by measuring the DNA fragments obtained by cleavage of the retroviral plasmid with EcoRI and BamHI (Roche Diagnostics). The expression of the transgene was tested by immunocytochemistry in transiently transfected C6 cells.
Because the initial experiments showed that ADSCs transduced only with the GFP transgene disappeared from the subarachnoidal space between the 9th and the 14th day after transplantation, but they were present at 14 days in rats immunosuppressed with cyclosporine A, and because it has been published that this reporter transgene elicits immune responses (2,39), the GFP sequence was removed from the retroviral plasmid for the remaining experiments, leaving empty the second cloning site. For that purpose, retroviral plasmid construct was cleaved with SwaI and SalI followed by blunting the ends with T4 DNA polymerase, and recircularizing the plasmid with T4 ligase.
Infective particles, pseudotyped with the vesicular stomatitis virus G protein (VSV-G) envelop and containing the retroviral plasmid, were produced in 293T packaging cells using the Retrofect kit (Genetrix). Supernatants were aliquoted and stored at −80°C until use. Viral titers were checked by transfecting NIH/3T3 cells following the manufacturer's protocols.
Transduction of ADSCs
Fifty thousand ADSCs were seeded per well on 12-well plates and cultured in α20 for 24 h to reach a 70–80% confluence. Then, the cells were incubated in 1 ml of retroviral supernatant for 1 h at 37°C in a centrifuge with plate adaptors (Eppendorf) spinning at 1000 × g. Thereafter, the retrovirus-containing medium was replaced by fresh α20 medium for an additional 24 h. The cells were then trypsinized and expanded in 75-cm2 culture flasks except for an aliquot of 3000 cells that was seeded on three coverslips to quantify transfection efficiency by immunocytochemistry.
Immunodetection
Immunocytochemistry
ADSCs were cultured on glass coverslips inside 24-well plates for at least 24 h. Cells were fixed in buffered 4% paraformaldehyde for 10 min and washed in PBS pH 7.4. followed by blocking (5% goat serum + 0.1% Triton X-100 in PBS) and incubation for 1 h at room temperature with anti-TH antibodies—either mouse monoclonal (Chemicon MAB5280, 1:100 in blocking solution) or rabbit polyclonal (Serotec AMP931or Abcam ab112, both 1:1000) antibodies—and/or rat monoclonal anti-GCH1 antibody (clone 6B6, 1:50, generously provided by C. Hesslinger and E. Kremmer, from Munich, Germany). Reaction with fluorescently conjugated secondary antibodies (Alexa anti-mouse or anti-rabbit IgGs, 1:500, Molecular Probes, or rhodamine anti-rat IgG, 1:200, Rockland) was performed for 45 min at room temperature in the dark. Coverslips were mounted on slides using a glycerol/PBS (9: 1) mounting medium, containing p-phenylenediamine 0.1%, as antifading reagent, and bis-benzimide (Hoechst 33342) at 3 × 10−5 M (both from Sigma-Aldrich), for fluorescently staining all nuclei. Fluorescent samples were analyzed and photographed using an Olympus BX51 microscope.
Immunohistochemistry
Transplanted animals were intracardially perfused with 4% paraformaldehyde. Meninges and nerve roots were obtained by carefully opening the vertebral canal and dissecting them out. These tissues were processed in toto for immunofluorescence on flotation and examined under an inverted microscope equipped with fluorescence optics and video camera or by confocal microscopy (Bio Rad 1024). Also, lumbosacral pieces of the spinal cord were extracted and sectioned on a cryostat. In other transplanted animals, pieces of L3—L4 vertebrae were decalcified in Surgipath Decalcifier I for 24–48 h and either sectioned in cryostat or paraffin embedded, and then sectioned in a microtome. Immunohistochemistry for TH was performed on sections adhered to slides. The primary antibodies were a rabbit anti-TH (Abcam, 1:2000 overnight, at 4°C, in blocking solution containing 5% goat serum and 0.3% Triton X-100), a mouse anti-SMA (smooth muscle actin, 1:400, Sigma-Aldrich), and/or mouse anti-rat CD3 (eBioscience, 1:100), and the secondaries were Alexa-conjugated anti-IgG (Molecular Probes) for 45 min at room temperature (RT). The sections were mounted in glycerol-based nonfade solution and examined for fluorescence, as above. Alternatively, biotinylated anti-rabbit or anti-mouse Ig (Dako Cytomation, 1:500 in blocking solution, 1 h at RT) were used. Then, the sections were reacted with either streptavidin-HRP or streptavidin-alkaline phosphatase (both from Dako Cytomation) for 30 min, followed by reaction with HRP-DAB or AP-Red kits (both from Zymed), respectively. DAB-reacted sections were dehydrated and mounted in DePeX (Serva) and AP-Red-reacted sections were rinsed with water and mounted in Aquatex (Merck).
Western Blotting
Proteins were obtained by scraping cells from flasks in cold 0.9% NaCl, followed by centrifugation at 1000 × g, resuspending the pellet in lysis buffer (Triton X-100 0.5%, SDS 0.1%, Tris-HCl 10 mM, KCl 10 mM, DTT 1 mM, EDTA 1 mM, PMSF 1 mM, leupeptin 10 μg/ml, aprotinin 1 μg/ml, NaF 20 mM, and sodium orthovanadate 8 mM, 50 μl/flask), sonicating for 30 s at 4°C, centrifuging 12,000 × g, at 4°C for 30 min, and keeping the supernatant. Protein content was determined by BCA kit (Pierce) in a TECAN spectrophotometer.
Denaturalized protein solutions were run in polyacrylamide gels (10 μg/lane) and were then electrotransferred to nitrocellulose membranes (Hybond-ECL, Amersham-GE). The membranes were blocked in Tris-saline, containing 0.1% Tween-20 and 5% skimmed milk, for 1 h at RT. They were incubated overnight at 4°C in primary antibodies (mouse anti-TH, 1:5000, rat anti-GCH1, clone 6H11, 1:1000, rabbit anti-TurboGFP, AB513, 1:20000, from Evrogen or mouse anti-β-actin, clone AC-15 from Sigma Aldrich, 1:5000) prepared in blocking solution. Secondary antibodies (anti-mouse IgG-HRP from ECL kit, Amersham-GE, 1:1000 or anti-rat IgG-HRP, from Jackson ImmunoResearch, 1:2000) were reacted for 1 h at RT. The membranes were reacted with the ECL kit and exposed to medical X-ray film (Fuji) and/or digitalized and quantitated with Versadoc 4000 (Bio-Rad).
