Abstract
BACKGROUND:
Wernicke’s encephalopathy (WE), a neurological disorder due to the deficiency of thiamine, is often underdiagnosed in the pediatric population. The classic triad of mental status changes, oculomotor abnormality, and ataxia is observed in only 16–21% of all pediatric presentations. Wernicke’s is most often associated with alcohol dependence, but also malignancy, parenteral nutrition, and gastrointestinal (GI) malformations. The correlation between following a Ketogenic diet and acquiring Wernicke’s, however, has not previously been reported.
CASE REPORT:
A 16-year-old previously healthy male presented with an eleven-day history of neurological deficits and GI upset. The patient had recently lost one hundred pounds while following a “ketogenic” diet. He was subsequently diagnosed with Wernicke’s, received intravenous thiamine, and was transferred to the inpatient rehabilitation, where he received extensive diet education. After making significant functional improvement, he was discharged home.
RELEVANCE:
This case illustrates the importance of including Wernicke’s in the differential diagnosis when a pediatric patient presents with neurological deficits after rapid weight loss. Wernicke’s encephalopathy may be fatal in the pediatric population, therefore, it must be treated immediately if clinically suspected. Children presenting with Wernicke’s would benefit from early intervention, intensive inpatient rehabilitation, and comprehensive education regarding the role of food and exercise on weight loss and health.
Introduction
Wernicke’s encephalopathy (WE) is an acute neurological disorder caused by a nutritional deficiency of thiamine, also known as vitamin B1. It typically is characterized by the classic triad of mental status changes, oculomotor abnormality, and ataxia (Table 1). It is observed in 16–21% of all pediatric patients who come to the emergency room. The most common presenting symptom is altered mental status followed by vomiting. Altered mental status may range from disorientation to an altered level of consciousness. It is estimated to be present in 82% of pediatric patients with Wernicke’s encephalopathy [1–4]. Oculomotor symptoms of diplopia, lateral rectus palsy, and nystagmus are present in approximately 68% [2, 4], and ataxia, the least common symptom from the triad, is estimated to be present in 