Abstract
OBJECTIVE:
To evaluate evidence supporting physical and occupational therapy interventions used to improve sensory and motor outcomes for children 0–6 years with central hypotonia.
METHODS:
Four electronic databases were searched from 1996 to March 2017. Level of evidence and study conduct was evaluated using American Academy of Cerebral Palsy and Developmental Medicine criteria. Traffic lighting classification identified interventions that were green (proven effective), yellow (possibly effective) or red (proven ineffective or contraindicated).
RESULTS:
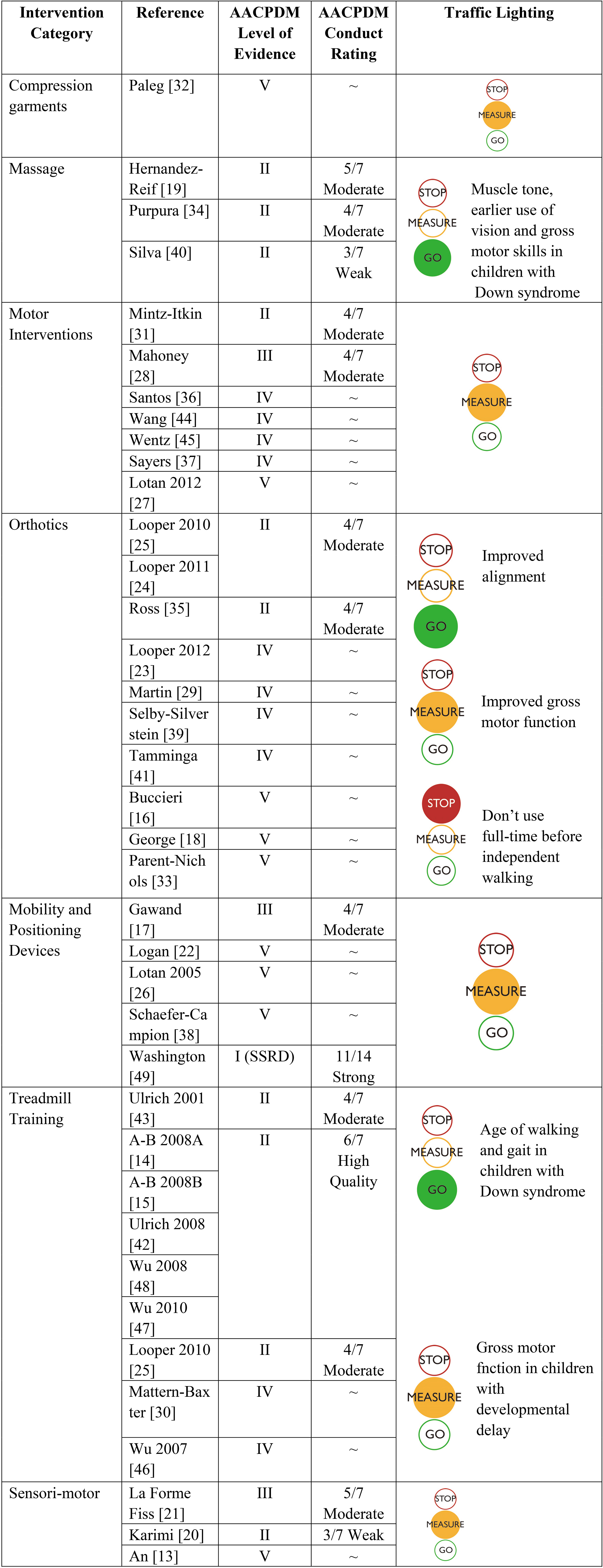
Thirty-seven articles were included. Nine studies measured orthotic interventions while four distinct studies published over nine articles measured treadmill interventions. Remaining studies measured impact of compression garments, massage, motor and sensori-motor interventions, positioning and mobility interventions.
CONCLUSIONS:
Green light evidence supports treadmill training (to promote ambulation and gait characteristics) and massage (to positively affect muscle tone, motor development and use of vision) for infants with Down syndrome. These interventions are considered Yellow (possibly effective) for other populations. Green light evidence supports impact of orthoses on foot alignment for ambulatory children with hypotonia, while impact on gait characteristics is Yellow light and motor development may be negatively impacted (Red light) in pre-ambulatory children. All other interventions rated Yellow (possibly effective) and therapists should monitor using sensitive outcome measures.
