Abstract
Embryonic stem (ES) cell-derived cell products may serve as a source of cells for regenerative medicine. Currently available technologies for the induction of ES cells into neural lineage cells require extended culturing in vitro and complex procedural manipulations, with variable yields of heterogeneous cells, which have hindered the prospective use of cell derivatives for treatment of ischemic stroke. We established a simple and efficient method to derive mouse ES cells into neural lineage cells using an 8-day coculture with the bone marrow stromal cells MS5, followed by a 6-day propagation culture and a 4-day selection culture. The protocol generated a relatively high yield of neural lineage cells without any mesodermal and endodermal lineage commitment. In in vivo study, these derived cells could improve the cognitive function of ischemic stroke mice. Three weeks after transplantation, migration of implanted cells to lesioned areas was noted. It was also evident of a normalization of pyramidal neuron density and morphology in hippocampal CA1 region. One (1/17) episode of teratoma development was noted. Data suggested that MS5 cells may exert a neurotrophic effect to enhance neural differentiation of ES cells and MS5-induced ES cell-derived cells appeared to be applicable to cell therapy for ischemic stroke.
Introduction
Stroke, particularly ischemic stroke, strikes millions of people worldwide. Current therapies are mainly for primary or secondary prevention (16,19). There is no effective treatment to improve the neurological functions of patients after stroke pertaining to cell death or degeneration. A majority of the survivors suffer from long-term to permanent neurological disorders. Embryonic stem (ES) cells characterized by pluripotency have been found to in vitro differentiate into many clinically relevant cell types including neurons, glias, cardiomyocytes, insulin-secreting cells, and others (30). Thus, cell therapy may represent a novel treatment. The challenge remains on how to effectively direct pluripotent ES cells into a specific cell fate for clinical application.
As for targeted neural differentiation of ES cells, it has been shown that neural fates emerge in the absence of serum and some growth factors in multicellular aggregates or in suspension culture of single cells, though the efficiency is very low (31–33). It was reported that higher yields of neural cells could be attained by using retinoic acid (2). Stromal cells PA6 were shown to induce ES cells into neural lineage cells in coculture systems suggesting the potential of employing feeder supports mimicking the microenvironment of injured or reactive brain for ES cell induction (11). However, the retinoic acid that was used at concentration far beyond the physiological dose is teratogenic. PA6 was noted to induce a heterogeneous population of ES cell-derived neural cells at various stages of differentiation together with some nonneural lineage cells and undifferentiated ES cells (21), thus limiting its application in cell therapy.
The murine bone marrow stromal cells MS5 have the capacity to support the growth of hematopoietic stem cells (10). The neurotrophic effect on ES cells was largely understudied. Here we report the use of MS5 in a noncontact coculture system to differentiate ES cells into neural lineage cells. MS5-induced ES cell-derived cell product implanted onto the brain of ischemic stroke mice was able to improve the cognition function, suggesting its therapeutic potential for stroke.
Materials and Methods
ES Cell Cultures
Mouse ES cell lines, D3 [CRL-1934, American Type Culture Collection (ATCC), Manassas, VA] and E14TG2a (CRL-1821, ATCC) were used. Undifferentiated ES cells were maintained on 80-Gy γ-irradiated primary mouse embryonic fibroblasts preestablished on gelatinized tissue culture dishes (10 cm in diameter) in high-glucose KnockOut Dulbecco's modified Eagle's medium (KO-DMEM, Invitrogen, Carlsbad, CA) supplemented with 15% ES cell-qualified fetal calf serum (FCS, Invitrogen), 2 mM L-glutamine (Invitrogen), 100 U/ml penicillin, and 100 μg/ml streptomycin (Invitrogen), 0.1 mM nonessential amino acid (Invitrogen), 0.2 mM β-mercaptoethanol (Sigma, St. Louis, MO), and 1000 U/ml leukemia inhibitory factor (LIF, Millipore, Billerica, MA). ES cells were passaged on alternate day with 0.05% trypsin containing 0.5 mM ethylene-diamine-tetra-acetic acid (EDTA, Invitrogen).
MS5 Cocultures
Confluent culture of the murine bone marrow stromal cells MS5 were trypsinized and mitotically inactivated by using 80-Gy γ-irradiation. Cells at 1 × 105 in RPMI (Invitrogen) supplemented with 2 mM L-glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, and 10% FCS were seeded onto tissue culture inserts housed to 12-well tissue culture plates (ThinCerts, Frickenhausen, Germany). Neuroblastoma cell lines, SK-N-AS (CRL-2137, ATCC), SH-SY5Y (CRL-2266, ATCC), and SK-N-MC (HTB-10, ATCC), were plated at 5 × 103 cells/well in 12-well plates with tissue culture inserts of inactivated MS5 and cultured for 8 days in supplemented RPMI. Culture medium and inserts of newly prepared feeder cells were changed every 4 days.
Preparation of MS5-Conditioned Medium (CM)
Mitotically inactivated MS5 cells at 1 × 107 cells in 10 ml serum replacement medium (SRM) made up of KO-DMEM with 15% serum replacer (SR, Invitrogen), 2 mM L-glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin, and 0.1 mM nonessential amino acid were seeded onto 75-cm2 tissue culture flasks and maintained at 37°C for 4 days. The spent medium was collected daily for 4 consecutive days and centrifuged at 400 × g for 10 min to remove cellular debris. The supernatant was kept at −80°C and six batches of CM were pooled for use thereafter.
