Abstract
Ischemic wounds are chronic wounds with poor blood supply that delays wound reconstruction. To accelerate wound healing and promote angiogenesis, adipose-derived stem cells (ADSCs) are ideal seed cells for stem cell–based therapies. Nevertheless, providing a favorable environment for cell proliferation and metabolism poses a substantial challenge. A highly sulfated heparin-like polysaccharide 2-N, 6-O-sulfated chitosan (26SCS)-doped poly(lactic-co-glycolic acid) scaffold (S-PLGA) can be used due to their biocompatibility, mechanical properties, and coagent 26SCS high affinity for growth factors. In this study, a nano-scaffold system, constructed from ADSCs seeded on electrospun fibers of modified PLGA, was designed to promote ischemic wound healing. The S-PLGA nanofiber membrane loaded with adipose stem cells ADSCs@S-PLGA was prepared by a co-culture in vitro, and the adhesion and compatibility of cells on the nano-scaffolds were explored. Scanning electron microscopy was used to observe the growth state and morphological changes of ADSCs after co-culture with PLGA electrospun fibers. The proliferation and apoptosis after co-culture were detected using a Cell Counting Kit-8 kit and flow cytometry, respectively. An ischemic wound model was then established, and we further studied the ability of ADSCs@S-PLGA to promote wound healing and angiogenesis. We successfully established ischemic wounds on the backs of rats and demonstrated that electrospun fibers combined with the biological effects of adipose stem cells effectively promoted wound healing and the growth of microvessels around the ischemic wounds. Phased research results can provide a theoretical and experimental basis for a new method for promoting clinical ischemic wound healing.
Introduction
Ischemic wounds are a fundamental challenge in postoperative care after skin injury1–3. Normally, during wound healing, angiogenic capillary sprouts invade fibrin/fibronectin-rich wound clots and organize them into a microvascular network throughout the granulation tissue within a few days4,5. The new microvascular network is necessary to transport oxygen, nutrients, immune cells, and other important factors carried by the blood to the wound site6–8. Failure of microvascular network reconstruction is one of the main causes of ischemic wounds9,10. However, recombinant proangiogenic growth factors therapy commonly associated with inappropriate blood vessel growth, including genetic modifications and anti-apoptotic proteins, also are not cost-effective. In addition, traditional pro-vascularized implants cannot easily form anastomoses with the host vasculature 11 . Thus, low-cost and efficient strategies that promote wound angiogenesis and accelerate wound healing may revolutionize the treatment12–14.
Considerable research has shown that stem cell therapy can be used to treat diseases that cannot be treated by traditional methods. Transplantation of stem cells enhances angiogenesis, and re-epithelization, and accelerates wound healing, mainly through the secretion of various bioactive factors, including exosomes, growth factors, and chemokines, which has provided an alternative in clinical practice. Adipose-derived stem cells (ADSCs) have emerged as promising candidates for wound-healing cell therapy due to their abundance and relative ease of isolation and expansion 15 . In terms of wound healing, ADSCs can not only proliferate and differentiate into skin cells to replace damaged or dead cells but also play an important role in activating cell regeneration and tissue reconstruction via autocrine and paracrine pathways 16 . There is evidence that ADSCs can survive for a few weeks in and around ischemic sites, with some differentiating into capillaries; and several studies have highlighted the capacity of ADSCs to promote wound pro-vascularization 17 . ADSCs fulfill the generally accepted criteria for cell-based therapies but still need further investigation into their efficiency, taking into consideration the host environment and patient-associated factors. These results indicate that ADSCs seeded within a 3D scaffold may improve wound healing by promoting vascularization. Specifically, tissue-engineering strategies aim to accelerate wound healing by providing support to ADSCs for adhesion, migration, differentiation, and proliferation.
Poly(α-hydroxyester)s-type biodegradable polymers, such as lactide-co-glycolide (PLGA), possess adjustable degradation rates, good biocompatibility, and suitable mechanical properties. They are also approved by the Food and Drug Administration (FDA) 18 . The fibers prepared by electrospinning technology can achieve an extremely fine diameter, typically in the nanoscale. The microstructure of these fibers mimics the characteristics of natural collagen fibers, contributing to a wound covering, and could provide a high surface-to-volume ratio, which not only enables maximal cell-material interactions and material-mediated signaling but also promotes rapid hemostasis, which can mimic extracellular matrix (ECM) 19 . Furthermore, the nanoporous nature aids in nutrient and waste exchange while creating a barrier to external pathogens 20 . Sulfated glycosaminoglycans (GAGs) are widely distributed in the ECM, cell surface, or intracellular vesicles 20 . Due to a large number of negatively charged sulfated modifications, extracellular sulfated GAGs bind to positive or polar amino acid residues on target proteins, affecting the interaction of receptor proteins with their ligands or inducing receptor oligomerization, and are also thought to be co-ligands of some membrane receptors 21 . Our previous study first synthesized highly sulfated heparin-like polysaccharide 2-N, 6-O-sulfated chitosan (26SCS)-doped PLGA electrospun scaffold (S-PLGA) which has been proven to interact with numerous proteins to regulate physiological and pathological processes, including inflammation, blood coagulation, lipid metabolism, and angiogenesisc 22 . Owing to its high affinity for angiogenic growth factor, 26SCS can efficiently induce angiogenesis 23 . However, ischemic wounds lack nutrients and bioactive molecules that promote healing. While S-PLGA can only enrich but not secrete, we aim to expand the functions of the scaffold. This is to assist in promoting the healing of ischemic wounds and building new microvascular systems that resemble the properties of natural skin.
Building upon the regenerative properties of ADSCs, we designed S-PLGA nanofibers seeded with ADSCs (ADSCs@S-PLGA) as a multifunctional skin substitute 24 . Previous studies demonstrated that PLGA scaffold promotes wound regeneration 22 . Our study aims to confirm that the structure of S-PLGA is suitable for incorporating ADSCs for delivery to wound beds, which can stimulate the collective migration of keratinocytes and enhance the formation of granulation tissue and new blood vessels. For our proof-of-concept study, we employed a skin substitute with improved properties as a wound dressing. The functionality of this substitute was confirmed in vivo by using a full-thickness excised ischemic wound. In summary, our results strongly suggest that ADSCs@S-PLGA holds great promise for application in wound healing research.