20% of pediatric patients [2, 4]. However, up to 19% of them will not have any of the classic symptoms. In those with WE, Magnetic Response Imaging (MRI) diffuse weighted T2 images may reveal hyperintense signaling as well as axial flair sequences of the mammillary bodies, basal ganglia, medial thalamus, periaqueductal gray matter, and tectal plate (Fig. 1) [5–8]. If MRI is unavailable, a cranial computed tomography (CT) scan may reveal hypodensities in the basal ganglia or thalamus. However, a CT scan is far less sensitive than an MRI [2, 5]. Lesions are typically symmetric and similar in both adult and pediatric cases of nonalcoholic WE [1–3, 5–7]. Imaging abnormalities on MRI or CT may be present in 55% of pediatric patients with WE [2].
Symptomatology in Wernicke’s Encephalopathy, including clinical triad of oculomotor abnormality, ataxia, and confusion. Both Central and Peripheral Nervous Systems are affected
Symptomatology in Wernicke’s Encephalopathy, including clinical triad of oculomotor abnormality, ataxia, and confusion. Both Central and Peripheral Nervous Systems are affected
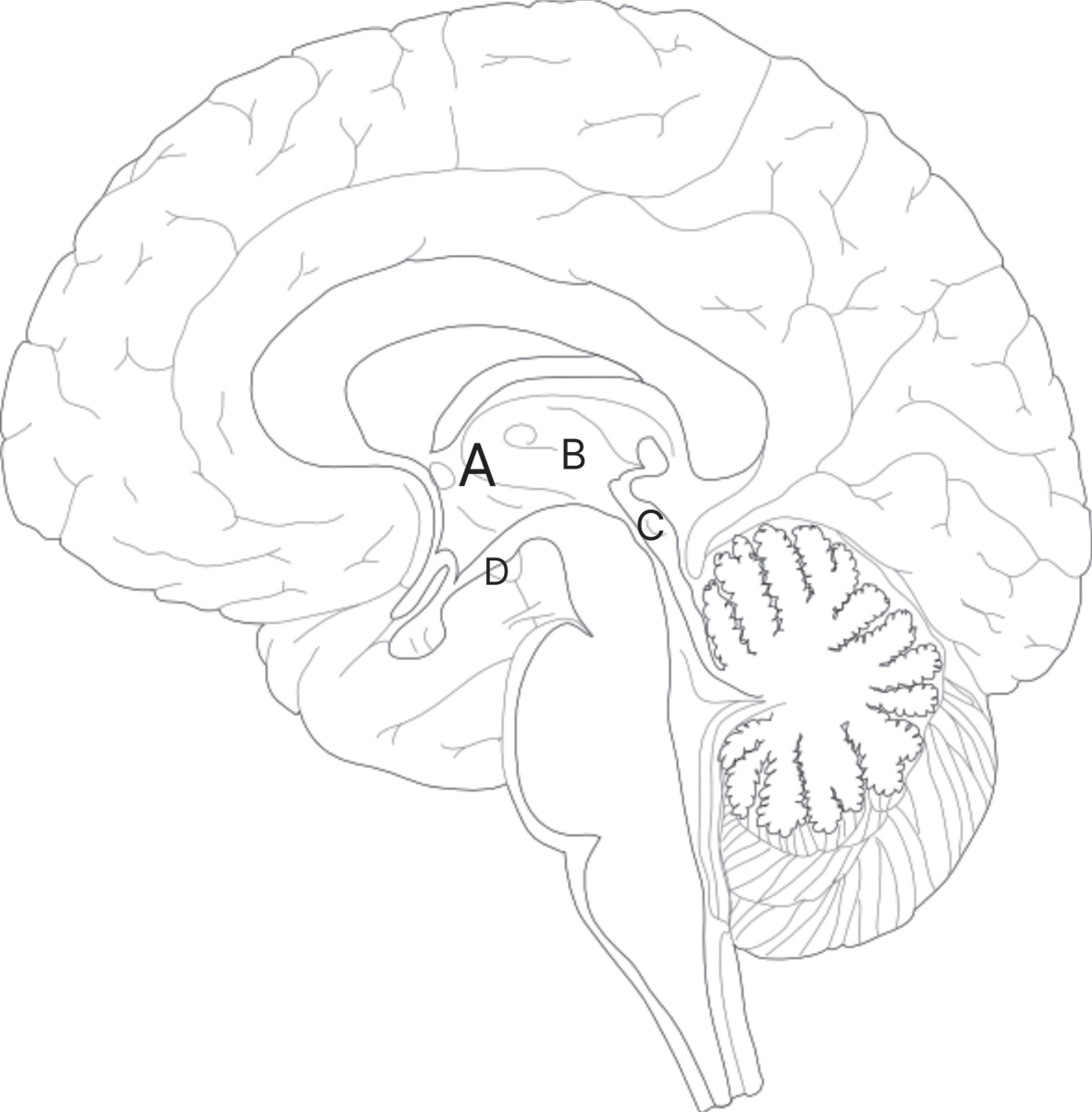
Mid-sagittal view of the brain demonstrating area of possible involvement with Wernicke’s Encephalopathy. Basal ganglia (A) overlies the thalamus (B), tectal plate (C) and mammillary bodies (D).
Wernicke’s encephalopathy is often underdiagnosed in both the pediatric and adult populations, but the frequency appears to be similar among both cohorts [3]. Wernicke’s encephalopathy is most often associated with chronic alcohol abuse, however, there are other etiologies including malignancy, inadequate diet, extended use of parenteral nutrition, and congenital or acquired gastrointestinal (GI) malformations, such as bariatric weight loss surgery, that can lead to the disorder due to malnutrition or GI losses [1, 8–10]. In children, malignancy is the most common known cause of WE [2]. The Ketogenic diet, which consists of high-fat, high-protein, and low-carbohydrate ratios, has been used in the treatment of pediatric epilepsy, but studies have also shown that it is a safe and effective weight loss regimen in morbidly obese adolescents [11]. Although multiple nutritional causes of WE have been described in the literature [1], the correlation between following a Ketogenic diet and acquiring WE has not previously been reported.