Transgene Expression
ADSCs transduced with GCH1 and/or TH genes were seeded as 100-μl drops in α20 on glass coverslips (2000 cells per coverslip) placed in 24-well plates that were enclosed inside a CO2-equilibrated, humidified container. After the cells attached to the surface, 400 μl of α20 were added to the well and incubated for 3 days. The medium was withdrawn and the cells were rinsed twice with HBSS and kept in either α20 (control undisturbed) or αMEM (serum-free culture) for 3 additional days. Treatments were added to αMEM and the cultures kept for an additional 2 or 6 h. For experiments of culturing ADSCs in CSF, the cells were kept undisturbed in the 100-μl α20 drop for 3 days, then carefully rinsed twice with HBSS after which 100 μl of either α20, αMEM, or CSF extracted from control animals (see below) was added. The coverslips were kept in the humidified container for 3 additional days with daily inspection. Then the cultures were fixed and processed for immunofluorescence.
For semiquantitative studies on changes in transgene expression, GCH1- or GCH1/GFP-trasduced cells were seeded on flasks and cultured for 3 days in α20, after which the cultures were approximately 90% confluent. The cells were rinsed twice with HBSS and refed with either α20 (control) or αMEM (serum-free culture). The treatments (20% FBS, 1 mM dbcAMP, or 1.5 mM valproic acid +100 nM trichostatin A) were added to the corresponding cultures for an additional 6 h. The flasks were then placed on ice and processed for Western blotting.
Animal Surgery
Rat Model of Neuropathic Pain
Peripheral mononeuropathy was produced in young adult rats by the chronic constriction injury (CCI) method developed by Bennett and Xie (4) with little variations. Briefly, the left sciatic nerve was exposed by blunt dissection of muscle and four loose ligatures, 1 mm apart, were done with 6/0 silk. The ligatures barely compressed the nerve so that blood circulation was maintained. The muscles were repositioned with one resorbable stitch and the skin was closed with staples that were withdrawn 7 days later. In our experience, the animals typically show signs of neuropathic pain by the first week after CCI and maintain stabilized signs for several weeks thereafter. These signs include tactile and cold allodynia and heat hyperalgesia. Pretransplantation tests were performed 14 days post-CCI.
Cell Transplantation and CSF Collection
Subarachnoidal cell grafting was performed the 15th day after CCI by a direct lumbar puncture method that we have described in detail elsewhere (9,10). Briefly, the animal was anesthetized with isoflurane and placed on an elevated surface with hind limbs hanging out to widen the intervertebral space between L3 and L4 vertebrae. The back was shaved, a small incision was made in the skin, and the fascia overlying L3—L4 was blunt dissected to clean the intervertebral entry site. A 25-gauge × 1 inch neonatal lumbar puncture needle (Becton-Dickinson, ref. 405243) was advanced into the subarachnoidal space and CSF was allowed to flow out into the needle cup until 50 μl was collected (the collected CSF was centrifuged for 2 min at 1000 × g to discard contaminating cells, pooled, and stored at −20°C for further use in culture experiments).
In parallel to the animal preparation, ADSCs were detached from flasks with trypsin, counted, resuspended in Leibowitz L-15 medium, distributed in sterile tubes (5–10 × 105 cells per tube) and kept on ice until use. Just before transplantation, the cells were pelletized by 1-min centrifugation at 1000 × g, and resuspended in L-15 to a final volume of 15–20 μl. Immediately after collecting CSF, the cell suspension was placed into the needle cup, a 1-ml syringe connected, and the cells pushed into the intrathecal space, under visual control, by a small bolus of air. The needle was then immediately withdrawn. The skin incision was closed with staples and the animal recovered from anesthesia in 5–10 min. CSF collection of postimplanted animals (or vehicle-injected rats) was accomplished also by direct lumbar puncture but, in this case, only 35 μl was extracted, so that the CSF originated mostly from the spinal regions. Liquids were immediately centrifuged for 2 min at 1000 × g, 4°C, and 30 μl of clean supernatant was frozen at −20°C until analysis by HPLC-ED.
Pain Tests
Rats were tested for tactile and cold allodynia. Basal measurements were performed before any surgery, so as to discard animals that showed to be hyperreactive to the tests. Only rats that showed a post-CCI tactile withdrawal response below 4 g or a cold withdrawal response above 4 s were considered candidates for transplantation. For behavioral tests, the animals were housed in transparent square cages (17 × 22 × 14 cm) with a grid bottom made of 7-mm wire mesh. The rats were allowed to accommodate to the cage for at least 10 min. The stimuli were applied from the bottom and the response was observed directly or with the help of a mirror.
Tactile allodynia was assessed by estimating the threshold for leg withdrawal in response to the mid-plantar application of von Frey nylon calibrated monofilaments for 6 s, as proposed by Chaplan et al. (6). The sequence of filament strengths was 0.4, 0.6, 1, 2, 4, 6, 8, and 15 g. The threshold was calculated by the “up and down” method described by Dixon (11), starting with a filament strength of 2 g and changing to a higher strength if there was no reaction or to a lower strength if the rat had lifted the paw. The test was stopped when five filaments had been tested after the first change of direction or if the test limits (0.4 or 15 g) had been reached. The test was recorded as a series of “O” (when there was no response) or “X” (when there was a lifting response) and that record was compared with Dixon's table to obtain a value (D50) that could be translated into the estimated tactile threshold, following the formula: threshold (in g) = 10(Xf+kδ)/10,000, where Xf is the value (in log units) of the final von Frey filament used, k is Dixon's tabular value for pattern of positive/negative responses, and δ is the mean difference (in log units) between stimuli (here 0.224).
