Abstract
Adipose-derived stem cells (ASCs) appear to be a useful stem cell population, which has been shown to possess multipotentiality. The aim of this study was to evaluate the utility of ASCs in tissue-engineered cartilage using a hybrid scaffold from fibrin-modified PLGA scaffold. ASCs were isolated from rabbit adipose tissue. The PLGA scaffold was prepared by low-temperature deposition technology and the hybrid scaffold was fabricated by a freeze-drying method. When ASCs were seeded onto fibrin-modified PLGA scaffold in vitro, enhanced cellular viability was observed compared to unmodified PLGA scaffold. The analysis of proteoglycan and collagen II revealed that fibrin-modified scaffold succeeded in inducing ASCs to differentiate into chondrocytes in vitro. A preliminary study on cartilage regeneration was also performed in vivo. Observation of histology and immunoblotting demonstrated that ASCs containing the hybrid scaffold promoted cartilage regeneration in the defects of articular cartilage much better than other groups. These results indicated that ASCs containing the hybrid scaffold are a more effective way to potentially enhance articular cartilage regeneration.
Introduction
Intact articular cartilage is very important for providing the joint with a frictionless articulating surface. However, due to very slow turnover at the cellular and molecular level, articular cartilage has a limited capacity for self-repair. Over the past several decades many resolutions, such as drilling and microfracture, have been used to restore normal function of the damaged cartilage (9, 14). Although these approaches provide some benefits, none is believed to be optimal for repairing cartilage lesions. Recently, the method of cell-based therapy, such as cartilage tissue engineering, has proved to be one of the most promising alternative therapies for repairing damaged cartilage. Successful cartilage tissue engineering requires cells capable of undergoing chondrogenic differentiation upon treatment with appropriate biochemical factors and a three-dimensional (3D) scaffold capable of providing a favorable environment for chondrogenic cell growth and new cartilage-specific extracellular matrix formation.
In cartilage tissue engineering, chondrocytes and stem cells were commonly used for cartilage regeneration and the choice of cell type determines the strategy of cartilage tissue engineering. Previously the use of autologous chondrocytes was believed to be preferable. But the present studies have suggested that the proliferative capacity of the terminally differentiated chondrocytes was decreased with in vitro expansion. In addition, the procedure required to harvest autologous cells may initiate joint degeneration (7). Because of the drawbacks of chondrocytes-based cell therapy, attempts have been made to employ stem cells for cartilage tissue engineering. Recently, studies have indicated that stem cells from human adipose tissue contain multipotent progenitor cells that can differentiate into osteogenic, chondrogeinc, myogenic, and neurogenic cells when induced by the appropriate biological factors in vitro (4, 13, 20). Our studies demonstrated that adipose-derived stem cells (ASCs) from rabbits were successfully isolated and were induced to differentiate into chondrocytes by transform growth factor-β1 (TGF-β1) or bone morphogenetic protein-2 (BMP-2) (16). Because ASCs are own to stable maintenance of undifferentiated status the pluripotency does not decrease in a growing body during the expansion in vitro, and adipose tissue is obtained by the extraction from the patient in a less invasive manner, and provides a large quantity of autologous cells, ASCs represent a fascinating cell source for regenerative medicine and will open new avenues for therapeutic approaches.
The use of ASCs in cartilage tissue-engineering applications creates a need for more research in cell scaffolds. As physical support and template, the scaffold plays an important role in tissue engineering by supplying a 3D substrate for cell growth and tissue regeneration. The biodegradable scaffold can be divided into two kinds: synthetic and natural material. Synthetic scaffold allows a better control of shape, surface morphology, mechanical and physicochemical properties, and possesses predictable biodegradation kinetics. Poly(DL-lactic-co-glycolic-acid) (PLGA), which belongs to one of the synthetic scaffolds, has been widely investigated to serve as the substitute of tissue regeneration and approved by the food and drug administration (FDA) of the US for certain clinical applications. However, PLGA does not present a favorable surface for cell adhesion, proliferation, and differentiation because of the hydrophobic surface properties and lack of specific cell-recognizable signals (10). To overcome this drawback, an alternative approach is to create a hybrid scaffold using a multifunctional biological protein and PLGA. Because the hybrid scaffold can be used to create a biomimetic cellular environment by balancing the structural and biofunctional element, the advent of biosynthetic hybrid scaffold signifies a major achievement in the fields of tissue engineering (1).