A 16-year-old obese male with no significant past medical history presented to the emergency department with an eleven-day history of diplopia, recent falls, headache, epigastric pain, nausea, and vomiting. The patient’s father noted increased fatigue as well. His father also noted that symptoms started after eating pork rinds with salsa and then elaborated that the patient had lost over one hundred pounds (going from 330 to 220 pounds) during the previous five months while following a Ketogenic diet, consisting primarily of meat, occasional vegetables, and rarely pork rinds. On exam, the patient was sluggish, had diffuse abdominal tenderness, a positive Murphy’s sign, tenderness at McBurney’s point, negative Rovsing sign, nystagmus, diplopia with confrontation, diminished sensation to bilateral lower extremities, and ataxic gait. The initial workup, including blood tests, urine analysis, toxicology screen, right upper quadrant ultrasound and MRI (Fig. 2) was unremarkable except for dehydration. During his acute hospital admission, the patient was evaluated by a registered dietician. Per their report, he was eating 2–3 meals a day consisting of veggies, avocados, omelets, and meat. He started eliminating these foods to enhance weight loss. Approximately 1.5 months before his admission, he hit a weight loss plateau; moreover, he reduced his intake to eating one meal a day, which primarily consisted of beef sticks, hot dogs, or buffalo chicken wings. Subsequently, the patient was diagnosed with WE after he was found to have significant thiamine and vitamin D deficiency. He required a five-day course of intravenous (IV) thiamine three times a day followed by oral thiamine and vitamin D supplementations for several weeks after discharge until symptoms resolved.
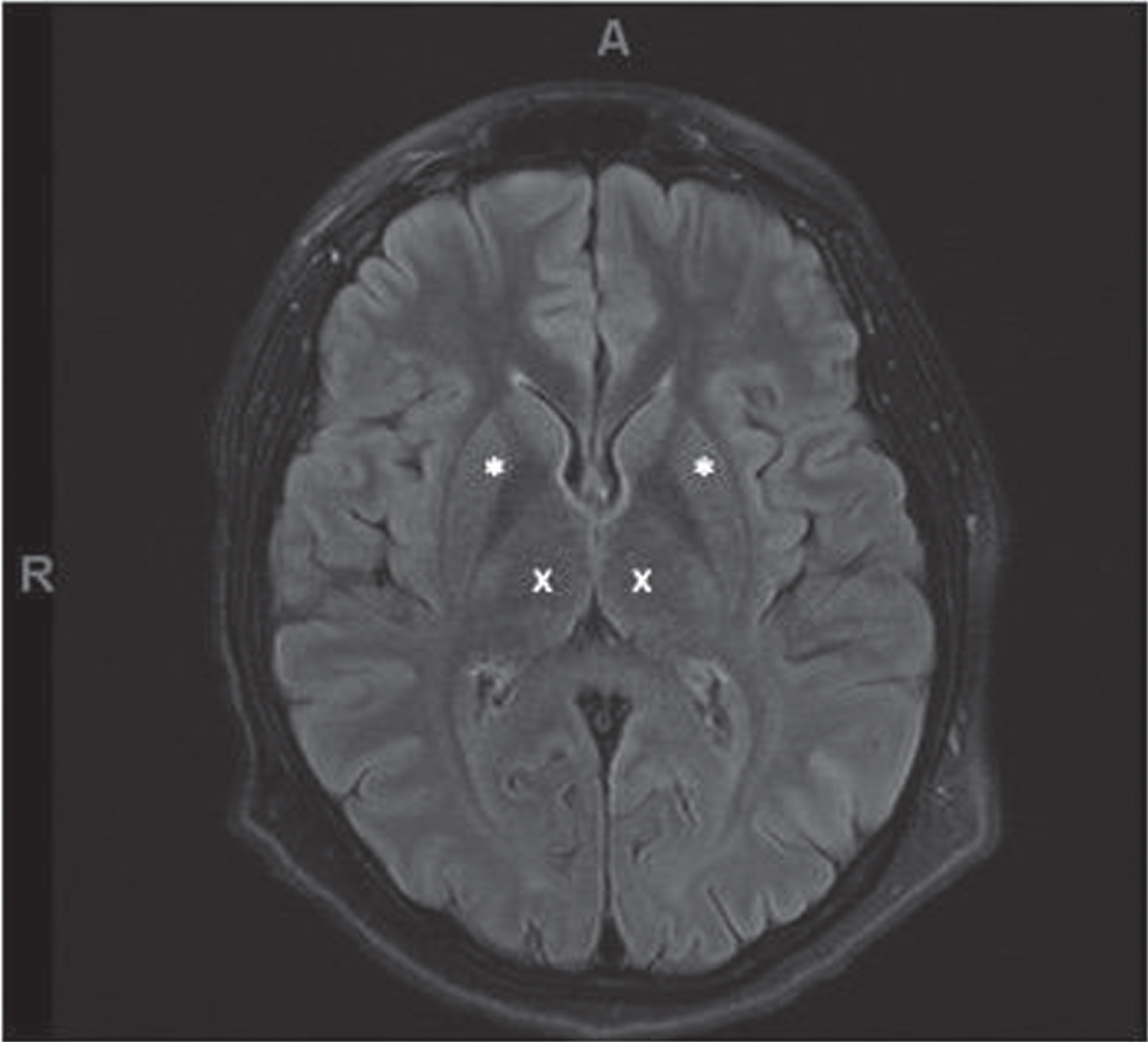
T2 flair magnetic resonance imaging (MRI) of the patient’s brain with normal thalamus and basal ganglia on initial presentation to the emergency department. * demarcates the putamen of the basal ganglia. x demarcates the thalamus.