Induction, Propagation, and Selection Cultures of ES Cells
In the induction culture of ES cells, under the influence of MS5 cocultures, SRM was used to culture 1 × 105 ES cells and refreshed every 2 days. Upon completion of the 8-day induction, ES cell-derived cells were enzymatically segregated using trypsin-EDTA and 1 × 105 cells were further cultured in serum-free medium (KO-DMEM with 2 mM L-glutamine, 100 IU/ml penicillin, and 100 μg/ml streptomycin) supplemented with insulin (5 μg/ml, Sigma), transferrin (5 μg/ml, Sigma), selenium (5 μg/ml, Sigma), and fibronectin (10 μg/ml, Sigma) for 6 days, and finally in neurobasal medium (Invitrogen) supplemented with 100 U/ml penicillin, 100 μg/ml streptomycin, 0.2× N2, and 0.8× B27 (Invitrogen) for 4 days. The medium was changed every other day.
Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
Total RNA was extracted from stromal cells MS5, undifferentiated ES cells, ES cell-derived cells, and control mouse tissues/cells [ischemic brain, embryonic heart and liver, embryos, embryonic fibroblasts and neural precursor cell line C17.2 derived from neonatal mouse cerebellum (a kind gift from the late Dr. David Walsh, University of New South Wales, Australia)] using an RNeasy Extraction Kit (Qiagen, Hilden, Germany) and the manufacturer's instructions. cDNA were transcribed from 2 μg total RNA with 100 ng random hexamers (Invitrogen) and 200 U Superscript reverse transcriptase (Invitrogen) in reverse-transcription reaction mix.
Five microliters of the reverse-transcription product of the first strand cDNA were used for amplification of a specific gene sequence in 25 μl of reaction mix containing 20 mM Tris-HCl buffer (pH 8.4), 50 mM potassium chloride, 1% Tween 20 (Sigma), 100 μM dNTP, 6 pmol of each primer, 0.5 U Platinum Taq DNA polymerase (Invitrogen), and 1.6–2.5 mM magnesium chloride. Details of primer pairs, thermal profiles, cycle numbers, and product sizes of Oct-4, Otx-1, Pax-6, Nestin, Nurr-1, Brachyury, Myf-5, Nkx-2.5, α-fetoprotein, GATA-4, CXCR-4, ciliary neurotrophic factor (BDNF), nerve growth factor (NGF), glia-derived neurotrophic factor (GDNF), neurotrophin-3 (NT-3), brain-derived neurotrophic factor (CNTF), glial fibrillary acidic protein (GFAP), insulin-like growth factor-I (IGF-I), insulin-like growth factor-II (IGF-II), erythropoietin (EPO), vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), CXCL-12, and Wnt-3A were reported previously (7). RNA integrity was confirmed by RT-PCR of a ubiquitous mRNA glyceraldehyde-3-phosphate dehydrogenase (GAPDH). Results were confirmed in at least two separate analyses.
Cell Labeling
ES cell-derived cells were labeled with 10 μM bromodeoxyuridine (BrdU; Roche, Basel, Switzerland) in supplemented neurobasal medium for 24 h prior to transplantation to ischemic animals. Cells were then trypsinized, washed, and adjusted to 1 × 105/μl in Dulbecco's phosphate-buffered saline (PBS; Invitrogen).
Preparation of Mice for Cerebral Ischemia
Animals were cared for and handled according to Guidelines of Laboratory Animal Services Centre in The Chinese University of Hong Kong. Forty adult male ICR mice of a mean age of 12 weeks, weighing 25–30 g, were anesthetized by intraperitoneal injection of ketamine (100 mg/kg; FarmaVet, Alfasan, Romania) and xylazine (10 mg/kg; FarmaVet). A cervical incision was sterilely made on the midline of the ventral neck. Bilateral common carotid arteries were exposed and then occluded with aneurysm clips for 20 min. The incision was sutured after removal of the clips. Eight control mice were sham operated but bilateral common carotid arteries were not occluded. Mice were nurtured in microisolator cages for 3 days prior to cell transplantation.
Cell Transplantation
Ketamine- and xylazine-sedated mouse was fixed onto the stereotaxic frame (Kopf, Tujunga, CA). An incision was sterilely made on the midline of the head and two burr holes were drilled. With a Hamilton micro-syringe fitted with a 26-gauge blunt needle, 5 × 105 ES cell-derived cells in 5 μl PBS were slowly injected into each caudate putamen (0.5 mm anterior to bregma; 2 mm mediolateral to the mean line, and 4 mm ventral to the dura) of 17 mice at a rate of 1 μl per minute. Equal volume of PBS was injected into seven ischemic control mice. The needle was slowly withdrawn in 10 min. The skull holes were cemented and the skin was sutured.
