Abstract
Adipose tissue contains a mesenchymal stem cell (MSC) population known as adipose-derived stem cells (ASCs) capable of differentiating into different cell types. Our aim was to induce hepatic transdifferentiation of ASCs by sequential exposure to several combinations of cytokines, growth factors, and hormones. The most efficient hepatogenic protocol includes fibroblastic growth factors (FGF) 2 and 4 and epidermal growth factor (EGF) (step 1), hepatocyte growth factor (HGF), FGF2, FGF4, and nicotinamide (Nic) (step 2), and oncostatin M (OSM), dexamethasone (Dex), and insulin-tranferrin-selenium (step 3). This protocol activated transcription factors [GATA6, Hex, CCAAT/enhancer binding protein α and β (CEBPα and β), peroxisome proliferator-activated receptor-γ, coactivator 1 α (PGC1α), and hepatocyte nuclear factor 4 α (HNF4α)], which promoted a characteristic hepatic phenotype, as assessed by new informative markers for the step-by-step hepatic transdifferentiation of hMSC [early markers: albumin (ALB), α-2-macroglobuline (α2M), complement protein C3 (C3), and selenoprotein P1 (SEPP1); late markers: cytochrome P450 3A4 (CYP3A4), apolipoprotein E (APOE), acyl-CoA synthetase long-chain family member 1 (ACSL1), and angiotensin II receptor, type 1 (AGTR1)]. The loss of adipose adult stem cell phenotype was detected by losing expression of Thy1 and inhibitor of DNA binding 3 (Id3). The reexpression of phosphoenolpyruvate corboxykinase (PEPCK), apolipoprotein C3 (APOCIII), aldolase B (ALDOB), and cytochrome P450 1A2 (CYP1A2) was achieved by transduction with a recombinant adenovirus for HNF4α and finally hepatic functionality was also assessed by analyzing specific biochemical markers. We conclude that ASCs could represent an alternative tool in clinical therapy for liver dysfunction and regenerative medicine.
Introduction
Orthotopic liver transplantation is major effective treatment for patients with fulminant and chronic liver failure. However, this clinical practice is highly limited by lack of donors and organ rejection. Applying strategies similar to those tissues use to renovate damaged cells after injury is the main idea behind new concepts of cell therapy and regenerative medicine.
Having identified stem cell plasticity (66), multiple reports raised hopes that tissue repaired by stem cell transplantation could be available in the near future. Therefore, replacing diseased hepatocytes and the stimulation of regeneration by stem cells are the main aims of liver-directed cell therapy.
Human multipotential mesenchymal stem cells (hMSC) have been reported to possibly differentiate into a variety of mesenchymal tissues (i.e., osteoblasts, chondrocytes, adipocytes, myoblasts, and cardiomyocytes), and into various types of tissue cells derived from other embryonic layers, including neural and hepatocyte-like cells (26,27,63,67,78). Although bone marrow was the first source reported to contain hMSC (bone marrow stem cells, BMSC), reports indicate that hMSC may also be isolated from different tissues, including adipose tissue (17,28,37,58,73,96,102). The stromal-vascular cell fraction of adipose tissue has increasingly become the focus of stem cell research (8,69). Adipose tissue contains a multipotent cell population called adipose-derived stem cells (ASCs) (12,37,45,87,102). This cell population can be isolated from human lipoaspirates and differentiated toward osteogenic, adipogenic, neurogenic, myogenic, and chondrogenic lineages (55,68,71,72,101). Recently, its hepatogenic differentiation potential has been demonstrated (3,4,76,85,87,94). Our previous studies demonstrated that, compared with BMSC, ASC population provides an abundant and accessible source of adult stem cells with minimal patient discomfort and, therefore, represents a rich source of MSC that also possesses the longest culture period and the highest proliferation capacity (37,87).
The ability for ASCs to give rise to hepatocye-like cells depends on the induction strategy selected. However, the studies published to date have used distinct hepatocyte differentiation media based on previous protocols applied to non-ASC progenitor cells. Consequently, analyzing the status of the derived cells is not always trivial and can lead to various conclusions. Thus, a thoroughly comparative study defining the best differentiation protocol is needed. Moreover, a battery of cell-specific markers needs to be defined and the cell-type differentiation status proven to study differentiation toward a specific cell type. In addition, because the differentiation process is gradual, it is necessary to discriminate between the genes expressed at each stage of the process. The genes expressed at the early development stages may not be representative of fully differentiated cells.
Here we have examined different hepatogenic induction assays based on the sequential exposure of ASCs to cytokines, growth factors, and hormones reflecting their temporal expression during in vivo hepatogenesis. Our differentiation protocol is based on the use of prohepatogenic signals and mediators of liver development. We have established and optimized a three-step hepatogenic induction protocol by analyzing the role of epidermal growth factor (EGF), fibroblastic growth factors 2 and 4 (FGF2 and FGF4), and bone morphogenic proteins 2 and 4 (BMP2 and BMP4) at the commitment step; hepatocyte growth factor (HGF), FGF2, FGF4, and nicotinamide (Nic) at the differentiation step; and finally HGF, oncostatin M (OSM), dexamethasone (Dex), and insulin-transferrin-selenium (ITS + premix) (28) at the maturation step. Additionally, we have conducted an extensive gene expression characterization of the ASC population at different hepatic differentiation stages, determining not only early and late hepatic markers, but also temporal activation and the role of transcription factors. Hepatic functionality was also assessed by analyzing glycogen synthesis and accumulation, plasma protein synthesis and secretion, and P450 activities as specific biochemical markers.
Materials and Methods
Isolation and Culture of ASC
Human adipose tissue was obtained from discarded tissue after suction-assisted lipectomy (i.e., abdomen liposuction) (10 patients; mean age 43.2 ± 12.1 years). Protocols were approved by the institutional Review Board. Tissues were obtained according to the hospital's Ethics Committee and after obtaining informed consents from patients. ASCs were isolated and cultured as previously described (87).
Isolation and Culture of Human Hepatocytes
Hepatocytes were isolated using a two-step perfusion technique and cultured as described in detail elsewhere (22). All liver samples (n = 5) were obtained in conformity with the rules of the hospital's Ethics Committee and all donors were not suspected of harboring any infectious disease and tested negative for human immumo-deficiency virus and hepatitis.