Materials and Methods
Materials
S-PLGA electrospun films were fabricated at The State Key Laboratory of Bioreactor Engineering, East China University of Science and Technology (Shanghai, China) 23 . Sprague-Dawley (SD) rats were provided by the Experimental Animal Center of the Fourth Military Medical University (Xi’an, China). Adipose stem cells (ADSCs) were isolated from rat epididymal fat tissue using a standard procedure. ADSCs were cultured in OriCellTM SD ADSCs growth medium (Cyagen, Santa Clara, CA, USA). ADSCs were characterized by cytofluorometric analysis using fluorescein isothiocyanate or phycoerythrin-conjugated antibodies against CD29, CD34, CD44, CD45, and CD90 (Miltenyi Biotec, Bergisch Gladbach, Germany). Cell Counting Kit-8 (CCK-8) was obtained from Yeasen Biotech Co. Ltd. (Shanghai, China). Anti-CD31 and anti-Ki67 antibodies were purchased from Abcam (Cambridge, UK).
ADSCs Culture and Preparation of ADSCs@S-PLGA Scaffolds
All cells were cultured in a 5% CO2 atmosphere at 37°C, and ADSCs at passages 3–5 were used for experiments. For in vitro cell experiments, after digestion and centrifugation, the concentration of ADSCs was adjusted to 2 × 105 cells/mL. Five hundred microliters of the cell suspension was then added to the surface of a circular scaffold with a diameter of 1.2 cm and incubated for 2 h at 37°C. A complete medium (2 mL) was added for longer culture conditions. Before further analysis, the scaffolds seeded with ADSCs were transferred to a sterile six-well plate and washed gently with phosphate-buffered saline (PBS).
Adhesion and Morphological Observation of ADSCs Seeded on Scaffolds
In the process of preparing samples for scanning electron microscopy (SEM), we fixed the cells with glutaraldehyde and performed gradient dehydration. Subsequently, a platinum sputter coating was applied to the scaffolds. The growth state of the ADSCs on the scaffolds and their cell morphology were then characterized using SEM (JEM-1230, JEOL Ltd., Tokyo, Japan).
To examine cell adhesion to the scaffolds, red fluorescent protein (RFP)-ADSCs were seeded onto PLGA and S-PLGA for fluorescence imaging. Cells were visualized using a fluorescence microscope (Nikon C2 confocal microscope, Tokyo, Japan).
Toluidine Blue Assay
Toluidine blue (TB) staining was utilized for visual confirmation of 26SCS immobilization on the scaffold. To replicate in vivo release conditions, stained films were immersed in a cell culture medium containing 10% fetal bovine serum (FBS) and incubated within a cell incubator for varying durations. Following this, the S-PLGA scaffolds underwent approximately 1 h of incubation in a TB solution, 0.005% TB solution in 0.01 M hydrochloric acid with 0.2% (w/v) sodium chloride, at room temperature. The development of a purple color on the scaffolds visually confirmed the successful immobilization of 26SCS. For quantitative analysis, a colorimetric assay was employed. After incubation, S-PLGA scaffolds were immersed in 0.5 mL of PBS, followed by the addition of 0.5 mL of TB solution. After subjecting the samples to 1 h of vibration, 1 mL of hexane was introduced and vigorously mixed through vortexing to induce phase separation. The absorbance of the aqueous layers at 631 nm was then measured using a microplate reader. The quantification of the 26SCS content on the surface of S-PLGA scaffolds was determined by referencing a standard curve of absorbance at 631 nm constructed with different concentrations of 26SCS prepared through the same method. Each experimental group underwent a minimum of three replicates, and the results were expressed as mean ± SD.
Cell Viability of ADSCs Seeded on Scaffolds
The cells were divided into three groups: control, ADSCs@PLGA, and ADSCs@S-PLGA. Cell viability was evaluated using a CCK-8 assay (Hanbio, Shanghai, China). When the ADSCs were 80% confluent, cells were trypsinized, and the cell density was adjusted to 5 × 104/ml before seeding into 24-well plates containing the scaffolds (PLGA and S-PLGA) on the bottom of the wells. On the first, third, fifth, and seventh days of cell culture, the cells were digested and collected. Cytotoxicity was determined using the CCK-8.
ADSCs Differentiation Potential Assay
Rat primary ADSCs were cultured at 37°C in DMEM (GIBCO, Grand Island, NE, USA) supplemented with 10% FBS (GIBCO) in a 5% CO2 environment. After 12 h, when the cell density reached approximately 80%, the cells were isolated by trypsin digestion. Subsequently, the immunophenotypic characterization of rat ADSCs was conducted using flow cytometry. Specifically, ADSCs were assessed by Fluorescence-Activated Cell Sorting (FACS) for markers CD29, CD90, CD34, CD45, and CD44 (BioLegend, San Diego, CA, USA).
Establishment of an Ischemic Wound Model
All rats received food and water ad libitum and were housed individually to prevent fighting and attacks on the wounds. All animal experiments were conducted following the National Institute of Health Guide for the Care and Use of Laboratory Animals and were approved by the Animal Research Ethics Committee of Xijing Hospital (Xi’an, China). Before treatment, all animals were quarantined for 1 week. As shown in the figure, two double-pedicle flaps, taking the spine as the axis of symmetry, were made on the back of each rat. The pedicles were located in the head and tail directions. The subcutaneous tissue and the vascular network under the flaps were completely separated. After the skin flap was completely lifted and the bleeding stopped, it was sutured intermittently in situ. The incision was disinfected with iodophor, and erythromycin ointment was applied to prevent infection.
Surgical Treatment and In Vivo Assessment
After establishing the ischemic wound model, two full-thickness circular skin sections (diameter = 8 mm) were made in the middle of the flaps. Rats were then randomly divided into three groups (n = 6 each): control, S-PLGA, and ADSCs@S-PLGA. All the scaffolds had the same diameter of 12 mm and were implanted under the wound.