Fibrin, an excellent natural polymer, has drawn significant interest in tissue engineering. Fibrin presents several important features for the scaffold material: 1) it is an FDA approved material and has been widely used in clinical setting due to its high affinity; 2) it possesses hydrophilicity, biocompatibility, and biogradation; 3) it is rich in fibrinogen protein, which is a well-characterized extracellular matrix (ECM) molecule with a central role in tissue remodeling and chondrocyte–ECM interaction.
In this study, we fabricated the hybrid scaffold from fibrin-modified PLGA by a freeze-drying method and hypothesized that the combination of the hybrid scaffold and ASCs was suitable for cartilage regeneration. Our study is to evaluate the capacity of differentiation of ASCs into chondrocytes on the PLGA scaffold with and without fibrin in vitro and the reparative capacity of ASCs hybrid scaffold used to repair cartilage defects in vivo.
Materials and Methods
Preparation of PLGA and PLGA-Fibrin Scaffolds
PLGA [50:50 wt% poly lactic acid (PLA)/poly glycolic acid (PGA); 12,000–16,500 kDa; Polysciences Inc.) copolymer scaffold was fabricated via low-temperature deposition (LDM) as we reported previously (19). The hybrid scaffold was prepared by forming fibrin microsponges in the openings of a macroporous PLGA scaffold as described below. The fibrinogen solution (75 mg/ml 0.5 ml) was poured into the PLGA scaffold. Then the thrombin (400 IU/ml 0.5 ml) was also poured into the PLGA scaffold to set a fibrin gel. The composites were frozen at −80°C overnight, then lyophilized for 72 h to make the PLGA-fibrin scaffold. The scaffolds were sterilized with ethylene oxide and stored until used.
Scanning Electron Microscopy Observation
The structure of the PLGA and PLGA-fibrin scaffolds was observed by scanning electron microscopy (SEM). Samples were fixed in 2% phosphate-buffered glutaraldehyde solution for 24 h. Then the scaffolds were washed with phosphate-buffered saline (PBS) and were freeze dried. Before analysis, the dried samples were mounted on aluminum supports and sputter-coated with gold.
Water Absorption of the Scaffolds
The water absorption ratios were measured following the procedure described in a previous report (18). Briefly, each scaffold, cut into 1 × 1 × 1-cm3 pieces, was weighed (Wi) before immersion in PBS. Then each group of the scaffolds was incubated in PBS and maintained in a humidified incubator at 37°C/5% CO2 for 14 days. The scaffolds were then removed from the PBS, gently blotted with filter paper to remove surface water, and immediately weighed (Ws). The scaffolds were dried in an oven for 2 days to completely remove the water, after which they were weighed (Wd) a third time. The water absorption ratios were calculated using the following equation: water absorption ratio = (Ws – Wd)/Wi × 100.