The patient lived in a third-story apartment with his father and brother. Before the onset of symptoms, he was independent with activities of daily living (ADLs) and mobility but reported multiple falls days before admission. Physiatry was consulted on day 12 of the acute care stay given his continued motor control problems, impaired balance, visuospatial deficits, and decreased activity tolerance. Physiatry recommended that he be admitted to inpatient rehabilitation (IPR) after an interdisciplinary meeting with him, his father, the Neurologist, the Psychiatrist, and the Physiatrist.
On the initial occupational therapy evaluation, the patient required minimum assistance with lower body dressing, toilet transfers, and functional mobility. His sensation was intact to light touch with 4/5 strength in bilateral upper extremities. Although he was noted to have horizontal nystagmus with tracking and fine motor examination, testing did not reveal apraxia, ataxia, or dysmetria. The patient was seen by occupational therapy five times a week for a month.
At the time of his physical therapy evaluation, he presented with distal weakness, gastrocnemius tightness, and impaired sensation to light touch and pinprick of bilateral lower extremities. He was noted to have a wide-based stance with a flexed trunk on standing which required contact guard assist with ambulation for dynamic balance instability. While ambulating, the patient had a wide-based, shuffling gait and attempted to keep his knees in full extension to prevent them from buckling. This led him to be seen by physical therapy seven times a week for a month.
A speech therapy evaluation revealed word-fin-ding, recall, and problem-solving to be within normal limits. Therefore, speech therapy services were not needed.
During his IPR stay, he required significant functional endurance training, dynamic balance activities, ADL retraining, and overall strengthening. With the consultations of the Neuropsychologist, Child Life specialists, and Nutritionists, he also received extensive education and counseling about the importance of a balanced, nutritious diet, along with incorporating exercise into his daily routine. Cognitive Behavioral Therapy (CBT) played an important role throughout his stay in IPR. Through regular sessions with Neuropsychology, the patient was provided individual therapy focusing on coping strategies, self-esteem, and adjustment. On the day of discharge, the patient still required supervision for ADLs due to decreased safety awareness. He had impaired balance and fatigue, requiring contact guard assist for higher-level mobility (such as stairs). He did not require the use of any assistive devices. After twenty-six days, he was discharged home with outpatient therapies and a home exercise program to improve overall strength, coordination, balance, activity tolerance, and safety awareness. He was also provided with clinic referrals for outpatient Psychological therapy and Nutrition.
Discussion
Thiamine, also known as vitamin B1, is a water-soluble vitamin which is obtained solely from dietary sources such as poultry, beef, beans, and whole grains [11]. The recommended daily value is 1.2 mg [12]. It is an essential cofactor in the biochemical reactions leading to adenosine triphosphate (ATP) production via the Krebs cycle and pentose phosphate shunt [8]. It is converted to the active form of thiamine pyrophosphate in neuronal and glial cells. Thiamine pyrophosphate is a coenzyme for several biochemical mechanisms in the central nervous system including carbohydrate metabolism (for ATP production), lipid metabolism (myelin sheath production), and production of neurotransmitters [3, 13]. At a more cellular level, it is involved with the enzymes alpha-ketoglutarate dehydrogenase, pyruvate dehydrogenase, and transketolase, which are an essential part of the tricarboxylic acid cycle (also known as the Krebs cycle) and pentose-phosphate pathway [8, 15]. These enzymes can be affected within four days of a thiamine deficient state which leads to cellular edema, accumulation of lactate, and altered synthesis of amino acids in neurons and glial cells. Glial cells are the first to be affected by thiamine deficiency after only four days due to decreased alpha-ketoglutarate dehydrogenase, which results in edema, a buildup of lactate, and alters the synthesis of essential amino acids [14]. To obtain an energy source, myelin is broken down for the formation of ketones [14]. Recent studies suggest this is the most likely mechanism of reversible injury of WE; hence emergent intervention is required [12–14]. Thiamine has a short half-life of approximately 18 days with the body storing up to 30 mg at one time [12–14]. Given the short half-life and low volume of storage, thiamine stores may be quickly depleted.