Introduction
Search terms and strategy
Search terms and strategy
Hypotonia, or reduced resistance to passive movement [1] may be identified in 4.25% of neonates born at or above 35 weeks gestation [2]. While congenital hypotonia may be central or peripheral in origin [3], central hypotonia represents 60% to 80% of cases [4]. Central hypotonia is concurrent with many genetic disorders such as Down, Rett, Joubert and Prader-Willi syndromes, but a similar presentation is also seen with children where no underlying cause can be identified. There are reports in the literature of children with hypotonic cerebral palsy (CP), but some surveillance registers exclude hypotonia as a CP sub-type [5] while others describe the prevalence as high as 5.1% [6].
Many children with idiopathic as well as other causes of hypotonia are referred to physical therapy (PT) and/or occupational therapy (OT) services with concerns regarding developmental and motor delays. Although believed to be beneficial [4], the effectiveness of PT and OT interventions for children with central hypotonia is unclear and clinical practice guidelines for young children combine low-level evidence with expert opinion [7]. Therapeutic interventions by OTs and PTs for children with hypotonia often focus on achieving developmental milestones, improving posture and postural stability, strength, endurance and enhancing function and activity [8].
While hypotonia from metabolic, genetic and neurologic pathologies may differ from idiopathic hypotonia, interventions for children with central hypotonia (regardless of specific etiology) should be based on the best available evidence and, in the absence of strong research with the specific population, interventions should be based on best-practice for children with similar impairments [9]. Accordingly, the purpose of this review was to identify and evaluate the evidence supporting interventions used by PTs and/or OTs to enhance motor and sensory outcomes for children aged birth to 6 years-of-age with central hypotonia.
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement was used to structure this review [10]. An electronic data-base search was undertaken in July 2016 and updated March 2017 for articles published from 1996 to present. We included studies conducted in the last 20 years, as these would meet similar reporting standards. Databases included CINAHL; Medline; EMBASE; and EBM Reviews. See Table 1 for search terms and a sample search strategy. No limits were placed on study design or publication status in the initial search. Primary research studies published in English in peer-reviewed journals were included if 50% or more participants were explicitly described as hypotonic or diagnosed with syndromes associated with hypotonia (e.g. Down syndrome) and measured impact of PT and/or OT interventions on motor or sensory outcomes. Exclusion criteria included; studies focused on other types of outcomes (e.g. behavioral, communication, activities of daily living); interventions not commonly used by OTs or PTs in typical early intervention settings (e.g. robotic trainers); interventions requiring an advanced professional certification (e.g. hippotherapy); studies involving children with hypotonia not central in origin (e.g. Spinal Muscular Atrophy, Spina Bifida, Spinal Cord Injury, Brachial Plexus Injury, etc.); and studies published as abstracts only.
Data were extracted from articles meeting inclusion criteria (see Table 2). American Academy of Cerebral Palsy and Developmental Medicine (AACPDM) [11] criteria was used to identify evidence level and AACPDM quality rating was used for individual studies evidence level I–III. AACPDM levels are as follows: Level I – systematic review (SR) of randomized controlled trials (RCTs), and large RCTs; Level II – smaller RCT’s and SR of cohort studies; Level III – cohort studies with concurrent controls, and SR of case control studies; Level IV – case series, cohort studies with historical controls, and case-control studies; Level V evidence – expert opinion or case studies. The traffic lighting classification system [12] was used to identify interventions that were green (proven effective), yellow (possibly effective) or red (proven ineffective or contra-indicated). All three authors agreed on studies to be read full text, articles meeting inclusion criteria, content of evidence table, level of evidence and quality ratings. Disagreements were resolved through discussion at all stages.
Included articles
Included articles
PRISMA flow diagram of the search results. From: Moher D, Liberati A, Tetzlaff J, Altman DG, The PRISMA Group (2009). Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med 6(6): e1000097. doi:10.1371/journal. pmed1000097.