Behavioral Assessment
The spatial learning and memory ability of the mice were evaluated by using the water T-maze system (Model SH-1, Shanghai Shibo Biotechnology, China) as reported previously (7). The water T-maze was a rectangular labyrinth, with 8-cm-wide pathways and four blind alleys diverging from the through-path. A mouse was placed at the starting point facing the wall and allowed to reach the ladder to exit and escape from the water. Mice that could get out of the maze in approximately 2 min were recruited to in vivo studies. Two weeks posttransplant, mice were trained daily for 3 days prior to formal assessments on 2 consecutive days. The time latency in the water maze and episodes of entering blind alleys were recorded. The randomization of sham-operated normal mice and mice with and without cell therapy was blind to the investigators to avoid conscious and subconscious bias during behavioral assessment.
Immunofluorescence Study
To prepare cells for immunofluorescence, cells (1 × 105) were either cyto-spun onto a glass slide or allowed to grow on poly-L-ornithine-coated (Sigma) and laminin-coated (Sigma) coverslips. They were fixed in 4% paraformaldehyde (Sigma) for 20 min. Paraffin-embedded brain sections were dewaxed and antigen retrieval was conducted by microwaving for 20 min. Cells/sections were permeabilized for 5 min with 0.3% Triton X-100 (Sigma) in PBS and then rinsed three times with PBS. Nonspecific binding was blocked with 10% normal goat serum (NGS; Zymed, London, UK) in PBS for 10 min. Cells/sections were washed with 1% NGS in PBS and incubated overnight at 4°C with the following primary antibodies diluted in PBS containing 1% NGS: IgM anti-stage-specific embryonic antigen-1 (SSEA-1, 1:100; Santa Cruz Biotechnology, Santa Cruz, CA), rabbit polyclonal anti-Musashi-1 (1:100; Millipore, Billerica, MA), IgG1 anti-nestin (1:400; BD Biosciences, San Francisco, CA), IgG1 anti-class III β-tubulin (TuJ-III, 1:1,000; Exbio, Prahy, Czech), IgG1 anti-GFAP (1:50; Zymed), IgG2b anti-myelin basic protein (MBP, 1:100; BD Biosciences).
After three washes with PBS, cells/sections were incubated for 30 min with the corresponding secondary antibody: fluorescein isothiocyanate (FITC)-conjugated goat anti-mouse IgM (1:100; Zymed), FITC-conjugated goat anti-mouse IgG (1:100; Zymed), or FITC-conjugated goat anti-rabbit antibody (1:100; Zymed). Washed cells were counterstained with propidium iodide (PI; Sigma) and visualized using an inverted fluorescence microscope. The percentage of immunoreactive cells was enumerated by scoring five randomly selected fields in each culture of three to five independent cultures. To visualize BrdU-labeled cells, cells/sections were incubated for 30 min with FITC-conjugated anti-BrdU antibody (1:1,000; Roche) and then counterstained with 4′,6′-diamidino-2-phenylindole hydrochloride (DAPI; Sigma). Cells/sections without primary antibody incubation were processed in the same manner as controls of false positivity.
Tissue Processing and Histology
Mice were transcardially perfused with 4% paraformaldehyde. Excised brains in either coronal or lateral plane were embedded in paraffin for sectioning at 4 μm thickness. Sections were dewaxed and stained with hematoxylin and eosin or thionin. Images of every fifth serial coronal sections of thionin-stained CA1 region of the hippocampus 1.8–2.0 mm posterior to bregma were captured and overlaid to an image of a counting frame 1 × 0.25 mm derived from the Neubauer hemocytometer. The preponderance of pyramidal neurons with large nuclei, discrete nucleoli, and clear cell periphery derived from the hippocampal CA1 region were enumerated as reported by Nakatomi et al. (23). The morphology of the hippocampal CA1 region was graded in a single-blind manner (0: no necrosis or <10% of total cells with pyknotic morphology within the counting frame; I: scattered single cell necrosis or 10–40% of total cells with pyknotic morphology; II: 40–70% of total cells with pyknotic morphology; III: almost complete cell necrosis or >70% of total cells with pyknotic morphology) (15).
Statistical Analysis
Data derived from at least three independent experiments were presented as mean ± SD, unless stated otherwise. The t-test was used to compare cell viability and relative frequencies of neural cell subtypes derived from cocultures with and without MS5. ANOVA in combination with Newman-Keuls post hoc analysis was used to evaluate the time latency and error incidence within the water maze, and the neuron density in the hippocampal CA1 regions of normal mice, ischemic mice, and transplanted mice. Ranked data of the histological grading of pyramidal neurons in the hippocampi of ischemic mice with and without cell therapy were compared using Kruskal-Wallis test followed by revised t-test or Mann-Whitney test. The software SPSS 13.0 (SPSS Inc., Chicago, IL) was used and values of p ≤ 0.05 were considered statistically significant.
