Abstract
BACKGROUND:
Lung cancer is prone to metastasize to the brain, which is difficult for surgery and leads to poor prognosis due to poor chemotherapy efficacy.
OBJECTIVE:
Our aim is to evaluate the efficacy and safety of stereotactic body radiotherapy (SBRT) for brain multi-metastases.
METHODS:
In the retrospective study, a total of 51 non-small cell lung cancer (NSCLC) patients with brain multi-metastases (3–5 metastases) receiving SBRT in the local hospital between 2016 and 2019 were enrolled for analyzing the efficacy and safety of SBRT. The primary endpoints included 1-year local control rate, radiotherapy toxicity, overall survival and progression-free survival.
RESULTS:
The median follow-up for the enrolled patients was 21 months, and the 1-year and 2-year OS rates were 82.4% and 45.1%, respectively. Demographic analysis showed no significant differences between SBRT alone and combination with whole brain radiotherapy in clinical characteristics including age, gender and Eastern Cooperative Oncology Group performance status. The 1-year local control rate was 77.3% (17/22) for SBRT alone, which was comparable to 79.3% (23/29) of combined radiotherapy. Cox proportional hazard regression demonstrated that the prognostic benefit of combining WBRT was not significantly superior to SBRT alone (HR
CONCLUSION:
The current research suggested that SBRT alone could effectively relieve tumor burden and improve the prognosis and quality of life for NSCLC patients with brain multi-metastases, which should be validated in further prospective clinical trials.
Introduction
Lung cancer patients are prone to develop brain metastasis, more than half of which are multiple brain metastases when diagnosed [1, 2, 3]. Multiple brain metastases can cause central nervous system dysfunction and rapid progression, leading to very poor prognosis and early relapse, whose natural course is less than 3 months [4, 5]. Furthermore, chemotherapeutic efficacy is unfavorable due to the blood-brain barrier and radical resection is difficult to achieve because of its special location [6]. Therefore, radiotherapy is the most recommended treatment for advanced patients with brain metastases [7, 8, 9].
The current radiotherapies mainly consist of whole brain radiotherapy (WBRT) and stereotactic body radiotherapy (SBRT). The application of WBRT can effectively relieve the neurological symptoms and improve biological functions of advanced patients [10, 11, 12, 13]. WBRT also hold prognostic benefits, but the improve survival is limited to 6 months [14, 15]. Another limitation of WBRT is that it’s difficult to produce curative effects on metastases with a diameter of more than 2 cm, and radiological damage is often inevitable to normal brain tissue [16, 17, 18]. SBRT is a radiation therapy method based on stereotactic principle, combined with target localization system and three-dimensional treatment strategy to achieve precise high-dose convergence to targeted lesions [19, 20]. It can not only precisely generate high-dose irradiation to the targeted lesions, but also protect the normal brain tissues from radiological damage, contributing to reduced radiotherapy toxicity, improved curative effects, and relieved compression and neurological symptoms [21, 22, 23, 24, 25].
Therefore, in current clinical practice, SBRT has been increasingly applied in treating brain metastases [26, 27, 28]. It can be used alone or in combination with WBRT, but its efficacy and safety are still inconclusive. Therefore, our research aims to evaluate the efficacy and safety of applying SBRT alone in NSCLC patients with brain multi-metastases and the necessity of combining WBRT.
Methods
Patients and data collection
NSCLC patients with brain multi-metastases who were treated with SBRT in Radiotherapy Department of Baoji High-tech Hospital between January 1, 2016, through December 31, 2019 were enrolled in our study. The specific inclusion criteria were listed as follows: (I) the diagnosis of NSCLC was confirmed by positive histology examination other than by autopsy or a death certificate; (II) brain multi-metastases (3–5 metastases) were confirmed by comprehensive imaging examination (brain magnetic resonance imaging (MRI), positron emission tomography/computed tomography (PET/CT), and/or CT); (III) patient age was between 18 and 80 years old; (IV) the Eastern Cooperative Oncology Group performance status (ECOG PS) was at least 2 before the onset of SBRT; (V) all lesions were suitable for high-dose radiotherapy. The exclusion criterions were set as follows: (I) patients were diagnosed with other synchronous primary tumors; (II) patients who had received radiotherapy for the primary lesions or metastases before onset of SBRT; (III) patients were diagnosed with additional synchronous metastasis other than brain.
