Abstract
BACKGROUND:
Lung cancer is a major burden to global health and is still among the most frequent and most lethal malignant diseases. Macrophage migration inhibitory factor (MIF) is a proinflammatory cytokine involved in a variety of processes including tumorigenesis, formation of a tumor microenvironment and metastasis. It is therefore a potential prognostic biomarker in malignant diseases.
OBJECTIVE:
In this study, we investigated the applicability of MIF in serum samples as a biomarker in lung cancer.
METHODS:
In a retrospective approach, we analyzed the sera of 79 patients with non-small-cell lung cancer (NSCLC) and 14 patients with small-cell lung cancer (SCLC) before the start of chemotherapy, as well as before the second and third chemotherapy cycle, respectively. Serum MIF levels were measured using a sandwich immunoassay with a sulfo-tag-labelled detection antibody, while pro-gastrin releasing peptide (proGRP) levels were determined with an enzyme-linked immunosorbent assay.
RESULTS:
No difference in serum MIF levels between responders and non-responders to chemotherapy was observed at all time points, while proGRP levels were significantly lower in responders before the second chemotherapy cycle (
CONCLUSIONS:
From the present results, there is no indication that serum MIF may serve as a biomarker in prognosis and monitoring of response to therapy in lung cancer. Limitations of this study include its retrospective design, the inclusion of a larger NSCLC and a smaller SCLC subgroup, the classical chemotherapeutic treatment, the use of a non-diagnostic immunoassay (RUO-test) for MIF measurement and the lack of a validation cohort. Strengths of the study are its highly standardized procedures concerning sample collection, preanalytic treatment, measurements and quality control of the laboratory assays.
Keywords
Introduction
Lung cancer is a major health burden worldwide and is being held accountable for 18% of all cancer deaths [1]. The major cause of lung cancer is tobacco smoking [2]. Although new therapeutic possibilities such as immunotherapy have considerably improved the treatment of lung cancer, overall survival remains relatively low, especially in advanced stages [3–5].
Blood-based biomarkers can play an important role in the diagnosis, prognosis and monitoring of therapy response of malignant diseases, as they possess several advantages compared to other diagnostic tools. They are readily available in most laboratory medicine environments without the need for additional specialized equipment. On the other hand, blood-based biomarkers are easily obtained on a virtually non-invasive basis, in contrast to procedures such as tissue biopsies or lumbar punctures.
Due to the great impact of lung cancer on public health, a multitude of biomarkers has been studied. Some of the most widely studied biomarkers in lung cancer are cytokeratin fragment 19 (CYFRA 21-1), carcinoembryonic antigen (CEA) and pro-gastrin releasing peptide (proGRP) [6–8]. In studies with lung cancer patients, these markers showed promising sensitivities and specificities, but did not reach a level of reliability sufficient for routine application in a clinical setting. Moreover, when a potential use as a screening tool is considered, positive and negative predictive values are low due to the overall low prevalence of lung cancer, which is estimated to be in the range of 1–4% in high risk groups [9]. Although combinations of biomarkers can further increase sensitivities and specificities [8, 11], the search for new biomarkers and their clinical evaluation remain crucial factors to facilitate early diagnosis of lung cancer.
The macrophage migration inhibitory factor (MIF) is a proinflammatory cytokine with an additional tautomerase activity which is expressed by a variety of cells including T cells, monocytes, macrophages, dendritic cells, B cells, granulocytes, epi- and endothelial cells, fibroblasts and cells of the anterior pituitary gland [12, 13]. Its active physiological form is a homotrimer with a molecular weight of 37.5 kDa [14, 15]. MIF is highly pleiotropic and hence exerts a multitude of effects on the immune system and the general inflammatory response. It can counteract the immunosuppressive effects of glucocorticoids [16], is an activator of macrophages and T cells [12], stimulates the release of proinflammatory molecules such as TNF, IFN-
In this study, we sought to evaluate the potential of MIF serum levels as a predictive factor of therapy response and a prognostic factor for survival. Moreover, the results are compared to established biomarkers of lung cancer so as to better assess the potential role ofMIF.