The recommended treatment of WE in the pediatric population includes high dose IV or intramuscular thiamine supplementation for at least three to five days [8]. This is followed by daily oral thiamine supplementation, comprehensive education regarding a balanced diet and exercise, and involvement in physical and occupational therapy. With this, most neurological symptoms start to resolve within 24 hours of IV thiamine administration. Altered mental status has been reported to be the last symptom to resolve, typically within 2–3 weeks of treatment. If untreated, mental status deficits may become permanent and can be fatal. The prognosis is dependent on the implementation of thiamine with a continuous supply and is therefore critical for clinical outcomes [16].
Malnutrition is one of the most common pediatric causes of WE. It may be the result of multiple micronutrient deficiencies of an extensive diet, anorexia, or increased GI losses [9, 18], which may be due to acute illness or chemotherapy-induced. Vomiting has been reported to be the most common precipitating event [2]. With its short half-life, this supply of thiamine can be quickly depleted through GI losses [16]. Thiamine deficiency is referred to as absolute or relative. An absolute deficiency is due to insufficient dietary intake of thiamine concerning total caloric and carbohydrate intake while a relative deficiency occurs when glucose supplementation is out of proportion to thiamine supplementation such as intravenous parenteral nutrition [6, 13]. According to the NIH, whole grains, some meats, and fish have high thiamine content. Pork chops and bottom round steak have the highest amount of thiamine for meat with 33% and 8%, respectively, of the daily recommended value. White rice and breakfast cereals, on the other hand, are fortified and contain 100% of the daily recommended value [19]. In an unbalanced diet such as the Ketogenic diet, signs, and symptoms of WE may be precipitated after three to four weeks of deficiency due to the quick depletion of stored thiamine [13].
The Ketogenic diet is a high-fat, low-carbohydrate diet that has been used in pharmacoresistant epileptic children since 1920. In most children on this diet, the ratio of fat to protein and carbohydrates is 4 : 1 [20]. The average clinical Ketogenic diet restricts patients to 20 g–50 g of carbohydrates per day with the primary source being starchy vegetables (Fig. 3). Eating a high-fat, low-carbohydrate diet results in the production of ketone bodies consisting of acetoacetate, beta-hydroxybutyric acid, and acetone [20]. Fat cannot directly be utilized by the central nervous system as a primary energy source when glucose is unavailable. If done without adequate counseling, it can lead to severe malnutrition. While following a Ketogenic diet, primary energy production is via the anaerobic cycle leading to the buildup of lactate while ketones are the new energy source for the brain [20]. Developing thiamine deficiency results in further accumulation of lactate, leading to neuronal death [15]. In the Ketogenic diet, fortified carbohydrates are not included, limiting thiamine intake. In addition, high protein meat including fish contains less than adequate thiamine further limiting thiamine intake (Fig 3) [19]. This risk of malnutrition is significantly increased in patients following any unbalanced fad diet. Eating a healthy balanced diet provides adequate macronutrients and micronutrients to prevent neurologic complications as a result of malnutrition such as Wernicke’s encephalopathy.