The included patient characteristics were as follows: demographic characteristics (patient age at treatment, gender), tumor features (primary tumor size, number of brain metastases), radiotherapy information (whether combination with WBRT, total radiotherapy dose and PTV size), length of survival (overall survival status and time, progression status), and radiotherapy toxicity (type, grade).
This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the local institutional review board. The patients were included in this study only after obtaining orally or written informed consent.
Radiotherapy procedure
Prior to SBRT, patients underwent four-dimensional CT (4D-CT) and/or MRI simulation. Intravenous and/or oral contrast and fusion of simulation with imaging examinations were taken into judgement by the treating radiation oncologist (RO) to contour the gross tumor volume (GTV) and organs at risk (OARs) [29]. An internal target volume (ITV) was generated by combining GTV volumes in all binned respiratory phases. Then the planning target volume (PTV) was created by expanding the GTV or ITV in an isotropic manner by 0–3 mm, which was based on the accuracy of patient setup, extent of target motion, quality of simulation images and precision of image fusion.
SBRT was performed using a dedicated Cyberknife G4 system (Accuray, Sunnyvale, California, U.S.A.) according to the institutional treatment protocol. Standard prescription of SBRT was total dose of 30 Gy in 3 fractions with the planning intent of achieving 95% coverage of the PTV with 100% of the prescription dose, and the radiation dose and fraction schedule could be modified according to established guidelines for dose-volume limits to OARs. Patient positioning was verified based on offline CT and the Synchrony Respiratory Tracking System was used to track tumor movement in real time.
Follow-up and evaluation
Patients underwent follow-up CT or MRI examination one month after the initiation of SBRT and then every 3 months in the first two years or every 6 months after two years.
The primary end point of this study was the 1-year local control rate evaluated independently for each brain metastases lesion using Response Evaluation Criteria in Solid Tumors (RECIST) criteria v1.1 [30]: Complete response (CR) defined as the disappearance of the target lesion; Partial response (PR) defined as at least a 30% decrease of diameter of the target lesion; Stable disease (SD) defined as less than a 30% decrease or less than a 20% increase of diameter of the target lesion; Progressive disease (PD) defined as at least a 20% increase of diameters of the target lesion.
The secondary end points were 2-year overall survival (OS), 2-year progression-free survival (PFS) and radiotherapy toxicity. OS was defined as the total survival time from the initiation of radiotherapy to death or the last date of follow-up. PFS was defined as the survival time from the initiation of radiotherapy to the day PD of the irradiated tumor was first noticed. Radiotherapy toxicities consist of acute reactions and late toxicities, which were evaluated according to the Common Terminology Criteria for Adverse Events version 5.0 (CTCAE v5.0) standard.
Statistical analysis
The enrolled patients were divided into two groups according to receipt of types of radiotherapy, and their clinical-pathological characteristics were compared using
Most statistical analysis was performed using R software (version 4.0.1). All
Results
Patient characteristics
A total of 51 advanced NSCLC patients with 193 brain metastases lesions treated with SBRT were enrolled in the study. The summary statistics for the demographics and clinic-pathologic characteristics of NSCLC patients are shown in Table 1. There are 28 male patients (54.9%) and 23 female patients (45.1%), and their age ranged from 31 years to 75 years (median age 62 year). 42 patients (82.4%) were treated with the schedule 30 Gy in 3 fractions, and 9 patients (17.6%) received 36 Gy in 3 fractions. The median follow-up was 21 months (IQR, 12–28 months), and the1-year, and 2-year OS rates were 82.4% and 45.1%, respectively. The1-year and 2-year PFS rates were 78.4% and 31.4%, respectively. Among the 51 patients, 22 (43.1%) received SBRT alone and 29 (56.9%) received SBRT combined with whole brain radiotherapy (WBRT). Demographic analysis showed no significant differences between the two groups in clinical characteristics including age, gender and ECOG PS score (Table 1).