Results
Neurotrophic and Neuroprotective Effects of MS5 Cells
Figure 1 shows RT-PCR for genes encoding BDNF, CNTF, GDNF, NGF, NT-3, IGF-I, and IGF-II. Neuroprotective factors, EPO and VEGF, were also detected. Besides, it was noted that MS5 cells expressed the growth-promoting bFGF and the chemokine CXCL-12, but not Wnt-3A. These data suggested that MS5 cells may exert neurotrophic and neuroprotective effects. The hypothesis was attested in 8-day noncontact cocultures of neuroblastoma cell lines, SK-N-AS, SH-SY5Y, and SK-N-MC, with MS5. The mean numbers of viable SK-N-AS, SH-SY5Y, and SK-N-MC in four separate experiments of 16 replicate cocultures showed 2.0-, 1.6-, and 2.2-fold increase, respectively, compared to control cultures run in parallel. Immunostaining demonstrated significantly higher numbers of class III β-tubulin+ (TuJ-III+) neurons derived from cocultures of neuroblastoma cells with MS5 (SK-N-AS: with vs. without MS5, 34.8 ± 4.2% vs. 25.9 ± 9.2%, p = 0.027; SK-N-MC: 23.7 ± 4.0% vs. 16.8 ± 4.0%, p = 0.004), indicating the neurotrophic effect of MS5.
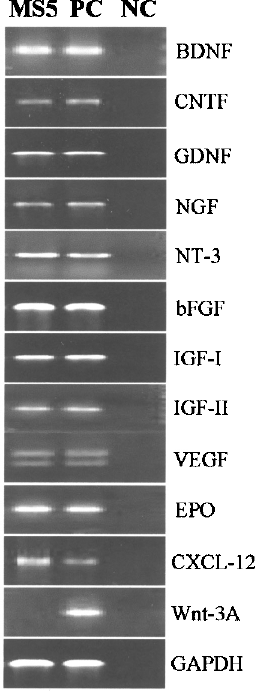
Ethidium bromide-stained agarose gel of electrophoresed RT-PCR products. PC: positive control (mouse ischemic brain for BDNF, GDNF, NGF, NT-3, bFGF, IGF-I, IGF-II, VEGF, CXCL-12, Wnt-3A, and GAPDH; mouse embryonic fibroblasts for EPO and CNTF); NC: negative control (reagent blanks).
MS5-Mediated ES Cell Induction
In four separate experiments of 16 replicate noncontact cocultures of ES cell D3 with MS5 for 8 days, a progressive increase in the number of colonies with protruding processes was noted (Fig. 2A and B). There were upsurges of 2.0- and 2.2-fold of nestin+ and Musashi-1+ neurospheres, respectively, compared to those derived from control cultures (Fig. 2C and D). The cell viability was comparable among cocultures with and without MS5. However, the total numbers of viable cells derived from cocultures with MS5 were significantly higher than those of the control cultures (D3 with vs. without MS5: 3.23 ± 0.38 × 105 vs. 1.19 ± 0.27 × 105, p = 0.0003), suggesting the growth-enhancing effects of MS5 coculture on ES cell-derived cells. Immunocyto-chemistry on enzymatically segregated D3-derived cells revealed TuJ-III+ neurons, GFAP+ astrocytes, and MBP+ oligodendrocytes accounting for 10.6 ± 2.0%, 3.8 ± 1.5%, and 3.7 ± 1.4%, respectively, whose incidences were significantly higher than those of ES cell D3 control cultures without MS5 induction (Fig. 3). Similar scenarios were noted in noncontact cocultures of ES cell E14TG2a with MS5 (11.7 ± 2.0% TuJ-III+ neurons, 4.5 ± 1.4% GFAP+ astrocytes, and 2.5 ± 1.2% MBP+ oligodendrocytes).

Morphology and immunostaining of nestin and Musashi-1 on ES cell D3-derived cell colonies cocultured with MS5 for 8 days. (A) Day 4 culture, (B) day 8 culture, (C) nestin (green fluorescence) expressed by cell with nuclei counterstained by propidium iodide (red fluorescence), and (D) Musashi-1 (green fluorescence) expression. Scale bars: 100 μm (A and B), 200 μm (C and D).

The prevalence of TuJ-III+ neurons, GFAP+ astrocytes, and MBP+ oligodendrocytes derived from neural induction of ES cells D3 with and without MS5 support. ∗p < 0.05.
In four individual experiments of 16 replicate studies on culturing ES cells with MS5 CM, data revealed the cell viability and numbers of D3-derived cells in 50% CM for 8 days were comparable to those in 25% CM (viability: 50% CM vs. 25% CM, 90.6 ± 5.5% vs. 92.8 ± 3.2%, p = 0.173; cell number: 2.85 ± 0.32 × 105 vs. 3.08 ± 0.4 × 105, p = 0.073), but significantly higher than those in 75% CM (viability: 50% CM vs. 75% CM, 90.6 ± 5.5% vs. 70.9 ± 8.5%, p = 0.0001; cell number: 2.85 ± 0.32 × 105 vs. 2.01 ± 0.25 × 105, p = 0.0003). Data suggested that dose-dependent growth-inhibiting metabolites accumulated in the CM could be negated by 50% dilution. Compared to MS5 cocultures, cultures in 50% CM showed a significantly less number of nestin+ colonies (nestin+ colonies: CM culture vs. MS5 cocultures; 43.5 ± 5.5% vs. 61.5 ± 4.3%, p = 0.0004).
