Abstract
BACKGROUND:
Chronic hyperglycemic conditions can activate aberrant metabolic pathways causing neurotoxicity.
OBJECTIVE:
This study aimed to explore the effect of ethanol extract of
METHODS:
To ascertain the neuroprotective effect, Neuro-2a cells were pre-treated with HS followed by high glucose and assessed for cell viability, reactive oxygen species (ROS) generation, alterations in mitochondrial membrane potential (
RESULTS:
High glucose exhibited toxicity in Neuro-2a cells by ROS generation, disrupting
CONCLUSION:
Our results propose that HS with its active constituents can be considered a promising therapeutic agent to treat hyperglycemia associated neurodegenerative diseases.
Introduction
The term neurodegenerative diseases (NDDs) symbolize a range of neurological dysfunction with diverse clinical and pathological symptoms affecting specific brain areas and halting their everyday functions [1]. With the increase in the life expectancy of people due to the advancement in medicine and given the correlation between ageing and neurodegeneration, it can be envisaged that the prevalence and incidence of neurodegenerative disorders are increasing progressively [2]. In addition to ageing, recent studies have exposed possible associations between metabolic diseases like diabetes mellitus (DM) and neurodegeneration. Being a high energy-consuming organ, the brain depends on glucose to fuel its respective functions. However, chronic hyperglycemic conditions promote oxidative stress and activate aberrant metabolic pathways causing neurotoxicity [3, 4]. Several case studies suggest that individuals with DM have a significantly increased risk of developing dementia, Alzheimer’s disease (AD) and related pathologies [5–8]. The long-term hyperglycemic condition has been reported to increase amyloid beta (Aβ) production and accumulation in experimental animals by inducing its synaptic release, altering amyloid precursor protein (APP) processing by stimulating β-secretase, GSK-3β or reducing the degradation through an insulin-degrading enzyme (IDE) [9–12]. Further, hyperglycemia could also exacerbate tau hyperphosphorylation and aggravate α-synuclein aggregation leading to cognitive impairment [13–15].
With the link between hyperglycemia and neurodegenerative diseases intensified, assessment of anti-diabetic drugs like insulin, metformin, and thiazolidinediones for cognitive improvement was demonstrated through pre-clinical and early clinical studies [16–21]. Further, reports from cellular and animal models of diabetic-associated neurodegeneration have exposed the role of oxidative stress and the deterioration of the antioxidant defense system during the progression of the disease. In this regard, bioactive compounds, phytochemicals, and plant extracts with antioxidant potentials are extensively focused on managing diabetic-associated neurodegenerative diseases [22–26].
Materials and methods
Chemicals
All the cell culture mediums used in the study were procured from HyClone, USA. Monoclonal rabbit primary antibodies (JNK, p-JNK, AKT, p-AKT, SIRT-1, cleaved Caspase-3, Nrf-2 and β-actin) were obtained from Cell Signaling Technology, USA. Anti-rabbit IgG- HRP linked secondary antibody, D-Glucose, Acetylcholinesterase enzyme (AChE), acetylthiocholine iodide (ATCI), and dithionitrobenzoic acid (DTNB) were purchased from Sigma Aldrich, USA. ECL Western blotting substrate was obtained from GE Healthcare, USA. The murine neuroblastoma cells, Neuro-2a were procured from Health Science Research Resources Bank, Osaka, Japan. The chemicals required for
Plant collection and extraction
The red calyxes of
Compound identification by LC-MS/MS analysis
The ethanol extract of HS was subjected to LC-MS/MS analysis to identify bioactive compounds. Separation was performed using a Thermo Scientific C18 column (AcclaimTM Polar Advantage II, 3×150 mm, 3
Cholinesterase inhibitory assay
Acetylcholinesterase inhibitory activity for ethanol extract of HS was measured according to the standard protocol [41]. To 0.1 U (10
Neuro-2a cells (2×105 cells) were seeded in a 96-well plate (DMEM containing 4.5 g/L glucose, 10% Fetal bovine serum, 1X Penicillin-Streptomycin) and allowed to grow overnight. The cells were treated with various doses of glucose (50–200 mM) and HS extract (20–100
Measurement of reactive oxygen species (ROS) level
The generation of ROS upon glucose treatment was measured both qualitatively and quantitatively. Neuro-2a cells (2×105 cells) were grown in a 96-well black plate for quantification. Cells were pre-treated with HS extract (7.5, 10
Measurement of mitochondrial membrane potential (ΔΨ m)
Cells were treated in 96-well black plates as mentioned in section 2.5. After treatment, cells were incubated with 5
Real-Time PCR analysis
After treatment of cells, as mentioned earlier, the cells were collected, and total RNA was extracted using the standard Trizol procedure and quantified. The extracted RNA (1000 ng) was reverse-transcribed real-time PCR (qPCR) analysis was performed with Green Star PCR Master Mix (Bioneer, Korea) with gene-specific primers (Table 1), and the data were normalized to the endogenous control (GAPDH) [40].