Patient and tumor characteristics of NSCLC patients grouped by radiotherapy modality (
51)
Patient and tumor characteristics of NSCLC patients grouped by radiotherapy modality (
Values are expressed as
In the first year of post-radiotherapy follow-up, 22.7% (5/22) patients treated with SBRT alone exhibited MRI-monitored brain metastases progression based on RECIST criteria v1.1, while 20.7% (6/29) patients treated with SBRT combined with WBRT exhibited progression. Thus the one-year local control rates were 77.3% and 79.3% for patients receiving SBRT alone or combining SBRT and WBRT, in which
One-year local control rate of NSCLC patients receiving radiotherapy
One-year local control rate of NSCLC patients receiving radiotherapy
Univariate and multivariate Cox regression analysis were conducted to identify the clinic-pathological factors of OS and PFS (Tables 3 and 4). In univariate analysis for OS, patient age, ECOG PS and PTV
Prognostic factors for overall survival (OS) in NSCLC patients
Prognostic factors for overall survival (OS) in NSCLC patients
Values are expressed as hazard ratios (HR) with 95% confidence intervals unless otherwise indicated.
Prognostic factors for progression-free survival (PFS) in NSCLC patients
Values are expressed as hazard ratios (HR) with 95% confidence intervals unless otherwise indicated.
As for PFS (Table 4), the independent prognostic factors were patient age (
Summary of radiotherapy toxicities of NSCLC patients
Kaplan-Meier curves showing overall survival (A) and progression-free survival (B) by radiotherapy modality.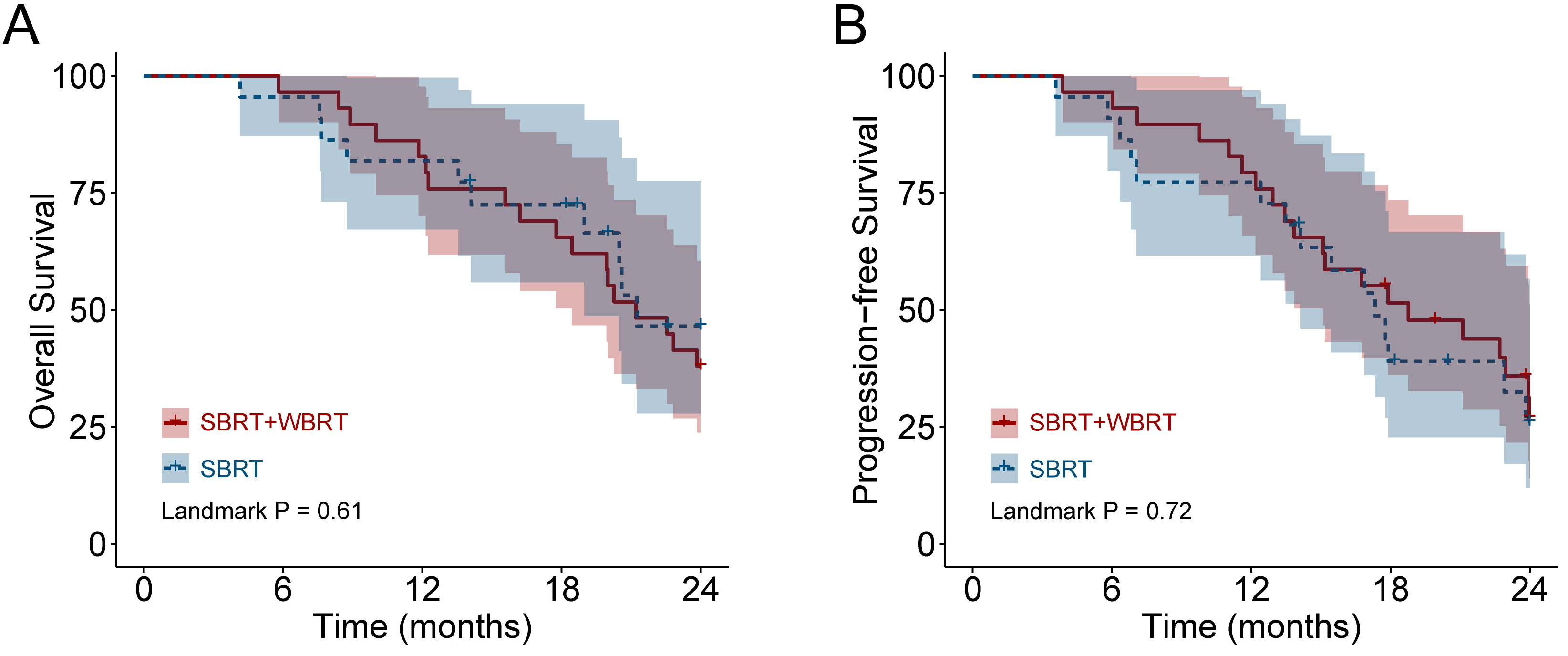
Among the entire cohort of 51 patients, a total of 16 patients had reported radiotherapy toxicity according to the CTCAE v5.0 standard (Table 5). At the time of data analysis, grade 1–2 toxicities (six with nausea, three with slight myasthenia, two with dizzy, and two with allotriosmia) occurred in 13 patients, grade 3 toxicities occurred in 3 patients (two with leukopenia and one with thrombocytopenia), and no grade
Discussion
NSCLC patients with brain multi-metastases usually hold natural course less than 3 months [4, 5]. Due to the blood-brain barrier, chemotherapeutic efficacy is very poor for brain metastases, and radical surgery is also very difficult. In addition to unfavorable therapeutic efficacy, brain metastases can cause severe central nervous system dysfunction and progress rapidly, leading to poor prognosis [3, 14]. On the other hand, the technological advances of SBRT over the past decade have provided a promising therapeutic approach for advanced NSCLC patients with brain metastases [31]. In a retrospective research including 46 cancer patients with oligo-metastatic NSCLC, SBRT treatment showed 1-, 2- and 3-year local control rates of 93%, 93% and 78%, respectively [32]. Another study reported that SBRT could reduce disease progression of end-stage 34 advanced patients with extracranial progression [33].
