Abstract
Combining stem cells with biomaterial scaffolds serves as a promising strategy for engineering tissues for both in vitro and in vivo applications. This updated review details commonly used biomaterial scaffolds for engineering tissues from stem cells. We first define the different types of stem cells and their relevant properties and commonly used scaffold formulations. Next, we discuss natural and synthetic scaffold materials typically used when engineering tissues, along with their associated advantages and drawbacks and gives examples of target applications. New approaches to engineering tissues, such as 3D bioprinting, are described as they provide exciting opportunities for future work along with current challenges that must be addressed. Thus, this review provides an overview of the available biomaterials for directing stem cell differentiation as a means of producing replacements for diseased or damaged tissues.
INTRODUCTION
Since the initial publication of our STEMBOOK chapter in 2008 [1], the field of stem biology has advanced rapidly as such regenerative medicine strategies move into clinical trials for a variety of health disorders [2]. Stem cells possess two novel properties – the ability to produce additional stem cells and the capacity to become multiple cell types [3]. Table 1 lists the major types of stem cells and their associated properties. These types include adult, embryonic, fetal, induced pluripotent, and mesenchymal stem cells. In particular, the discovery of induced pluripotent stem cells (iPSCs) in 2006 catalyzed the fields of tissue engineering and regenerative medicine as they represented a major advance in stem cell technology [4]. These iPSC lines can be generated from patient-derived cells, making it possible to obtain personalized stem cell lines.
Different types of stem cells and their associated properties
Tissue engineering combines biomaterial scaffolds with these different types of cells to replace damaged organs. The differentiation potential of stem cells makes them a valuable tool for such applications. In 1987, a group of experts defined the word biomaterial as “a non-viable material used in a medical device, intended to interact with biological systems” [5]. This definition reflected the state of the field at the time, which focused on the development of materials and coatings to prevent the rejection of implantable medical devices. Since 1987, this field of study has expanded to include the design and development of implantable scaffolds with defined properties produced from a wide variety of biomaterials. Such biomaterial scaffolds promote the viability and differentiation of stem cells seeded inside depending on the intrinsic properties of the material as well as the incorporation of specific chemical and physical cues into the material. A wide range of natural and synthetic biomaterials have been evaluated as substrates for controlling stem cell behavior. First, the different types of scaffold formulations are defined and critically analyzed. Next, specific examples will be discussed for the three major categories of biomaterials: natural, synthetic, and ceramic-based. The advantages and drawbacks of each material type will be detailed along with relevant examples of how such scaffolds can influence stem cell behavior. This review focuses on three-dimensional (3D) scaffolds as they mimic the environment found in the human body, making them highly relevant when engineering tissues, including the use of 3D printing tissues using biomaterial-based inks. This review summarizes the current knowledge of using biomaterials in combination with stem cells for tissue engineering applications, including cell delivery to repair damaged regions of the body.
TYPES OF SCAFFOLD FORMULATIONS
Both natural and synthetic biomaterials can serve as the starting point for generating bioactive scaffolds for controlling stem cell differentiation into the desired tissue type. These scaffolds can take several different forms, which in turn have unique features. This section will detail the properties of the following types of scaffolds: hydrogels, micro and nanofibers, and micro and nanospheres (Fig. 1). Each type of scaffold formulation possesses certain advantages and disadvantages, which will be discussed. These scaffolds can also be combined to yield novel hybrid materials as often certain formulations enable better cell survival with others providing a more desirable time course for drug delivery.
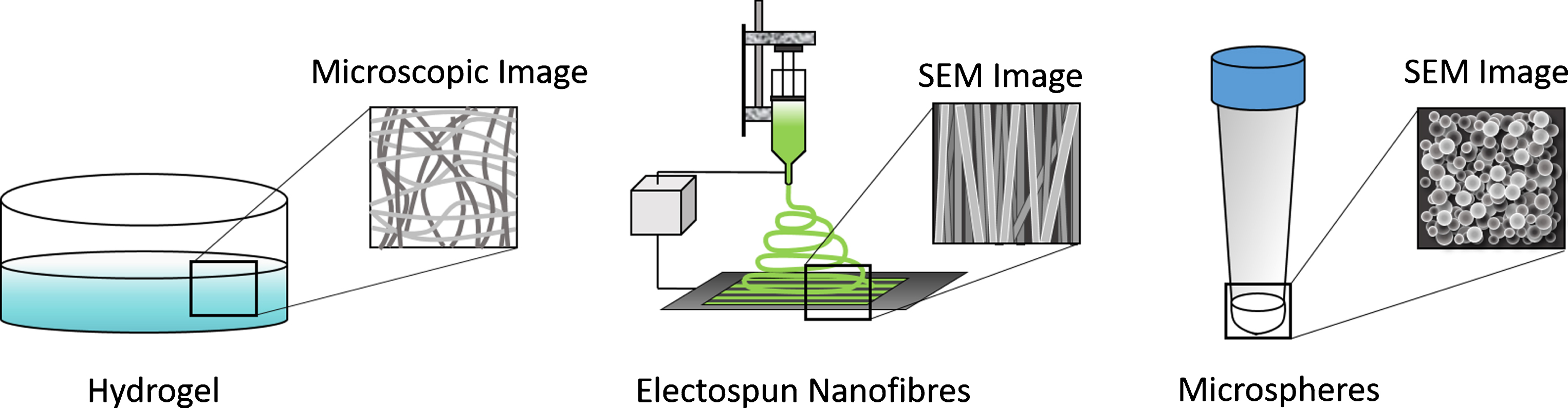
Commonly used scaffold formulations for engineering tissues from stem cells include hydrogels, electrospun scaffolds, and nano/microspheres.