Cell Lines
Human hepatoma HepG2 cells (ECACC No 85011430) were cultured as previously described (87).
Hepatic Differentiation Protocol
Passage 2 ASC cultures at confluence were used to assess different hepatogenic induction assays over a 2-week period. It is estimated that at least 50 hepatocyte-like cells from each initial ASC are obtained. A three-step differentiation protocol with serum-free DMEM low-glucose medium (Gibco, Paisley, UK), supplemented with 0.3 mg/ml Matrigel (lot 354234, BD Biosciences, Madrid, Spain) and sequential addition of factors and hormones, was used (Fig. 1A). In the commitment step, cells were cultured in medium supplemented with 20 ng/ml EGF (Sigma-Aldrich Química SA, Madrid, Spain) and 10 ng/ml FGF2 (Invitrogen, Barcelona, Spain) (protocol A), or 20 ng/ml EGF, 10 ng/ml FGF2, and 10 ng/ml FGF4 (Sigma-Aldrich Química SA) (protocol B), or 20 ng/ml EGF, 10 ng/ml FGF2 and FGF4, and 50 ng/ml BMP2 and BMP4 (Sigma-Aldrich Química SA) (protocol C). Thereafter, cells were cultured until day 9 with the same media containing Nic 4.9 mmol/L (Sigma-Aldrich Química SA) and 20 ng/ml HGF (PeproTech EC Ltd., London, UK). The maturation step of hepatocyte-like cells was performed with protocol B cultures, assaying three different maturation protocols (B1, B2, and B3) (Fig. 1A). Cells were cultured with the same medium as in step 2 (protocol B1), or with 20 ng/ml HGF, 20 ng/ml OSM (PeproTech EC Ltd.), 1 μmol/L Dex (Merck Pharma, Mollét del Vallés, Spain), and ITS + premix (BD Biosciences, Madrid, Spain) (protocol B2) and finally with 20 ng/ml OSM, 1 μmol/L Dex, and ITS + premix (protocol B3).
Hepatogenic induction protocols for ASC with sequential addition of growth factors, cytokines, and hormones and ASC cell morphology changes during hepatogenic induction. (A) Confluent undifferentiated ASC cultures at passage 2 were treated with different combinations of exogenous factors over a period of 16 days. Phase-contrast microscopy analysis of ASC on day 0 (B), day 9 of protocol B (C), and on day 16 of protocol B3 with (D) or without (E) supplementation of culture medium with matrigel. Original magnification of 20x for all the pictures (scale bars: 50 μm). Abbreviations: ASC, adipose-derived stem cells; EGF, epidermal growth factor; FGF, fibroblastic growth factor; BMP, bone morphogenic protein; HGF, hepatic growth factor; Nic, nicotinamide; Dex, dexamethasone; ITS, insulin-transferrin-selenium.
Transduction of ASC with the Adenoviral Vector Encoding HNF4α
ASCs on day 7 of differentiation protocol B were infected with recombinant adenovirus for the expression of HNF4α (Ad-HNF4α) for 120 min at a multiplicity of infection (MOI) of 4 plaque-forming units/cell (9,31,49). Thereafter, cells were washed with PBS and fresh medium (protocol B) was added. Cells were analyzed 48 h posttransduction.
Flow Cytometry Analysis
A FACScalibur flow cytometer (Becton Dickinson, Madrid, Spain) and CellQuest (Becton Dickinson) software program were used for flow cytometric analysis, as previously described (54). To analyze the surface marker expression, cells from the initial sample, cultured cells and nonadhered cells, were labeled with monoclonal antibodies against human antigens CD13-PE, CD34-APC, CD45-FITC, CD90-APC (Becton Dickinsonn) and CD105-PE (Serotec, Madrid, Spain). Actinomycin D (7-AAD) (Sigma-Aldrich, Madrid, Spain) was used to exclude nonviable cells from the analysis for nonspecific staining.
RNA Extraction and Real-Time Quantitative PCR
Sequence of Primers Used for RT-PCR and Length of Fragments

Gene Expression Profiling by Microarray Analysis
RNA Preparation
Total RNA was purified from matched-pair untreated (control, day 0) and treated (day 16, protocol B3) ASCs from three different donors, and their expression profile was analyzed with the Human Genome U133 Plus 2.0 Array (Affymetrix, Santa Clara, CA). Total RNA was extracted and purified with Trizol reagent following the manufacturer's recommendations. The amount and integrity of purified RNA was estimated by microcapillary electrophoresis (2100 Bioanalyzer, Agilent Technologies, CA), and purity was assessed by the absorbance ratio 260:280 nm.
Amplification and Hybridization
Double-stranded cDNA was generated from total RNA and transcribed to obtain biotin-labeled cRNA according to the Affymetrix standard protocol. cRNA (15–20 μg) was fragmented, combined with hybridization and spike controls, and hybridized with a preequilibrated Affymetrix chip for 16 h. Following hybridization, arrays were washed and stained with streptavidin phycoerythrin conjugate using an automated GeneChip® Fluidics Station 450, and were then scanned with a GeneChip® Scanner 3000 using a 570 nm excitation wavelength laser. Target sample preparation and microarray hybridization and scanning were performed at the Gene Analysis Service, Central Research Unit (UCIM), Faculty of Medicine, University of Valencia (Spain).
Quality Control, Data Analysis, and Statistics
Microarray quality control assessment and data acquisition were performed with the GeneChip® Operating Software (Affymetrix). To permit the correct alignment of the software analysis grid, a control oligo (B2 oligo) was hybridized and used to indicate the boundaries of the probe area. The other quality control metrics used included the average background and noise (Raw Q) of the measured probe cell intensities, the ratio of expression of the 3′ probe set to the 5′ probe set of the housekeeping control gene GAPDH, and the percentage of all the probe sets in the array detected as present. Perfect match (PM) and mismatch probe intensities were used to generate a detection call (present, marginal, or absent) for each probe set, according to the microarray suite version 5 (MAS5) algorithm. Normalization among the different microarray data files and comparative data analysis were performed with DChip (Microarray Analysis Software Package, Harvard School of Public Health). The one-way analysis of variance (ANOVA) followed by Tukey's post hoc test was used to detect significant differences in gene expression between biological conditions (Bonferroni post hoc testing gave similar results). In addition, unsupervised hierarchical clustering analysis and a principal component analysis (PCA) were performed with the DChip software to establish nonforced groups of samples.