As for brain multi-metastases, SBRT is usually performed to relieve neurological symptoms and improve quality of life as a palliative therapy, and is often combined with WBRT to achieve better outcome. Although WBRT can help to relieve the tumor-mass effects and improve biological functions, its prognostic benefit is limited to 6 months. Besides, WBRT shows no curative effects on big metastases (
In the present study, we analyzed the therapeutic efficacy and safety of applying SBRT in advanced NSCLC patients with brain multi-metastases, including local control rate, prognosis and radiotherapy toxicity. 51 eligible patients with 193 brain metastases lesions were enrolled in this research, whose 1-year and 2-year OS rates were 82.4% and 45.1%, respectively. In survival analysis, we found older patient age and greater PTV volume were the independent prognostic factors for OS and PFS, consistent with previous reports [35], while no prognostic differences were identified between SBRT alone and combination with WBRT. The local disease control rates were also comparable in the two groups (77.3% vs. 79.3%).
Another controversy of applying radiotherapy in treating brain metastases is that imprecise irradiation could generate severe acute toxicities to the central nervous system [13, 16]. In our study, the radiotherapy encountered in the SBRT alone group were mild and limited to a satisfactory extent. The most common toxicities were grade 1–2 reactions including nausea, dizzy and slight myasthenia, while grade 3 reaction leukopenia occurred in only one patient. By contrast, severe toxicities were more frequent in the SBRT
Despite evidence supporting SBRT for advanced NSCLC patients with brain multi-metastases, our research need to be interpreted with caution and several limitations should be acknowledged. First, the main drawback of this study is that it’s a retrospective study in a single institution. Patients selection bias is inevitable in choosing specific radiotherapy modality. Additionally, our cohort only enrolled 51 patients, which is relatively small number to draw a solid conclusion. Prospective, randomized, clinical trials in large population and multiple institutions are needed to carry out to define the role of SBRT in the treatment of NSCLC patients with brain multi-metastases.
Conclusion
The current research suggested that SBRT alone could effectively relieve tumor burden and improve the prognosis and quality of life for NSCLC patients with brain multi-metastases, which should be validated in further large-scale prospective clinical trials. Regardless of the limitations, these findings can offer suggestions to physicians and patients to aid in making personalized and interactive therapy decisions.
Footnotes
Acknowledgments
The authors have no acknowledgments.
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
The authors report no funding.