Patients, materials and methods
The cohort reported in this study and the applied chemotherapeutic agents have already been described elsewhere [34], and is therefore covered only briefly here. In a retrospective approach, serum samples of 93 patients suffering from newly diagnosed or recurring lung cancer were investigated, comprising 79 patients with NSCLC (53 male, 26 female) and 14 SCLC (8 male, 6 female). NSCLC patients were classified as UICC stage 3 (
Response to chemotherapy was determined by CT-based staging after cycle 2 according to the WHO classification, thereby defining
MIF concentrations were determined using the Human MIF Assay (Meso Scale Diagnostics, Rockville, MD, USA) on a Meso Quickplex SQ120 imager (Meso Scale Diagnostics, Rockville, MD, USA). This method is based on a sandwich immunoassay with a sulfo-tag-labelled detection antibody. Detection is conducted by electroluminescence using tripropylamine and [Ru(bipyridyl)3]2 + as catalyst. Regeneration of the Ru(II) complex is controlled by an anodic current. Serum levels of CEA and CYFRA 21-1 were measured by electrochemiluminescence on the Cobas Elecsys E 411 platform (Roche Diagnostics, Mannheim, Germany), ProGRP by enzyme-linked immunosorbent assay (ELISA, IBL, Hamburg, Germany).
Statistical analyses were performed with IBM SPSS Statistics (version 23, International Business Machines Corporation, Armonk, NY, USA). Medians, interquartile ranges (IQRs) and ranges are used in tables and diagrams as measures of central tendency and measures of variation, respectively. Differences in MIF and proGRP between groups were calculated with Mann-Whitney-U tests. Correlations between biomarkers were assessed with Spearman’s rank-order correlation, whereas correlations of biomarkers with therapy response were assessed with point-biserial correlations. To evaluate the diagnostic power of MIF and proGRP, receiver operating characteristic (ROC) curves with tumor progression as state variable were computed. Areas under the curves (AUCs) as well as sensitivities at specificities of 90% (Sens90) and 95% (Sens95), were calculated from ROC curves. Prognostic value of MIF and proGRP was investigated by univariate log-rank tests and visualized with Kaplan-Meier curves. The significance level was set to
Results
Serum levels of MIF and proGRP
Pretherapeutic concentrations of MIF and proGRP in responders and non-responders with NSCLCs are given in Table 1, while the respective concentrations in SCLCs are shown in Table 2. MIF levels did not differ significantly between responders and non-responders at all time points both in NSCLCs and SCLCs, as determined by Mann-Whitney-U tests. Concentrations of proGRP only differed between responders and non-responders with NSCLCs at the second chemotherapy cycle, with responders possessing a lower concentration than non-responders.
Serum concentrations of MIF and proGRP in responders and non-responders with NSCLC
R: responders, NR: non-responders, IQR: interquartile range. Concentrations are given in ng/mL (MIF) and pg/mL (proGRP), respectively.
Serum concentrations of MIF and proGRP in responders and non-responders with NSCLC
R: responders, NR: non-responders, IQR: interquartile range. Concentrations are given in ng/mL (MIF) and pg/mL (proGRP), respectively.
Serum concentrations of MIF and proGRP in responders and non-responders with SCLC
R: responders, NR: non-responders, IQR: interquartile range. Concentrations are given in ng/mL (MIF) and pg/mL (proGRP), respectively.
Spearman correlation of pretherapeutic levels of MIF with pretherapeutic levels of proGRP in NSCLC cases showed a positive correlation coefficient (
As given in Tables 1 and 2, changes in biomarker concentrations from the start of chemotherapy to the second and third chemotherapeutic cycle did not differ significantly between responders and non-responders for both biomarkers and in both subgroups. In SCLC, median levels of proGRP were considerably higher in non-responding patients and kinetics differed between both response groups, too. However, the numbers of patients investigated were quite low preventing an acceptable level of significance.
Influence of histopathology in NSCLC
Pretherapeutic serum concentrations of MIF and proGRP in the most prominent histopathological entities, i.e. adenocarcinomas and squamous cell carcinomas, were compared to the remaining NSCLC cases, respectively. According to the results shown in Table 3, no significant differences were found.
Pretherapeutic serum concentrations of MIF and proGRP stratified for histopathological entities in NSCLC cases
Pretherapeutic serum concentrations of MIF and proGRP stratified for histopathological entities in NSCLC cases
As MIF expression levels in tissue samples of squamous cell carcinomas (SCC) have been found to be associated with tumor progression [33], further analyses of MIF in patients with SCC were performed. No differences in MIF concentrations between responders and non-responders to chemotherapy within the SCC cohort were observed in pretherapeutic samples (
Results from ROC curve analyses of all three time points are shown in Table 4, while ROC curves are depicted in Fig. 1. While proGRP was able to significantly discriminate between therapy response and tumor progression at cycle 2, MIF failed to distinguish responders from non-responders at all time points. Accordingly, Sens90 and Sens95 values were well below 20% and 10%, respectively, with the exception of proGRP at cycle 2 reaching values above 40%.