Induction of Fat Overload in HepG2 Cells and Human Hepatocytes and Determination of Lipid Content
HepG2 and 24-h cultured hepatocytes were exposed to a free-fatty acid (FFA) mixture (2:1 ratio of oleate and palmitate; Sigma) (23). A 1 mM FFA solution was prepared in culture medium containing 1% bovine serum albumin. Cells were incubated with the FFA mixture for 14 h. Lipid content was determined fluorimetrically by Nile red (Sigma) staining in HepG2 and 24-h cultured hepatocytes exposed and nonexposed to FFA and in ASC at day 16 of protocol B3, as described in detail elsewhere (23).
Glycogen Content Evaluation
Synthesis and intracellular glycogen accumulation was triggered in ASC at day 0 and at day 16 of protocol B3 by a 6-h incubation with a fresh culture medium containing 1 μM insulin and 30 mM glucose. Glycogen content was determined by an enzymatic assay, described in detail elsewhere (24).
Drug Metabolism Assessment
P450 activity assays were performed at different time points by the direct incubation of ASC at day 0 and ASC at day 16 of protocol B3 with specific substrates for P450 isozymes (42). Those metabolites that formed during the activity assays were quantified by using HPLC-MS/MS (Micromass Quattro Micro; Waters, Milford, MA, USA) in the electrospray ionization mode interfaced with an Alliance 2795 HPLC (Waters Chromatography), as described previously (43). Activity values were expressed as pmoles of product formed per hour and per milligram of protein.
Immunoblotting
Cell lysates and extracellular culture media of ASC at days 0, 2, 9, and 16 of protocol B3 were analyzed for APOE, C3, and α2M by Western blot analysis. Proteins were resolved by 7.5% and 12% SDS-PAGE (30 μg protein/lane), transferred to Immobilion membranes (Millipore Corporation, Billerica, MA, USA), and incubated with anti-APOE (1:3000, Chemicon, Millipore Corporation), anti-α2M (1:1000, DakoCytomation, Denmark), and anti-C3 (1:1000, Quidel, San Diego, CA, USA). After washing, blots were developed using an enhanced chemiluminescence kit (Amersham Biosciences UK, Ltd., Little Chalfont, Buckinghamshire, UK).
Results
Flow Cytometry Analysis of ASC From Human Adipose Tissue
To characterize the ASC population, we examined the cell surface marker expression of ASCs from 10 patients undergoing lipectomy using flow cytometry. The major cell population obtained in primary culture was positive for the mesenchymal markers CD13 (89.6 ± 13.9%), CD90 (94.1 ± 6.3%), and CD105 (77.6 ± 28.9%), whereas it was negative for the hematopoietic marker CD133 and only a small percentage of the population (10.9 ± 10.1%) was positive for the hematopoietic marker CD45. The characteristic surface stem cell marker, CD34, showed a higher percentage of positive cells in the biopsy (87.3 ± 8.4%) than in the primary culture (56.9 ± 33.7%), and CD34-positive cells decreased with culture time. Concerning the expression of embryonic markers, the primary culture was negative for SSEA-4, Oct-4, and Sox2. Briefly, our ASC isolation procedure results in a majority MSC population positive for CD90, CD105, and CD13 and negative for the hematopoietic markers CD45 and CD133 and negative for the embryonic markers SSEA-4, Oct-4, and Sox2, which coincides with our previous findings (87).
Morphological Changes of ASCs During Hepatic Transdifferentiation
The morphology of ASCs was analyzed at day 0, at the end of differentiation (day 9), and the maturation (day 16) steps. On day 0, cells exhibited a fibroblastic-like morphology that was observed until day 9 (Fig. 1B, C). OSM and Dex treatment induced a noticeable change to a polygonal shape, characteristic of mature hepatocytes (Fig. 1D). Adding Matrigel to the culture medium maintained the cell culture up to 1 month, showing a higher cellular proliferation rate and longer survival compared with Matrigel-free cultures (data not shown). Besides, Matrigel induces a more pronounced polygonal shape of ASC-derived hepatocyte-like cells (Fig. 1E).
Induction of Early Hepatogenic Differentiation of ASC by FGFs, BMPs, and HGF
To investigate which factors act at defined stages to control hepatogenic differentiation of ASCs, we assessed the expression of multiple transcription regulators. Total RNA from ASC was isolated on days 0, 2, 9, and 16 of culture. Undifferentiated cells (day 0) were used as controls. Expression levels of homeobox protein Hex (Hex) and GATA binding protein 6 (GATA6), CCAAT/enhancer binding protein β (CEBPβ) and alpha (CEBPα) and peroxisome proliferator-activated receptor gamma, co-activator 1α (PGC1α) significantly increased in the first differentiation protocol steps. As Figure 2 illustrates, similar increases in the expression levels of the five hepatic transcription factors were observed in the three differentiation media (A, B, and C), and the maximum was reached at the end of the differentiation step (day 9). However, the expression was similar or slightly lower in the maturation step (data not shown), suggesting that these transcription factors may play a role in the early transdifferentiation process of ASCs toward a hepatic phenotype.