Isolation and Culture of Adipose-Derived Stem Cells
ASCs were isolated from cervical adipose tissue of New Zealand white rabbits (age 4 months) using a method reported previously (17). In brief, the tissue obtained was washed with PBS to remove red cells. The adipose tissue was minced finely using surgical scissors, and the extracellular matrix was digested for 1 h at 37°C with 0.15% collagenase (type I; Sigma, St. Louis, MO, USA) in PBS. Once digested, enzyme activity was neutralized with culture medium containing DMEM (Gibco, Paisley, UK), 10% fetal bovine serum (FBS, Sijiqing Biological Engineering Materials Co., Hangzhou, China), penicillin 100 U/ml, and streptomycin 100 μg/ml. The samples were filtered through a 500-μm mesh filter to remove tissue debris. The cell suspension was centrifuged at 800 × g to obtain a pellet, and the pellet was resuspended in culture medium. The cells were seeded in 25-cm flasks (Corning-Costar, Acton, MA, USA) at density of 4 × 105 cells/cm3 and incubated at 37°C/5% CO2. The medium was changed after 24 h and then three times a week. At a confluence of more than 80%, cells were detached with 0.25% trypsin and 0.1% EDTA. The cells were passaged three or four times prior to use. The cells harvested were kept separated for each rabbit during in vitro expansion in order to be implanted in the rabbits. Chondrogenic differentiation was induced by culturing ASCs on each scaffold in chondrogenic medium (CM) [consisting of 10 ng/ml TGF-β1, 1% FBS, 1 × ITS (6.25 μg/ml insulin, 6.25 μg/ml transferrin, 6.25 μg/ml selenious acid, 1.25 μg/ml bovine serum albumin, 5.35 μg/ml linoleic acid, BD Biosciences, USA), penicillin 100 U/ml, and streptomycin 100 μg/ml] for 14 days. Every third day this medium was changed.
To seed the cells into PLGA scaffolds with and without fibrin (thickness: 3 mm; diameter: 5 mm), 40 μl of cell suspension containing 1 × 106 cells was loaded onto an upper side of each prewetted scaffold and allowed to penetrate into the scaffold. Each sample was transferred to a 24-well tissue culture plate and then incubated at 37°C/5% CO2 for 4 h to allow cells to diffuse into and adhere to the scaffolds before the addition of 2 ml chondrogenic media. Chondrogenic media were changed every 2 days and the culture plates were shaken.
Histological Analysis
At 14 days, samples were fixed for histology with 4% paraformaldehyde in PBS, embedded in paraffin, and sectioned using standard histochemical techniques. Sections were stained with hematoxylin and eosin (H&E).
Cell Viability Study
MTT viability of the cells in the scaffolds was measured after injecting 1 ml MTT (5 mg/ml) solution into the scaffolds and culturing for 4 h at 1, 7, and 14 days. The scaffolds were then immersed in 5 ml DMSO until the formation pigment dissolved completely. Pigment solution (200 μl) was added to the 96-well ELISA plate and vortexed for 10 min, and the MTT absorbency values of the resulting solution were measured using an ELISA reader at the optical density (OD) of 490 nm.
Sulfated Glycosaminoglycan (sGAG) and DNA Content
The total GAGs of the scaffolds were extracted at 1, 7, and 14 days following a method as a modification of a previous report (3). Briefly, samples were harvested, washed with PBS, and digested with 1 ml papain (Sigma) solution (1 mg/ml in 50 mM sodium phosphate, pH 6.5, containing 2 mM N-acetyl cysteine and 2 mM EDTA) for 20 h at 65°C. GAGs were measured by its reaction with 1,9-dimethylmethylene blue (DMMB; Sigma) using shark chondroitin sulfate (Sigma) as standard at 525 nm.
The same papain implant digest was also used to measure the total amount of DNA with the Hoechst 33258 fluorescent dye assay (6). A 100-μl aliquot was diluted in 1 ml of dye/buffer solution and the fluorescence of the samples was evaluated for excitation at 365 nm and emission at 458 nm by a spectrofluorometer. A standard curve was prepared from known concentrations of herring sperm (Sigma).