Cold allodynia was assessed by the duration of the paw-lifting response during the first minute after spraying 0.5 ml acetone to the foot with the help of a syringe with a blunt needle. Acetone evaporation produces an innocuous cooling of the paw that is felt as painful only in a neuropathic limb, so the animal lifts that leg one or more times as a withdrawal response to pain. The total time spent by the rat with the limb raised from the floor during the first minute can be thus considered as a measurement of perceived pain.
HPLC-ED Measurements
Quantification of levodopa, monoamines, and their metabolites was performed by HPLC equipped with a nucleosyl 5C18 column (Kontron) and an electrochemical detector (Coulochem 5100, ESA) as described previously (9). For measuring in vitro levodopa production by transduced or control ADSCs, the cells were cultured on six-well plates in α20 until full confluence. The medium was withdrawn and, after two rinses with HBSS, 600 μl of αMEM supplemented with NEAA was added per well. The cultures were incubated for an additional 18 h. Then the medium was collected and filtered (0.45 μm) and 66 μl of 4 N perchloric acid was added before storing frozen. The cells were simultaneously scraped in 300 μl of 0.4 N perchloric acid, sonicated, and centrifuged for 20 min at 12,000 × g at 4°C. The supernatant was stored frozen until analysis. Sibling wells were used for cell counting. For measuring CSF levels of levodopa and monoamine metabolites, collected liquids were diluted 1:3 in 0.4 N perchloric acid and filtered (0.45 μm) before running HPLC.
Statistical Analyses
As discussed elsewhere (Cobacho et al., submitted), the data obtained from the tests of tactile and cold allodynia, as we perform them, do not follow a Gaussian distribution and, furthermore, have upper and lower limits, so these data cannot follow parametric statistics. A Kruskal-Wallis one-way analysis of variance by ranks was used to analyze all data groups, followed by Wilcoxon signed rank test to compare pre- and postgrafting for each implant type, using GraphPad Prism 4 software. The rest of experimental data were expressed as mean ± SD.
Results
Transduction of ADSCs
Highly efficient (60–95%), stable transduction of GFP, TH, or GCH1 genes was obtained in rat ADSCs using replication-incompetent retroviruses as gene vectors (Fig. 1B). Constitutive, high-level transgene expression was driven by CMV promoter and lasted longer than 6 weeks in culture.
Survival of ADSCs in the Subarachnoidal Space
ADSCs were first tested for their ability to survive and integrate into the subarachnoidal space. Intrathecal grafting of GFP-transduced ADSCs was performed via direct puncture at lumbar levels. Microscopic exams of whole mount meningeal membranes and nerve roots revealed the presence of clusters of fluorescent cells adhered to their surface at 7 or 9 days postimplantation (DPI) (Fig. 2a–c), but not at 14 DPI (not shown). When animals were daily treated with the immune suppressor cyclosporin A, GFP-expressing ADSCs were found on meninges and nerve roots at 14 DPI, which raises the possibility of immunological rejection of cells due to the expression of an invertebrate protein on their membrane. If untransduced stromal cells were labeled with two fluorochromes (PKH26 and Hoechst 33342) previous to their implantation, doubly stained cells could be found on the pial surface and septae of spinal cord sections at 3 months after grafting (Fig. 2d, e). Histological staining of paraffin sections of the vertebral canal at 3 months after grafting nonlabeled ADSCs showed the presence of clusters of fibroblastic cells that were interpreted as being the implanted ADSCs (Fig. 2f). Taken together, these observations suggest that ADSCs are able to survive intrathecally for long periods, although immune rejection might take place if they are made to express xenoproteins. To prevent GFP-driven immune rejection, this transgene was removed from the retroviral constructs so transduced ADSCs did not express GFP for the remaining transplantation experiments.