The wound area was calculated after 7, 14, and 21 days of treatment. The skin wounds of the six groups were photographed, and the area of the unhealed wound was determined using the ImageJ software (National Institutes of Health, CA, USA). The percentage of the wound closure was calculated according to %wound closure = (Amax − Ad)/Amax × 100%, where Amax is the area of the original wound, and Ad is the area of the actual wound.
Histological Analysis
At scheduled time points, wound tissues were excised and immediately fixed in 10% formaldehyde/PBS solution overnight. The samples were dehydrated using a graded series of ethanol solutions and seeded in paraffin. Tissues were then cut into 5-μm sections and stained with hematoxylin and eosin (H&E, Sigma-Aldrich, MA, USA) and Masson’s trichrome (Sigma-Aldrich, MA, USA) and visualized using an optical microscope. Collagen fibers were stained blue, and muscle fibers were stained red using Masson staining, with a minimum of six measurements per group. Contraction gap, average epidermal thickness, and collagen mean optical density were calculated using Image J software (National Institutes of Health, CA, USA).
Immunohistochemistry and Immunofluorescence
Immunofluorescent staining was conducted to label the vascular structure using CD31 (1:100; Abcam, Cambridge, UK). Immunohistochemical staining was performed to assess cell proliferation based on Ki67 (1:500; Abcam, Cambridge, UK). Representative hotspot areas (n = 3) were obtained from each mouse and processed for histological analysis.
Statistical Analysis
Data are expressed as mean values ± standard deviation (SD). For quantitative comparison and analysis, values were subjected to Student’s t test or one-way analysis of variance. Statistical significance was defined as */#P < 0.05, **/##P < 0.01, ***/###P < 0.001.
Results
Characterization and Biocompatibility of ADSCs@S-PLGA
As depicted in Fig. 1A, ADSCs were intentionally seeded onto the scaffold surface to create a therapeutic nanofibrous scaffold that resembles the properties of natural skin. Given the critical role of ADSC growth in enhancing tissue regeneration, we further explored the effects of ADSC adhesion and morphology on PLGA and S-PLGA in vitro. In Fig. 1B, the SEM image illustrates the attachment of cells to both PLGA and S-PLGA after 2 days of culture. For enhanced observation of cell attachment and morphology on the scaffold in a bigger picture, we employed RFP-ADSCs for fluorescent imaging of the seeded cells. We took a view from the edge of the scaffold to observe the morphology of RFP-ADSCs. Fluorescence microscopy revealed a uniform distribution of cells across the entire scaffold (Fig. 1C). Despite the modification of S-PLGA with sulfated chitosan (SCS) on the original PLGA electrospun membrane, it does not impede cell adhesion and spreading on both materials.
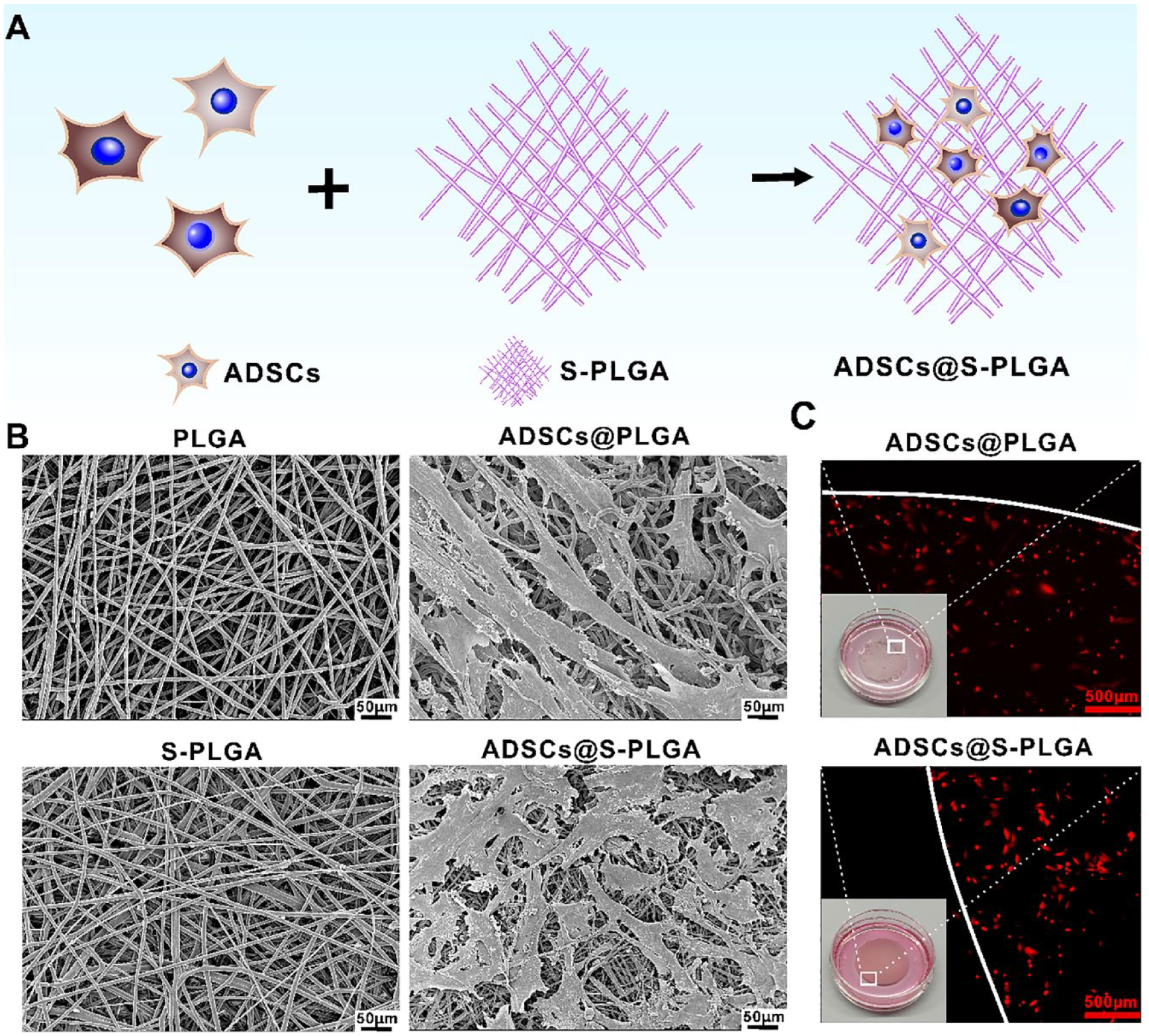
Characterization of ADSCs@S-PLGA as cell-laden scaffolds. (A) Schematic illustrations of ADSCs@S-PLGA structure. (B) Scanning electron microscope images depicting PLGA, S-PLGA, ADSCs@PLGA, and ADSCs@S-PLGA. ADSCs were cultured for 24 h on both PLGA and S-PLGA. Scale bars: 50 μm. (C) Optical images showing RFP-ADSCs seeded on the PLGA and S-PLGA scaffold on day 2 of culture. Scale bars: 500 μm. Inset: appearance details of ADSCs@PLGA and ADSCs@S-PLGA. ADSCs: adipose-derived stem cells; S-PLGA: poly(lactic-co-glycolic acid) scaffold; RFP: red fluorescent protein.
