Abstract
Objective:
To investigate the feasibility of hippocampal-sparing whole-brain radiotherapy and reduction in neurocognitive function impairment after radiotherapy.
Methods:
Forty-three patients with brain metastases were selected. Whole-brain radiotherapy was performed in 22 patients, with 3-dimensional conformal radiotherapy in parallel opposed fields. Twenty-one patients had significant difference. Planning parameter values and neurocognitive function scores in 2 groups were statistically analyzed.
Results:
Homogeneity index in the 3-dimensional conformal radiotherapy group and tomotherapy group was 0.12 ± 0.02 and 0.36 ± 0.03, respectively, with a significant difference (P < .05). The homogeneity of target doses was better in the tomotherapy group than in the 3-dimensional conformal radiotherapy group. There was a statistical significance in Dmean and Dmax between the 2 groups (P < .05). The dose in hippocampal was less in the tomotherapy group than in the other group. Dmean and Dmax of the left hippocampus declined to 20.14% and 35.39% of prescription dose, respectively, and Dmean and Dmax of the right hippocampus declined to 19.92% and 35.14% of prescription dose, respectively. Neurocognitive function score between the 2 groups before treatment and 1 month after treatment had no significant difference (P > .05), while there was a significant difference in 3 and 6 months after treatment (P < .05). Neurocognitive function score was higher in the tomotherapy group than in the other group. No level III or above adverse reactions were observed.
Conclusions:
Hippocampal-sparing whole-brain radiotherapy in brain metastases treatment is feasible. Homogeneity index value is higher in the tomotherapy group than in the 3-dimensional conformal radiotherapy group. All V95% in the 2 groups meet the requirements. Hippocampal sparing could avoid neurocognitive function impairment to some extent.
Introduction
Brain metastasis (BM) is one of the most common brain tumors in adults, 1 whose incidence is 10 times of primary intracranial malignant tumors. 2 Over the past 4 decades, the incidence of BM is close to 1/10 000. 3,4 Smedby and other Swedish scholar 3 analyzed 15 517 patients with BM and then made a conclusion: From 1987 to 2006, the incidence of BM increased from 7/100 000 to 14/100 000 and doubled in 20 years. According to the 2010 census, about 21 000 to 43 000 patients in the United States are diagnosed with BM each year. 5 Although the exact incidence has not been determined, the number of patients with BM is increasing with the development of imaging techniques (such as magnetic resonance imaging [MRI]) and the clinical application of systematic therapies. 6 At the time of initial diagnosis of extracranial malignant tumors, 20% to 40% of patients had progressed to BM. 7 The main symptoms of BM include intracranial hypertension, changes in mental state, seizures, and so on, which seriously affect the quality of life of patients. 2 The prognosis of BM is very poor. For example, in lung cancer BM, only dexamethasone, mannitol, and other symptomatic supportive treatments are given, and the median overall survival (MOS) is 1 to 3 months. 8 The best treatment for BM is to be individualized (considering the general situation of patients, the pathological type of tumor, immunophenotype, primary site, etc), and combined with multidisciplinary treatment, the ultimate goal is to improve the patients’ survival prognosis and reduce or prevent life-threatening complications. 9 As one of the standard treatment models of BM, 10 whole-brain radiotherapy (WBRT) plays an important role in BM therapy, and its clinical application is still developing rapidly. 11 The local control rate of WBRT is 0% to 71%, and MOS is 4 to 6 months. 12 The neurocognitive function (NCF) disorder caused by WBRT has also attracted clinical attention and has become one of the hotspots in recent years. 13 The previous view believed that the NCF injury after radiotherapy was caused by intracranial lesions. With the deepening of related studies and the updating of radiotherapy equipment, the decrease in NCF after WBRT was associated with hippocampal (HP) injury after radiotherapy. 14,15 The probability of NCF decrease was 40% to 50% in patients with malignant brain tumors who survived after radiotherapy for more than 1 year. 16 At present, the impact of NCF on the quality of life of patients and its importance are recognized as secondary to the survival period of clinical trials. 17 However, hippocampal-sparing whole-brain radiotherapy (HS-WBRT) has a protective effect on HP, thus avoiding NCF obstruction. The development of helical tomotherapy (TOMO) makes HS-WBRT become possible; it can avoid the NCF injury in patients with BM after radiotherapy.