In four separate experiments of induction culture, immunostaining of the undifferentiated ES cell marker SSEA-1 revealed a significant decrease of immunoreactive cells in ES cell-derived cell products that had been cocultured with MS5 for 8 days (SSEA-1+ cells: with vs. without MS5 support; 9.3 ± 1.5% vs. 13.1 ± 2.1%; p = 0.001) (Fig. 4). Readouts suggest the prodifferentiative effect of MS5.

Immunostaining of the stage-specific embryonic antigen-1 (SSEA-1). (A) A representative staining of SSEA-1 (green fluorescence) of ES cell D3-derived cells with nuclei counterstained by propidium iodide (red fluorescence). Scale bar: 20 μm. (B) Percentage of SSEA-1+ cells derived from cultures with and without MS5 supports upon completion of 8-day induction, 6-day propagation, and 4-day selection in four separate experiments of 16 replicate analyses. ∗p < 0.05 versus corresponding control.
Propagation Culture and Selection Culture of ES Cell-Derived Cells
Upon completion of 8-day induction culture with and without MS5, ES cell-derived cells were allowed to propagate in serum-free medium supplemented with insulin, transferrin, selenium, and fibronectin for 6 days and further selected in N2B27-supplemented neural basal medium for 4 days. Immunostaining displayed a progressive loss of SSEA-1 expression in MS5-mediated D3-derived cells after 18 days, which was in contrast to the persistent existence of SSEA-1+ cells in control cultures (Fig. 4B). Semiquantitative RT-PCR demonstrated no expression of the octamer-binding transcription factor-4 (Oct-4; undifferentiated ES cell marker), Brachyury (mesodermal), and α-fetoprotein (endodermal) genes in the ES cell-derived cells derived from cocultures (Fig. 5).

Ethidium bromide-stained agarose gel of electrophoresed RT-PCR products derived from ES cell-derived cells. (A) Gene expression of cell derivatives of ES cell D3 upon completion of 8-day induction culture (I), 6-day propagation culture (P), and 4-day selection culture (S). Coc: with MS5 coculture; Ctr: control culture without MS5 coculture. (B) Gene expression of ES cell D3-derived cells (D3) and E14TG2a-derived cells (E14) after 18-day neural induction in cocultures. PC: positive controls (undifferentiated mouse ES cells for Oct-4; mouse ischemic brain for Nestin, Pax-6, Otx-1, Nurr-1, GFAP, CXCR-4, and GAPDH; mouse embryos for α-fetoprotein; mouse embryonic liver for GATA-4; mouse embryonic fibroblasts for Brachyury; mouse embryonic hearts for Myf-5 and Nkx-2.5); NC: negative controls (reagent blanks).
A progressive increase of nestin+ cells was noted in D3-derived cells at three different phases of coculture (data not shown). Figure 6 shows the preponderance of nestin+ stem cells, TuJ-III+ neurons, GFAP+ astrocytes, and MBP+ oligodendrocytes derived from D3/MS5 cocultures over control cultures. On day 18, neural stem cells, neurons, astrocytes, and oligodendrocytes accounted for 73.1 ± 3.0%, 14.2 ± 1.9%, 6.2 ± 1.7%, and 5.2 ± 1.5%, respectively, of 10,000–20,000 cells scored in four separate experiments. Notably, cultures extended for 4 more days in N2B27-supplemented neural basal medium resulted in a significant drop of cell numbers (1.38 ± 0.08 × 106 on day 18 vs. 1.16 ± 0.16 × 106 on day 22, p = 0.003) and viability (67.6 ± 2.6% on day 18 vs. 61.7 ± 5.3% on day 22, p = 0.015), suggesting the 18-day optimally defined protocol.
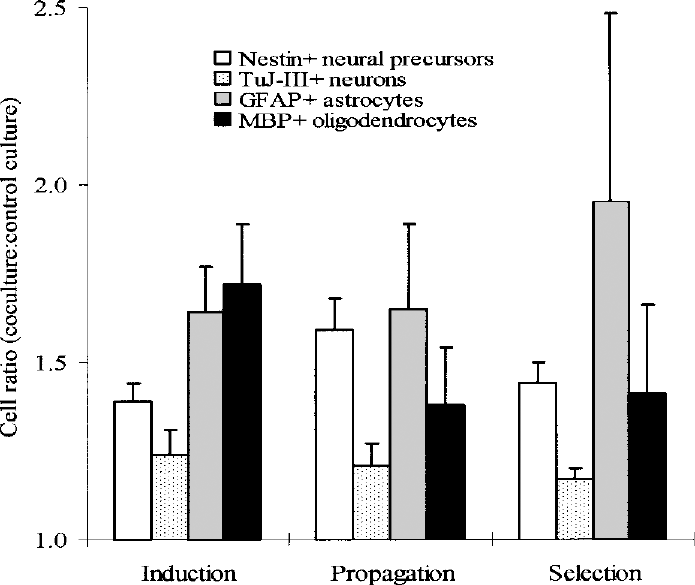
Mean ± SD ratios of the relative frequencies of nestin+ neural stem cells, TuJ-III+ neurons, GFAP+ astrocytes, and MBP+ oligodendrocytes derived from ES cell D3 upon completion of induction, propagation, and selection in four separate experiments of four replicate cultures with to without MS5.