RT-PCR for Collagen II and Aggrecan
Total RNA was isolated from ASCs-PLGA and ASCs-fibrin-PLGA with 800 μl Trizol reagent. After addition of 160 μl chloroform to the homogenized samples, RNA was precipitated using 400 μl of isopropanol. RNA pellets were dissolved in 20 μl of RNase- and DNase-free water and RNA yields were estimated based on A260. First-strand cDNA was reverse transcribed from 3 μg of total RNA by using Superscript™ reverse transcriptase (Invitrogen, CA, USA). The resulting cDNA was used as a template in the polymerase chain reaction, using Taq polymerase and the primers 5′-GACCCCAT GCAGTACATG-3′ (forward) and 5′-GACGGTCTTG CCCCACTT-3′ (reverse) designed to amplify rabbit collagen II, and the primers 5′-CCTTGGAGGTCGTGGT GAAAGG-3′ (forward) and 5′-AGGTGAACTTCTCT GGCGACGT-3′ (reverse) designed to amplify rabbit aggrecan, and the primers 5′-GGTGAAGGTCGGAGT CAACGG-3′ (forward) and 5′-GTCATGAGTCCTTC CACGAT-3′ (reverse) designed for GAPDH, the internal loading control. Amplification was performed for 35 cycles and a total reaction volume of 10 μl PCR products were resolved by agarose gel electrophoresis.
Repair of Articular Defects
A cylindrical, full-thickness defect (thickness: 3 mm; diameter: 5 mm) was created using a hand drill with a 5-mm drill bit, penetrating the articular cartilage in the patellar groove of the distal femur in 24 rabbits (the ASCs from them had been prepared as mentioned above). The scaffold (the pure PLGA scaffold or the fibrin-modified PLGA scaffold) containing ASCs (1 × 106 cells) was cultured in chondrogenic media at 37°C/5% CO2 for 14 days before transplantation experiment. The rabbits were operated on by injection of 2 ml/kg Sumianxin (Agricultural University, Changchun, China) into the muscle. The rabbits were classified into the following four groups: the empty group (no insertion group); the PLGA scaffold insertion group; the ASCs-PLGA group; and the ASCs-fibrin-PLGA group. All rabbits were allowed to move freely after surgery without plaster immobilization. The committee on animal experimentation of Fourth Military Medical University approved all experiments, which met the national institute of healthy (NIH) guidelines for the care and use of laboratory animals.
Histological Evaluation
Samples were harvested 12 weeks postoperatively. The distal parts of femurs were removed and fixed in 10% paraformaldehyde, decalcified in 10% EDTA and embedded in paraffin, sectioned at 6 μm in thickness, and stained with safranin O. Cartilage repair of the defects (six sections taken from each defect) was evaluated microsopically and scored according to a histological grading scale modified after Wakitani et al. (15) and Pineda et al. (12), consisting of five categories with a total score ranging from 0 to 20 points (Table 1).
Histological Grading Scale for Defect Cartilage

Western Blot Analysis for Collagen II
Protein extracts from the native articular cartilage and the repair tissues after digestion were used for Western blot analysis. The protein concentration of the extracts was assessed using a Bradford assay kit. The optical density was measured at 595 nm using a spectrophotometer. The protein concentration was interpolated from a standard curve obtained from a serial dilution of bovine serum albumin at a concentration of 0.8 mg/ml. The cell extracts, normalized for total protein content, were resolved using polyacrylamide gel electrophoresis and electrophoretically transferred to a supported nitrocellulose membrane (Amersham, USA). Afterwards, the membranes were blocked in 5% nonfat milk Tris-buffered saline Tween (TBST) for 2 h, and incubated with primary antibodies, mouse monoclonal anti-collagen II (1:100, Chemicon, USA) at 4°C overnight. Membranes were then washed three times for 10 min each with TBST, followed by the addition of peroxidase-linked anti-mouse IgG (1:500) and enhanced chemiluminescence (ECL) visualization of the bands.
Statistical Analysis
All the data were expressed as the mean ± SD. The values for in vitro study were analyzed using ANOVA test (LSD posttest, n = 5). In vivo study histological scores among groups were analyzed with Mann-Whitney test and the Bonferroni method (n = 6). A value of p < 0.05 was considered to be statistically significant.