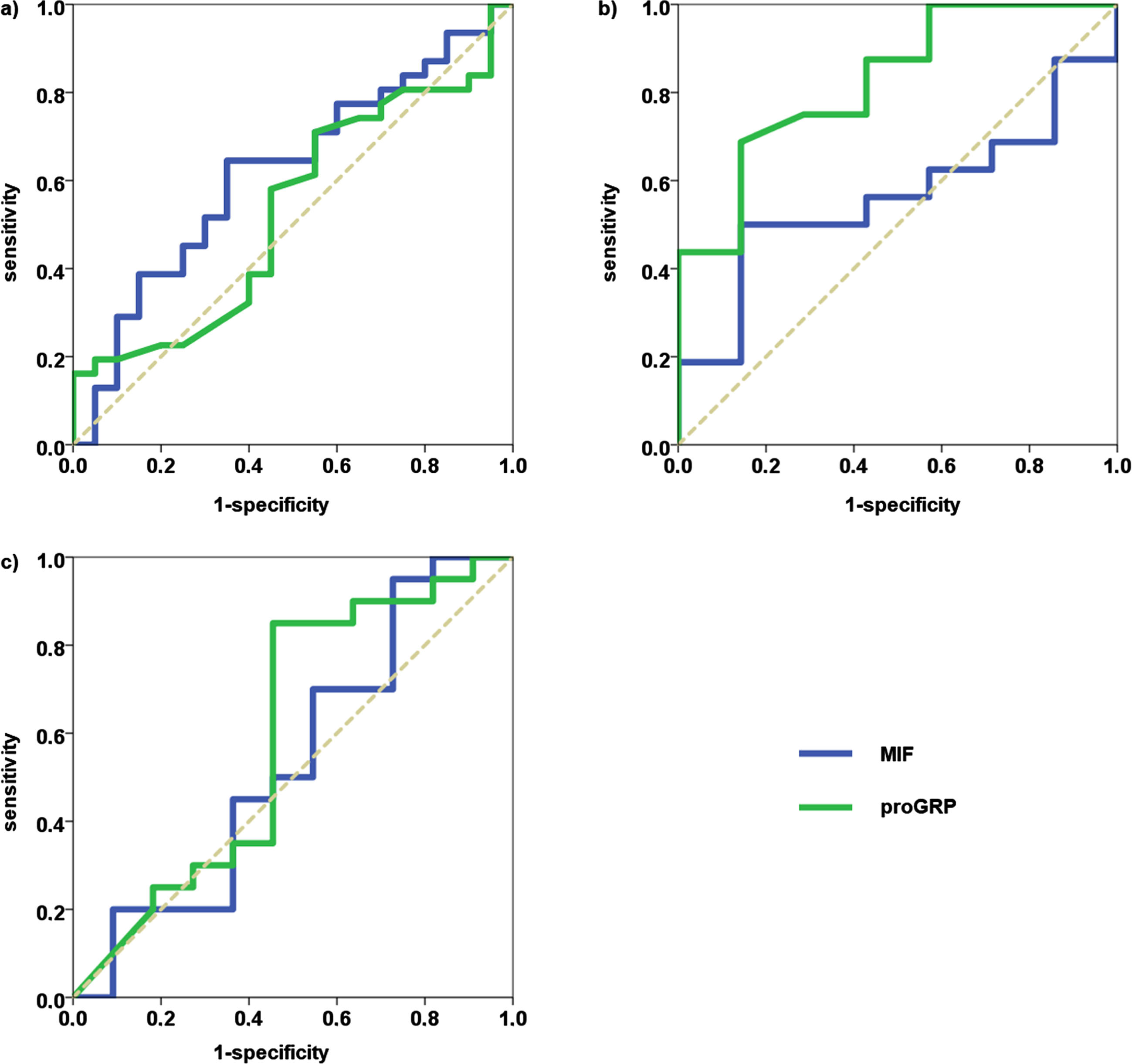
ROC curves of MIF (blue) and proGRP (green) in NSCLC patients with tumor progression as state variable. a) pretherapeutic biomarker concentrations, b) concentrations at cycle 2 and c) concentrations at cycle 3.