Molecular Analyses of ES Cell-Derived Cells
MS5-induced ES cell-derived cells were characterized for lineage commitment by studying specific gene expressions using RT-PCR. A progressive loss of expressions of Oct-4 (undifferentiated status-related), Brachyury (mesodermal), and α-fetoprotein (endodermal) genes was noted in D3-derived cells upon completion of induction, propagation, and selection cultures with MS5, compared to the persistent expressions in control cultures without MS5 support (Fig. 5A). These data indicated that the coculture system did not favor nonneural commitment. On day 18, Pax-6 and Otx-1 (neuroectodermal), nestin and GFAP (neural stem cell-related), Nurr-1 (neuronal), and CXCR-4 (chemokine-related) genes were detected in MS5-induced D3- and E14TG2a-derived cells, respectively, whereas Myf-5 and Nkx2.5 (mesodermal) and GATA-4 (endodermal) were not detected (Fig. 5B). It was suggestive that ES cells cocultured with MS5 cells induced a hierarchical population of neural lineage cells, but selected against mesodermal, endodermal, and undifferentiated ES cells. Besides, gene expressions of neurotrophins, neuroprotective factors, and chemokines, BDNF, CNTF, GDNF, NGF, NT-3, bFGF, IGF-I, IGF-II, VEGF, EPO, and CXCL-12, were also evident in 18-day-derived cell products (data not shown).
In Vivo Study of MS5-Induced ES Cell-Derived Neural Lineage Cells
It has been documented that the hippocampus is the most vulnerable part of the brain to ischemic injury. Thionin staining demonstrated that a remarkable number and extent of pyknotic cells and necrosis, respectively, in both hippocampal CA1 regions at 12 h shortly after ischemic attack (Fig. 7). The numbers of morphologically intact pyramidal neurons with large nuclei, discrete nucleoli, and clear cell periphery dropped extensively on day 3 postinduction of ischemia, compared to those derived from intact mice and mice at 12 h and 1 day having undergone induction of ischemia. There was no significant difference in the numbers of intact pyramidal neurons on day 3 and 5.

Scoring of thionin-stained pyramidal neurons with large nuclei, discrete nucleoli, and clear cell periphery in every fifth serial coronal sections of the CA1 region of the hippocampus 1.8–2.0 mm posterior to bregma of 12 mice 12 h to 5 days after ischemic stroke and corresponding sham-operated control mice. Data were derived from counting cells in 24 frames of 1 × 0.25-mm dimension from 8–10 coronal sections per mouse. ∗p < 0.05.
Immunostaining of BrdU in ES cell-derived neural lineage cells showed that the labeling efficiency was 64.6 ± 4.8%. In four separate experiments, trypan blue dye exclusion tests demonstrated that the mean ± SD cell viability of enzymatically segregated BrdU-labeled D3-derivrd cells was 67.2 ± 4.4%. Upon completion of the time course of 16-day culture (8-day induction coculture, 6-day propagation culture, and 4-day selection culture), MS5-induced ES cell-derived neural lineage cells (5 × 105) were implanted bilaterally onto the caudate putamens of 17 ischemic mice 3 days after stroke, at which time an extensive loss of approximately 80% of pyramidal neurons was evident.
Thirty-six mice that were able to find way out of the water maze in 154 ± 18 s were recruited to in vivo studies. Having undergone ischemic induction, 85.7% (24/28) mice survived and exhibited signs of disturbance of consciousness, drowsiness, paucity of movement, and coma. Two weeks posttransplant no death was encountered and mice did not display abnormal behavior or develop dyskinesia, which may suggest any adverse reactions of intracranial implantation of ES cell-derived cells into the caudate putamen.
On day 5 of behavioral assessment (19 days posttransplant), the episodes of transplanted ischemic mice entering blind alleys of the water maze were significantly less than that committed by ischemic control mice (episodes of error committed by ischemic mice with vs. without cell therapy, 4 ± 3 vs. 11 ± 3, p = 0.0001), but comparable to that of sham-operated normal mice (episodes of error: transplanted ischemic mice vs. sham-operated normal mice: 4 ± 3 vs. 2 ± 1, p = 0.074). Figure 8 shows the time latency of 17 ischemic mice with cell therapy and eight sham-operated normal mice to escape from water over 5 days of behavioral assessment. Seventeen transplanted ischemic mice exhibited a progressive reduction of retention time in the water maze, compared to a relatively constant time latency derived from seven ischemic control mice. On day 5 of behavioral assessment, transplanted mice took significantly less time to get out of the maze [ischemic mice with (n = 17) vs. without (n = 7) cell therapy, 62.2 ± 17.9 s vs. 174.6 ± 33.7 s, p = 0.0002]. There was no difference in the time latency between transplanted ischemic mice and eight sham-operated normal mice [transplanted ischemic mice (n = 17) vs. sham-operated normal mice (n = 8):62.2 ± 17.9 s vs. 54.9 ± 15.4 s, p = 0.439). Motor functions of transplanted ischemic mice and sham-operated normal control mice were comparable as evident by a similar swimming speed tracked on an 80-cm straight path of the water maze on day 5 of testing (transplanted ischemic mice vs. sham-operated normal control mice; 27.2 ± 3.5 cm/s vs. 29.0 ± 4.5 cm/s, p = 0.304). Taken together, these data suggested that cell therapy helped improve spatial learning and memory ability of ischemic mice.