Results
Morphology of Scaffolds
SEM observation showed that the pure PLGA scaffold with pore sizes of 500 μm was smooth and had a larger, interconnected open-pore structure (Fig. 1A). The surface of fibrin-modified PLGA scaffold was rough and the web-like fibrin microsponges were firmed in the opening of the synthetic PLGA scaffold (Fig. 1B). The image at a high magnification indicated that the fibrin microsponges were cross-linked by the fibrin fibers and possessed a homogeneous, well-interconnected porous structure with an average pore size of 13 μm (Fig. 1C, D).

SEM photomicrographs of the PLGA scaffold at the original magnification of (A) 70×; the PLGA–fibrin composites at the original magnification of (B) 70×; top surface of the PLGA–fibrin composites at the original magnification of (C) 700×; and cross section of the PLGA–fibrin composites at the original magnification of (D) 1000×.
Water Absorption of Scaffolds
High water absorption of the scaffold contributes to nutrition transport, cell attachment and growth inside the scaffold. The water absorption capacity of the PLGA scaffold was 41.8 ± 1.3%, and the PLGA-fibrin scaffold was 95.7 ± 2.8%. There was significant difference for the PLGA scaffold with and without fibrin sponges (p < 0.01) (Fig. 2).
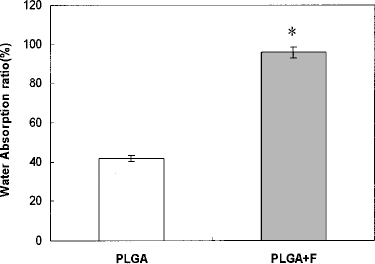
Water absorption of the PLGA scaffold without and with fibrin sponges (scaffold size: thickness 3 mm, diameter 5 mm) at day 14 (n = 5). *Statistically significant relative to the PLGA control (p < 0.05).
Histology
The morphology of 14-day ASCs was examined with H&E staining. The finding suggested that the cells in both scaffolds mainly showed round shape, and sparse elongated cells were found. In addition, an unequal distribution of the differentiated ASCs was showed and the difference of cell number was visible between the PLGA scaffold with and without fibrin (Fig. 3).
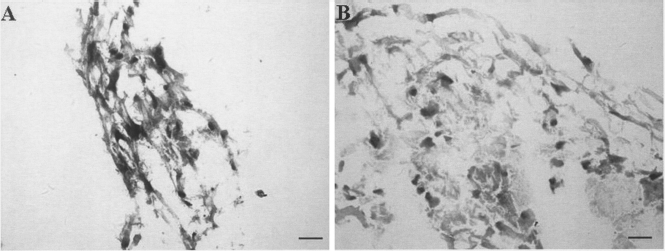
H&E staining of ASCs in the PLGA scaffold and the PLGA–fibrin scaffold (scaffold size: thickness 3 mm, diameter 5 mm) maintained in a chondrogenic medium for 14 days. Scale bars: 50 μm.
Cell Viability
At day 1, the PLGA-fibrin scaffold showed higher cell viability (0.89 ± 0.06 OD) compared to the pure PLGA scaffold (1.06 ± 0.04 OD), but no significant difference was observed in both constructs (p > 0.05). This trend continued as cell viability was much greater in the PLGA-fibrin scaffold than that in the pure PLGA scaffold at day 7 (1.25 ± 0.14 and 2.25 ± 0.09 OD, respectively) and day 14 (2.5 ± 0.26 and 4.3 ± 0.72 OD, respectively), and significant difference was showed between both constructs at different time points (p < 0.05) (Fig. 4).