Upregulation of transcription factors in the early hepatic differentiation of ASC. Specific mRNA levels of Hex, GATA6, CEBPα, CEBPβ, and PGC1α were measured by real-time quantitative RT-PCR. The results were normalized to the housekeeping PBGD quantification values. Data were expressed as being relative to undifferentiated cells (day 0) and represent the mean ± SD (n ≥ 4). Abbreviations: ASC, adipose-derived stem cells; Hex, homeobox protein Hex; GATA6, GATA binding protein 6; CEBPβ, CCAAT/enhancer binding protein β; CEBPα, CCAAT/enhancer binding protein α; PGC1α, peroxisome proliferator-activated receptor gamma, coactivator 1α; PBGD, porphobilinogen deaminase; RT-PCR, real time-polymerase chain reaction.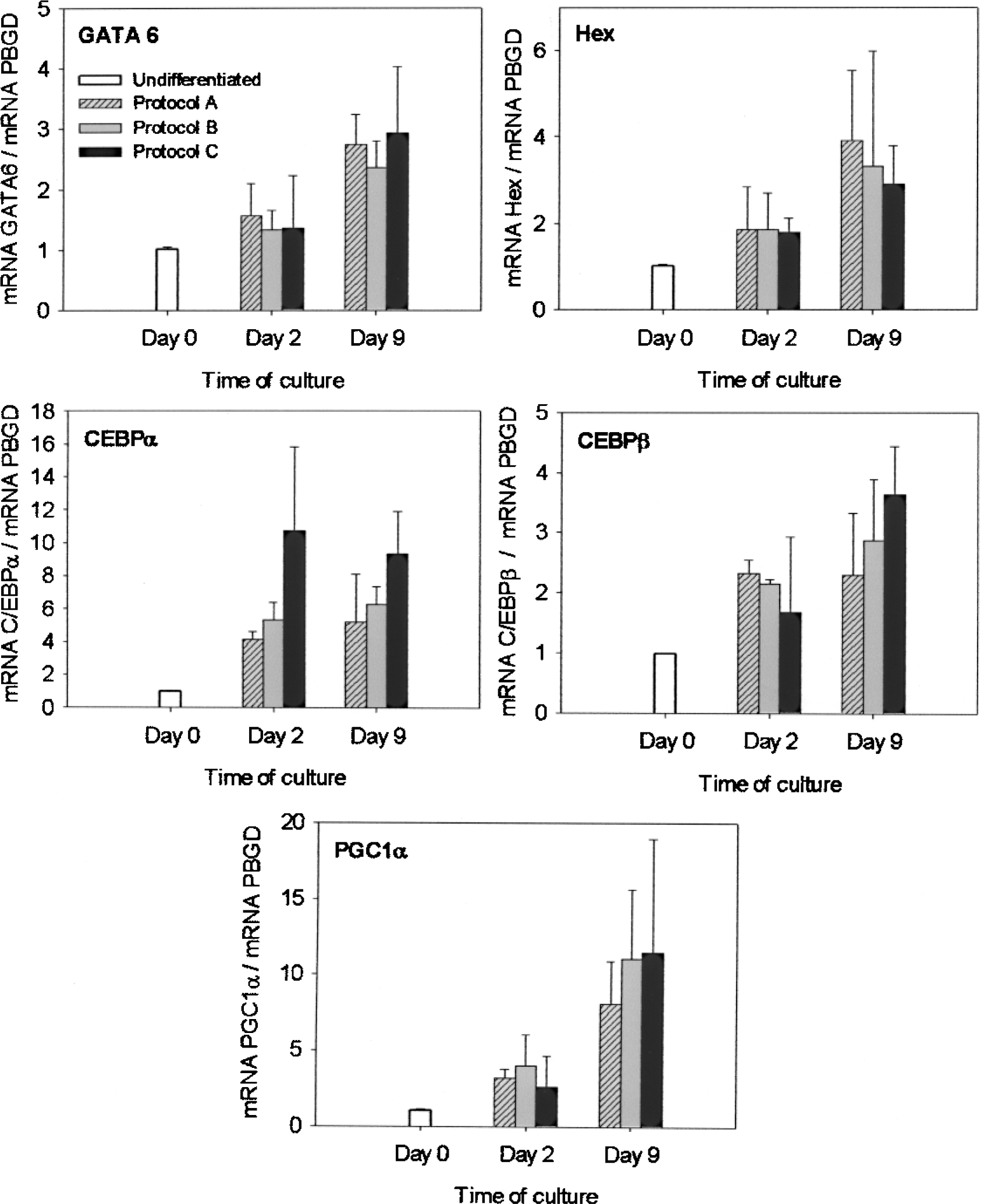
To further demonstrate that these transcription factors are associated with the upregulation of typical hepatic genes, we determined the mRNA expression level of a battery of hepatic genes by RT-PCR and found that several could be selected as early markers of hepatocye differentiation: albumin (ALB), early hepatic marker regulated by CEBPα and CEBPβ, α-2-macroglobuline (α2M), an acute phase protein predominantly synthesized in the liver, complement protein C3 (C3), synthesized by the liver and involved in the complement system and acute phase response, and selenoprotein P 1 (SEPP1) secreted by the liver to act as an antioxidant. The expression levels of ALB, α2M, C3, and SEPP1 markedly increased in cells cultured with the protocols A and B, reaching maximal levels on day 9 (Fig. 3); increases of the ALB expression coincided with the maximal expression of CEBPα and CEBPβ but the cells in the presence of FGF4 (protocol B) showed a higher rate of survival and proliferation (data not shown). However, cells cultured with medium supplemented with BMPs (protocol C) generally showed a smaller increase in the mRNA level of all the hepatic markers on days 2 and 9. This probably suggests that BMPs do not play a critical role in hepatic transdifferentiation of ASC to the hepatic phenotype.
Induction of early hepatic markers during the hepatogenic induction of ASC. Specific mRNA levels of ALB, α2M, C3, and SEPP1 were measured by real-time quantitative RT-PCR. Results were normalized to PBGD values. Data were expressed as being relative to undifferentiated cells (day 0) and represent the mean ± SD (n ≥ 4). Abbreviations: ASC, adipose-derived stem cells; ALB, albumin; α2M, α-2-macroglobulin; C3, complement 3; SEPP1, selenoprotein P plasma 1; PBGD, porphobilinogen deaminase; RT-PCR, real time-polymerase chain reaction.
Induction of Hepatic Maturation of ASC by HGF, OSM, and Dex
The results presented in Figure 3 show that protocols A and B seem to satisfy the requirements to induce an early hepatogenic differentiation of ASCs. However, we selected protocol B based on reports showing that several FGFs, including FGF4, are involved in the regulation of liver development. Thus, the addition of FGF4 (protocol B) would likely enhance the transdifferentiation process. Then, we investigated different conditions for hepatocyte maturation in ASCs cultured with protocol B. A battery of hepatocyte-specific genes were analyzed by quantitative RT-PCR and found that several could be selected as late markers of hepatocye differentiation: cytochrome P450 3A4 (CYP3A4), apolipoprotein E (APOE), acyl-CoA synthetase long-chain family member 1 (ACSL1), angiotensin II receptor type 1 (AGTR1), and HNF4α. As Figure 4 depicts, undifferentiated ASC (day 0) expressed very low or negligible levels of the five late hepatic markers, whereas an increase was already seen on day 9 after commitment and differentiation. ASCs cultured with the same medium until day 16 (protocol B1) showed no further increase in the expression levels of any late hepatic marker. The addition of OSM and Dex combined with HGF and ITS (protocol B2) induced a remarkable increase in ACSL1, but the other markers showed no further upregulation. The largest increase in the mRNA level of CYP3A4, APOE, AGTR1, and HNF4α occurred in cells cultured with medium supplemented with OSM, Dex, and ITS (protocol B3), but deprived of HGF (Fig. 4). These results suggest that the most potent hepatogenic induction is produced by protocol B3.