Hydrogels
Hydrogels consist of cross-linked hydrophilic polymer networks that swell upon exposure to water – hence the name [6]. The extracellular matrix (ECM) serves as a complex naturally occurring hydrogel that provides the microenvironment that supports the stem cells found resident in tissues, making this scaffold formulation highly desirable for tissue engineering applications using stem cells. Many different factors play roles in determining the properties of these hydrogels, including the properties of the polymer, degree of cross-linking, chemical functionalization, and other parameters. Hydrogels are often modified to incorporate drug delivery systems, which can enhance the survival and differentiation of the stem cells seeded inside [7–9]. They also have been used successfully to deliver stem cells in vivo and they can often be formulated to be injectable, which is highly desirable for clinical applications [10]. Recent reviews have summarized the wide body of work detailing how hydrogels have been used to successfully deliver stem cells in vivo for a variety of applications, including vascularization, tendon repair, healing kidney damage, and for neural regeneration [11–14] Ensuring long-term stability can be challenging as well as matching the desired release rate for molecules embedded in the hydrogel matches the desired degradation rate. However, both natural and synthetic hydrogels remain popular tools for engineering tissues from stem cells.
Electrospun scaffolds
Electrospun scaffolds serve as another popular option as scaffolds for promoting the culture and differentiation of stem cells into tissues as detailed in a recent book [15]. The process of electrospinning requires the application of a high voltage field to a polymer solution, which draws out thin fibers that are collected in a specialized fashion. Electrospinning set-ups are often inexpensive to implement and thus it has become an increasingly popular scaffold fabrication technique. Different parameters including the polymer solution being spun, the collection distance and method, the applied voltage field, and the ambient conditions all influence the size, topography, and consistency of the fibrous scaffolds produced when electrospinning [16]. The properties of these fibrous scaffolds can influence how stem cells behave, including their topography and their ability to generate controlled release of biomolecules [17]. Electrospinning can generate fibrous scaffolds ranging in size from nanometers to micrometers and the size and orientation of these fibers play an important role in controlling the behavior of the cells seeded upon such scaffolds [18]. Such topography can direct stem cells to form functional tissues, including cardiac, bone, and nerve [19–21]. Bioactive factors, such as small molecules, polysaccharides, and growth factors, can be added to the solution being spun to generate controlled release for directing stem cell behavior [22, 23]. Topography, fiber size, and fabrication parameters can all affect the release rate of such molecules and these factors can be tuned accordingly to generate a desirable release course necessary for achieving the desired effect on stem cell behavior. Some limitations of electrospinning include the reproducibility of these scaffolds as fiber size can vary significantly even within a scaffold and scale-up [20]. In conclusion, electrospun scaffolds can serve as multifunctional scaffolds for generating tissues from a variety of stem cells.
Nano and microparticles
Particles ranging in size from the nanoscale to microscale can serve as effective tools for drug delivery for a number of reasons [24, 25]. The use of such particle-based drug delivery system has several advantages. The ability to tune the encapsulation efficiency of the drug contained in the particles and the number of particles used for a particular application enables a high degree of control over the concentration and time course of the target molecule being delivered. Their size also means they can be delivered in vivo through injections and certain formulations of nanoparticles can even cross the blood-brain barrier to provide controlled drug release [26]. Such particles are often combined with hydrogels [27] and electrospun scaffolds [28] to provide additional bioactivity and deliver additional factors when engineering complex tissues. Accordingly, drug-releasing particles on both the nano and micro scale can be used to control stem cell differentiation – both alone and in combination with other scaffold formulations [29]. Different combinations of drug-releasing particles can be incorporated into such systems to yield a multifunctional construct for promoting stem cell differentiation into specific types of tissues. Such particles can also be directly incorporated into stem cell aggregates to ensure more homogenous differentiation as well [30]. While these applications highlight the desirable properties of drug-releasing particles, they also have certain issues associated with their use. These issues include determining proper dosage to ensure the desired effect on stem cell behavior. Also, controlling the size distribution and morphology of the particles produced depends on the method of fabrication and large variations can be observed, which is similar to limitations associated with electrospun scaffolds. However, such particle distributions can be adjusted using sieving.
NATURAL BIOMATERIALS
The proteins and polysaccharides found in the ECM provide an obvious starting point when developing scaffolds derived from natural biomaterials. These bioactive molecules perform many roles in vivo, and often contain sites for cellular adhesion while displaying inherent biocompatibility. These materials can exhibit variability depending on their source and may induce an immune response if not properly purified. These scaffolds often exhibit a limited range of mechanical properties and often their scaffold formulations need to be optimized for stem cell culture. The following sections will first focus specifically on scaffolds made from purified proteins or polysaccharides and then highlight the emerging area of using decellularized ECM for directing the differentiation of stem cells.
Protein-based biomaterials
Proteins play an important role in providing structure to tissues, making them a logical choice for tissue engineering applications requiring stem cell differentiation and transplantation. This section highlights some of the most commonly used protein-based biomaterials, including collagen, fibrin, silk, fibronectin, and vitronectin, and their application to engineering tissues from stem cells. Proteins can be isolated from human or animal sources and care must be taken to ensure that animal-derived proteins do not trigger an immune response if clinical translation remains the end goal. For example, collagen, a commonly used biomaterial, can be isolated from a variety of tissues, including skin, tendon, or bone. It can also be processed into other forms, including gelatin and gelatin methacrylate. The protein fibrin plays an essential role in the blood clotting process. It can be isolated from blood, and it is commonly used as a surgical sealant in clinical studies and as a biomaterial scaffold. Insects and worms secrete silk, which is another protein commonly used for generating tissue-engineered scaffolds. Scaffolds made of silk or silk fibroin have slow degradation rates and desirable mechanical properties, providing an alternative to collagen and fibrin. Scaffolds made from silk fibers can be fabricated into a variety of structures, such as mats, sponges, meshes and membranes, expanding the possible applications. We will also discuss the use of other proteins found in the ECM as scaffolds materials for directing stem cell differentiation, including fibronectin, vitronectin, and laminin. The following sections will highlight specific examples of these scaffolds being used in combination with stem cells for a range of different tissue engineering applications.