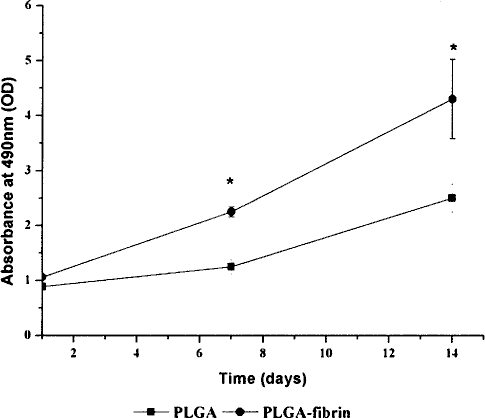
Cell viability on the pure PLGA scaffold and the PLGA–fibrin scaffold (scaffold size: thickness 3 mm, diameter 5 mm) at day 1, 7, and 14 (n = 5). *Statistically significant relative to the PLGA control (p < 0.05).
sGAG Synthesis
After being normalized against DNA content, the result of sGAG production indicated that constructs from the pure PLGA scaffold and the PLGA-fibrin scaffold accumulated extracellular matrix over the 14-day culture period and both scaffolds exhibited a continued increase in sGAG production. At day 1, both scaffolds (0.161 ± 0.004 and 0.171 ± 0.002 mg/μg, respectively) exhibited no significant change in sGAG/DNA (p > 0.05). The GAG values of the pure PLGA scaffold were 0.196 ± 0.003 and 0.230 ± 0.005 mg/μg, and that of the PLGA-fibrin scaffold were 0.261 ± 0.006 and 0.332 ± 0.004 mg/μg at day 7 and 14. Significant difference was shown between both scaffolds at day 7 and 14 (p < 0.05) (Fig. 5A, B).

GAG content of cells on the pure PLGA scaffold and the PLGA–fibrin scaffold (scaffold size: thickness 3 mm, diameter 5 mm) at day 1, 7, and 14 (n = 5). *Statistically significant relative to the PLGA control (p < 0.05).
RT-PCR for Collagen II and Aggrecan
The mRNA expression of collagen II and aggrecan was tested to evaluate the differentiated status of ASCs on the pure PLGA scaffold and the PLGA–fibrin scaffold after 14 days of chondrogenic induction. The expression of collagen II and aggrecan on the PLGA–fibrin scaffold (75% and 61%, respectively) was significantly higher than that in the pure PLGA scaffold (62% and 50%, respectively) at day 14 (p < 0.05) (Fig. 6A, B).

The mRNA expression of collagen II and aggrecan for the differentiated ASCs on the pure PLGA scaffold and the PLGA–fibrin scaffold (scaffold size: thickness 3 mm, diameter 5 mm) at 14 day (n = 5). *Statistically significant relative to the PLGA control (p < 0.05).
Histological Evaluation
Histological study (Fig. 7) showed that in the ASCs–fibrin–PLGA group the defects were completely filled with reparative tissue and the surface and integration of the newly formed cartilage with host cartilage were the best among these groups. In addition, extensively meta-chromatic staining was shown. In the ASCs–PLGA group, the surface of reparative tissue was irregular and the thickness of the tissue was smaller than normal cartilage tissue, and the intensity of safranin O declined. The tidemark was recognized in the tissue-engineered cartilage (the ASCs–fibrin–PLGA group and the ASCs–PLGA group). The scaffold was degraded and the surface of the defects was filled with a thin fibrous tissue in the PLGA scaffold insertion group. The defects were filled with fibrous tissue in the empty group. The means in Wakitani's score of the cell-containing scaffold group (ASCs–PLGA: 8.3; ASCs–PLGA–fibrin: 6.5) exhibited significantly better cartilage regeneration than those of the other two groups (control: 17.1; PLGA: 17.6) at 12 weeks (Table 2). Compared with the ASCs–PLGA group, the ASCs–fibrin–PLGA group showed much better regeneration of cartilage tissue.