Induction of late hepatic markers during the differentiation of ASC. Specific mRNA levels of ACSL1, AGTR1, APOE, CYP3A4, and HNF4α were measured by real-time quantitative RT-PCR. The results were normalized to PBGD values. Data were expressed as relative to undifferentiated cells (day 0) and represent the mean ± SD (n ≥ 4). Abbreviations: ASC, adipose-derived stem cells; ACSL1, acyl-CoA synthetase long-chain family member 1; AGTR1, angiotensin II receptor, type 1; APOE, apolipoprotein E; CYP3A4, cytochrome P450 3A4; HNF4α, hepatocyte nuclear factor 4 a; PBGD, porphobilinogen deaminase; RT-PCR, real time-polymerase chain reaction.
Loss of Adipose Adult Stem Cell Phenotype
To investigate the loss of adipose adult stem cell phenotype during the hepatogenic transdifferentiation process, we assessed the mRNA expression level of both Thy1 (CD90), and the inhibitor of DNA binding 3 (Id3), until day 16 (Fig. 5). ASC-derived hepatocyte-like cells obtained with protocol B1 expressed similar levels of Thy1 and Id3 to undifferentiated ASC (Fig. 5). However, the expression levels of both markers significantly decreased in cells cultured with HGF, OSM, Dex, and ITS (protocol B2), while a much more significant decrease was induced by medium supplemented with OSM, Dex, and ITS (protocol B3), particularly in Id3 mRNA. The results suggest that OSM and Dex are apparently critical to induce the loss of the MSC phenotype to levels as low as those in the human liver (data not shown). However, the presence of HGF caused no major effect on the downregulation of Thy 1 and Id3.
Loss of undifferentiated stem cell phenotype in hepatocyte-like derived cells. Specific mRNA levels of Thy1 and Id3 were measured by real-time quantitative RT-PCR. The results were normalized to PBGD values. Data were expressed as relative to undifferentiated cells (day 0) and represent the mean ± SD (n ≥ 4). Abbreviations: Thy1, CD90; Id3, inhibitor of DNA binding 3; PBGD, porphobilinogen deaminase; RT-PCR, real time-polymerase chain reaction.
Gene Expression Patterns in ASC-Derived Hepatocyte-Like Cells
To gain deeper insight into the molecular events during the transdifferentiation of ASC, we compared three matched-pair untreated (day 0) and treated (day 16, protocol B3) ASCs by whole human genome expression analysis. The ANOVA test detected a differential gene expression between biological conditions. We selected genes whose expression varied at least twofold between undifferentiated and differentiated cells, and whose value was p < 0.05. The comparative analysis showed a total of 657 differentially expressed probes representing 498 different genes.
Hierarchical clustering of samples using the differentially expressed gene set segregated samples into two major clustering branches. All the replicate samples of the untreated group self-clustered into one branch while the differentiated samples self-clustered into another (Fig. 6A). The PCA of the whole gene profiles grouped samples into only two clusters, as in the hierarchical clustering analysis (Fig. 6B). Clustering and PCA analyses demonstrated that the transdifferentiation process leads to a cellular population with a substantially different gene expression pattern from the same cell population before treatment.
Global expression microarray analysis of ASC during hepatogenic induction. Total RNA was purified from matched-pair untreated (day 0) and treated (day 16, protocol B3) ASCs from three different donors and their expression profile analyzed with the Human Genome U133 Plus 2.0 Array (Affymetrix). (A) Hierarchical clustering analysis of differentially expressed genes filtered by ANOVA with a value of p < 0.05 (light gray, downregulation genes; dark gray, upregulation genes). (B) Nonforced grouping of samples by principal component analysis (light gray, untreated cells on day 0; dark gray, hepatocyte-like cells on day 16, protocol B3). Abbreviation: ASC, adipose-derived stem cells.
List of Gene Ontology (GO) Categories Significantly Overrepresented in ASC Hepatocyte-Like Cells

In order to identify GO categories with significantly enriched gene numbers, the distribution of genes in the selected gene list in each GO category is compared to those in the reference gene list (i.e., all genes in the Human Genome U133 Plus 2.0 Array). If K out of the N genes in the reference list, and k out of the n genes in the selected list, are in category X, then, if the n selected genes are effectively a random sample uniformly selected from the reference gene list, the expected value of k is ke = (n/N)K (expected gene number). If, on the other hand, the observed k (Observed gene number) exceeds the above expected value, category X is said to be enriched, with a Ratio of enrichment R = k/ke. GOTM software reports only those enrichments that are statistically significant as determined by hypergeometric test (Significance of enrichment).
These results demonstrate that our sequential protocol triggers the adjustment of multiple pathways involved in differentiation, morphogenesis, survival, and hepatic-associated functions in ASCs.
Reactivation of Hepatic Genes by Adenovirus-Mediated Reexpression of HNF4α in ASCs
HNF4α is an essential transcription factor for the constitutive expression of key hepatic genes such as phosphoenolpyruvate corboxykinase (PEPCK), apolipoprotein C3 (APOCIII), aldolase B (ALDOB), and cytochrome P450 1A2 (CYP1A2) (49). Even though a noticeable increase of HNF4α expression was observed on day 16 of the differentiation protocol (Fig. 4), the mRNA levels of these particular HNF4α target genes were negligible (data not shown), suggesting that the expression level of HNF4α was insufficient to induce a functional transcriptional activation of all its hepatic target genes.