Long-term survival of intrathecally implanted ADSCs. (a–c) GFP-transduced ADSCs could be found adhering to meninges (a, b) and nerve roots (c) at 9 days postimplantation (DPI) in these in toto preparations of nonimmunosuppressed rats. (d, e) In cryostat sections of the lumbar spinal cord at 3 months after their transplantation, control stromal cells, prelabeled with fluorescent stains PKH26 (red) and Hoechst 33342 (blue nuclei), could be found on the spinal surface and septae. (f) Paraffin section (5 μm thick) of decalcified vertebral columns at spinal L6 level, stained for toluidine blue/methylene blue, showing clusters of fibroblastic cells (black arrows), mostly in relation to blood vessels or meningeal sheets. nr, nerve root; sc, spinal cord; dsv, dorsal spinal vein. Scale bars: (a–d) 50 μm; (e) 200 μm; (f) 80 μm.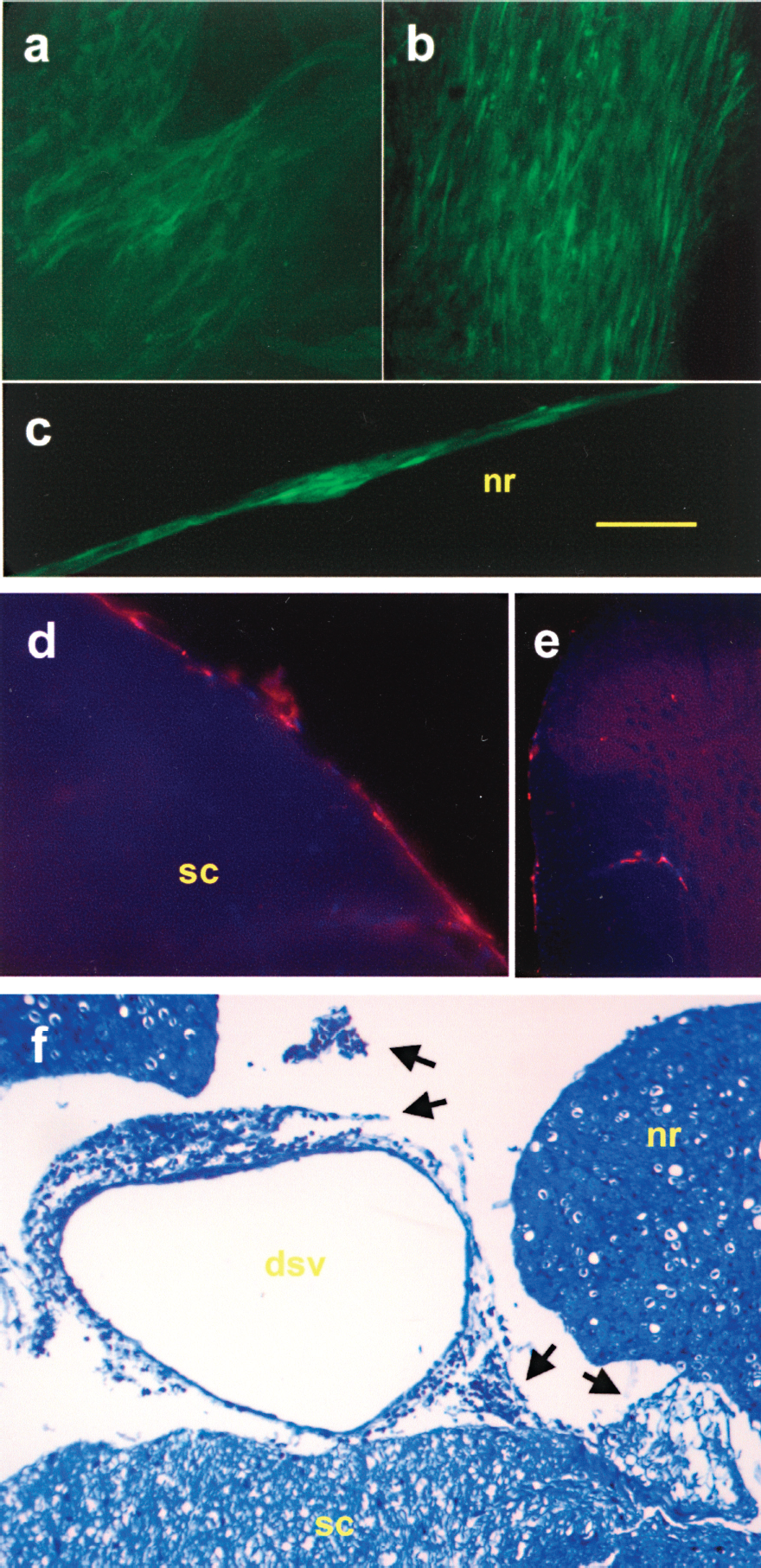
The survival of intrathecally grafted TH- and GCH1-transduced ADSCs was examined at various time periods without immune suppression. Immunohistochemical staining of decalcified vertebral columns showed clusters of fibroblastic cells in the subarachnoidal space that were lightly positive for TH at 6 DPI (Fig. 3a). Immunostaining for smooth muscle actin (SMA), which is expressed in most ADSC, showed the presence of SMA-positive but TH-negative cell clusters lying between nerve roots at 10 DPI (Fig. 3b). Taken together, these observations suggest that transduced ADSCs survive for more than 7 days (i.e., longer than the time period at which their analgesic action was tested). However, the progressive loss of immunostaining of grafted cells might be an indicative of decreased transgene expression.
Progressive disappearance of TH expression in transduced ADSC after grafting. Immunofluorescent staining for TH (a) and TH + SMA (b) of intrathecally implanted ADSCs transduced with the TH and the GCH1 genes. (a) A large cluster of weakly TH-expressing cells is located in between nerve roots (nr) at 6 days postimplantation (DPI). (b) A cluster of SMA-positive cells (in red, arrowhead), presumably ADSCs, is located dorsally to the L6 level of the spinal cord at 10 DPI, but the cells are not stained for TH (in green), which suggests transgene expression silencing; as control for the staining, blood vessels (bv) stain for SMA and catecholaminergic fibers in the spinal cord (arrows) stain for TH. Scale bars: (a) 100 μm; (b) 50 μm.
Levodopa Production by Transduced ADSCs
Levodopa production by transduced ADSCs was measured in culture by analyzing intracellular contents of cells maintained for 18 h in defined medium (αMEM + NEAA) by means of HPLC with electrochemical detection. Cells expressing the TH transgene did not produce levodopa in significant amounts but, when 100 μM BH4 was added to the medium, high levels of levodopa were measured (Fig. 4). It is thus clear that the presence of BH4 is required for the synthesis of levodopa by the TH enzyme and also that ADSCs were not able to spontaneously produce this cofactor by themselves. On the other hand, large quantities of levodopa were also present extracellularly in the culture medium (Fig. 4D), which suggests that levodopa was released from cells.
HPLC-ED analysis of cultured cells. (A) Standard chromatogram containing levodopa (DOPA), dopamine (DA), and dihydroxyphenylacetic acid (DOPAC). (B, C) Intracellular contents of TH-transduced cells without (B) and with (C) the addition of BH4 100 μM to the medium. (D) Quantification of levodopa contents in cells (right) and in the culture medium (left) of control cells and TH transduced cells in the absence and presence of BH4. For the experiment, the confluent cultures were washed twice with HBSS and maintained in αMEM + NEAA for 18 h. Values represent mean ± SD of 3–4 cultures.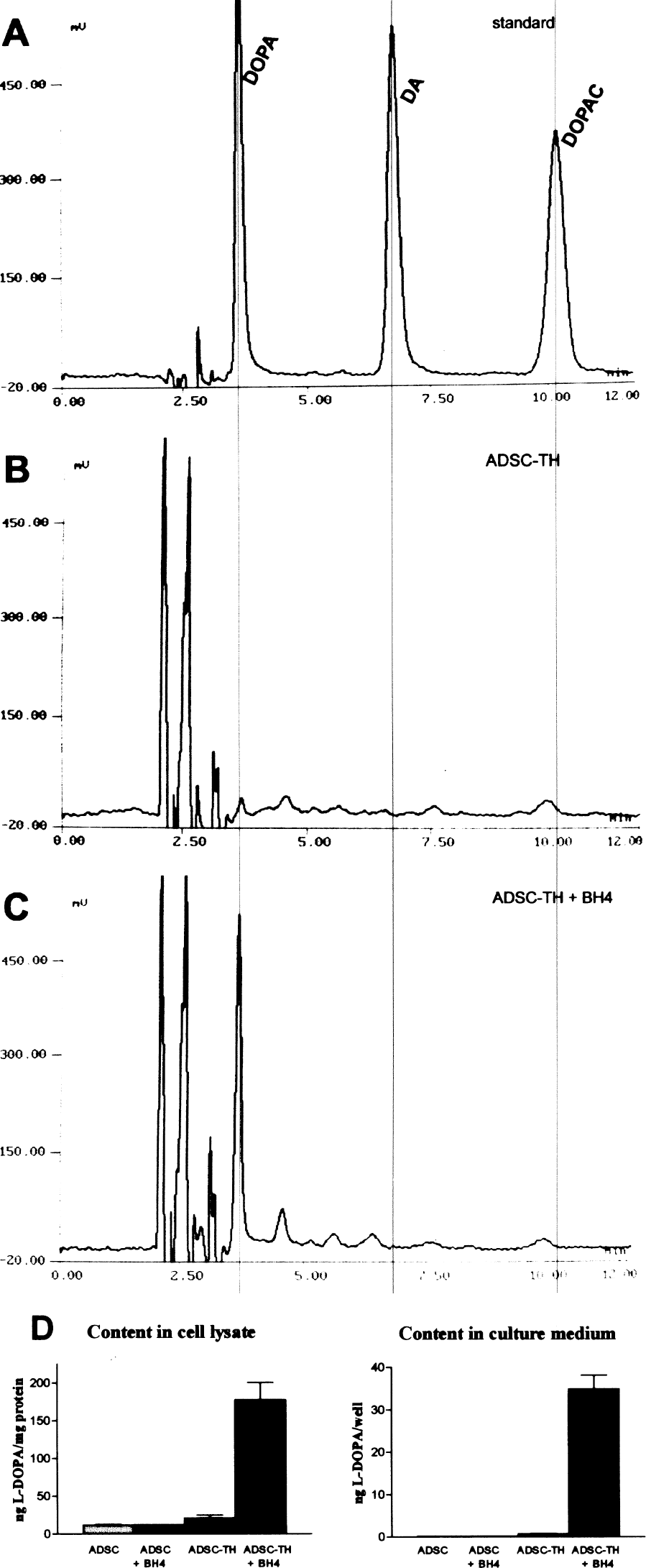
Levodopa Contents in Culture

Intracellular and extracellular levodopa contents after 18-h culture in defined medium, in nanomoles per million cells. The data are expressed as mean ± SD (n = 3). n.m.: not measurable (below detection threshold at the dilutions that were used).
When cells expressing both transgenes were rinsed twice with isosmolar Krebs-Ringer buffer, either with or without potassium, the intracellular contents of levodopa decreased to less than one tenth of the original levels (Table 1). These results suggest that levodopa, a catechol amino acid, passively diffuses through cell membranes to equilibrate intra- and extracellular concentrations and, thus, the release of levodopa to the medium depends mostly on its rate of synthesis. Consequently, if levodopa is constitutively produced by doubly transduced ADSCs, the number of intrathecally grafted cells would determine the dose of levodopa being delivered to the CSF. Thus, estimated releases of approximately 10 nmol levodopa/h could be provided by 1 million ADSCs that had been retrovirally transduced with TH and GCH1 genes driven by CMV promoter.