Collagen
Collagen comprises 25% of the total protein found in the human body, making it a logical choice for tissue engineering applications [31]. Collagen contains sites for cell adhesion and its natural materials properties are similar to that of soft tissue [32]. It comes in several types with Type 1 being the most commonly used to form hydrogels as it is the most abundant in tissues. Hydrogels made from collagen can be modified to enhance their chemical and mechanical properties, which can be tailored to the desired tissue to be engineered [33]. 3D collagen hydrogels can culture a wide variety of stem cells for different tissue engineering applications, including neural tissues, blood vessels, cardiac tissue, liver and cartilage [34–44]. Collagen can also be denatured to produce gelatin, which can be modified to produce gelatin methacryloyl (GelMa) [45]. GelMA hydrogels can be produced by photo-crosslinking and its mechanical and chemical properties are easily tunable depending on the application. GelMa also exhibits decreased antigenicity as it is denatured, enhancing its relevance for clinical applications [46]. Engineering tissues by combining stem cells with GelMa hydrogels has been gaining in popularity recently, especially with 3D bioprinting approaches using GelMa as a novel bioink [47–49]. An interesting paper from the Khademhosseini lab showed how 3D printing different types of GelMa-derived bioinks can serve as a valuable tool for determining an optimal formulation for directing human mesenchymal stem cells (MSCs) to form bone tissue [50]. The printing set-up and experimental design can be seen in Fig. 2.

A) A robotic microarray spotter was used to rapidly print droplets consisting of hMSCs, gelatin methacrylate (GE)-based prepolymer solution and various ECM proteins on TMSPMA functionalized glass slide. The printing step was followed by a 15|sec UV light exposure to form the miniaturized cell-laden constructs. Following printing, cell-laden gel microarrays were placed inside sealed chambers (Illustration made by Jeffrey Aarons). B) Various combinations of ECM proteins and media formulations were used to conduct the microarrays experiments. The concentration of LN and FN was selected to be 40|μg/ml while OCN was printed at two concentrations of 20|μg/ml and 40|μg/ml. C) Fluorescence images of the encapsulated proteins within the hydrogel constructs after 24 hours in solution. D) hMSCs viability within 48 combinatorial 3D microenvironments in normal (control) media after 7 days of culture along with color-diagram displaying the quantified cell viability (n = 3– 9). This figure was reproduced under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. Taken from [50].
The properties of collagen can be further manipulated on the micro and nanoscale using techniques such as electrospinning and fabrication of microspheres [51]. Electrospinning poses challenges in terms of maintaining the bioactivity of the protein, but it can be achieved under appropriate conditions [52, 53]. Collagen is often added to synthetic polymer scaffolds to enhance their bioactivity and enable stem cells to bind to these scaffolds [54, 55]. Both collagen and gelatin microspheres can be seeded with stem cells as a novel approach to tissue engineering. Collagen microspheres are commonly used to deliver a variety of stem cells ranging from mesenchymal stem cells to oligodendrocyte progenitors [56–58]. A recent study also highlighted how GelMa microspheres containing bone marrow-derived stem cells could be injected in vivo for bone tissue engineering application [59]. Thus, collagen and the related polymer – GelMa - serve as powerful tools for engineering tissues from stem cells.
Fibrin
Similar to collagen, fibrin exhibits natural biocompatibility and contains sites for cell adhesion [60]. Accordingly, significant work has been performed evaluating fibrin scaffolds for directing the differentiation of different types of stem cells. As mentioned earlier, fibrinogen circulates in the bloodstream and becomes polymerized into fibrin clots during the wound healing process [61]. Surgeons use fibrin as surgical glue, making it a desirable material for clinical translation [62]. It can be used to generate hydrogels, electrospun scaffolds, and microspheres that serve as cell carriers for a wide variety of tissue engineering applications [63–65]. Additionally, its mechanical properties can be manipulated to ensure a proper environment for stem cell differentiation as seen in Fig. 3 [66]. Many groups have investigated a variety of fibrin hydrogel formulations for engineering neural tissue from stem cells, including both embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), in vitro and for delivering stem cells into the damaged nervous system in vivo. Other groups have examined the behavior of MSCs seeded inside of fibrin clots and treated with growth factors for use in engineering bone. In terms of other applications, fibrin scaffolds seeded with stem cells have also been used for engineering cartilage [78, 79] and for promoting vasculature formation [80–82]. This body of work suggests that a variety of stem cell lines can be cultured inside of fibrin scaffolds for many different tissue engineering applications. While fibrin can be electrospun to generate scaffolds for cell culture, such scaffolds have not been evaluated for use in combination with stem cells for tissue engineering applications [83, 84], providing an interesting avenue for future work. Similarly, the use of fibrin microspheres seeded with stem cells also provides an opportunity for future investigation. Finally, hybrid bioinks containing fibrin have shown promise for use in 3D printing functional tissues from stem cells [85].