To investigate this possibility, ASCs were transduced with an adenoviral vector encoding the transcription factor HNF4α (Ad-HNF4α) on day 7 of the differentiation protocol B. Infection of ASCs with a control adenovirus expressing the GFP demonstrated 80–90% transduction efficiency. Moreover, transduction of ASCs with Ad-GFP or Ad-pAC (insertless adenoviral vector) neither modified the morphology nor the expression of differentiation marker genes (data not shown). Adenoviral-mediated transfection of HNF4α in ASCs caused a highly significant increase in the expression of HNF4α compared with the mRNA levels induced by protocol B3 (the most hepatogenic protocol) (2416 ± 399% vs. 520 ± 254%, percentage of day 0 undifferentiated ASCs). Concomitantly, a significant increase in the expression of its target genes, APOCIII, ALDOB, and CYP1A2, and particularly PEPCK, was observed. As a reference, we compared the levels attained with those in the differentiated hepatoma HepG2 and found comparable or, as with PEPCK, much higher values (Fig. 7). This finding suggests that the forced expression of HNF4α after early hepatogenic differentiation of ASC can effectively improve maturation and the hepatic phenotype.
Transcriptional activation of liver genes by the forced expression of HNF4α ASCs was transduced with an adenoviral vector encoding the transcription factor HNF4α (Ad-HNF4α on day 7 of the hepatogenic protocol B and were collected on day 9. Specific mRNA levels of PEPCK, ALDOB, APOCIII, and CYP1A2 were measured by real-time quantitative RT-PCR and were compared with those in the differentiated hepatoma HepG2. The results were normalized to PBGD values. Data were expressed as relative to undifferentiated cells (day 0) and represent the mean ± SD (n + 4). Abbreviations: ASC, adipose-derived stem cells; HNF4α, hepatocyte nuclear factor 4 a; PEPCK, phosphoenolpyruvate carboxykinase; ALDOB, aldolase B; APOCIII, apolipoprotein C3; CYP1A2, cytocrome P450 1A2; PBGD, porphobilinogen deaminase; RT-PCR, real time-polymerase chain reaction.
Lipid Content in ASC-Derived Hepatocyte-Like Cells
Some of the identified marker genes at early and late differentiation could also be associated with adipogenesis. In order to rule out that our hepatogenic differentiation protocol is inducing concomitant adipogenesis, we investigated whether ASC-derived hepatocyte-like cells accumulate intracellular lipids. To confirm that lipid droplets did not develop while the transdifferentiation process took place, we compared the lipid content in ASC-derived hepatocyte-like cells at day 16 in culture under protocol B3, with control (non-FFA-overloaded), and FFA-overloaded HepG2 cells and human hepatocytes. Fat-overloaded HepG2 and hepatocytes showed a fivefold increase of lipid content, while differentiated 16-day ASC presented similar negligible levels to the control HepG2 cells and human hepatocytes (Fig. 8).
Lipid content evaluation of ASC under protocol B3 for hepatogenic transdifferentiation. Lipid content in FFA-overloaded HepG2 and human hepatocytes was compared with that in 16-day ASC and nonoverloaded HepG2 and hepatocytes. Lipid content was estimated by Nile Red staining after a 14-h induction in overloaded samples. The results are expressed as fluorescence arbitrary units/mg protein. Abbreviations: ASC, adipose-derived stem cells; FFA, free-fatty acid.
Hepatic Functionality Assessment
The results show that glycogen synthesis, after incubating cells with insulin and high concentrations of glucose, was negligible in ASC at day 0 (1.1-fold increase), while differentiated ASC (day 16 of protocol B3) synthesized and stored glycogen at similar levels to the 24-h cultured human hepatocytes (6.3- and 4.1-fold increase, respectively). To demonstrate the functional capacity of differentiated ASC, the activity of several enzymes involved in drug metabolism was analyzed. Table 3 shows an increase in the levels for diclofenac hydroxylation, bufuralol 1′-hydroxylation, S-mephenytoin 4-hydroxilation, chlorzoxazone 6-hydroxylation, and ethoxyresorufin O-deethylation at day 16 of protocol B3. To further confirm the functionality of ASC-derived hepatocyte-like cells, we determined the APOE, α2M, and C3 protein levels in both intracellular and extracellular compartments during the differentiation protocol by immunoblotting analysis. ASC-derived hepatocytes were able to synthesize and secrete these plasma proteins in a time-dependent manner, as shown in Figure 9.
Immunoblotting analysis of hepatocyte specific markers during the hepatogenic transdifferentiation of ASC. Cells were cultured under protocol B3 and monolayers were washed and fresh medium was added at the indicated times. After 6-h incubation, aliquots of the culture medium were taken and the cellular protein of the monolayers was extracted by means of a ready-to-use cell lysis solution (M-PER, Pierce). Total cell protein (30 μg) or 30 μl of extracellular medium were loaded per lane. Coomasie blue staining of membranes was performed to check for equal loading (not shown). Western blot analysis indicated that protocol B3 induced the synthesis and secretion of APOE, C3, and α2M in ASC-derived hepatocyte-like cells in a time-dependent manner. Abbreviations: ASC, adipose-derived stem cells; APOE, apolipoprotein E; α2M, α-2-macroglobulin; C3, complement 3. Enzyme-Selective P450 Activities in ASC Before and After Hepatic Transdifferentiation

Discussion
Human ASCs have been reported to transdifferentiate in vitro into several cell lineages (20,102), including hepatocyte-like cells (3,5,76,87). Therefore, the multipotent nature of ASCs, together with their easy extraction and isolation, make these cells a promising alternative for many tissue engineering strategies, autologous cell-based therapies, and preclinical drug testing.
Great effort has been made in the last decade to identify the factors regulating the onset of hepatogenesis and embryonic liver development (14,99). Based on these findings, different strategies have been attempted to induce hepatogenic transdifferentiation of ASCs into functional hepatocyte-like cells. A first approach was the use of a cocktail of exogenous factors (39,76), while other reports (3–5,80), including ours (87), used multistep transdifferentiation protocols by sequential exposure of ASC to growth factors, cytokines, and hormones, reflecting their temporal expression during liver embryogenesis in vivo. All these reports have demonstrated the hepatogenic potentiality of ASC by the detection of classical hepatic markers and biochemical functions (3–5,76,78,80,87). However, the hepatogenic media used and the differentiation markers determined in these previous studies differed remarkably.
Induction of hepatogenic transdifferentiation in vitro of ASC is a process that likely differs from embryonic liver development. Consequently, in vitro differentiation protocols attempting to experimentally reproduce the embryonic development of the liver may not adequately mimic the cross-lineage commitment between ASCs and hepatocytes during dedifferentiation–transdifferentiation processes.