Action of Transduced Cells on Neuropathic Pain
Five hundred thousand to 1 million transduced cells were intrathecally implanted in rats with CCI. To preclude any possibility of immune rejection of transduced cells, rats were given a daily dose of 10 mg/kg of cyclosporin A, SC, starting the day before implantation. Rats were randomly divided into three experimental groups of nine rats each: group 1, implanted with GCH1-transduced cells; group 2, implanted with TH-transduced cells; group 3, implanted with GCH1- and TH-transduced cells. Only group 3 was expected to release levodopa and, hence, to show analgesia.
Tactile and cold allodynia tests were performed before cell grafting and at 2 DPI (to see early effects) and 6 DPI (to see maintained effects). As shown in Figure 5, none of the experimental groups showed analgesic effects on allodynia, at any time postimplantation. Thus, either chronic treatment with levodopa was not effective on neuropathic pain or levodopa was not being released to the CSF at the expected rates.
Effects of intrathecally grafted cells on neuropathic pain. Sensory tests for cold and tactile allodynia in mononeuropathic rats that were intrathecally implanted at lumbar levels with transduced ADSCs. Left legs were subjected to CCI (neuropathic), while right legs served as unoperated controls. The columns represent the hind leg withdrawal time or the tactile threshold (see Materials and Methods for details) at basal (i.e., prelesion), post-CCI (i.e., preimplantation values at 14 days postsurgery), and 2 or 6 DPI of cells transduced with TH, GCH1, or both transgenes. Each column represents the median value ± interquartile range of nine rats. Cut-off extremes for tactile tests are 0.4 and 15 g. As shown, statistically significant differences (p < 0.001) were found only between basal values and the rest of the values in operated legs.
Levodopa Measurements in CSF of Grafted Rats
Thirty-five microliters of CSF was extracted after the last pain test. Additional rats were grafted with TH- and GCH1-transduced or control (untransduced) ADSCs without immune suppression and 35 μl of CSF was collected at the 3rd day postimplantation (sampling CSF at the 2nd day postimplantation is usually less reliable) and at the 6th day postimplantation. At 3 DPI, HPLC-ED analysis showed levodopa as well as increased dopamine metabolites in CSF of all animals that had been grafted with doubly transduced ADSCs and not in those animals that had been grafted with the same amounts of control cells (Fig. 6). This result demonstrated that doubly transduced grafted cells were actually releasing levodopa to the CSF environment and that levodopa was increasing the dopamine synaptic release in the lumbar spinal cord, which was in agreement with the aim of the present work. However, the concentration of levodopa found in CSF at 3 DPI (0.1 nmol/ml CSF) was much lower than expected for a release that had been estimated in 5–10 nmol/h. Furthermore, at 6 DPI no levodopa was measurable in any of the animals, including those that had shown levodopa at 3 DPI, and the levels of dopamine metabolites were back to normal values (Fig. 6). The low concentration of levodopa at 3 DPI and its definite absence at 6 DPI might suffice to explain the lack of analgesic activity of doubly transduced ADSCs that we are reporting above. But we need to explain now why levodopa release is lost so early in grafted animals. It is unlikely that the cells disappear from the subarachnoidal space since, as we show above, transduced ADSCs are present at least at 7 DPI even in nonimmune suppressed animals (Fig. 2). However, we also observed that immunostaining of grafted cells was weak (Fig. 3), so we undertook a series of in vitro experiments to check if changes in the cell environment might have an early effect on the expression of transgenes that were driven by CMV promoter.