Whether it is BM itself or the decline in NCF after radiotherapy, they both have seriously affected the quality of life of patients, so to explore a way of performing WBRT while avoiding NCF reduction of new treatment measures has become the focus of recent years in the radiotherapy.
Materials and Methods
Patients
Forty-three patients with BM admitted from August 2015 to August 2016 in the First Affiliated Hospital of Zhengzhou University were selected as participants, who were diagnosed with malignant tumor with cytology and pathology, including 22 cases of non-small cell lung cancer (NSCLC), 15 cases of small-cell lung cancer (SCLC), 3 cases of breast cancer, 2 cases of choriocarcinoma, and 1 case of esophageal cancer. Brain metastases were confirmed by MRI, the average number of metastases was 2, 1 –6 the largest diameter of metastasis was 3.2 cm, the mean diameter was 1.56 ± 0.79 cm, and all metastases were not in HP. There were 24 males and 19 females, aged 21 to 69 years old, and the median age was 47.5 years. Among these, 3 patients had clinical symptoms of intracranial hypertension, specifically headache and nausea, and showed evidence of tolerance after taking dexamethasone, mannitol, or glycerol fructose. All patients’ Karnofsky Performance Status (KPS) score was ≥70; their liver and kidney function and blood routine were basically normal, and there were no evidence of hypertension, cardiovascular and cerebrovascular disease, and other basic diseases and no history of head surgery and radiotherapy. The Mini-Mental State Examination (MMSE) score was normal for all patients before radiotherapy. The study not only received informed consent from all patients but also received the support of the ethics committee of the First Affiliated Hospital of Zhengzhou University.
Methods
Simulation Positioning
All patients were simulated with computed tomography (CT), and CT scan thickness was 3 mm. In the same position, the enhanced sagittal, coronal, and transverse MRI was performed, and the slice thickness was 3 mm. The simulated CT images were fused with the enhanced MRI T1-weighted images and would sketch by the same radiotherapy physician in the Eclipse System (Varian Medical Systems, Palo Alto, CA) CT-MRI T1-weighted fusion image on the target.
Target Outline and Plan Design
The computer automatically sketched the clinical target volume (CTV) of the whole brain, and CTV expanded 3 mm to form the planning target volume (PTV). The outline of the HP referred to the Radiation Therapy Oncology Group (RTOG) 0933 report 14 ; the HP expanded 5 mm to form a planning risk volume (PRV), called hippocampal planning risk volume (HP-PRV). The outline of organs at risk (OARs) was described in International Commission on Radiation Units (ICRU) 83 report, 15 including crystals, eyeballs, optic nerve, optic chiasm, pituitary, brain stem, and temporal lobe. All radiotherapy programs were developed by the same senior physician, and the plan was reviewed by the same radiologist at the target area.
The prescription dose was 36 Gy/2 Gy/18F, 5 times for each week. OAR limit standard: the maximum HP less than 17Gy, and the mean HP less than 10Gy. Of the 43 patients, WBRT was performed in 22 and these patients were planned with 3-dimensional conformal radiotherapy (3D-CRT), referred to as 3D-CRT or WBRT group; HS-WBRT was performed in another 21 patients, referred to as TOMO or HS-WBRT group. There was no significant difference in gender, age, and pathological type between the 2 groups (Table 1).
Basic Data of Patients.