BMPs are not only important for liver development, but also for hepatic specification of mouse and zebrafish embryonic stem cell-derived definitive endoderm (25, 79). However, and in agreement with our findings, a lack of requirement for BMP4 addition was demonstrated in the generation of hepatocytes from human embryonic stem cells (hESCs) (1). The disparity between these observations could reflect species variations. Alternatively, it has been shown that addition of HGF restored growth in mutant mouse liver explants impaired in BMP signaling via TGF-β receptors, suggesting that HGF may provide parallel signals to TGF-β-induced pathways (92).
Different FGF signals appear to initiate distinct liver development phases during mammalian organogenesis (33). The positive influence in hepatic differentiation of FGF2 (45,61,87) and, more recently FGF4, either alone or combined with EGF (16) or HGF in ASCs, have been reported (3–5,80). FGF4 is one of the factors secreted by septum transversum mesenchyme and cardiogenic mesoderm at the early stages of endoderm formation in liver development in vivo, suggesting that related molecules probably function in a redundant capacity (77). In addition, exogenous FGF4 has been recently reported to increase the rate at which MSCs proliferate, and has no significant effect on MSC pluripotency (18). Our results show that the most consistent early hepatic transdifferentiation of ASCs is achieved with the differentiation medium supplemented with both FGF2 and FGF4 combined with EGF, HGF, and Nic (protocol B).
HGF has been reported to play an essential role in the early stages of hepatogenesis (38,75) and in liver regeneration after partial hepatectomy (60). Therefore, HGF and Nic, which enhances the in vitro differentiation of fetal liver cells (51,74), were included in our hepatogenic protocol, as in others (3,39,45,76,87). Our results point to a crucial role for HGF in the commitment and differentiation of ASCs, but it is detrimental for the last maturation step. Some studies show induction of hepatocyte-like phenotype after exposure of hMSC to HGF alone (91), or in cocktails combined with other factors (39,76,80). However, many reports like ours propose multistep hepatogenic protocols in which hMSC are exposed to HGF to induce hepatic commitment and differentiation, but not at further maturation stages of hepatocyte-like cells (3–5,61,80,87).
Our results also demonstrate a critical role for OSM and Dex during the maturation step. This finding is consistent with the role reported for OSM to promote hepatoblasts differentiation to mature hepatocytes (56,86), that OSM in combination with glucocorticoids is required to induce maturation of liver stem and progenitor cells (34,35), and that hormones, glucocorticoids, and insulin are involved in the late maturation stage leading to increased liver-specific gene expressions (11).
Another key point investigated was the relevance of the extracellular matrix (ECM). It has been reported that ECM components contribute to hepatoblast differentiation (14,86). Our results show that supplementing culture medium with Matrigel induced a higher cellular proliferation rate and longer survival of ASC-derived hepatocyte-like cells.
The term transdifferentiation refers to the phenotypic change of one differentiated cell type to another lineage. It has been suggested that precommitted cells could change their phenotype during transdifferentiation by dedifferentiating into a primitive stem cell stage, perhaps through genome reprogramming (81). We did a whole genome expression analysis and observed a large readjustment in the expression profile of ASC during hepatogenic transdifferentiation with many genes related to development, proliferation, and differentiation undergoing important expression adjustment, which suggests a profound reprogramming event. Most of the findings agree with those previously reported by Yamamoto et al. (94).
Several transcription factors involved in liver development have been characterized, many of which were originally identified as regulators of liver-specific genes in the adult liver. GATA6 is the most critical player to regulate the differentiation of hepatoblasts (83,99). Hex, a homeobox transcription factor, is the earliest identified marker for endoderm that will form liver (7). Both are essential factors in hepatic specification and the commitment to hepatogenic cell fate (36,98,99). However, medium supplementation with FGF4 and BMPs during steps 1 and 2 led to no noticeable differences in the induction of GATA6 and Hex, thus suggesting that FGF4 and BMPs, present in protocols B and C, do not likely play a critical role in the induction of GATA6 and Hex during ASC transdifferentiation.
C/EBP factors are master regulators of liver development, and the early expression of C/EBPα and C/EBPβ exclusively in the early liver bud has been reported (89,93). It has also been reported that HGF induces the expression of C/EBP factors (86). PGC1α is an important coactivator for the control of metabolic homeostasis, and developmental regulation of PGC1α coactivation can be an essential step to build up the appropriate regulatory machinery for liver energy metabolism. In the mouse embryo liver, the expression of PGC1α is high (97) and parallels that of HNF4α and HNF6 (6). They are present in early development but their concentration steadily increases in late development up to a threshold level that is functional for some liver-specific target genes (6). Thus, synergism, interdependency, and threshold concentrations of transcription factors and coactivators could determine the time-specific expression of hepatic-specific genes during ASC transdifferentiation to hepatocytes.
Like GATA6 and Hex, CEBPα, CEBPβ, and PGC1α expressions during hepatogenic induction of ASCs revealed no significant response to FGF4 or BMPs. Moreover, the expression of these five transcription regulators increased during the first differentiation protocol steps (steps 1 and 2), but none showed further increase during step 3 (maturation). In this sense, the only transcription factor analyzed showing a significant upregulation during the maturation step was HNF4α.
HNF4α is essential for hepatocyte differentiation during later development and crucial for metabolic regulation and liver function (46). The prominent role of HNF4α during hepatocyte late development is consistent with a moderate increase of HNF4α expression at the end of the differentiation step (step 2), which becomes more noticeable in step 3 (maturation) of protocols B2 and B3. Our results are also consistent with previous observations, showing that GATA6 regulates HNF4α during development in a transcriptional cascade that controls the differentiation of the visceral endoderm (57). Moreover, C/EBPα and HNF3β are also important for sustaining the high HNF4α expression during liver development (15,41).
More recently, studies have shown that HNF4α is critical for hepatic morphogenesis. HNF4α deficiency in embryonic mouse liver results in abnormal tissue architecture and a lack of appropriate cell–cell contacts (62), while its overexpression induces cell polarity in F9 embryonic carcinoma (10), H5 hepatoma (82), and even in NIH3T3 fibroblast (62) cells. During ASC transdifferentiation, we observed a mesenchymal–epithelial transition. In steps 1 and 2, cells developed a fibroblastic-like flattened shape, but changed to a polygonal morphology in the presence of OMS and hormones in step 3 when the HNF4α expression reached the highest level.