Determination Combining Capacity of 26SCS on Scaffolds
In the qualitative analysis, TB staining was employed to assess the distribution of 26SCS on S-PLGA scaffolds. As depicted in Fig. 2A, the blue color progressively lightened on the S-PLGA during the 0- to 7-day period. To precisely measure the quantity of immobilized 26SCS on the S-PLGA scaffolds, colorimetric assays were conducted. The results depicted in Fig. 2B indicate a decreasing trend in the level of 26SCS immobilization.
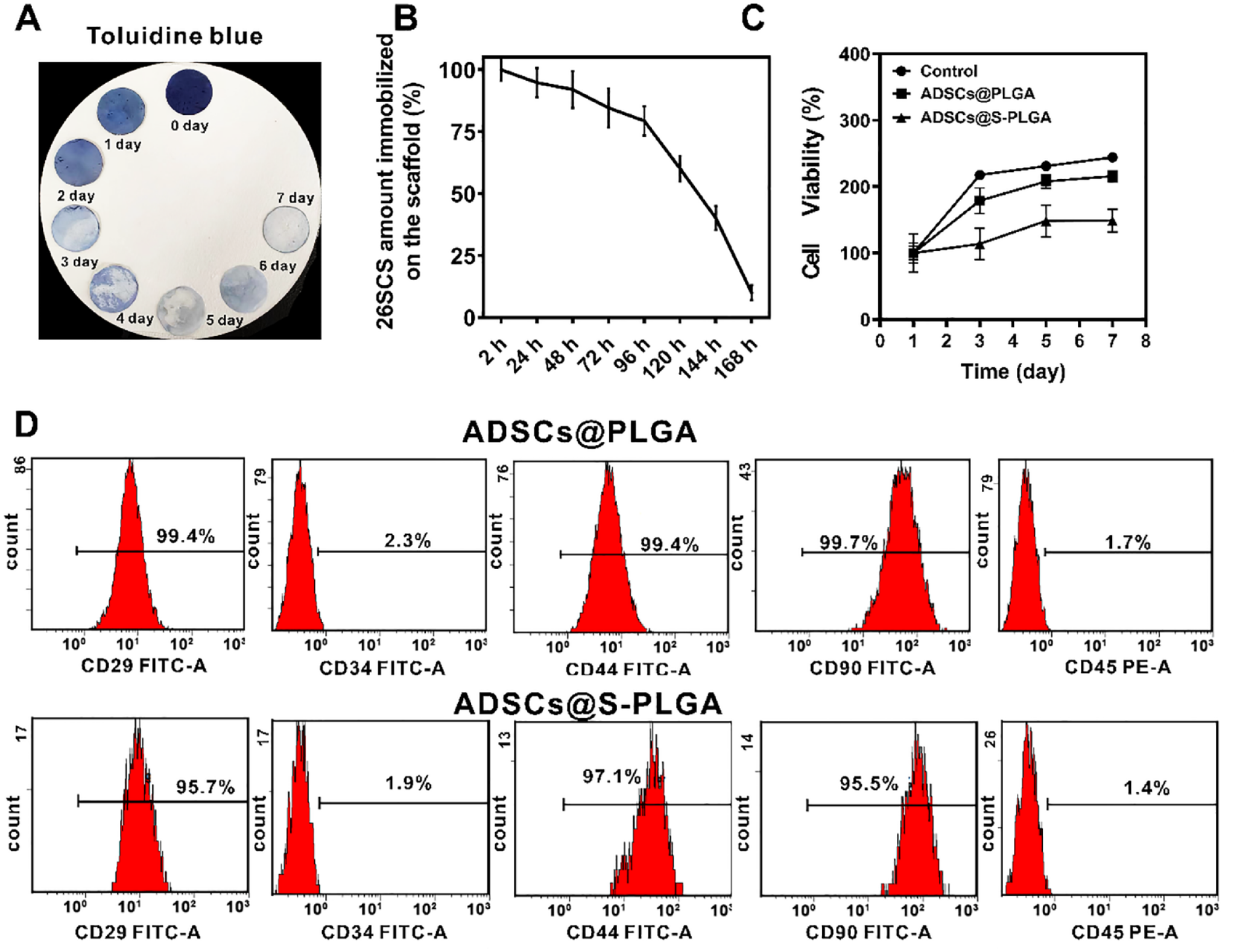
Characterization of biocompatibility of ADSCs@S-PLGA and differentiative capacity of ADSCs seeded on S-PLGA. (A) Assessment of ADSCs survival following co-culture with PLGA and S-PLGA for 1, 3, 5, and 7 days, determined using the CCK-8 Kit. (B) Visualization of 26SCS on scaffold surfaces using toluidine blue staining assay. (C) Detection of the 26SCS amount immobilized on the scaffold by simulating an in vivo environment. (D) Flow cytometry analysis of CD29, CD34, CD44, CD45, and CD90 expression in ADSCs after coculturing with S-PLGA for 1 day and 7 days. ADSCs: adipose-derived stem cells; S-PLGA: poly(lactic-co-glycolic acid) scaffold; CCK-8: Cell Counting Kit-8; 26SCS: 2-N, 6-O-sulfated chitosan.