HPLC-ED analysis of rat CSF obtained before intrathecally grafting TH- and GCH1-transduced cells (preimplant) and at 3 or 6 days postimplantation (DPI). The chromatograms belong to the same rat and show elevated levels of levodopa and dopamine metabolites DOPAC and HVA at 3 DPI, while these traits disappear at 6 DPI. Noradrenaline metabolite, 3-methoxy-4-hydroxyphenylglycol (MHPG), also appears to be increased at 3 DPI. The peaks were identified and quantitated by parallel chromatograms of standards containing levodopa (DOPA), MHPG, dihydroxyphenylacetic acid (DOPAC), homovanillic acid (HVA), and 5-hydroxyindoleacetic acid (5HIAA). The asterisk marks an unidentified peak that appears in all chromatograms. Arrowhead points to a peak appearing in a few CSF samples, irrespective of their experimental condition, that may be related to blood contamination. The table shows CSF levels of levodopa, DOPAC and HVA in animals grafted with TH + GCH1-transduced or control (untransduced) ADSCs. Data are mean ± SD in pmol/ml. nd, nondetectable.
Changes of Transgene Expression in ADSCs in Culture
We first checked in culture if ADSCs survived well in the absence of serum and in CSF. ADSCs were seeded on coverslips (13 mm diameter) and kept growing for 3 days in a drop of 100 μl of maintenance medium (α20). Then the medium was withdrawn, the coverslips were rinsed twice with HBSS, and the cultures were kept for 3 additional days in 100 μl of either α20 (which contained 20% FBS in αMEM-NEAA), αMEM-NEAA, or CSF collected from control rats. As early as 18 h later, most of the cells maintained in CSF appeared floating and at 48 h all cells were apoptotic or dead. Culturing cells in αMEM-NEAA produced some initial cell detachment and stopped cell division but a near-confluent monolayer of cells remained for at least 3 days. Cells cultured in α20 kept proliferating.
Interestingly, immunocytochemical staining for TH and/or GCH1 in cultures of transduced ADSCs showed that serum deprivation for 3 days produced an inhibition of transgene expression, which was light in TH-transduced cells and drastic in GCH1-transduced cells (Fig. 7a, b).
Reversible changes of transgene expression in culture. (a, b) Double immunostaining of TH (in green) and GCH1 (in red) of mixed cultures of cells transduced with TH and cells transduced with GCH1. Nuclei are stained in blue. Scale bar: 50 μm. In (a), the cells are kept for 6 days in the maintenance medium, α20, which contains 20% FBS; in (b), the cells were kept for the last 3 days in αMEM, without added serum. After this period of serum depletion, GCH1 contents are close to the immunofluorescent detection threshold, while TH labeling is still visible in many cells. (c, d) Reversion of GCH1 transgene downregulation by diverse treatments. GCH1-transduced ADSCs kept in α20 for 6 days, αMEM for the last 3 days, or in αMEM for 3 days supplemented for the last 6 h with 20% FBS, dibutyryl cAMP, or histone deacetylation inhibitors [trichostatin A (TSA), valproic acid (VPA)]. (c) Western blots. (d) Fluorescence photomicrographs in fixed exposure conditions (objective: 20x, F: 4.0, exposure time: 2 s). Transduced cells are brightly immunofluorescent when maintained in α20, but GCH1 expression is strongly downregulated when cells are kept for 3 days in αMEM. Addition of FBS at this point for 6 h intensifies GCH1 immunostaining. Addition of TSA also produces a slight recovery of GCH1 immunostaining. Addition of dbcAMP for 6 h produces an increase in GCH1 immunostaining, but this time it is accompanied by morphological changes in many cells. Scale bars: 50 μm.
It has been reported that serum deprivation in cultures is a stressor that produces a general decrease in protein translation (22,48). Additionally, it has been reported that serum deprivation produces inactivation of CMV promoter by mechanisms that may imply methylation and/or deacetylation of histones (5). We thus checked various treatments to test if the observed decrease of transgene expression could be reverted. Densitometric analysis of Western blots from GCH1-transduced ADSCs showed that serum deprivation for 3 days actually decreased 70% the GCH1/β-actin protein ratio. Furthermore, a 6-h treatment with 20% FBS to GCH1-transduced ADSCs that had been serum deprived for 3 days doubled the ratio of GCH1/β-actin protein, while a 6-h treatment with inhibitors of histone deacetylases (valproic acid + trichostatin A) or dibutyryl cAMP produced a small but consistent increase of that ratio (Fig. 7c).
Recovery of transgene expression was also tested by immunocytochemistry in GCH1-transduced ADSC cultures. Cells were grown on coverslips in α20 for 3 days and then the medium was withdrawn. After two rinses in HBSS, the cells were maintained in complete medium (α20) or in serum-depleted medium (αMEM + NEAA) for 3 additional days. At this time, FBS (20 %), dbcAMP (1 mM), valproic acid (1.5 mM), trichostatin A (100 nM), or valproic acid + trichostatin A was added to the culture medium for an additional 6 h. Immunocytochemistry for GCH1 showed that transgene expression was barely detectable in serum-deprived medium, but cells were clearly stained after FBS addition for 6 h, although less intensely than those kept all the time in α20 (Fig. 7d). Treatment with dbcAMP also induced recovery of GCH1 expression, accompanied by morphological changes producing ramified cells. Treatment with trichostatin A or valproic acid produced tenuous perinuclear labeling of cells, and the addition of both histone deacetylase inhibitors appeared to enhance that staining.