Genipin crosslinking influences the morphological and mechanical properties of fibrin scaffolds. The effect of genipin concentration on fiber diameter (biological n = 3, technical n = 10, shown on the top left). The effect of genipin concentration on pore size of fibrin scaffolds (biological n = 3, technical n = 30, shown on the top right). The effect of genipin on elastic modulus (shown on the bottom). The elastic modulus for spinal cord tissue is taken from 37. Each group is represented by its mean average with error bars indicating the S.E.M. *Indicates p < 0.05 compared to control (0 mM). This figure was reproduced under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. Taken from [61].
Silk
The secondary structure of silk can vary based on the source, but in general it tends to contain a number of β sheets, giving it crystalline properties and relatively slow degradation rate in comparison to mammalian ECM proteins, such as fibrin and collagen [86]. Silk scaffolds can also be modified to enhance their chemical properties and enable controlled delivery of molecules [87, 88]. The properties of silk make it attractive for engineering bone and ligament tissue [89]. Extensive research has been done using 3D silk scaffolds in conjunction with mesenchymal stem cells for these applications by David Kaplan’s lab as well as others. [90–98]. Often these silk scaffolds are electrospun to improve their ability to form functional tissues from stem cells [99, 100].
Combining silk scaffolds with stem cells can produce replacements for damaged cartilage. Meinel and colleagues showed that silk scaffolds promoted more extensive chondrogenesis compared to collagen scaffolds when seeded with human mesenchymal stem cells [96]. More recent efforts have used the combination of stem cells with silk scaffolds to engineer skin as reviewed recently [101]. Silk offers superior biocompatibility in comparison to other biomaterials based on in vivo testing. An interesting study demonstrated that silk microspheres could be combined with a novel hydrogel as a novel injectable way to deliver cardiac-derived stem cells [102]. This study opens intriguing areas for future work. Silk has also recently been evaluated as a potential bioink [103], making the 3D printing stem cell-derived tissues possible.
Laminin
Laminin, a high molecular weight protein ranging in size from ∼400 kDa-900 kDa, contains numerous sites for cell adhesion as it is found in the ECM [104]. It performs critical roles in several stem cell niches, including neural, cardiac, and kidney [105–107]. While laminin is not often used in 3D hydrogel form, it still can play a valuable role in controlling stem cell behavior. For example, laminin-coated surfaces support the maintenance and differentiation of stem cells [108]. Often times other scaffold materials are functionalized with laminin-derived peptides to enhance their ability to support stem cell adhesion and differentiation [109]. Similarly, electrospun scaffolds can be coated in different laminin fragments to enhance stem cell binding and differentiation [110, 111]. Further application of self-assembling laminin-derived peptides will be discussed in the section on synthetic biomaterials.
Fibronectin
Another large glycoprotein found in the ECM is fibronectin, which contains sites for both cells and proteins to bind [112]. Fibronectin enhances the differentiation of neural stem cells into mature neural cells and it can serve as a scaffold for delivering stem cells into the damaged nervous system [113, 114]. It also plays an important role in chondrogenesis [115] and the loss of fibronectin correlates with decreased muscle regeneration capacity [116]. Another common strategy is to conjugate fibronectin or peptide motifs derived from fibronectin to functionalize scaffolds to enable them to bind stem cells for tissue engineering [117–119]. This strategy is often used to functionalize electrospun scaffolds produced from synthetic materials to enhance their bioadhesive properties, which enables them to support cell culture [120, 121]. Thus, fibronectin serves as an important tool for engineering a variety of tissues from stem cells and it could serve as a valuable additive when developing novel bioinks for printing stem cells.
Vitronectin
Another glycoprotein vitronectin can be found in the serum and bone in addition to the ECM [122]. It often plays a role in tissue repair, including promoting angiogenesis after injury. Similar to laminin and fibronectin, vitronectin contains sites for integrins expressed by cells to bind, as well as sites for growth factors and other proteins to adhere [123]. Undifferentiated human pluripotent stem cells are often cultured on vitronectin surfaces as it provides a defined substrate in comparison with the traditionally used Matrigel (a mixture of proteins secreted by a mouse tumor cell line), reducing the possibility of immune response if these cells are transplanted into human subjects [123]. It also plays a role in promoting the differentiation of mesenchymal stem cells into osteoblasts, indicating its potential for stem cell applications [124]. While it tends not to be used as the sole scaffold material, other synthetic scaffolds have been decorated with vitronectin and its associated binding domains to produce engineered tissues from stem cells, including bone and neural tissue [125, 126]. Similar to fibronectin, the addition of vitronectin enhances the ability of different types of scaffolds to bind and influence stem cell behavior.
Polysaccharide-based biomaterials
Polysaccharides consist of sugar monomers, and these molecules perform important roles in maintaining the structure and function of the ECM. The structure and monomer composition contribute the properties of the specific polysaccharides. These polymers tend to be branched and they can be sourced from plants or animals. The source and method of isolation for a polysaccharide determines its level of immunogenicity and thus must be carefully chosen [127]. Polysaccharide-based scaffolds can often be formulated to gel rapidly, allowing for injection into the injury site – a desirable feature when engineering tissue and when delivering stem cells for clinical applications. Accordingly, these materials have been investigated for use as a potential scaffold material for stem cell transplantation. This section will review some of the most commonly used polysaccharide-based scaffolds, including agarose, alginate, hyaluronan, and chitosan, that have been used for the culture and differentiation of stem cells.