The Survival of ADSCs on S-PLGA In Vitro
In Fig. 2C, we measured the cell survival of ADSCs on PLGA and S-PLGA scaffolds. The results indicated that cells proliferated on both PLGA and S-PLGA scaffolds. Surprisingly, the growth rate of ADSCs on S-PLGA was not the fastest.
ADSCs Differentiation Potential
The expression of CD29, CD34, CD44, CD45, and CD90, key regulators for maintaining self-renewal and multipotency of ADSCs, was verified using flow cytometry. As shown in Fig. 2D, cells were positive for CD29, CD44, and CD90, while being negative for CD34 and CD45 when seeded on PLGA and S-PLGA scaffolds. These results indicate that both PLGA and S-PLGA scaffolds contribute to maintaining the stemness of ADSCs until implantation under the skin.
Macroscopic Evaluation and Wound Contraction Rate
We assessed the improvement in skin regeneration using a combination of ADSC@S-PLGA nanofibrous scaffolds. Fig. 3A illustrates the process of establishing an ischemic wound on the back of a rat. Fig. 3B presents representative images of the full-thickness ischemic skin wound healing process in SD rats within the control, S-PLGA, and ADSCs@S-PLGA groups at 0, 7, 14, and 21 days. On the 21st day, when the ischemic wound did not heal completely, it was evident from the photos that wound healing was fastest in the ADSCs@S-PLGA group, followed by the S-PLGA group. Wound contraction rates were calculated using the formula: Wound contraction (%) = (Amax − Ad)/Amax × 100%, where Amax is the area of the original wound, and Ad is the area of the actual wound. On day 7, wound contraction of the ADSCs@S-PLGA group exhibited the most significant reduction at 50.19%, while for the S-PLGA group, it was 31.19%, and for the control group, 17.96%. By day 14, the ADSCs@S-PLGA group demonstrated a wound closure ratio of 93.59%, indicating nearly complete healing of the ischemic wounds. In contrast, the S-PLGA group still exhibited a closure ratio of 76.70%, and the control group exhibited 65.03%. On the 21st day, the wound contraction of ADSCs@S-PLGA, S-PLGA, and control groups was 97.28%, 88.63%, and 80.21%, respectively. In the ADSCs@S-PLGA group, the wound size decreased most significantly, showing a significant difference compared to the other two groups according to Fig. 3C.
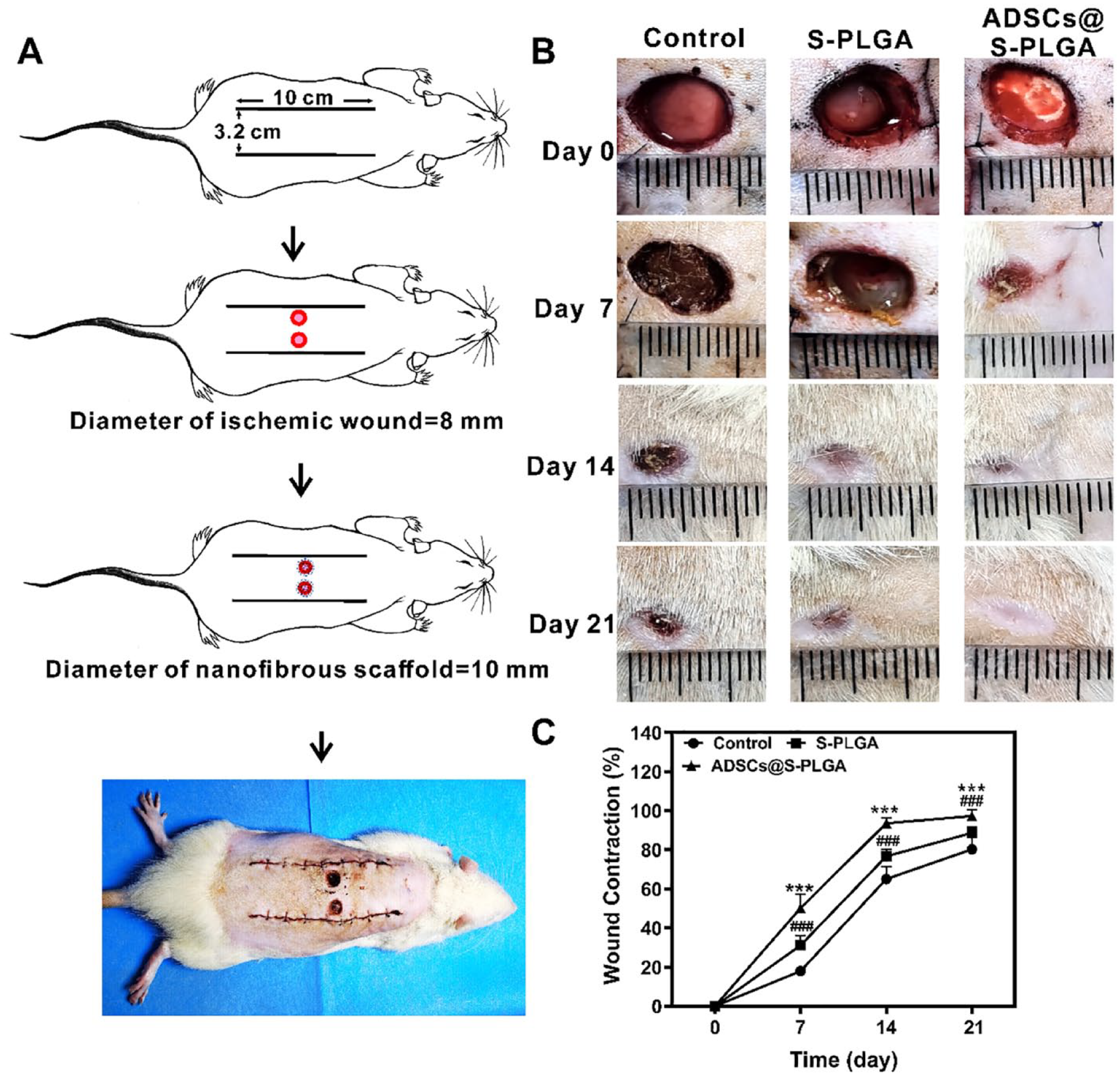
Evaluation of the nanofibrous scaffold in wound healing in vivo. (A) Illustration of the steps taken to establish the ischemic wound healing model. (B) Representative images of full-thickness ischemic skin wound healing process in SD rats with control, S-PLGA, and ADSCs@S-PLGA groups at 0, 7, 14, and 21 days. (C) Wound contraction percentages for the different groups at 7, 14, and 21 days. Statistical significance was defined as */#P < 0.05, **/##P < 0.01, ***/###P < 0.001. (Significance of the comparison between the control group and ADSCs@S-PLGA group was indicated by *P, and the significance of the comparison between the S-PLGA group and ADSCs@S-PLGAc group was indicated by #P.). SD: Sprague-Dawley; S-PLGA: poly(lactic-co-glycolic acid) scaffold; ADSCs: adipose-derived stem cells.