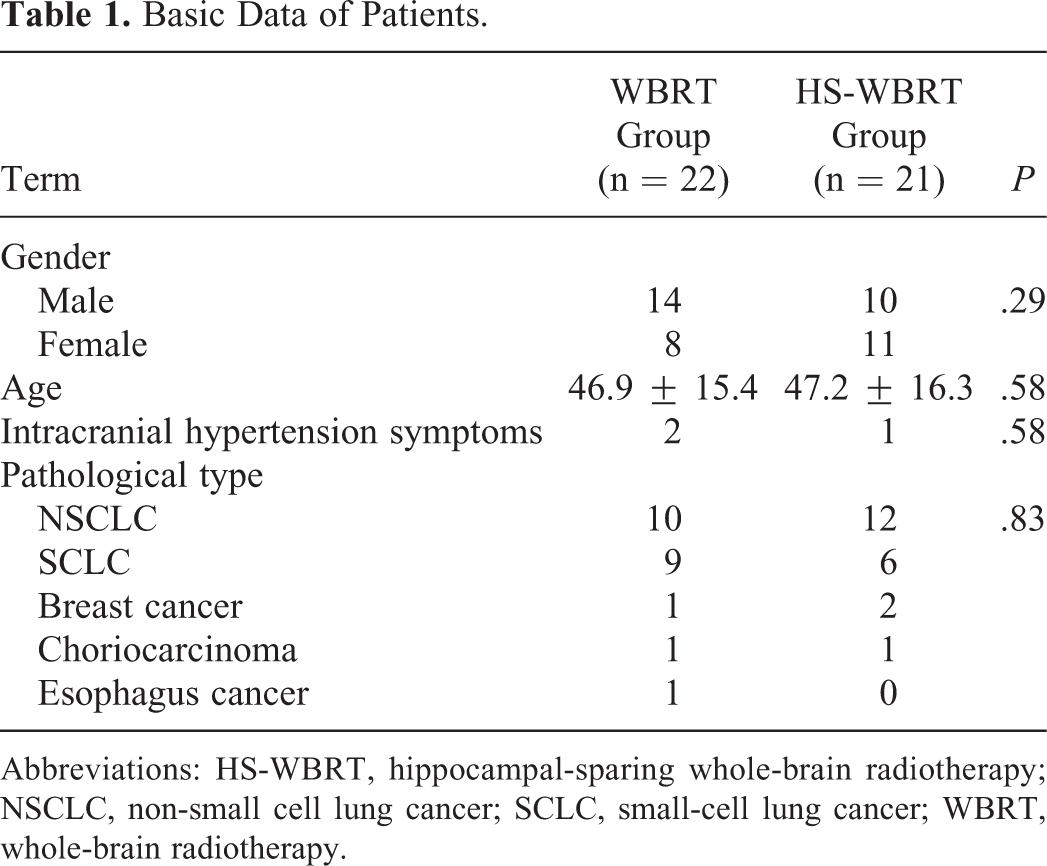
Abbreviations: HS-WBRT, hippocampal-sparing whole-brain radiotherapy; NSCLC, non-small cell lung cancer; SCLC, small-cell lung cancer; WBRT, whole-brain radiotherapy.
Data Collection
Dosimetric data statistics: Obtained the required data in the dose–volume histogram.
Neurocognitive function score: Seventy-two hours before radiotherapy and 1 month, 3 months, and 6 months after radiotherapy, MMSE was administered for NCF evaluation.
Statistical Approach
SPSS 21.0 software was used to analyze the data and make a statistical treatment: the dose difference between the 2 groups, NCF score comparison, t test, or correction of the t test. When the data didn’t obey the normal distribution, the Wilcoxon rank sum test can be used. The NCF scores were compared between the groups before and after treatment: paired t test or rank sum test. Test standard: There is a significant difference at P < .05.
Results
Dosimetric Comparison of PTV Parameters
There was no significant difference in PTV between the 2 groups (P > .05). It was noted that TOMO PTV = (whole-brain CTV + externally expanded 3 mm) − HP-PRV, whereas 3D-CRT PTV = (whole-brain CTV + externally expanded 3 mm). Homogeneity index (HI): 3D-CRT was 0.12 ± 0.02 and HI: TOMO was 0.36 ± 0.03. The difference between them was significant (P < .05). The TOMO was superior to 3D-CRT in the dose uniformity of the target. The Dmean of 3D-CRT was 38.00 ± 0.56 Gy, and the Dmean of TOMO was 37.89 ± 0.74 Gy (P > .05); the Dmax were 40.77 ± 0.80 Gy and 40.55 ± 0.92 Gy (P = .42), respectively. There was no significant difference (P > .05). There was no difference between Dmean and Dmax. HI = (D2% − D98%)/D50% (Table 2).
Dosimetric Comparison of PTV Parameters in 2 Groups of Patients.

Abbreviations: D2%, 2% of the target volume received by the dose; HI, homogeneity index; PTV, planning target volume; TOMO, tomotherapy; 3D-CRT, 3-dimensional conformal radiotherapy.