AACPDM
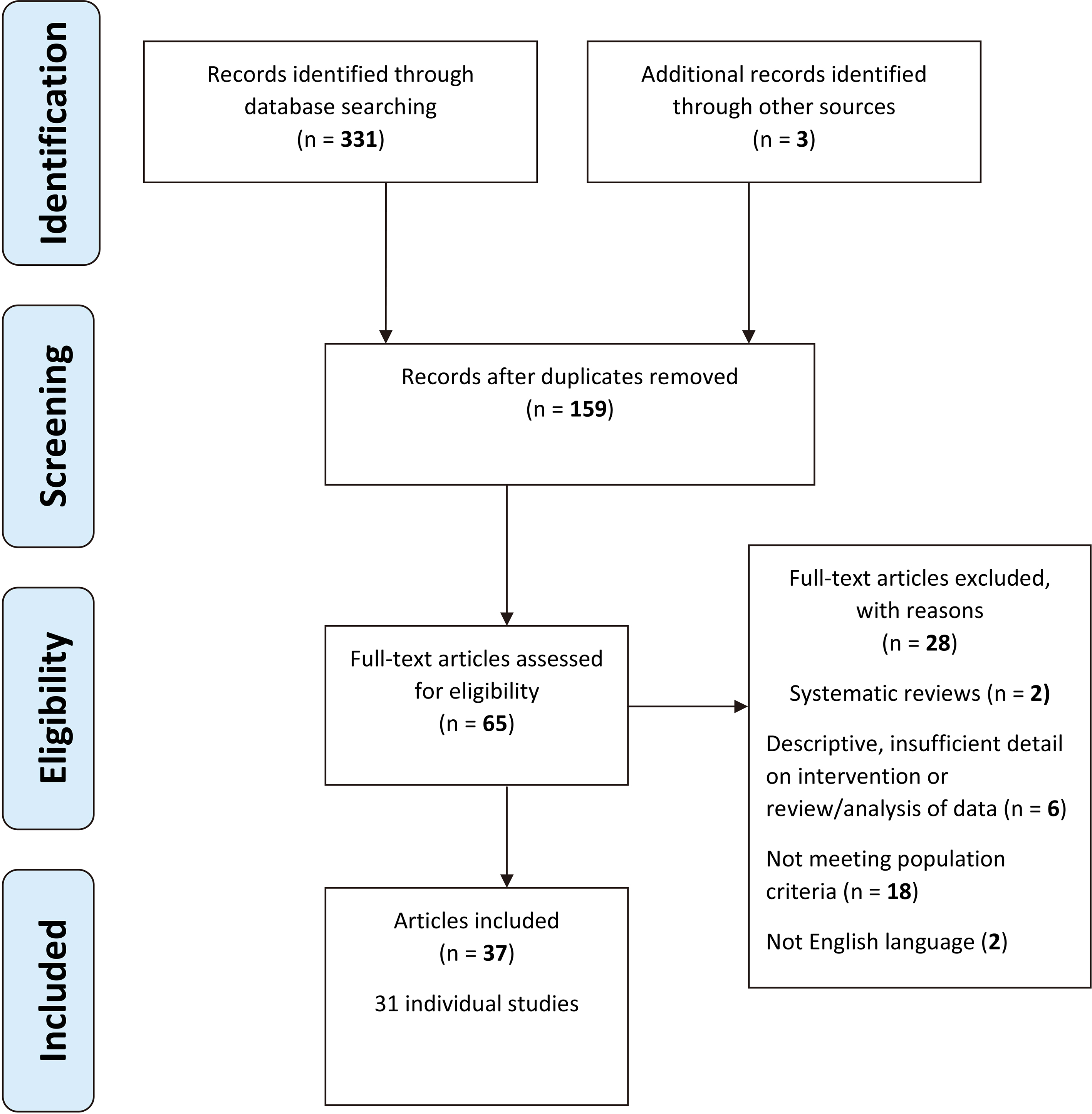
Thirty seven articles met inclusion criteria [13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49] (see Fig. 2 for PRISMA flow chart) and are summarized in Table 2. See online appendix A for details of excluded studies.