Histological Analysis
To gain further insight into tissue regeneration at the wound site, H&E staining was performed to reveal morphological changes in the skin layers during wound healing (Fig. 4A, C). On day 14, wound healing was markedly accelerated in wounds treated with ADSCs@S-PLGA (Fig. 4B) compared to the other groups. H&E staining at postoperative day 21 revealed the average thickness of the newly formed epidermal layers in the ADSCs@S-PLGA group was significantly larger than that in the S-PLGA and control groups (Fig. 4D). In addition, the ADSCs@S-PLGA group exhibited more abundant and better-organized granulation tissue at the wound site than the other groups, which is crucial for tissue repair and regeneration. Masson staining showed that ADSC@S-PLGA treatment resulted in relatively intact skin regeneration (Fig. 4E). As shown in Fig. 4E, quantitative analysis of the collagen staining showed that the ADSCs@S-PLGA group had a better ability to regenerate collagen than the other groups.
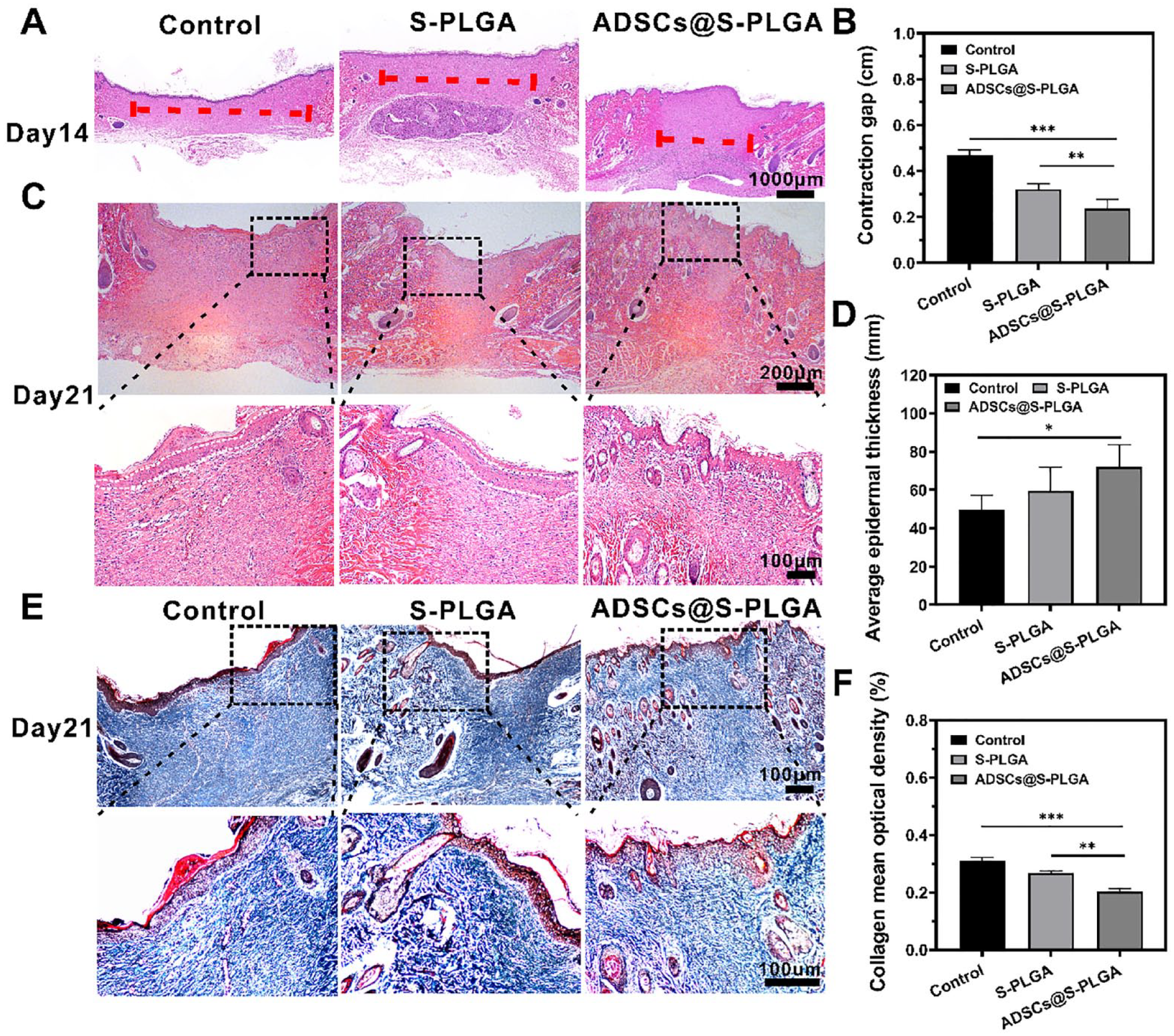
Histological analysis and investigation of the biochemical characteristics of wound healing after treatment with control, S-PLGA, and ADSCs@S-PLGA. (A) H&E staining of the wound regions to observe wound healing on day 14. The red dotted line indicates the wound surface that did not cover the new epithelia. Scale bar = 1,000 μm. (B) Wound contraction was quantified by measuring the gap between the wound margins. (C) H&E staining of the wound to observe epidermal thickness. Scale bar = 200 μm/100 μm. (D) Quantification of the thickness of the epidermis. (E) Masson staining for evaluation of collagen fibers deposited at the wound sites. Scale bar = 100 μm. (F) Quantification of the collagen density. S-PLGA: poly(lactic-co-glycolic acid) scaffold; ADSCs: adipose-derived stem cells; H&E: hematoxylin and eosin. Statistical significance was defined as *P < 0.05, **P < 0.01, ***P < 0.001.