Comparisons of the 2 Groups of Parameters of Planned HP
There was no significant difference in the volume of HP between the 2 groups (P > .05). The Dmean of 3D-CRT and TOMO of the left HP was 37.43 ± 0.59 Gy, 7.25 ± 0.57 Gy, respectively, whereas the Dmean of the right HP was 37.44 ± 0.58 Gy and 7.17 ± 0.55 Gy, respectively. And the Dmax of the left HP was 37.72 ± 0.61 Gy and 12.74 ± 1.81 Gy, respectively, whereas the Dmax of the right HP was 37.72 ± 0.61 Gy and 12.65 ± 1.49 Gy, respectively. There was a significant difference between the 2 groups in Dmean and Dmax of HP (P < .05). The HP dose in the TOMO program was significantly lower than that in the 3D-CRT, and the Dmean and Dmax of the left HP dropped to the prescription dose of 20.14% and 35.39%, respectively, whereas right HP dropped to the prescription dose of 19.92% and 35.14% (Table 3).
Comparisons of Parameters of 2 Groups of Planned HP.

Abbreviations: D2%, 2% of the target volume received by the dose; HP, hippocampal; TOMO, tomotherapy; 3D-CRT, 3-dimensional conformal radiotherapy.
Comparison of the 2 Groups of OAR
There was a significant difference in Dmax of all OARs (P < .05), and the dose in the 3D-CRT group was lower than that in the TOMO group. In contrast to Dmean, there was no significant difference in the comparison of pituitary, brain stem, brain stem PRV, optic chiasm, and frontal lobe (P > .05), while there were significant differences in the comparison of eyes, crystal, PRV crystal, optic nerve, cochlea, and spinal cord (P < .05). The Dmean of the above organs in the 3D-CRT group was lower than that in the TOMO group (Table 4).
Dmax and Dmean Comparison of 2 Groups of OAR.a

Abbreviations: PRV, planning risk volume; OAR, organs at risk; TOMO, tomotherapy; 3D-CRT, 3-dimensional conformal radiotherapy.
a “-” indicates that no data have been calculated in the plan.
Comparison of the 2 Groups of NCF Score
The comparison of 2 groups of NCF score indicated that there was no significant difference 1 month before (P = .64) and after (P = .21) treatment, while there was a significant difference 3 months (P = .001) and 6 months (P = .00) after treatment (P < .05). At 3 and 6 months after radiotherapy, the NCF score of the TOMO group was better than that of the 3D-CRT group.
Within the comparison of 2 groups of NCF score, there was no significant difference before and after treatment in the TOMO group (P > .05), and there was either no significant difference 1 month before or after treatment in the 3D-CRT group (P > .05), while there were significant differences 3 and 6 months after treatment compared with before treatment (P < .05; Table 5).
Comparison of 2 Groups of NCF Score.

Abbreviations: NCF, neurocognitive function; TOMO, tomotherapy; 3D-CRT, 3-dimensional conformal radiotherapy.
All patients with 3-month and 6-month brain treatment were assessed to be better or more stable, and some patients showed progress in the external brain lesions between 3 and 6 months (2 cases of SCLC and 1 case of NSCLC), but the symptoms were not significant and the patients’ NCF score was not affected.
Adverse Reactions
Adverse reactions are mainly manifested as cerebral edema and cranial hypertension, including headache, dizziness, nausea, vomiting, fatigue, and so on; blood system toxicity is mainly leukopenia, in which level I reacted 81% (35/43), level II reacted 14% (6/43), and level III reacted 5% (2/43). After the administration of mannitol or glycerol fructose and dexamethasone to reduce intracranial pressure, protect stomach, and stop vomiting and after leucogenic supportive treatment, all patients were successfully completed WBRT and, after the end of radiotherapy, were given the treatment of primary tumor according to the specific circumstances of patients.
Discussion
With the development of medical science and technology, as well as people’s higher requirements for quality of life after radiotherapy, the prevention and treatment of NCF caused by radiotherapy have been paid more and more attention. The previous view that NCF injury after radiotherapy is caused by intracranial lesions, along with the deepening of related research and the update of radiotherapy equipment, it is thought that the decrease in NCF after radiotherapy is related to the damage of HP. Tomotherapy therapy (HS-WBRT) for the treatment of BM is more widely used.