Results from ROC curve analyses in NSCLC patients with tumor progression as state variable
AUC: area under the curve, CI: confidence interval.
To assess the prognostic value of MIF and proGRP in NSCLC patients, the NSCLC group was stratified according to the respective median serum concentrations. Results of subsequent analyses of overall survival with Kaplan-Meier curves are given in Table 5 and Fig. 2. At cycle 2, proGRP levels above the median value of 13.0 pg/mL were associated with significantly shorter overall survival. At other time points, and at all time points in the case of MIF, no significant differences in survival times between low and high biomarker concentrations were obtained.
Kaplan-Meier analyses of NSCLC patients
CI: confidence interval.
Kaplan-Meier analyses of NSCLC patients
CI: confidence interval.

Kaplan-Meier curves of NSCLC patients of a) pretherapeutic MIF concentrations, b) MIF levels at cycle 2, c) MIF levels at cycle 3, d) pretherapeutic proGRP concentrations, e) proGRP levels at cycle 2, f) proGRP levels at cycle 3.
In ROC curve analyses, neither proGRP nor MIF could differentiate significantly between responders and non-responders before the start of chemotherapy (proGRP:
In Kaplan-Meier analyses after stratification by median serum concentrations of proGRP, no difference in survival times before chemotherapy (
Discussion
Lung cancer is still a severe burden to public and individual health. It is among the most frequent and most lethal types of cancer both in industrial and in developing countries. Although novel treatment options, such as immune checkpoint inhibitors expand therapeutic strategies, success rates remain relatively low in advanced stages [37]. Biomarkers have the potential to guide therapeutic decision making as well as provide a basis for prognosis. A biomarker with promising biochemical properties and a vital position within oncological pathways is MIF. Not only does MIF influence crucial processes such as apoptosis, it also serves as a link between tumorigenesis, inflammation and the immune system [38]. MIF has been shown to promote the tumor microenvironment, for instance, by stimulating tumor-associated macrophages [28, 29]. Additionally, MIF may affect the cell cycle and cell growth by interacting with pathways involving p53, Bcl-2 or PI3K/Akt [13, 18]. Additionally, MIF supports immunoevasion of tumor cells by promoting suppression of T-cell activation [39–41] and activation of immunosuppressive functions of myeloid-derived suppressor cells [42, 43].
It has already been shown that MIF may facilitate prognosis in lung cancer [32, 33]. In the present study, we investigated the possible use of MIF and proGRP for prediction of therapy response and stratification of overall survival in a cohort of lung cancer patients. Pretherapeutic concentrations of MIF and proGRP in NSCLCs and SCLCs did not differ between responders and non-responders to subsequent chemotherapy. Therefore, it was assumed that initially, the biochemical status concerning MIF and proGRP was similar in responders and non-responders. Moreover, the histopathology within the NSCLC group did not influence pretherapeutic biomarker levels. In contrast to expectations, no differences in MIF concentrations between responders and non-responders occurred in the course of chemotherapy. As MIF promotes tumor growth and tumor microenvironment, tumor progression in non-responders was thought to increase MIF secretion as compared to responders.
The only significant results were obtained for proGRP in the NSCLC group before the second chemotherapy cycle, with responders possessing lower serum concentrations of proGRP and accordingly longer survival times in patients with lower proGRP levels. A possible explanation for this interesting finding is that after the start of chemotherapy and the onset of therapeutic response (or lack thereof in non-responders) with reduction in tumor burden, an increasing difference in serum marker concentrations and better differentiation between responders and non-responders based on these concentrations may occur. However, these considerations do not account for the lack of statistical significance before the third chemotherapy cycle. A thorough explanation for this convergence of proGRP concentrations at cycle 3 cannot be given at the moment.