Microvascular Formation Analysis
To study angiogenesis during the healing process, the protein expression of CD31, a key factor related to microvascular development, was determined. As shown in Fig. 5A, C, ADSCs@S-PLGA treatment markedly increased the microvessel density in the dermis compared to the control group. Ki67 is a commonly used marker of cell proliferation, and its expression was significantly higher in the ADSCs@S-PLGA treatment group on day 14 (Fig. 5B, D). These results show that microvascular formation was higher in the ADSCs@S-PLGA-treated wounds, demonstrating a positive effect on angiogenesis in this group.
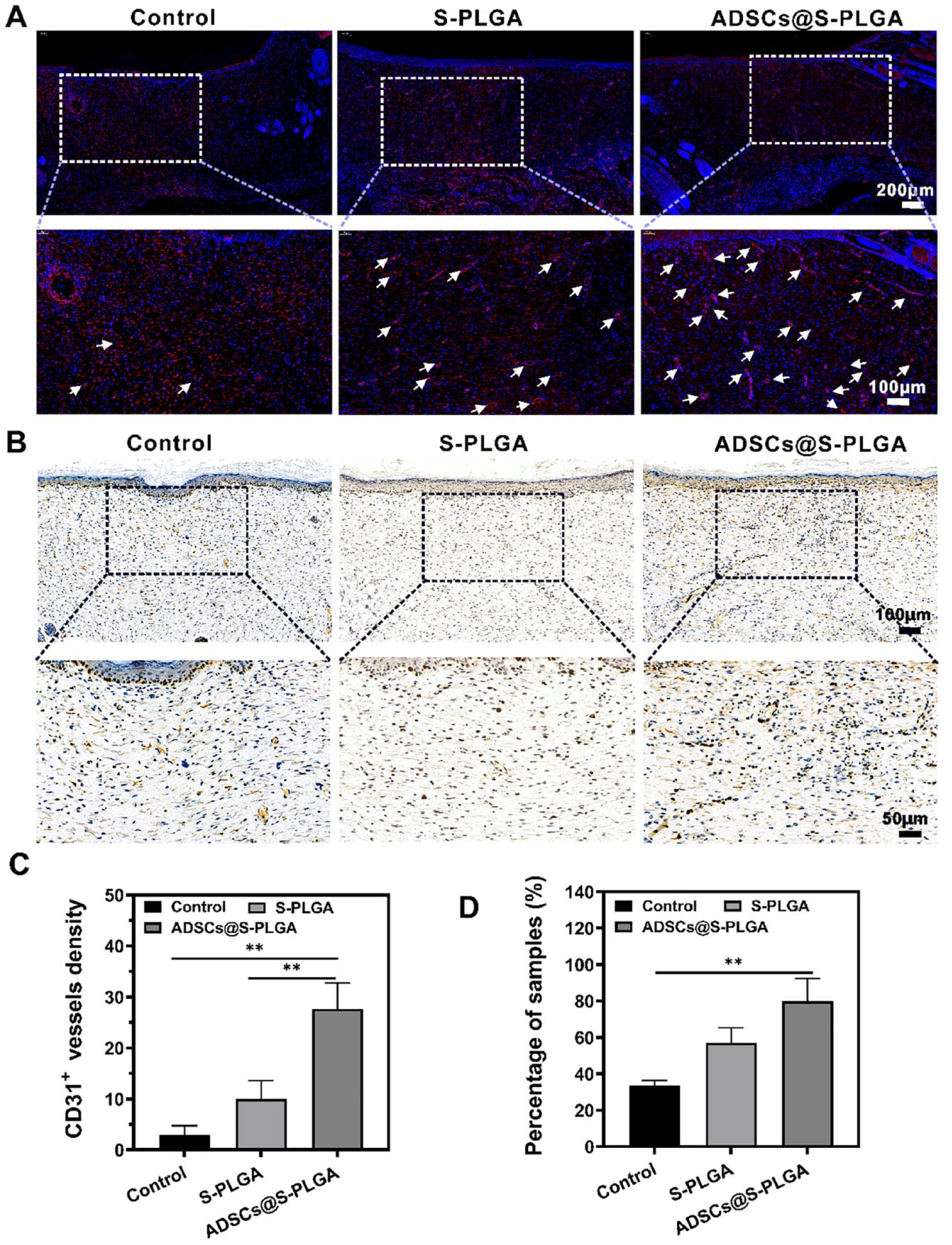
Angiogenesis and tissue proliferation assay on wound sections after different treatments. (A) Representative images of immunofluorescent CD31 staining of wound sections in control, S-PLGA, and ADSCs@S-PLGA groups at day 14 after treatment. Endothelial cells were stained with CD31 antibody (red), and the cell nucleus was stained with DAPI (blue) within wound sites. Scale bar = 200 μm/100 μm. (B) Expression of proliferating cell nuclear antigen Ki67 by immunohistochemical staining. Scale bar = 100 μm/50 μm. (C) Quantification of microvessels per microscopic field in different groups. (D) Quantitative analysis of immunohistochemistry. S-PLGA: poly(lactic-co-glycolic acid) scaffold; ADSCs: adipose-derived stem cells. Statistical significance was defined as *P < 0.05, **P < 0.01, ***P < 0.001.