Metastasis of HP and HP-PRV was rare. Sun et al 18 analyzed 1678 metastases in 314 patients and found that 4.1% of the patients had metastases at HP and 11.1% of the patients’ metastases were located at HP-PRV. Wu et al 19 studied 632 patients with 6064 metastases, and the results showed that 4.1% of patients’ BM was located at HP and 5.5% was located at HP-PRV. Similar results were obtained from the study by Wan et al. 20 After WBRT, the rate of recurrence or new onset of HP and HP-PRV metastasis is very low. RTOG 0933 21 studied and analyzed 1133 BMs in 371 patients after HS-WBRT and observed that no metastasis was found in HP, 3% of the patients had HP-PRV metastasis, and 8.6% of the metastases were located at HP-PRV. The study also found that patients mostly experienced levels I and II side effects. One patient had a level III toxicity reaction associated with radiotherapy, no level IV and V toxicity reaction was observed, and no decrease in quality of life score was observed in the study. No acute and chronic toxicity which is higher than level II was noted in the study of Oehlke et al. 22 The result was consistent with this research.
In the randomized trials of Sun et al, 23 MMSE score decreased sharply at 3 months after prophylactic cranial irradiation in patients with NSCLC. RTOG 0933 24 showed that 4 months later the WBRT group in the Hopkins Verbal Learning Test–Revised Delayed Recall fell 30% lower than the baseline, whereas the HS-WBRT group only decreased by 7%. In this research, the MMSE score of the 3D-CRT group was lower than that in the TOMO group at 3 and 6 months after radiotherapy. The MMSE score was lower than the baseline in the 3D-CRT group at 3 and 6 months after radiotherapy, and there was a significant difference (P < .05), indicating that the protection of HP can reduce the decline in NCF.
In the study of Giaj Levra et al, 25 the Dmax and Dmean of HP were 7.7 ± 0.3 Gy and 10.5 ± 0.5 Gy, respectively, and the whole-brain HI was 0.42 ± 0.12. In the experiment of Kim et al, 26 the whole-brain HI was 0.52 ± 0.16, and the Dmean of HP was 13.65 Gy. In the study of Prokic et al, 27 the HP of Dmean was 7.55 ± 0.62 Gy and HI was 0.54 ± 0.04. This study was not very different from the HI value of other studies, but the Dmean and Dmax values of HP were different. The reason may be that the prescription dose size of each study is inconsistent, and it also may be the differences between outline of the target, radiotherapy plan designer and personal technique, equipment, and so on.
Combined with previous studies and the results of this study, we found that HS-WBRT is feasible in the treatment of BM. The HI value of TOMO was more satisfying than 3D-CRT, both of their V95% can meet the requirements, so as to protect HP to some extent and avoid NCF damage.
There was a significant difference in Dmax of all OARs (P < .05), and there were significant differences in comparison of eyes, crystal, optic nerve, cochlea, spinal cord of Dmean (P < .05), and the OARs in Dmean of the 3D-CRT group were lower than those in the TOMO group. The result was proved indirectly by the study of Moon et al 28 : In the treatment of breast irradiation, TOMO and 3D-CRT were both able to obtain a valid PTV coverage. But in the TOMO plan, non-PTV breast target can get more effective protection at the cost of higher doses of the heart and lung X-ray exposure. The dose uniformity of TOMO target was better, but the cost of Dmean and Dmax in eyes, crystal, optic nerve, cochlea, and spinal cord was higher than those in the 3D-CRT group.
There are certain limitations to this study. First, the number of cases selected was limited to a single-center study. All patients were from the radiotherapy department of the First Affiliated Hospital of Zhengzhou University, and there may be bias in the selection; second, in the design of the target area and the plan, although there were relevant international standards for reference, the experience and subjectivity of the doctor’s target sketched were relatively strong, which may be one of the reasons for the existed differences between this research and others, multiple radiologists can be targeted, and multiple radiologists can evaluate the radiotherapy plan simultaneously; third, while scoring NCF, although we tried to avoid interference factors, it couldn’t exclude the possibilities due to the patients’ personal reasons such as not cooperating deliberately, resulting in inaccurate score, so taking multiple measures to select the average value and reduce the error is feasible; fourth, the survival rate of the selected cases was not observed because of the pathological differences; finally, the effects of patient’s gender, age, and chemotherapy were not taken into account. The results of this research still need to be validated by a larger sample of prospective studies.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