Two relevant systematic reviews [50, 51] were identified, but since they included some studies not meeting inclusion criteria for this review they are referenced only in the discussion. The 37 included articles represent 31 individual studies. Nine studies [16, 18, 23, 24, 25, 29, 33, 35, 39, 41]. measured orthotic interventions while four studies [14, 15, 25, 30, 42, 43, 46, 47, 48] measured treadmill interventions. One study on treadmill training was reported over six articles [14, 15, 42, 46, 47, 48] and another study investigating use of supra-malleolar orthoses (SMOs) with treadmill training was reported over two articles [24, 25]. Remaining studies measured impact of compression garments [32], massage [19, 34, 40], motor [27, 28, 31, 36, 37, 44, 45] and sensori-motor [13, 20, 21] interventions, positioning and mobility interventions [17, 22, 26, 38, 49]. Of 31 individual studies, 20 included children with Down syndrome, while children with Rett syndrome, CP with hypotonia and developmental delays were represented in fewer studies. A wide range of study designs were identified including nine individual randomized controlled trials (RCTs) or randomized crossover designs [19, 20, 25, 31, 34, 35, 40, 42, 43] as well as a number of non-randomized group designs such as pre-test post-test, repeated measure and case-control designs. Single-subject designs and case studies were also represented.
Results were divided into seven different types of clinical interventions; compression garments, massage, motor interventions, positioning and mobility interventions, orthotic interventions, treadmill training, and sensory interventions. Motor interventions were all different and included Conductive Education, Neurodevelopmental Therapy (NDT), ankle weights, jumping training and tummy time as well as different intensities of PT. Positioning and mobility devices included adaptive seating, standing and lying systems, walking aids and early power mobility. See Table 3 for a summary of positive versus inconclusive results across AACPDM evidence levels with details of outcome measures used. Statistically significant results are shown by bold type and mainly achieved in observational rather than experimental designs.
Evidence supporting the seven clinical intervention areas is summarized in Table 4 with AACPDM evidence level [11] and quality rating for individual studies achieving AACPDM [11] evidence levels I-III. See online appendix B for AACPDM [11] quality rating detail. Interventions supported by statistically significant positive results from moderate or high quality, level I or II group studies were accorded a green light or proven effective recommendation. Other than massage and treadmill training for infants with Down syndrome, other interventions achieved yellow light indicating that although results may be positive, outcomes should be measured to ensure benefit for individual children. Evidence supporting orthotic interventions is mixed, with possible negative effects for use of orthoses with children who are not yet walking independently [25], yellow evidence for type and timing of introduction of orthoses and green light for promoting better stability and foot structure and alignment in children who are walking independently.
Discussion
In this review, the strongest evidence was found to support massage (to improve muscle tone, use of vision and gross motor skills) and treadmill training (to accelerate onset of walking and improve gait parameters) for children with Down syndrome. Only one treadmill training study including children with a diagnosis other than Down syndrome met inclusion criteria for this review [30]. Treadmill training should be standard of care for infants with Down syndrome [52] and may be reasonably supported as an intervention for young children with other causes of hypotonia [53]. This was confirmed by a systematic review [50] despite having different inclusion criteria than this review. A major limitation however, is that of the nine primary studies that met our inclusion criteria, six used the same data set and this warrants caution in application of results.
Based on the best available evidence for effective treadmill training protocols for children with Down syndrome, treadmill training for children with hypotonia from other causes may benefit from home based treadmill training using speed of 0.33–0.67 miles per hour (or 0.15–0.35 meters per second), starting at a lower speed and increasing as the child gains skill. Starting around 10 months of age for 8–12 minutes per day, 5 days a week, increasing time as the child progresses and possibly adding light ankle weights to increase resistance may be effective. As with adapting interventions shown to be effective for a different population, therapists should be careful to measure outcomes with children to ensure desired outcomes. In the case of treadmill training, progress should be recorded with a valid and reliable outcome measure. Therapists should also monitor vital signs especially in children with cardiac concerns.