Agarose
Agarose, usually isolated from red algae and seaweed, consists of a galactose-based backbone and it possesses several desirable properties as a biomaterial for tissue engineering applications including being able to undergo reversible gelation in response to temperature [128]. The stiffness of agarose can be altered, allowing for tuning of the mechanical properties of the scaffold to specific tissue engineering applications [129]. Agarose hydrogels have been combined with stem cells for generating a variety of applications, including cartilage, heart, and nerve [130–134]. Bone and cartilage are commonly engineered by combining agarose with different types of stem cells. Stem cells can also be encapsulated into agarose microwells to form structures known as lockyballs where the interior of the structure consists of an aggregate of stem cells that is surrounded by a synthetic coating containing binding sites for other lockyballs. These lockyball structures can then be combined for tissue engineering applications as seen in Fig. 4 [135]. Finally, agarose can serve as a printable bioink with tunable properties for generating tissues from stem cells. Recent work illustrated how MSCs could be printed using an agarose-based bioink [136]. While some studies have explored electrospinning agarose fibers, these materials have not been evaluated in combination with stem cells [137]. In summary, agarose scaffolds and their different formulations provide a versatile platform for tissue engineering.

Biofabrication of human adipose-derived stem cell (ASCs) spheroids. A) ASCs spheroid fabricated in a confined space (resection) of micro-molded non-adhesive hydrogel has more regular shape and size than B) ASCs spheroid fabricated by the hanging drop method. Phase contrast. Bar size: 100 micrometers. C) Graph showing major (blue bar) and minor (red bar) diameters of spheroids generated in hanging drop and in resections of micro-molded non-adhesive hydrogel. Note that standard deviation in hanging drops is higher. A total of 45 spheroids were measured randomly. Graph represents the mean±standard error. D) Graph showing shape elongation coefficient (major/minor diameter of each spheroid) distribution of spheroids biofabricated by the two techniques representative from 1 micro-molded non-adhesive hydrogel and 1 petri-dish. (blue: spheroids fabricated in resections of micro-molded hydrogel; red: spheroids fabricated in hanging drops). This figure was reproduced under a Attribution 4.0 International (CC BY 4.0). Figure taken from [135].
Alginate
The cell walls of brown algae contain the polysaccharide alginate, a popular biomaterial that is often used as a bioink for 3D printing tissues [138]. These properties also have led to its application for microencapsulation of stem cells as alginate can be ionically cross-linked, which can be used to expand and differentiate these stem cells into a variety of tissue types [139–142]. Similar to agarose, many studies have combined both adipose-derived adult stem cells (ASCs) as well as bone marrow-derived MSCs with alginate hydrogels to generate replacement cartilage [132, 143– 145]. Alginate has also been used for neural tissue engineering applications with a variety of stem cell types [146, 147]. Tunable alginate scaffolds can be produced by incorporating enzyme releasing microspheres to control scaffold degradation and these scaffolds were successfully combined with neural stem cell progenitors to engineer neural tissues [148]. Alginate scaffolds have also been combined with ESCs to generate hepatocytes and vasculature [149, 150]. While alginate can be electrospun, such scaffolds have not been evaluated for their compatibility with stem cells [151]. The properties of alginate make it easy to bioprint. Accordingly, several different 3D bioprinted tissues have been produced using alginate bioinks in combination with stem cells, including neural tissue, cartilage, bone and liver [152–155]. This body of work shows that alginate serves as a highly desirable material for engineering a wide variety of tissues due to its unique properties.
Hyaluronan
Hyaluronan, also known as hyaluronic acid, serves as one of the major components of the extracellular matrix, [156] containing numerous sites for cell adhesion [156]. Hyaluronan also plays a key role in various stem cell niches, suggesting its suitability as a scaffold material for stem cell culture [157]. The Langer lab demonstrated that such scaffolds could be used for promoting both self-renewal of human ES cells as well as vascular differentiation [158]. Also, a recent review detailed how different hyaluronan scaffolds can be combined with MSCs to engineer a variety of tissues, including cartilage and bone [159]. Work from the Woodhouse group has also used such approaches to engineer adipose substitutes from stem cells [160, 161]. Other approaches have combined hyaluronan scaffolds with stem cells derived from keratinocytes and adipose for engineering skin and bone respectively [162, 163]. Such engineered tissues can be used as novel ways to promote wound healing as well as for repairing damaged nerve tissues [164–166]. Encapsulating stem cells into hyaluronic acid microcarriers is another popular tissue engineering strategy due to its ability to provide a supportive environment for stem cell survival [167, 168]. Similar to other polysaccharides, hyaluronic acid can be incorporated into electrospun scaffolds to improve their biocompatibility and enhance cell adhesion [169]. Finally, several recent studies have demonstrated that bioink formulations containing hyaluronic acid can be used to bioprint 3D tissues, making this an exciting area for future work [170–172].
Chitosan
Another commonly used polysaccharide, chitosan, can be derived by the deacetylation of chitin and it consists of glucosamine units [173]. Additionally, the rate of gelation of chitosan scaffolds can be controlled using pH [174] and the scaffold properties can also be modulated for stem cell applications through chemical cross-linking [175]. Chitosan has been evaluated extensively for tissue engineering applications, including in combination with stem cells. For example, 3D chitosan scaffolds containing coralline promoted osteogenic differentiation of mouse mesenchymal stem cells [176]. Two recent papers reviewed such efforts using different formulations of chitosan scaffolds for applications in developing replacement bone and cartilage [177, 178]. Other efforts have combined chitosan scaffolds with stem cells to treat spinal cord injury [179, 180] as well as wound healing [181]. Chitosan scaffolds have supported the culture of mouse ESCs and the expansion of cord blood-derived stem cells [182, 183]. Microencapsulation of MSCs inside of chitosan particles has also been evaluated as a way to engineer bone tissue engineering [184]. Chitosan and its derivatives can also be electrospun into substrates under the appropriate conditions [185]. Composite electrospun nanofibers containing chitosan support stem cell differentiation into nerve tissue, suggesting their utility for tissue engineering applications [186]. Chitosan can also be used as a bioink [187], but more work needs to be done to determine an optimal formulation for printing stem cell-derived constructs. Often these bioinks must be tailored to the specific application to ensure proper stem cell survival and differentiation.