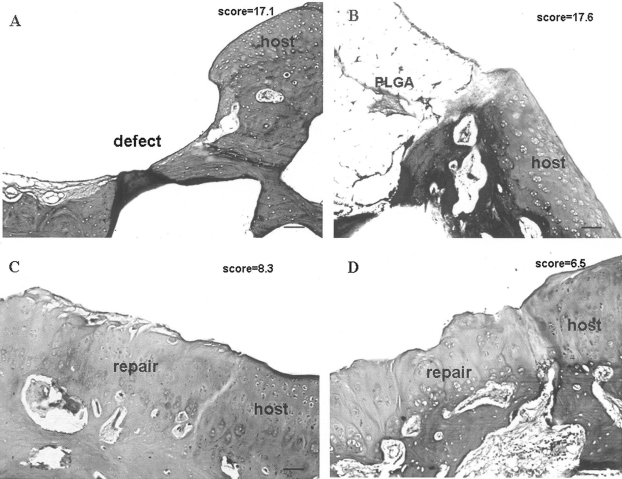
Histologcial observation of cartilage repair at postoperative 12 weeks. (A) Empty control group. (B) PLGA group. (C) ASCs–PLGA group. (D) ASCs–PLGA–fibrin group (safranin O staining). Scale bars: 100 μm.
Results of Histological Grading

Values are mean ± SD.
The scale has five categories assigning a total score ranging from 0 (best) to 20 (worst).
p < 0.05 compared with control group.
p < 0.05 compared with ASCs–PLGA group.
Behavioral Description
All rabbits limped after transplantation. The animals from the ASCs–fibrin–PLGA group and the ASCs–PLGA group exercised normally at day 7 but other animals from the empty group and the PLGA scaffold did not exercise during the observed periods and mental state of those animals was not well.
Expression of Collagen II Protein
Western blot suggested that collagen II protein levels was 0.46- and 0.78-fold, respectively, in the ASCs–PLGA group and the ASCs–fibrin–PLGA group compared with the normal cartilage tissue (Fig. 8).

Western blotting analysis for collagen II in cartilage repair.
Discussion
Production of a tissue-engineered hyaline cartilage has been a central tissue of cartilage tissue engineering, which requires a wide variety of studies including growth factor, cell sources, and scaffold biomaterials. A well-designed three-dimensional scaffold is the important factor to guide tissue formation in vitro and in vivo. Numerous attempts have been made for tissue reconstruction using PLGA based studies. In the present study, we successfully used LDM system to fabricate the PLGA scaffold. LDM system is based on rapid prototyping (RP) and phase separation technologies. RP technology could easily fabricate the scaffold with individualized profile, highly interconnected macropores, mechanical properties, and heterogeneous pore morphologies or materials. Although LDM system could better preserve bio-activity of the scaffold because of its nonheating liquefying processing, the PLGA scaffold fabricated on a LDM system is not good enough for cell attachment, which will further affect cell proliferation and differentiation. So a hybrid scaffold is required to solve this problem.
Using SEM, we found that the PLGA scaffold coated with fibrin sponges changed some of its surface characteristics (e.g., surface roughness, microstructure). Test of water absorption suggested that the hydrophilic property was significantly increased in the scaffold of PLGA-incorporated fibrin sponges. With these results, it is clear that the fibrin-modified PLGA scaffold enhanced the properties of the scaffold. Several studies have also demonstrated the above phenomena. For example, Park et al. suggested that NaOH-treated PLGA 3D scaffolds improved the characteristic of scaffold including a more hydrophilic surface, surface area increased, and porosity altered, which promoted chondrocyte attachment and growth compared to nontreated scaffolds (11). The main reason for the fact is that the hybrid scaffold mimics the nanostructure of cartilage in the body. Cartilage in the body possesses a high degree of namometer roughness because of the existence of proteins. So we think that surface roughness was a major contributor to affecting cellular function.