Comparison with established biomarkers
We have already reported results for the established biomarkers carcinoembryonic antigen (CEA) and cytokeratin-19 fragments (CYFRA 21-1) as well as for the immunogenic biomarker high mobility group box 1 protein (HMGB1) in the present cohort [34]. CYFRA 21-1 was well able to discriminate between responders and non-responders to chemotherapy in the case of NSCLCs, with significantly lower concentrations of CYFRA 21-1 in responders after the second and third chemotherapy cycle. In contrast, CEA allowed for a good differentiation between responders and non-responders in SCLCs. Furthermore, not only did CYFRA 21-1 show a good performance in ROC curves of NSCLC patients with areas under the curve above 0.7, but also yielded a significant prognostic value in Kaplan-Meier curves, with overall survival being 3.7–4.8 times higher in patients with a low CYFRA 21-1 concentration than in those with high serum levels. Similar results have also been obtained by other groups (reviewed in [44]). Clearly, both MIF and proGRP could not compete with these biomarkers. This is a remarkable result, since MIF has been linked to inflammatory processes which also occur within a tumor environment. Additionally, our study comprised patients in advanced tumor stages, tantamount to relatively high tumor mass and therefore not only a high relative, but also a potentially large absolute reduction of tumor mass in the case of responders to therapy. Hence, it was assumed that response to therapy would be associated with a decrease in serum levels of MIF, and that MIF in turn may serve as a significant factor in overall survival as determined by Kaplan-Meier analyses. It is unclear whether this is an intrinsic characteristic of lung cancer in general or a specific behavior of the cohort investigated here. However, as outlined above, studies concerning the role of MIF in cancer have found somewhat contradicting results. Our current data indicate that the subtype of NSCLCs does not change the secretion of MIF significantly, thereby ruling out the possibility of a histopathological sampling effect. It is unclear whether the lack of a change in MIF concentrations is influenced by chemotherapy itself in this cohort. Chemotherapeutic agents such as cisplatin or doxorubicin are known to promote inflammatory signal transduction [45]. However, due to the heterogeneous therapies and the general design of the present study, it was not possible to determine if the respective therapies themselves were able to elevate MIF concentrations and counterbalance a possible decrease of MIF serum levels in responders. In the SCLC subgroup, low MIF concentrations before the third chemotherapy cycle were associated with significantly longer survival times. This may indicate that serum MIF levels could be useful as a prognostic tool in SCLC patients. However, it must be emphasized that the significance of such a result is clearly limited due to the very limited number of SCLC patients in this study.
Therefore, more research is needed towards the effects and interlinkage of MIF in biochemical pathways involved in lung cancer progression. At present, we conclude that there is no indication that serum MIF levels may serve as a biomarker in prognosis and monitoring of response to chemotherapy in NSCLC. From the present data, no conclusions can be drawn regarding the applicability of MIF in immunotherapy of lung cancer, for instance with PD-1/PD-L1 inhibitors in advanced stages of NSCLC. Due to the constant advancement in therapeutic options with biologicals, more research towards the role of MIF and similar biomarkers in patients treated with such novel therapies is needed.
Obviously, there are some limitations of the study like the retrospective design, the inclusion of a larger NSCLC and a smaller SCLC subgroup, the classical chemotherapeutic treatment, the use of a non-diagnostic immunoassay (RUO-test) for MIF measurement and the lack of a validation cohort. However, samples were analyzed in a standardized procedure regarding collection, storage, treatment prior to measurement and analysis. High quality standards were obtained by standardized calibration curves, internal controls, measurement of serial samples within the same runs and final crossplate checks to minimize inter-assay variations. Radiological imaging analyses were performed by experienced, independent radiologists and data interpretation was done independently from data acquisition.
Conclusion
The present study unfortunately revealed the lack of clinical utility of soluble MIF in serum samples of advanced lung cancer patients for monitoring therapy response and estimating prognosis in both NSCLC and SCLC subtypes.
Footnotes
Acknowledgments
We thank the biobank of the Ludwig Maximilians-University (LMU) Munich for storage of serum samples.
Author contributions
CONCEPTION: S.H. and J.v.P.
DATA CURATION: S.H., J.v.P., S.B., N.T. and A.R.
ANALYSIS OF DATA: S.B., A.R. and S.H.
PREPARATION OF THE MANUSCRIPT: S.H. and A.R.
REVISION FOR IMPORTANT INTELLECTUAL CONTENT: J.v.P., S.B. and N.T.
SUPERVISION: S.H. and J.v.P.
Conflict of interest
S. H. is one of the editors of the special issue “Lung Cancer Tumor Markers” but had no involvement in the peer review process of this article. The other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Ethics Committee (IRB) of the Ludwig Maximilians-University (LMU) Munich (UE-Nr 114-13, date of approval: 05.08.2013). Anonymized biobanked serum specimens of the LMU Munich were used for this retrospective study as specified in the IRB approval (waiver for informed consent).