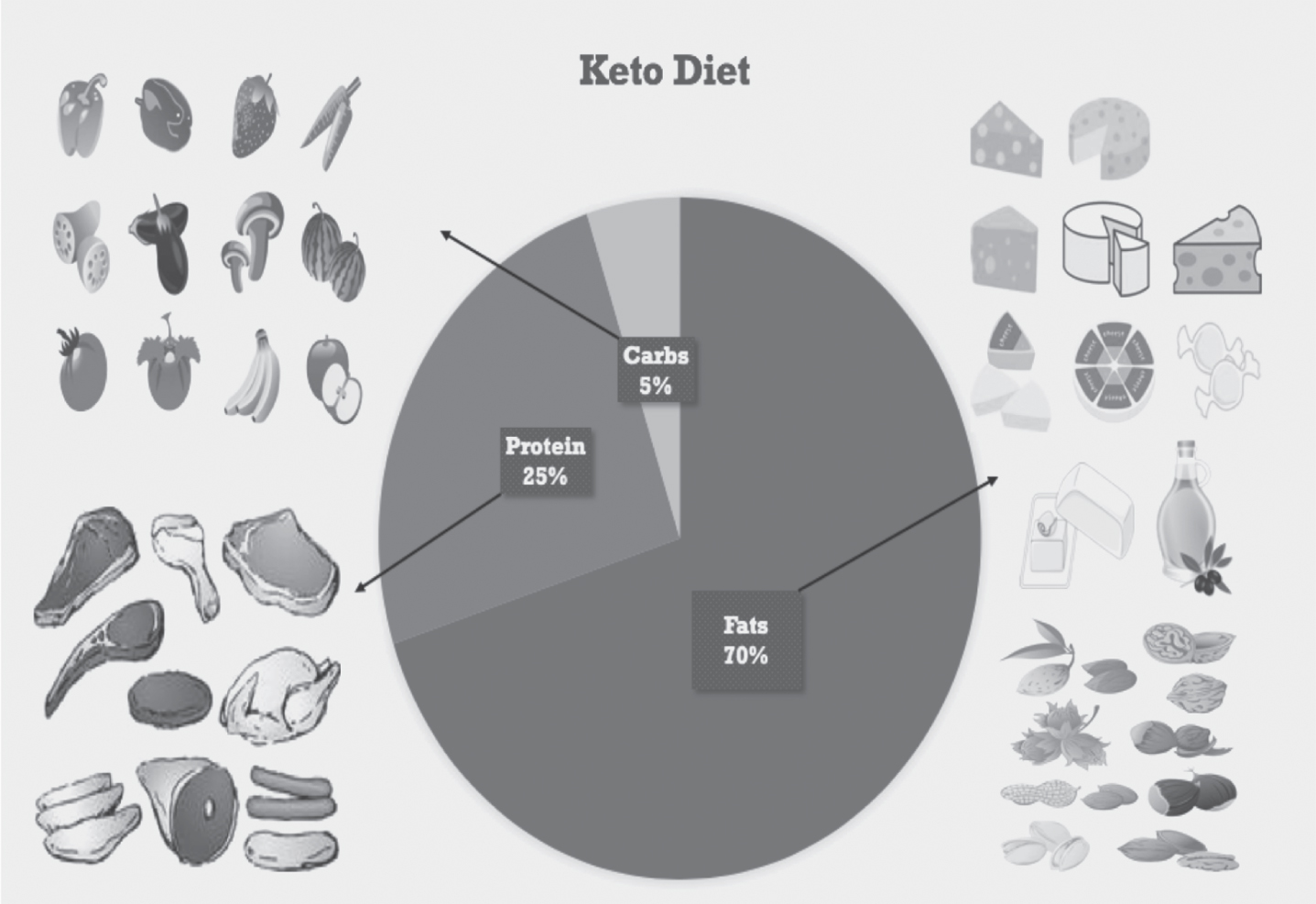
The macronutrient breakdown of a Ketogenic diet, including examples of food from each macronutrient.
The role of clinicians in the holistic approach to WE includes early diagnosis and treatment to prevent permanent disability and maximize functional recovery with a multidisciplinary approach. The team consists of the Physiatrist, Occupational, Physical, and Speech Therapies, Neuropsychologists, Nutritionists, and Child Life specialists who can provide support to the patient, caregivers, and family through extensive CBT and education on nutrition as well as incorporating a balanced diet and exercise into a healthy lifestyle.
This case illustrates the importance of including Wernicke’s encephalopathy in the differential diagnosis when a pediatric patient presents with unexplained neurological deficits in the setting of significant recent weight loss through the use of fad diets such as Keto. Wernicke’s encephalopathy may be fatal in the pediatric population, and therefore, must be treated immediately if clinically suspected. However, children that present with WE would benefit not only from early intervention through intensive multidisciplinary inpatient rehabilitation but also from comprehensive education regarding the role of a balanced diet and exercise on health.
Footnotes
Acknowledgments
The authors have no conflicts of interest to disclose. This study was completed independently without any financial subsidies to disclose.