Mean ± SD retention time over 5 consecutive days in the water maze taken by ischemic mice with (n = 17) and without cell therapy (n = 7) and sham-operated normal control mice (n = 8) 2 weeks post transplant. ∗Ischemic mice with vs. without cell therapy: p = 0.0002.

Histology of pyramidal neurons in the hippocampal CA1 region at 3 weeks after ischemic damage. (A) A representative coronal section of the entire thionin-stained hippocampus with illustration of a counting frame. (B) Immunostaining revealed the homing of few BrdU-labeled ES cell-derived cells (green fluorescent nuclei) to the injured hippocampal CA1 region. (C) A representative section derived from sham-operated normal control mice demonstrating intact pyramidal neurons. (D) A representative section derived from saline-injected ischemic control mice showing conspicuous necrosis and abundant pyknotic cells. (E) A representative section derived from transplanted ischemic mice exhibiting hippocampal CA1 neurons in orderly alignment. (F) Quantification of intact pyramidal neurons showed the numbers derived from transplanted ischemic mice were comparable to those of sham-operated normal control mice (CA1 neuron density in transplanted ischemic mice vs. sham-operated normal control: 177.0 ± 19.1 cells/0.25 mm2 vs. 189.9 ± 15.6 cells/0.25 mm2, p = 0.089), but were significantly higher than those of ischemic control mice with saline injection (ischemic mice with vs. without cell therapy: 177.0 ± 19.1 cells/0.25 mm2 vs. 65.6 ± 12.4 cells/0.25 mm2, p = 0.0008). Scale bars: 500 μm (A), 50 μm (B, C, D, E). ∗p < 0.05.
Mice were sacrificed 3 weeks posttransplant. Immunofluorescence staining of BrdU revealed a substantial number of the donor cells in the caudate putamen. BrdU+ cells were also evident in the cortex, corpus callosum, and hippocampus (Fig. 9). There were significantly greater numbers of intact pyramidal neurons in the hippocampal CA1 regions of ischemic mice with than without cell implants. The densities of pyramidal neurons were comparable among transplanted ischemic mice and sham-operated normal control mice (Fig. 9). A lesser degree of lesion of the hippocampal pyramidal layer in transplanted ischemic mice than nontransplanted ischemic mice was also seen (Table 1). Among 17 transplanted ischemic mice, one was noted to develop teratoma (p = 0.059). Data suggested that transplanted cells help protect the hippocampal CA1 region from ischemic damage and enhance regeneration.
Histological Grading of Pyramidal Neurons in CA1 Region of Hippocampus

p < 0.05: test versus test control, normal control versus test control.
p > 0.05: test versus normal control.
Discussion
In a noncontact coculture system we demonstrated that MS5 cells could induce ES cells toward the neural fate, and intracranial implantation of ES cell-derived neural lineage cells onto the caudate putamens of cerebral ischemic mice could help recover cognitive functions of spatial learning and memory ability.
MS5 have been demonstrated to support the proliferation of hematopoietic progenitor cells in long-term cultures (9). Recently, stromal cells derived from mouse bone marrow, PA6, M2-10B4, and MS5, were shown to induce neural differentiation of ES cells in coculture systems (4,17,21). In this study by using a noncontact coculture system it was noted that the neurotrophic effect of MS5 on ES cells was similar to the stromal cell-derived inducing activity (SDIA) of PA6 cells in contact cultures (11). MS5-conditioned medium also exerted the neural inducing effect, although to a lesser extent, implying the presence of labile soluble neurotrophic and neuroprotective factors, which were not well preserved at low temperature. In a previous study employing a noncontact coculturing system, C17.2 neural precursor cells were noted to induce ES cells into neural lineage cells (7). Taking the findings derived from the present and previous studies, the noncontact system implies that the SDIA may not be requisite for the induction of neural lineage cells and direct contact may not be essential to SDIA. Data also suggest the potential of employing feeder supports mimicking the microenvironment of injured or reactive brain for ES cell induction, despite that the underlying molecular mechanisms of directed differentiation into neural lineage cells are not fully known.
In normal developmental process, the neural fate in mice is thought to take place during gastrulation of E6.5 to E8. In vitro neural induction of ES cells was conducted by coculture with MS5 for 8 days to mimic the developmental microenvironment and the duration. Compared to other methods reported in the literature, the noncontact coculture system established in this study offered a simple and efficient procedure to differentiate ES cells into neural lineage cells and evaded using complex and procedural embryoid body-based differentiation protocols (3,8,11,22). Besides, nonneural feeder-mediated ES cell-derived cells are heterogeneous, consisting of neural cells at different stages of differentiation, nonneural lineage cells, and undifferentiated ES cells.