Given the particular relevance of HNF4α in the hepatic phenotype, we decided to further investigate the level of functional activation attained in our ASC-derived hepatocytes. Despite a noticeable increase of HNF4α on day 16 of the differentiation protocol, we observed that the mRNA levels of some typical HNF4α target genes were negligible (data not shown), suggesting that the HNF4α expression level reached was likely insufficient to induce fully functional transcriptional activity. To investigate this possibility, ASCs were transduced with an adenoviral vector encoding the transcription factor HNF4α on day 7 of differentiation protocol B. This upgrading caused a highly significant increase in HNF4α mRNA compared with those attained with protocol B3 and, concomitantly, a significant increase in the expression of its target genes, APOCIII, ALDOB, and CYP1A2, and particularly PEPCK, was observed. Therefore, the adenovirus-mediated expression of HNF4α could be an effective procedure to attain full maturation of ASC-derived human hepatocytes.
After the commitment and differentiation steps, we found robust increases in the expression levels of ALB, α2M, C3, and SEPP1. Transcription of ALB, α2M, and C3 is controlled by C/EBP factors (21,32,48,52,53) and, accordingly, their induced expression correlated with the upregulation of CEBPα and CEBPβ in ASC-derived cells. SEPP1 is another hepatic-specific gene that is likely responsive to GATA binding proteins (84,95). Our previous work demonstrated that undifferentiated cells stain negatively by immunocytochemistry for albumin and AFP, while homogeneous positive staining was reported in ASC-derived hepatocyte-like cells (87). Despite the extended use of ALB as an early hepatic marker, our results demonstrate that α2M, C3, and SEPP1 are more robust markers for early hepatogenic differentiation of ASC.
During the maturation step of ASC, no further significant upregulation of ALB, α2M, C3, and SEPP1 was observed, whereas a very significant induction of CYP3A4, APOE, AGTR1, and ACSL1 mRNAs was noted, suggesting that these latter genes could be considered markers of mature hepatogenic differentiation. CYP3A4 and APOE expressions in the liver are controlled, at least in part, by HNF4α (2,50,88,90), and in agreement with these previous studies, we found a correlation between the upregulation of HNF4α and the increase in APOE and CYP3A4 during hepatogenic maturation of ASC.
ACSL1 and AGTR1 can also be considered liver-specific genes. The activation of ACSL1 in ASC could be induced by OSM, as it has been demonstrated that this cytokine can induce the expression of the homolog family members ACSL3 and ACSL5 in human hepatoma HepG2 cells (100). The mechanism responsible for the remarkable induction of AGTR in ASC deserves further investigation. The measurement of the selected late hepatic markers under different conditions demonstrated that exposure of ASC to OSM, Dex, and ITS (protocol B3) triggers the more efficient maturation toward the hepatic lineage.
The loss of the adipose MSC phenotype during the hepatogenic transdifferentiation process has not been previously investigated by others. Our results show that ASCs decrease in both the expression of the adult mesenchymal cell surface marker Thy1 and the undifferentiated status gene Id3. Thy1-positive cells were seen to decrease during early fetal rat liver development (29). Thy1 is also expressed in fetal hepatic cells throughout liver development (19), but is only expressed in oval cells in the adult liver, and not in mature hepatocytes (13,29,44,65). Similarly, Id3 has been shown to be a positive regulator of hepatoblast proliferation, but is also an inhibitor of their differentiation into hepatocytes during the development of the chick liver (59). Id3 is active in undifferentiated tissues and organs that are undergoing the differentiation process, but with proceeding development, Id3 shows a decreased expression and becomes restricted to various tissues, such as olfactory epithelium, kidney, and teeth (30). In fact, the knockdown of endogenous Id3 sensitizes epithelial cells to BMP, leading to a robust growth inhibition and induction of transdifferentiation (40). The expression of other immature markers like AFP was well characterized in ASC in a previous study from our laboratory (86). The expression of Thy1 and Id3 was elevated in undifferentiated ASC until day 9, but a significant decrease of both markers was observed in step 3 in hepatocyte-like cells obtained with protocols B2, and particularly B3, suggesting glucocorticoids and/or OSM are likely mediators of their repression during maturation of ASC-derived hepatic-like cells. Alternatively, the suppression of FGFs could trigger Thy1/Id3 downregulation.
In spite of a certain overlap in the expression profile between the adipogenic and hepatogenic lineages, the hepatocyte-like population at day 16 of protocol B3 shows very similar levels of lipid content to those detected in HepG2 and primary hepatocyte cultures, thus confirming that lipid droplet accumulation does not occur while hepatic transdifferentiation takes place.
Under hepatogenic conditions, we have shown that ASCs differentiate into cells with the characteristic morphology and phenotype of hepatocytes. In addition, the acquisition of functional characteristics of hepatocytes by transdifferentiated ASCs has been shown: the activity of several P450 enzymes involved in drug metabolism (Table 3), the capability to synthesize and store glycogen in a time-dependent manner, and synthesis and secretion of plasma proteins to culture medium (Fig. 9). These results do not completely demonstrate that our protocol triggers differentiation into fully mature hepatocytes. Comparison with data from cultured human hepatocytes indicates that the levels attained in differentiated ASC are still below those found in the adult hepatocyte (data not shown). However, it has been demonstrated that it is not necessary (or is even detrimental) to obtain fully differentiated hepatocytes when cell therapy is the final goal (76). Most in vivo models have aimed to investigate cell therapy in diseased livers that require repopulation by infused cells, which may require a less differentiated phenotype that will prove advantageous to donor cells to engraft and proliferate into the injured liver parenchyma (47).
Collectively, our experimental evidence demonstrates that the selected sequential protocol not only induces a hepatic-specific expression profile, but also leads to the loss of the mesenchymal undifferentiated phenotype. Moreover our results suggest, for the first time, the relevance of specific transcription factors (Hex, GATA6, C/EBPs, PGC1α, and HNF4α) in promoting hepatocyte-derived MSC and uncover new informative robust markers for the step-by-step transdifferentiation of hMSC into hepatocytes.
Footnotes
Acknowledgments
The authors acknowledge the financial support from the ALIVE Foundation, Instituto de Salud Carlos III, Fondo de Investigaciones Sanitarias (PI 070550), and European Commission. A.L. gratefully acknowledges Ministerio Ciencia e Innovación/Instituto de Salud Carlos III for a Miguel Server contract.