To test if cap-dependent downregulation of protein synthesis in response to stress was involved in the silencing of GCH1 transgene expression we used ADSCs transduced with retroviruses incorporating GCH1–IRES—GFP construct. The rationale behind this experiment was that if IRES promoted cap-independent protein synthesis, then serum deprivation would downregulate GCH1 translation (as well as general protein translation) but GFP translation would proceed undisturbed. In fact, when GCH1–IRES–GFP-transduced ADSCs were placed for 3 days in αMEM, GFP expression, as evidenced by its autofluorescence, was expressed in medium-to-high levels while GCH1 expression, as evidenced by immunofluorescence, had almost disappeared in the same cells (Fig. 8c, d). Addition of FBS for 6 h at this point induced a visible recovery in GCH1 expression while GFP expression remained almost unchanged (Fig. 8e, f). Western blot analyses showed that GCH1/β-actin ratio largely decreased after serum withdrawal, as observed above, but GFP/β-actin ratio in the same cells did not show significant changes (Fig. 8). Addition of FBS for 6 h recovered GCH1/β-actin ratio but the GFP/β-actin remained almost constant. The GCH1/GFP ratio of cells cultured in αMEM was 21% with respect to cells kept in α20 medium, while the ratio of cells maintained in αMEM and then supplemented for 6 h with FBS was 105%. These results clearly demonstrated that IRES allowed GFP expression in serum deprivation conditions so that, when GCH1-IRES-GFP transduced ADSCs are placed in stressful conditions (as it happens when cells are intrathecally implanted), they keep expressing the fluorescent protein but silence expression of the GCH1 enzyme.
Protection from protein translation silencing by IRES, as shown by the differential expression of two transgenes cloned in a bicistronic vector (GCH1–IRES–GFP) driven by the CMV promoter in serum deprivation conditions. (a–e) Fluorescence photomicrographs in fixed exposure conditions (objective: 20x, F: 4.0, exposure time: 2 s). The left column shows the green autofluorescence due to the expression of GFP. The right column shows, in the same field, the immunofluorescent staining for GCH1. (a, b) Cells cultured for 6 days in α20; (c, d) sibling cultures maintained for the last 3 days in αMEM medium; (e, f) cultures maintained for the last 3 days in αMEM and then supplemented with 20% FBS for an additional 6 h. As shown, serum deprivation pronouncedly inhibited of GCH1 expression, while GFP expression was still elevated. Serum addition at this point increased GCH1 expression. Nuclei are stained yellow in the left column to mark all the cells in the culture. Scale bars: 50 μm. Arrowheads in (d) point to apoptotic cells. The lower panels show Western blot analyses of protein expression. Densitometric measurements of bands were normalized to values of transduced ADSCs maintained in α20 medium. Anti-TurboGFP staining produced a multiple band (28, 26, and 22 kDa) pattern; for quantification purposes, the sum of all the stained bands was considered. Control refers to nontransduced ADSCs kept in α20, to show lack of staining for either anti-GCH1 or anti-TurboGFP antibodies.
Taken together, these in vitro experiments imply that the absence of (unidentified) serum factors is able to produce a decrease of transgene expression, both at transcriptional and at translational levels, and that different actions can reverse partially such decrease. In relation to the cells that were intrathecally implanted, the lack serum factors in the CSF environment might produce a similar decrease of GCH1 and/or TH transgene expression and, consequently, result in a diminished or even absent levodopa production.
Discussion
The present report shows that ADSCs are useful vector cells to be transduced and employed as biologic minipumps delivering neuroactive products when implanted. ADSCs proliferate fast in culture and are easy to transfect with retrovirus, so large amounts of transduced cells might be readily available for autologous transplantation in a short period of time. These characteristics drove us to test them as a possible cell therapy strategy to treat chronic, unbearable neuropathic pain. We chose transducing ADSCs to produce levodopa, because we and others have shown that this catechol amino acid can reduce allodynia in an experimental rat model of neuropathic pain, acting through the descending dopaminergic system, and both levodopa and dopaminergic metabolites can be readily measured in CSF. Furthermore, there are other chronic conditions where intrathecally grafted levodopa-producing ADSCs might be considered as a strategy, like restless leg syndrome (7) and Parkinson's disease. We used a commercially available retroviral vector that contained a CMV promoter to drive the expression of two cloned genes with an IRES placed between them. This construct gave us the opportunity to have a high constitutive expression of one enzyme (either TH or GCH1) keeping a second site to clone a second gene, like a suicide gene, aimed at controlling the growth of transduced cells or terminating their presence if necessary.
BH4 was required for levodopa production in large amounts by TH-expressing ADSCs. Transduction of ADSCs with the BH4 synthesis limiting enzyme GCH1 enabled cells to produce this cofactor. A similar strategy had been used by others in rat marrow stromal cells (37) and fibroblasts (3) to efficiently produce levodopa in vitro. As shown in our study, TH-expressing ADSCs did not need to be cotransduced with the GCH1 gene to produce levodopa, because cultures containing a mixture of TH-ADSCs and GCH1-ADSCs were as efficient (or even more) than those of doubly transduced TH + GCH1-ADSCs.
In culture, ADSCs transduced with TH and GCH1 (either cotransfected or mixed cultures) produced and released levodopa in sufficient amounts as to deliver analgesic doses of levodopa to the spinal cord. A release of 5–10 nmol levodopa/106 cells/h was calculated for these transduced cells. As discussed elsewhere (Cobacho et al., submitted), despite the fast clearance of levodopa from CSF, there is a strong enhancement of dopaminergic activity when levodopa is chronically delivered to the subarachnoidal space, as it was evidenced by the elevated CSF concentrations of the dopamine metabolite DOPAC in those experiments (after 3 days of continuous infusion of 40 nmol/h levodopa through an intrathecal catheter, 38 nmol of levodopa and 4.46 nmol of DOPAC/ml of CSF were measured). Furthermore, because the release of levodopa by transduced ADSCs is volume free, local concentration of levodopa close to the grafted cells would be higher than the one that can be obtained by intrathecal delivery of a drug solution by injection or through a catheter.