By using a noncontact coculture system, ES cell-derived cells did not need to be enzymatically or mechanically segregated from the feeder support, in contrast to previously reported contact cocultures (25,28), hence minimizing cell loss and feeder cell contamination prior to transplantation. Subsequent cultures in serum-free medium enriched with insulin, transferrin, selenium, and fibronectin, and N2B27-supplemented neurobasal medium allowed the propagation and selection of neural lineage cells, without impairing their differentiation potential (18,24,26). Upon completion of the 8-day induction culture, 6-day propagation culture, and 4-day selection culture, stable yields of relatively purified neural lineage cells, including neural stem cells, neurons, astrocytes, and oligodendrocytes, could be achieved. Nestin+ neural stem cells accounted for 73% of the derived cells. RT-PCR revealed neuroectodermal and neural-related genes, Otx-1, Pax-6, Nestin, and Nurr-1, but no mesodermal and endodermal genes and ES cell transcriptional factor Oct-4. Apparently, the system did not support nonneural differentiation and maintenance of undifferentiated status of ES cells, which may attribute to a single episode of teratoma development among 17 transplanted ischemic mice examined.
Functional studies of ES cell-derived cells in animal models displayed contradictory results (13,29). The interaction of neural progenitors, neurons, and glial cells underlines the significance of transplantation of neural lineage cells to the ischemic brain, which is characterized by a severe loss of different neural cell types. Previous studies demonstrated that fully maturated neurons survived less efficiently than neural progenitors upon implantation to the brain, which might be related to the susceptibility of mature neurons to mechanical stress and harvesting for transplant (22). Cognitive impairment was found to correlate with the neuronal loss in the hippocampus (27). We performed cell transplant to mice on day 3 after stroke to explore the therapeutic potential of ES cell-derived neural lineage cells, at which time the cell loss was most severe, accounting for approximately 80% in the hippocampal CA1 region. The ES cell-derived cell grafts, which were composed of largely neural stem cells and some postmitotic neurons and glias (73.1 ± 3.0% nestin+ neural stem cells, 14.2 ± 1.9% neurons, 6.2 ± 1.7% astrocytes, and 5.2 ± 1.5% oligodendrocytes), were shown to improve the spatial learning and memory ability of ischemic mice significantly, as demonstrated by the water maze test.
It has been reported that, during normal development of the mouse, approximately 10 to 12 days postcoitus are required for the occurrence of neurons in the ventral mesencephalon of the brain (12). We speculated that transplanted ES cell-derived neural stem cells may exhibit their plasticity and further differentiate in vivo in response to the environmental cues in the host brain in similar time duration. Behavioral assessments of the animals were conducted 2 weeks post transplant. The reversal of lesion-induced neurological deficits observed in the test animals 2 weeks post transplant might be attributable to the newly formed functional neurons.
The hippocampus is the major site of memory functions. To a lesser extent the caudate putamen is also critical for the procedural memory (20). Injured caudate putamen was noted to sustain the interaction between transplanted neural cells and striatal neurons (6), making it a target of cell transplantation therapy for cerebral ischemic damage. Tracking of BrdU+ cells in the study demonstrated that a majority of the implanted cells were localized in the caudate putamen, while some were evident in the cortex, corpus callosum, and hippocampus. As BrdU intensity of proliferating cells will reduce and lose to an undetectable level, there may be an underestimation of migration, homing, and engraftment of donor cells to different localities of the ischemic brain. It was inconclusive that the small number of ES cell-derived cells engrafted to the lesioned hippocampus responded to the improved cognitive functions.
Data of the present and earlier studies demonstrated genes of neurotrophins, growth factors, and chemokines, including BDNF, CNTF, GDNF, NGF, NT-3, bFGF, IGF-I, IGF-II, VEGF, EPO, and CXCL-12, were expressed by neural stem cells, microglias, and astrocytes (1,7). The administration of neurotrophic factors and growth factors was also noted to recruit endogenous progenitors in the periventricular zone near the hippocampus to induce massive regeneration of CA1 pyramidal neurons after ischemia (14,23,34). In the study, readouts of the neuronal density and histological grading of pyramidal neurons in the hippocampal CA1 region supported the hypothesis of regeneration of neural stem and progenitor cells present in the periventricular region near the hippocampus subject to ischemic stress (5). A comparable degree of cell necrosis, incidence of pyknotic cells, and neuronal density was noted among sham-operated normal control mice and ischemic mice at 3 weeks posttransplant, while a significant extent was evident in ischemic control mice. It is suggestive that the small number of ES cell-derived neural stem and progenitor cells engrafted to the hippocampus might enhance endogenous regeneration through the donor cell-derived neurotrophic and neuroprotective factors.
In conclusion, the findings of the study suggested that MS5 cells might exert the neurotrophic and neuroprotective effects to induce the differentiation of ES cells into neural lineage cells, which could effectively improve neurological deficits in the mouse ischemic stroke model. Having implanted onto the caudate putamen of ischemic mice, MS5-induced ES cell-derived neural lineage cells not only enabled migration, homing, and engraftment to the injured brain parenchyma but also could enhance endogenous regeneration. The teratoma development in an animal having undergone ES cell-derived cell transplant instigates the prerequisite of safety. More works are needed for the prospective clinical application of stem cell therapy.
Footnotes
Acknowledgments
This study was supported in part by stem cell research funding from the Li Ka Shing Institute of Health Sciences, The Chinese University of Hong Kong, and by the Strategic Research Program SRP 2/02, The Chinese University of Hong Kong.