Cellulose and its derivatives
Cellulose serves as a major component of cell walls found in bacteria and plants and it, along with its chemically modified derivatives, have been evaluated for tissue engineering applications [188]. It is often modified to enhance its ability to support mammalian cell culture. One of the most commonly used derivatives is methylcellulose due to its ability to form hydrogels under different conditions [189], including injectable versions and thermally reversible formulations [190]. These scaffold formulations can be combined with a number of stem cell types for engineering a wide variety of tissues, ranging from bone to nerve [191, 192]. It can also be combined with other polysaccharides to yield printable bioinks [172]. Cellulose and the closely related methylcellulose have demonstrated significant promise for a variety of tissue engineering applications due to its versatile properties.
Decellularized extracellular matrix
An alternative strategy to using pure biomaterials requires the process of removing the cellular components of a tissue, leaving behind the decellularized extracellular matrix (dECM) [193]. This method takes advantage of the natural bioactive properties of tissue to produce a suitable scaffold for stem cell culture and differentiation. The methods used to process the tissue may vary and can result in different properties for the scaffold material obtained [193] and they vary based on the tissue targeted [194]. Such dECM scaffolds have been extensively characterized for tissue engineering applications involving MSCs [195, 196]. Even whole decellularized organs, such as kidneys and livers, can be repopulated into functional tissues after seeding with stem cells [197]. Such dECM materials can also be bioprinted, providing another avenue for tissue engineering [198]. While these materials show significant promise, they can show variability due to the source material and the isolation method, and could potentially trigger immune responses [199]. However, they are worthy of further investigation for applications in stem cell-based tissue engineering and potentially the engineering of whole organs.
SYNTHETIC BIOMATERIALS
Synthetic biomaterials provide an alternative to natural materials for engineering tissues from stem cells. These materials offer many advantages over their natural counterparts, including reproducibility due to their defined chemical composition, and the ability to control the mechanical properties, degradation rate, and shape independently. The mechanical properties of a scaffold play a large influence in how stem cells differentiate [200]. The ability to shape a material allows for production of scaffolds that conform to specifications of the injury or transplantation site. Producing scaffolds with a specific degradation rate serves as a key advantage of synthetic scaffolds over natural biomaterials, and they can also affect the release rate of drugs incorporated into such scaffolds. However, many synthetic biomaterials lack sites for cell adhesion and accordingly must be chemically modified to allow for stem cell adhesion and culture. Other considerations when choosing a biomaterial include the biocompatibility of the material and its suitability for transplantation in vivo, as well as whether the material and its byproducts can trigger an immune response. However, as the following section will attest – many synthetic materials demonstrate huge promise for stem cell-based tissue engineering.
Polymer-based biomaterials
This section discusses synthetic polymers and how they can be combined with stem cells for producing different tissues. These materials are chemically defined and can often be formulated to have targeted mechanical properties to replicate the microenvironment present in the tissue being engineered. They can also be chemically modified to contain cues to enhance their bioactivity and promote stem cell differentiation into the desired phenotypes. Potential issues with these polymers include a lack of sites for cell adhesion and the potential for toxic byproducts after degradation. This section focuses on the most commonly used polymer scaffolds for culture of stem cells and their different formulations.
Poly (lactic-co-glycolic acid)
Poly (lactic-co-glycolic acid) (PLGA) a copolymer that consists of monomers of glycolic acid and lactic acid connected by ester bonds, has been approved by the FDA for a range of applications, mainly focused on drug delivery as it can be formulated to control drug release rate [201]. Cells enzymatically degrade PLGA into monomers that can cause cell death due to their acidic nature [202]. PLGA can be formulated into scaffolds, nano and microparticles, and electrospun fibers [203]. The ability to create biocompatible scaffolds with tunable drug delivery properties makes PLGA an attractive material for stem cell-based tissue engineering.
PGLA scaffolds can be combined with different types of stem cells, including MSCs, ESCs, and iPSCs to engineer a wide range of tissues, including muscle, bone, cartilage, and nerve [204–209]. The scaffold porosity, topography, mechanical properties, and drug delivery capacity all serve as important variables when designing such scaffolds. Both PLGA microspheres and electrospun nanofibers have also been used for differentiating stem cells into mature tissue types as well [210, 211]. Intriguingly, despite its widespread adoption as a biomaterial for clinical applications as well as for engineering tissues in vitro, PLGA has not been adopted as a bioink for use with 3D printers. This knowledge gap provides an intriguing opportunity for further work.
Poly (ethylene glycol)
Poly (ethylene glycol) (PEG), and its high molecular weight counterpart poly (ethylene oxide) (PEO), resist protein absorption, making them a commonly used polymer for in vivo applications due to this biocompatibility [212]. Scaffolds made from PEG can be polymerized chemically or using photoinitiators. The amount of initiator used affects the properties of the resulting scaffolds and it also means PEG can be used for applications in 3D printing [213]. While unmodified PEG is typically inert, it can also be chemically modified to contain bioactive molecules, including peptides and heparin [214]. PEG hydrogel scaffolds have been combined with a variety of stem cell types for their suitability as potential replacements for bone, cartilage, nerve, liver and vasculature tissue [215–223]. Other groups have incorporated PEG into other polymer solutions for producing electrospun scaffolds for engineering tissues from stem cells [224, 225]. PEG microcarriers can also serve as effective vehicles for delivering stem cells for in vivo tissue engineering applications [226, 227]. More recent work has focused on using PEG-derived bioinks to print tissue constructs from stem cells, including cartilage and muscle tissues [228, 229]. Its versatility and ability to be modified make PEG an essential tool for tissue engineering.