Most early intervention programs encourage natural environments and routines and treadmill interventions may be challenging to incorporate into this model. The use of typical infant walkers, reverse postural control walkers, push toys and/or gait trainers have not been studied in this same population, but these interventions may be more effectively incorporated into natural environments and routines. Positive results have been demonstrated from gait trainer interventions with children with intellectual disability, many presenting with hypotonia [54]. More rigorous research is required with group designs to support the effectiveness of gait trainer or ‘over-ground’ training for children with hypotonia.
There is also good evidence supporting use of massage with infants with Down syndrome. Infant massage is also helpful for parent bonding with children who may have self-regulation, sensory and motor concerns [55]. Although the difference in visual acuity and stereopsis [34] was not maintained as the children got older, it could be argued that stimulating earlier use of vision might impact other outcomes like cognition or visual motor control and should be considered for chil-
Outcomes
Outcomes
AIMS
dren with other causes of hypotonia. Expert opinion suggests that early intervention providers, certified in infant massage, train parents and caregivers to provide traditional massage strokes given at a medium level [56]. This training should include attention to state (arousal versus relaxation), stress signals (e.g. finger splaying, yawning or eye aversion) and enhance caregiver child bonding as well as the child’s awareness of body segments.
Traffic lighting synthesis
The evidence surrounding use of orthotic interventions is more controversial, with a mix of positive, inconclusive and negative results. From a biomechanical perspective, orthoses should provide children with hypotonia a more neutral and stable foot position however, children should also be given barefoot time to explore the sensation of active weight bearing and weight shifting. Kathy Martin and Julia Looper (personal communication March 2017) suggest that children who are pulling to stand and have good exploratory behavior should be allowed to move without the restriction of full-time orthotic use. Those that do not have the stability at the foot and ankle to move and explore in the standing position, should be provided with orthoses that give them enough stability to explore their environment in order to maximize cognitive, motor and social development. When children pull to stand and cruise, a wearing schedule of 50% waking time is suggested.
The systematic review of orthotics was inconclusive [51], but did not divide the studies on children before and after the onset of independent ambulation. The authors questioned the reliability and validity of the outcome measure used to support the contention that SMOs and foot orthotics improved foot alignment [35]. However, numerous references suggest that the Staheli arch index [57] is reliable [58], has clinical utility and is sensitive in identifying flatfoot in preschool children [59] and may be considered a gold standard measure in individuals with Down syndrome [60].
The only study we identified that included only children with benign hypotonia (and excluded children with Down syndrome or CP) did not meet inclusion criteria as it was published in conference proceedings only. However, the researchers identified significantly improved gait characteristics when the children wore SMOs as opposed to a shoes only condition, and caregivers reported improved balance and functional skills such as ability to jump [61]. One case study [32] supporting use of compression garments with children with hypotonia met inclusion criteria. Therapists may consider inexpensive neoprene models before more expensive alternatives such as full body lycra garments.
No strong recommendations can be made regarding motor interventions since higher evidence level studies were inconclusive. However early implementation of tummy time, 90 min/day before 11 weeks of age [45] appears to promote motor development and should be combined with 2–4 sessions per month early intervention. When a child is not sitting by 8 months (adjusted age), early intervention therapists should consider the introduction of adapted toys, supported sitting and standing devices as well as increasing activity for participation in natural routines with an enriched environment [62].
Augmentative mobility (use of gait trainers and early manual and power mobility) is recommended for children who are not moving and exploring their environment between 9 and 12 months corrected age [54, 63]. Three included studies described use of a supported seating device for young children with hypotonia [17, 26, 49], while one also described other postural management interventions, such as stander, floor seat and a lying positioning system [26]. Although, the evidence supporting seating, standing and augmented mobility with children of any diagnostic group is lower level [54, 63, 64] evidence is mainly positive and these interventions should be considered for children with hypotonia who are not sitting, standing or walking at a similar age to their peers.