Cell viability was tested by MTT. Our study demonstrated that the PLGA–fibrin scaffold significantly increased cell viability when compared with the pure PLGA scaffold. In vitro results of H&E staining showed that many round cells were attached to PLGA scaffold modified with fibrin compared to PLGA scaffold at day 14. This also indirectly indicated that a higher cellularity and faster proliferation rate were observed in the hybrid scaffold. The above phenomena may be due to that the fibrin provided a more natural microenvironment for cell growth.
To further compare cellular function in both scaffolds, the differentiation of ASCs was evaluated. The synthesis of type II collagen and aggrecan has been considered to be sensitive metabolic markers to investigate the phenotype level, as has been described for chondrocytes isolated from hyaline cartilage tissue. Therefore, in this study, the synthesis of GAGs and type II collagen was evaluated as a metabolic marker of the phenotype of the differentiation of ASCs into chondrocytes. Biochemical analysis clearly showed that the overall GAG synthetic activity in the PLGA coated with fibrin sponges was obviously higher than that in the pure PLGA scaffold at each time point. RT-PCR suggested that the expression of collagen II and aggrecan was also obviously higher in the PLGA–fibrin scaffold than that in the PLGA alone at day 14. Such studies have provided evidence that the production of collagen II and aggrecan in the hybrid scaffold was affected by the fibrin, and the incorporation of the fibrin significantly promoted the differentiation of ASCs into chondrocytes. The reason for this was mainly due to that the fibrin promoted the chondrogenic matrix synthetic through the release of TGF-β1 in the culture medium and was able to accumulate the newly synthesized extracellular matrix components of hyaline cartilage in the intercellular space (5). In addition, the fibrin around the cells mimicked a more natural microenvironment for ASCs to enhance the production of proteoglycan and collagen II.
The present in vitro results showed the usefulness of the hybrid scaffold from fibrin-modified PLGA scaffold as a biomaterial for cultures of ASCs and for differentiation of ASCs into chondrocytes. Taking advantage of ASCs-hybrid scaffold, we performed a preliminary study on articular cartilage defects implanted with ASCs-containing hybrid scaffold in vivo.
Our studies suggested that 12 weeks postoperatively both ASCs–fibrin–PLGA group and ASCs–PLGA group stained positive for safranin O, which is specific for highly sulfated proteogylcans. Immunobloting for collagen II was also positive in both groups, indicating specific collagen production of the hyaline cartilage phenotype. In addition, histological observations of cartilage repair suggested that ASCs–fibrin–PLGA group resulted in better surface zone repair, subcondral bone connection, as well as deeper zone remodeling compared with ASCs–PLGA group. We speculated that ASCs seeded onto the hybrid scaffold should have better differentiation than those on the PLGA scaffold, which promoted the regeneration of cartilage tissue. At present, observations regarding the repair of chondral defects using ASCs scaffod have been reported. Masuoka demonstrated that the transplantation of autologous ASCs containing atelocollagen honeycomb-shape scaffold with a membrane sealing is effective in repairing articular cartilage defects (8). Dragoo also showed that induced ASCs supported in a fibrin glue matrix promoted cartilage repair (2). Although natural polymer such as collagen and fibrin contributed to cartilage regeneration, it is mechanically too weak to maintain the desired shapes and structures for a long time when used in the body. In addition, natural polymer is also easily degraded; the hybrid scaffold fabricated in the study appears to solve these drawbacks.
Conclusion
Our work has shown that the hybrid scaffold from fibrin-modified PLGA scaffold appears to be an effective biomaterial for the growth and differentiation of ASCs in vitro and tissue engineering using ASCs and the hybrid scaffold promoted cartilage regeneration in vivo. In the next phase of this work, we will carry out longer term analysis of at least 24 weeks for cartilage repair using ASCs and the hybrid scaffold to evaluate some properties of tissue-engineered cartilage, including biomechanics and degradation of regenerated cartilage.
Footnotes
Acknowledgment
This work was supported by a grant from the National Natural Science Foundation of China (No. 30801164).