Poly (caprolactone) (PCL)
Poly caprolactone (PCL), a popular low-cost polymer with an extended degradation rate, can be used to form a wide range of scaffolds with different properties, including novel topographies and controlled release, for applications in tissue engineering as reviewed recently [230]. These scaffolds are often fabricated using both solution and melt electrospinning, and then seeded with stem cells to engineer tissues, including cartilage, nerve, muscle, and bone [231–235]. Figure 5 shows an example of how such scaffolds can be combined with adipose-derived stem cells to produce fat tissue as well as how topographical cues can influence stem cell behavior [236]. Melt electrospinning requires that the PCL be heated until it becomes a liquid, which can then be used to directly write scaffolds with defined structures that can be seeded with cells [237]. Similar work has been performed using 3D printed PCL to produce defined structures which are then seeded with stem cells to engineer tissues such as bone [238]. Work still remains to create a PCL bioink that could directly print cells into defined structures. Other work has incorporated drug-releasing PCL microspheres in pluripotent stem cell aggregates to engineer neural tissues [239, 240]. Also, the mechanical stability and ability to generate controlled drug release over extended time periods means that PCL is often combined with other biomaterials to generate hybrid scaffolds for tissue engineering [241], making it a useful biomaterial for a range of applications.

Human adipose derived stem cells growing in PCL fiber matrices. A) Normalized intensities of the CARS (2845 cm– 1) signal of the fibers (violet, solid) and the MPEF signal (495– 530 nm) (black, dashed lines, one line for each cell) of the cells in the matrices in the z dimension for the aligned (left) and random (right) matrices. 5 cells were investigated for each condition, for the random matrix two fibers were shown to visualize the different fiber profiles in this matrix. B) 3D reconstructions of the MPEF signals of the cells on/in the fiber matrices for aligned (left) and random (right) matrices. Nuclei are indicated with circles and fibers with arrowheads (scalebar: 30μm). This figure was reproduced under a Attribution 4.0 International (CC BY 4.0). Figure taken from [236].
Polypyrrole (Ppy)
Polypyrrole (Ppy) possess the unique property of being a biocompatible conductive polymer, which makes it attractive for certain tissue engineering applications such as nerve and muscle [242]. It can also be doped with biological molecules, including polysaccharides, such as heparin and hyaluronic acid, and such biomaterials have been used to engineer bone from MSCs [243]. The use of electrical stimulation on stem cells seeded on Ppy scaffolds enhances their differentiation into neural tissue, including promoting the development of mature retinal cells [244] and neurons [244]. Electrical stimulation of stem cells seeded on Ppy scaffolds can also drive osteogenesis [245] and the formation of functional muscle tissues [246]. Ppy can also be electrospun to produce scaffolds for stem cell differentiation [247] as well as be combined with other materials to generate conductive hybrid scaffolds for stem cell-based tissue engineering [248].
Polydimethylsiloxane (PDMS)
Polydimethylsiloxane (PDMS), a silicon-based viscoelastic polymer, has been used in a wide range of biomedical applications due to its inertness [249]. It is often used to construct microfluidic devices, which serve as valuable tools for studying stem cell behavior [250]. Its inert nature makes an ideal material to create molds for replicating tissue structure, such as vasculature networks being derived from pluripotent stem cells [251] or for creating spheroids from different types of stem cells [252]. As a scaffold material, PDMS can be modified to contain chemical cues, including sites for cell adhesion. The mechanical properties of PDMS are easily manipulated and thus these scaffolds have been used to determine how mechanical properties of the microenvironment influence stem cell differentiation [200, 253]. Another interesting tissue engineering application requires the incorporation of PDMS microspheres in stem cell aggregates. Such microspheres can provide mechanoregulation cues to direct MSC differentiation into functional tissues [254]. Additionally, hybrid microparticles consisting of PDMS and PCL promoted ESCs to form cardiovascular tissue [255]. PDMS has been applied in unique ways to engineer tissues from stem cells as discussed here.
Peptide-based biomaterials
Peptide-based biomaterials consist of short sequences of amino acids, which can produce self-assembling scaffolds [256]. These scaffolds combine the functionality of protein-based scaffolds by using motifs derived from naturally occurring proteins with the reproducibility of synthetic scaffolds. Many of the peptide-based biomaterials can self-assemble into 3D scaffolds using amphiphilic peptides, which form aggregates in aqueous solutions. The Stupp lab was one of the first groups to use such self-assembling scaffolds for promoting the differentiation of murine neural progenitor cells into neurons [257]. These scaffolds contained the peptide sequence IKVAV (isoleucine-lysine-valine-alanine-valine) derived from laminin and this sequence had been shown previously to promote neurite outgrowth [258]. A similar approach was used to develop self-assembling peptide scaffolds seeded with mesenchymal stem cells for bone tissue engineering [259]. A variety of such self-assembling peptides have been evaluated for stem cell applications both in vitro and in vivo [260, 261]. Figure 6 shows representative images of different types of self-assembled peptide scaffolds as analyzed using atomic force microscopy.

Atomic Force Microscopy (AFM) images of: A) Self-assembling peptide hydrogel pristine (SAP) or enriched with conjugates between SAPs and adhesive peptides (called SAP-RGD and SAP-HVP) or decorated with a conjugate between SAP and Insulin-like Growth factor-1 (called SAP-IGF-1) at 4 10– 5 M; and B) SAP-RGD, SAP-HVP and SAP-IGF-1 at 4 10– 7 M on mica surface. This figure was reproduced under a Attribution 4.0 International (CC BY 4.0). Figure taken from [261].