Surveys of Physical (PT) and Occupational Therapists (OT) suggest that the definition of hypotonia should include decreased strength, decreased activity tolerance, delayed motor skills development, rounded shoulder posture with leaning onto supports, hypermobile joints and increased flexibility [8, 65]. However, for hypotonia to be classed as a diagnosis, these characteristics need to be further defined and quantified [66]. While there are reports of PTs and OTs using range of motion (ROM), manual muscle testing and tests of ligamentous laxity, [67] there currently exists no valid and reliable way to measure hypotonia [68]. The ALT Scale [19] attempts to quantify hypotonia using an inverse of the Modified Ashworth Scale [69]. Govender [70] has published a clinical algorithm based on a previous literature review [68], survey of relevant clinicians (OTs, PTs, neurologists and pediatricians) [71] and a Delphi consensus of experts [72]. This work requires further development and validation. Recently the Hammersmith Infant Neurological Examination (HINE), has been suggested as a valid and reliable tool to identify hypotonia between 3 and 24 months as well as to identify underlying biomarkers of cerebral palsy [62].
Most notably, the original intention was to search for evidence regarding interventions for children with idiopathic hypotonia. However, the majority of the research identified, and the highest levels of evidence apply to children with Down syndrome, and it is acknowledged that this evidence cannot necessarily be directly applied to children with hypotonia from other causes. Children with hypotonia from a variety of causes make up a significant portion of referrals to early intervention services and the lack of evidence supporting intervention for these children is challenging for therapists. Consequently we chose to search for evidence supporting interventions for children with all forms of central hypotonia to assist therapists in identifying interventions that could reasonably be applied to their clients with careful monitoring and appropriate measurement of outcomes.
It should also be noted, that the search conducted in this review was wide ranging as we included a broad range of populations and interventions. We sought to address the most common interventions conducted by OTs and PTs with the aim of influencing motor and sensory outcomes, but excluded a number of daily living, play and assistive technology interventions that may influence these outcomes. The search was systematic and covered several different databases, but inclusion criteria were limited to studies published in English, in peer-reviewed literature. We may have missed studies published in other languages or grey literature. We did not have access to translation services for the two studies we found where only the abstract was available in English.
The quality rating used in this review was only applicable to evidence levels I-III, and therefore, no quality rating is provided for 19 out of the 31 included primary studies. The low numbers of participants involved and low evidence level in the majority of studies included warrants caution in interpretation of results. Although a number of higher evidence level studies were identified in this review, a number of the publications include the same data set. We chose to rate the study as a whole, rather than providing separate ratings for each publication, and recommendations cannot be given the same weight as if these were separate studies. Additionally, the traffic lighting classification system [12] used provides a green light (proven effective) rating for interventions supported by positive results from evidence level II group studies. We chose to use this classification as it is appropriate for rehabilitation interventions and allows discrimination between smaller number and lower evidence level studies. If we had chosen a more stringent rating designed for discriminating between high number medical interventions such as GRADE [73], all evidence would have been rated as low or very low.
Conclusion
Green light evidence supports treadmill training (to promote ambulation and gait characteristics) and massage (to positively affect muscle tone, motor development and use of vision) for infants with Down syndrome only. Foot orthoses should be used with caution for pre-ambulatory children, but may be helpful in maintaining foot alignment for ambulatory children with hypotonia from various causes. For children with central hypotonia (other than Down syndrome), all therapeutic interventions discussed in this review can be considered yellow (possibly effective) and valid and reliable outcome measures should be used, in order to ensure appropriate use.
Lack of a clear diagnosis is often a barrier to implementation of early intervention services but when hypotonia is interfering with function and/or development, we recommend they be found eligible for early intervention services as ‘atypical’. Evidence-informed clinical practice requires that the clinician be able to identify the condition (based on agreed upon definition), measure the severity, apply an intervention and then re-measure the severity with a valid reliable tool. None of these conditions can be met for children with central hypotonia. Further research is warranted into all aspects of hypotonia including definition, measurement, assessment and intervention.
Footnotes
Acknowledgments
Sunny Hill Health Centre for Children Clinical Library services for assistance in conducting the search and retrieval of documents. Dr Meir Lotan for sharing his extensive research and Drs Julia Looper and Kathy Martin for their willingness to discuss their views on use of orthotic interventions in regards to this review.
Conflict of interest
The first author has worked as an educational consultant for various manufacturers and suppliers of gait trainers and standers. Funding from these sources did not influence or bias the content of this work. The second and third authors report no conflict of interest. The authors alone are responsible for the content and writing of the paper.