This notwithstanding, when doubly transduced cells were intrathecally grafted, no analgesic effect was observed at 2 or 6 DPI. HPLC analysis of CSF at 3 DPI showed that the implanted cells were actually releasing levodopa and, importantly, that the dopaminergic activity had been increased, because dopamine metabolites, DOPAC and HVA, were present in elevated concentrations relative to control CSF (Fig. 6). Nevertheless, CSF levels of levodopa and dopamine metabolites at 3 DPI were much lower than expected (see table in Fig. 6), which might explain the lack of analgesic effect at 2 DPI. Levodopa levels in CSF rapidly diminished to disappear by 6 DPI. It can be ruled out that the absence of levodopa was due to the death of grafted cells because they were histologically demonstrated at longer survival times and, additionally, other experiments had shown that they can survive in nonimmunosuppressed animals for at least 7 days even when expressing a xenoprotein like GFP. Other groups that have tried to graft transduced cells to deliver levodopa or dopamine in brains of parkinsonian rat models reported the downregulation of transgene expression a few days after transplantation (3,27,29,36,44) independently on the transduction vector or the promoter used. Several molecular mechanisms might account for silencing of the transgene expression. At the transcription level, CMV promoter might be inactivated due to DNA methylation (40), to chromatin reorganization by histone deacetylation (26,34), or to cell cycle or transcription factor-dependent transcriptional regulation (5). At translational level, cap-dependent silencing mechanisms in response to stress might be acting (22,48).
Intrathecally grafted cells are exposed to an environment containing most of the culture medium components except serum (ions, amino acids, glucose, multiple antioxidants, and vitamins), although in different amounts. However, while transplanted ADSCs survive intrathecally for prolonged periods, when cells were kept in culture in CSF they died in few days for unknown reasons, so the intrathecal conditions could not be reproduced in vitro and we had to use instead a defined culture medium for studying possible alterations of transgene expression.
We found that when ADSCs were cultured in serum-depleted medium expression of the GCH1 transgene was silenced in less than 3 days, thus mimicking what might be happening after intrathecal cell grafting and providing an explanation for the gradual disappearance of levodopa from CSF of transplanted animals. Additionally, treatment with serum or, less pronouncedly, dibutyril cAMP or histone deacetylase inhibitors for just 6 h induced a partial recovery of transgene expression. Silencing of GCH1 expression was not due to fasting of ADSCs in serum-depleted medium, because GCH1 content was decreased in relation to β-actin protein content. It rather appeared to be a consequence of the lack of some serum-induced transcription factor(s), given that serum addition recovered GCH1 expression to a great extent in just 6 h. It did not seem to depend on cell cycle either because, although ADSCs kept in serum-deprived medium did not divide, treatments that were not mitogenic, like dibutyril cAMP or histone deacetylase inhibitors, also produced a partial recovery of the GCH1 expression. The relatively short half-lives of the GCH1 transcript and protein (33) might explain why this transfected enzyme is more sensitive to expression downregulation in the absence of serum than TH.
Interestingly, intensely fluorescent GFP-transduced ADSCs were demonstrated intrathecally at 7 days post-transplantation or later, so it appears that GFP expression is not silenced in the same way as GCH1 or TH transgenes are. In our viral construct, the GFP gene was situated after an IRES sequence, while GCH1 and TH genes were cloned in the MCS (i.e., after the CMV promoter) (see Fig. 1). It is known that different adverse environmental conditions, like serum deprivation, inactivate protein translation from capped mRNA, but when an IRES sequence is present at the 5′-UTR, a cap-independent translation of the mRNA is allowed (22,48). So, in our viral constructs, while GCH1 and TH expression may be subjected to cap-dependent translation silencing in serum deprivation conditions, GFP expression is IRES mediated (and thus cap independent) and so it may escape translation silencing. Our experiments of differential transgene expression downregulation in cells transduced with a GCH1–IRES–GFP construct supported this hypothesis. In relation to total protein content, GFP was shown to remain quite constant along the experiment. Meanwhile, β-actin, a very stable protein (12), showed minor changes with respect to total protein content and GCH1 protein suffered a drastic reduction in serum deprivation, followed by a fast recovery after serum supplementation. The GCH1/GFP ratio in serum-deprived conditions was 21% of that of α20 medium, while serum supplementation returned GCH1/GFP ratio to previous values in just 6 h (Fig. 8). Although the contribution to these changes of the different protein stabilities cannot be overlooked, the rapidity of GCH1/GFP ratio recovery rather points to differential translation regulations of cap-dependent and cap-independent genes.
Additionally, transcriptional changes appear to take part also in the downregulation of transgene expression in serum-deprived conditions. The increase in GCH1 protein after addition of dbcAMP might be attributable to an augmented mRNA transcript stability (33) and the increase obtained when using histone deacetylase inhibitors might also indicate regulation of transgene transcription by histone acetylation. Further experiments are required in order to ascertain the contribution of each of these mechanisms on the silencing of transgene expression in grafted cells.
These experiments led us to hypothesize that retrovirally transduced cells are able to efficiently synthesize levodopa in culture conditions but, when these cells are intrathecally grafted, the lack of some serum components) in the CSF environment downregulates transgene expression to the point of turning cells inefficient to produce enough levodopa. Due to the fast clearance of levodopa from CSF, its availability in the spinal parenchyma would be so low that it would not be able to increase the dopaminergic turnover and thus no analgesia would be obtained. Nevertheless, the present experiments also suggest several ways of acting to enhance transgene expression of cells that are grafted in the nervous system environments, such as introducing an IRES at the 5′-UTR of the transgene sequence or pharmacologically enhancing transgene transcription.
In conclusion, although ADSCs can be transduced to efficiently produce neuroactive molecules, such as levodopa, their applicability to the treatment of CNS diseases like neuropathic pain will require the development of strategies that keep cells from transgene downregulation or silencing after grafting. On the other hand, serum deprivation of transduced ADSCs in culture provides an interesting in vitro model to study transgene silencing and the ways to tackle it.
Footnotes
Acknowledgments
This research was supported by Fondo de Investigaciones Sanitarias PI 05/1324 and Fundación UPSA grants. We thank K. O'Malley for the gift of TH clones, U. J. Kang for the gift of GCH1 plasmid, and C. Hesslinger for the gift of antibodies to GCH1. We also thank M. Serrano, S. Sacristán, A. Gómez, and P. Flórez for technical help, Drs. J. Menéndez-Cuervo, R. Gonzalo-Gobernado, D. González, D. Piñeiro, and V. M. González for methodological help, Dr. E. Martín for discussions on cap-dependent translation regulation, and C. Correa and A. I. Ortiz for veterinarian assistance.