One popular self-assembling peptide called RADA16-I has been combined with stem cells for tissue engineering applications. Such scaffolds were seeded with embryoid bodies derived from mouse ESCs in the presence of osteogenic medium to engineer bone tissue [262]. A follow-up study combined these self-assembling peptides with the ceramic hydroxyapatite, producing a more effective scaffold for differentiating the mouse ESCs seeded inside [263]. The Zhang lab altered the RADA16 sequence to incorporate 18 different peptide motifs to determine the most appropriate scaffold material for mouse adult neural stem cells [264]. More recent work has shown that RADA16 scaffolds can be combined with stem cells to engineer cardiac, neural, adipose and bone tissue [265–268]. As their properties can be modified based on changing their sequence, there are significant possibilities to extend such peptide scaffolds for a range of tissue engineering applications.
CERAMIC-BASED BIOMATERIALS
Ceramics, inorganic materials formed through treatment with heat, possess crystalline structures, meaning they are often porous and brittle [269]. Commonly used ceramics include bioactive glass, which consists of a mixture of silicon dioxide, sodium oxide, calcium oxide, and phosphate oxide, [270] and hydroxyapatite, a naturally occurring material found in bone [271]. Other ceramic-based materials exhibit similar properties and these materials, along with hydroxyapatite, have been investigated extensively in combination with stem cells for tissue engineering of bone [206, 272– 289]. Figure 7 shows an excellent example of how interconnected porous calcium hydroxyapatite (IP-CHA) scaffolds can direct MSCs to form bone tissue [290]. These studies include both in vitro and in vivo testing of these materials. The Caplan lab has done a great deal of work characterizing the culture of mesenchymal stem cells on ceramic materials as a way to engineer bone tissue [279, 286]. Other groups as well have performed similar studies as reviewed recently [291]. These ceramic-based materials can be combined with biodegradable polymers, such as those materials mentioned earlier in this review, to be used for bone tissue engineering applications [292–294]. The use of composite materials allows for the ability to deliver drugs and the fabrication of highly porous structures necessary for stem cell infiltration when tissue engineering. More recent work has focused on how to use additive manufacturing to bioprint ceramic scaffolds and related composites for tissue engineering applications, which is an important area for future work [295].

In vitro osteogenesis of interconnected porous hydroxyapatite (IP-CHA discs combined with rat MSCs. A) Alkaline phosphatase (ALP) staining of IP-CHA discs, which were combined with MSCs and cultured in vitro for 14 days in non-osteogenic or osteogenic differentiation medium. Each graph is composed of three groups: no osteogenic induction (left), plasma-untreated (center), and plasma-treated (right) IP-CHA discs. ALP activity, B) and content of total protein, C) per MMCs/IP-CHA composite. D) ALP activity normalized by the content of total protein. Data were represented as mean±SD of 2 independent triplicate experiments. This figure reprinted under a Attribution 4.0 International (CC BY 4.0) license. Figure taken from [290].
CONCLUSIONS AND FUTURE STUDIES
This review detailed the most commonly used biomaterial scaffolds for engineering tissues from stem cells by covering the types of materials available and their unique properties. This information allows readers to determine which material best suits their specific application. As mentioned in earlier in this review, many of these materials have not been fully optimized for specific tissue engineering applications and further work will continue to optimize these formulations for translation to the clinic for targeted applications. For example, optimized scaffolds could enhance the survival and differentiation of neural stem cells being transplanted into the diseased or damaged nervous system, which could lead to improved function. The type of material and the cues that are incorporated in the scaffold play a large role in directing the fate of the stem cells seeded inside as detailed in this review. The ability to further functionalize the materials discussed in this review in terms of their mechanical and chemical properties provides an excellent opportunity for future work, as such bioactive and instructive scaffolds can improve cell survival and differentiation into the desired phenotypes. The method of fabrication serves as an important parameter, which allows different types of patterns and architecture to be formed, which include hydrogels, microcarriers, fibers and 3D bioprinted constructs. In particular, 3D bioprinting offers the possibility of printing constructs to fill an injury site as well as the ability to produce tissues with complex structures containing multiple cell types. Additionally, work remains to make many of these biomaterials into printable formulations that can support stem cell survival and differentiation post printing. being able to incorporate or produce a vascular network inside of engineered tissues fabricated through traditional tissue engineering methods as well as using bioprinting remains challenging, but may be addressed through complex 3D printing strategies or through other strategies. The lack of a stable vasculature often restricts the size of the constructs due to concerns about maintaining cell viability in light of diffusion constraints. This review provides an important overview of the general concepts when combining biomaterial scaffolds with stem cells for tissue engineering applications.
DISCLOSURES
Dr. Willerth has on-going a collaborative research agreement with Aspect Biosystems to commercialize her neural tissue engineering technologies and a collaborative research agreement with iProgen Biotech for her reprogramming work.
STEMJOURNAL OPEN REVIEW
The evaluations from reviewers for this article are freely available and can be found as supplementary material here: https://dx.doi.org/10.3233/STJ-180001.
Footnotes
ACKNOWLEDGMENTS
Dr. Stephanie Willerth would like to acknowledge funding from the Canada Research Chairs program, the Natural Science and Engineering Research Council, the Stem Cell Network, and the British Columbia Innovation Council’s Ignite Program. Dr. Sakiyama-Elbert would like to acknowledge funding from the NIH (NINDS R01 NS090617).