Discussion
The electrospinning technique has several advantages: It is simple, elegant, reproducible, continuous, and scalable 25 . Therefore, many research groups have focused on creating improved scaffolds to simulate the functions of natural skin substitutes 26 . Over the past decade, numerous scaffolds have been developed for various tissue-engineering applications. PLGA is a biodegradable, elastomeric, biocompatible copolymer, and many studies have indicated that it can be used as a tissue-engineered scaffold for re-epithelialization, blood vessel construction, and wound closure. Electrospinning can produce PLGA fibers that can be used to accelerate wound healing and tissue reconstruction27–30. In the previous study, PLGA was doped with SCS, which has an affinity for different growth factors and has previously been used in skin tissue engineering scaffold researches31,32. However, these artificial synthetic skin substitutes cannot accomplish epithelial re-establishment in the face of refractory ischemic wounds, owing to the loss of complex biological functions. Several studies have indicated that ADSCs can differentiate into vascular endothelial cells, which may contribute to angiogenesis during the healing process after transplantation 33 . Furthermore, ADSCs secrete various growth factors and stimulate angiogenesis to enhance wound healing 34 .
In this study, we designed PLGA and S-PLGA nanofibers seeded with ADSCs as multifunctional skin substitutes. The morphology of the ADSCs on the S-PLGA or PLGA nanofibrous scaffolds was investigated after seeding for 2 days, and the flat morphology of ADSCs indicated that the scaffolds were biocompatible. The cell proliferation results demonstrated that S-PLGA slightly promotes cell proliferation, but not much more than the other two groups of active cells. This reduction in cell viability may be caused by the release of ADSCs from the ADSCs-containing scaffolds. The acknowledged biocompatibility of chitosan can be primarily attributed to the electrostatic attractive force between the positively charged amino groups on chitosan chains and the negatively charged cell membranes 35 . SCS is produced through the chemical modification of the C-2 and C-6 positions of the pyranose ring on chitosan, involving a β-linked d-glucosamine and N-acetyl-d-glucosamine copolymer 36 . Generally, GAGs are negatively charged linear polysaccharides 20 . These modifications significantly influence the electronegativity of the chitosan surface 23 , potentially facilitating the release of cells. Second, despite the good biocompatibility and cell adhesion observed in S-PLGA, we posit that cell release occurs as the surface-trapping agent is shed. To validate this hypothesis, we simulated the shedding process of the S-PLGA group in vitro. Through TB staining, we observed a noticeable decrease in the concentration of GAG on the membrane surface, supporting our conclusion. In addition, human ADSCs seeded on the S-PLGA scaffold showed differentiation potential into fibrovascular, endothelial, and epithelial components in the restored tissue. Overall, an improved therapeutic nanocomplex was obtained in this study, which not only maintained the cell proliferation ability but also preserved the differentiation potential of ADSCs.
Local tissue ischemia, characterized by reduced blood flow in the wound, can impede nutrient supply and hinder proper inflammation clearance, potentially leading to conditions such as venous ulcers, pressure sores, and chronic refractory wounds37,38. This disruption also affects the normal stages of wound repair and stands as a primary factor contributing to chronic wounds 39 . To mimic these conditions for further research, we established an ischemic model. In this study, we examined the effects of S-PLGA and ADSCs@S-PLGA scaffolds in a rat ischemic full-thickness wound model. Planimetric analysis revealed that the rats in the ADSCs@S-PLGA group exhibited the highest wound closure percentages. These findings align with recent reports demonstrating enhanced wound closure after the application of adipose-derived mesenchymal stem cells. H&E and Masson staining further indicated that the regenerated skin in the ADSCs@S-PLGA group possessed a complete epidermal structure with significant cuticular and corium layers. The planimetric analysis of wound closure suggested that ADSCs@S-PLGA combined the advantages of a scaffold for growth factor recruitment, while ADSCs contributed nutrition and growth factors to promote wound healing. Our study demonstrates that ADSCs@S-PLGA scaffolds create a healing microenvironment on the wound surface, supplying nutrition to the tissue and maintaining the differentiation potential of ADSCs, ultimately accelerating wound closure.
To study angiogenesis during the healing process, the expression of key protein factors related to vascular development, CD31, was measured. Immunofluorescence staining for CD31 also suggested that the number of angiogenic capillaries in the ADSCs@S-PLGA group was higher than that in the other groups. This demonstrated that the nanofibrous system presented significant advantages for ischemic wound healing by promoting vasculogenesis. Furthermore, immunohistochemical staining of Ki67 confirmed its role in promoting tissue proliferation.
One major repair phase of wound healing is angiogenesis, which requires cells to secrete massive amounts of angiogenic growth factors at an appropriate time at the wound margin to ensure the construction of a network of blood vessels 5 . We have ascertained that ADSCs@S-PLGA can increase growth factors, but their specific mechanism of action is uncertain. The characteristics of skin substitutes with good efficacy in animal wound-healing models may need further exploration.
In conclusion, we designed a multifunctional nano-scaffold consisting of PLGA doped with 26SCS loaded with ADSCs and successfully demonstrated its use to improve ischemic wound healing. As a delivery and retention system for ADSCs, S-PLGA supports their attachment, proliferation, and differentiation. In addition, ADSCs@S-PLGA significantly enhanced granulation tissue formation and ECM deposition and simultaneously triggered a pro-regenerative response, thereby accelerating wound healing by promoting angiogenesis. ADSCs@S-PLGA supported the delivery and engraftment of stem cells, as well as their differentiation into epithelial components, and demonstrates that S-PLGA is a promising delivery vehicle for stem cells in clinical reconstruction settings. Our study provides evidence that ADSCs@S-PLGA can promote wound healing and has the potential to become a new therapeutic alternative for skin tissue-engineered scaffolds.
Footnotes
Acknowledgements
The authors would like to thank Dr. Zhou Yu for the beneficial discussions (Xijing Hospital, Fourth Military Medical University).
Author Contributions
GuoFang Shen, Xi Zhang, and Yan Jiao designed the experiment. Tong Shen and Yuanman Yu participated in electrospinning synthesis. Juanli Dang and Yu Zhou participated in animal experiments. Yan Jiao wrote the manuscript. All authors contributed to the manuscript revision and approved the submitted version.
Availability of Data and Material
The data, results, and conclusions of this study are all original and authentic and have not been published in any other academic journals or conferences.
Ethical Approval
This study was approved by the Laboratory Animal Care and Welfare Committee of the Fourth Military Medical University, Shaanxi Province, China.
Statement of Human and Animal Rights
This study does not contain human subjects.
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a program grant from Xijing Hospital, Air Force Medical University (XJZT24CY57).