List of genes and sequences of primers used for Real-Time PCR analysis
List of genes and sequences of primers used for Real-Time PCR analysis
The cells after treatment were collected and lysed with NP-40 lysis buffer. Separation of proteins (50
Molecular docking analysis
The structures of the target proteins Acetylcholinesterase (4EY7), FOXO-1 (3CO7), Calpain-1 (2R9C), GSK-3β (1R0E), LXR-α (3IPQ), and LXR-β (1PQ6) were retrieved from PDB database. Later, the water molecules, other ligands and cofactors were removed from the protein structures, followed by energy minimization using Swiss-PDBViewer software [44]. The ligand structures were downloaded from Pubchem database. To gain insight into the possible mode of action of the active hits, the possible interactions with protein by docking simulations were studied using DockThor. This program uses a grid-based docking method to compute different modes of ligand binding on the protein. We have performed blind docking with standard mode feature of the tool [45]. Docking calculations were conducted with a grid generation of 40×40×40 grid size for the target protein and the results were compared with the respective positive controls for each protein.
ADMET prediction of potential phytochemicals from HS extract
Pharmacokinetic properties of drug like small molecules based on their molecular structures were predicted by SwissADME online server (http://www.swissadme.ch) [46]. Prediction for toxicity of the phytochemicals were performed ProTox-II webserver (https://tox-new.charite.de/protox_II/index.php?site=home) [47].
Caenorhabditis elegans strains and culture conditions
The wild type
Lifespan assay
Lifespan analysis of
Real-time PCR analysis
The CL2006 strains were grouped into five (Control, Glucose treated (500 mM), HS-30 + Glucose treated, HS-40 + Glucose treated, HS-40 treated alone) and incubated for five days. After the treatment period, the nematodes were collected for RNA extraction and Real-Time PCR analysis for
Statistical analysis
All the experiments were done in triplicates and expressed as Mean±SD. One-way ANOVA (SPSS 17, SPSS Inc., Chicago, IL) followed by Duncan’s
Results
Phytochemical screening of HS extract by LC-MS analysis
To identify the potential bioactive compounds in HS extract, LC-MS analysis have been performed. The list of possible compounds after analyzing the m/z peaks were listed in Table 2. The compounds were analyzed using the METLIN database and compared with previous literature regarding the phytochemical screening performed in Hibiscus sabdariffa. Compounds including Hibiscus acid, allo-Aromadendrene, N-Feruloyltyramine, Sinapoyl aldehyde, Isobetanin, Hibiscus acid hydroxyethylester, Anisocoumarin H, Ethyl chlorogenate, which have been already reported to be present in Hibiscus sabdariffa extracts have been identified.
List of compounds in HS extract identified by LC-MS/MS analysis
List of compounds in HS extract identified by LC-MS/MS analysis
Cholinesterase enzyme (ChE) is a potential therapeutic target in treating Alzheimer’s disease. The inhibitors of ChEs abrogate the enzymatic degradation of acetylcholine in the synaptic cleft and improve cognition and memory in AD individuals. As HS was evaluated for its neuroprotective efficacy, the impact of the extract against the acetylcholinesterase enzyme was assessed. HS extract showed significant dose-dependent inhibition of the enzyme at an IC50 value of 92.72±2.95
Further, the bioactive compounds identified in the HS extract were subjected to molecular docking analysis against acetylcholinesterase enzyme (AChE) with donepezil as the positive ligand. Donepezil showed a binding score of –8.32 kcal/mol. In contrast, the phytochemicals of HS, including N-Feruloyltyramine (–9.894 kcal/mol), 11-Hydroxycanthin-6-one (–9.194 kcal/mol), allo-Aromadendrene (–9.058 kcal/mol), showed higher binding score than the control indicating they have better inhibitory property against AChE. The mode of interaction of the top 3 hits and control against the target were represented in Fig. 1B–E.

(A) Acetylcholinesterase inhibitory effect of HS extract (100–500
Neuro-2a cells were treated with varying doses of glucose (50–200 mM) and subjected to the MTT assay to check the toxic effect of high glucose. Glucose exhibited dose-dependent cytotoxicity in Neuro-2a cells. After 24 h treatment, a significant (

Cell viability determination by MTT assay. (A) Effect of varying concentrations of glucose in Neuro-2a cells (B) Effect of HS extract in Neuro-2a cells (C) Protective effect of HS extract against high glucose (175 mM) (Significance at
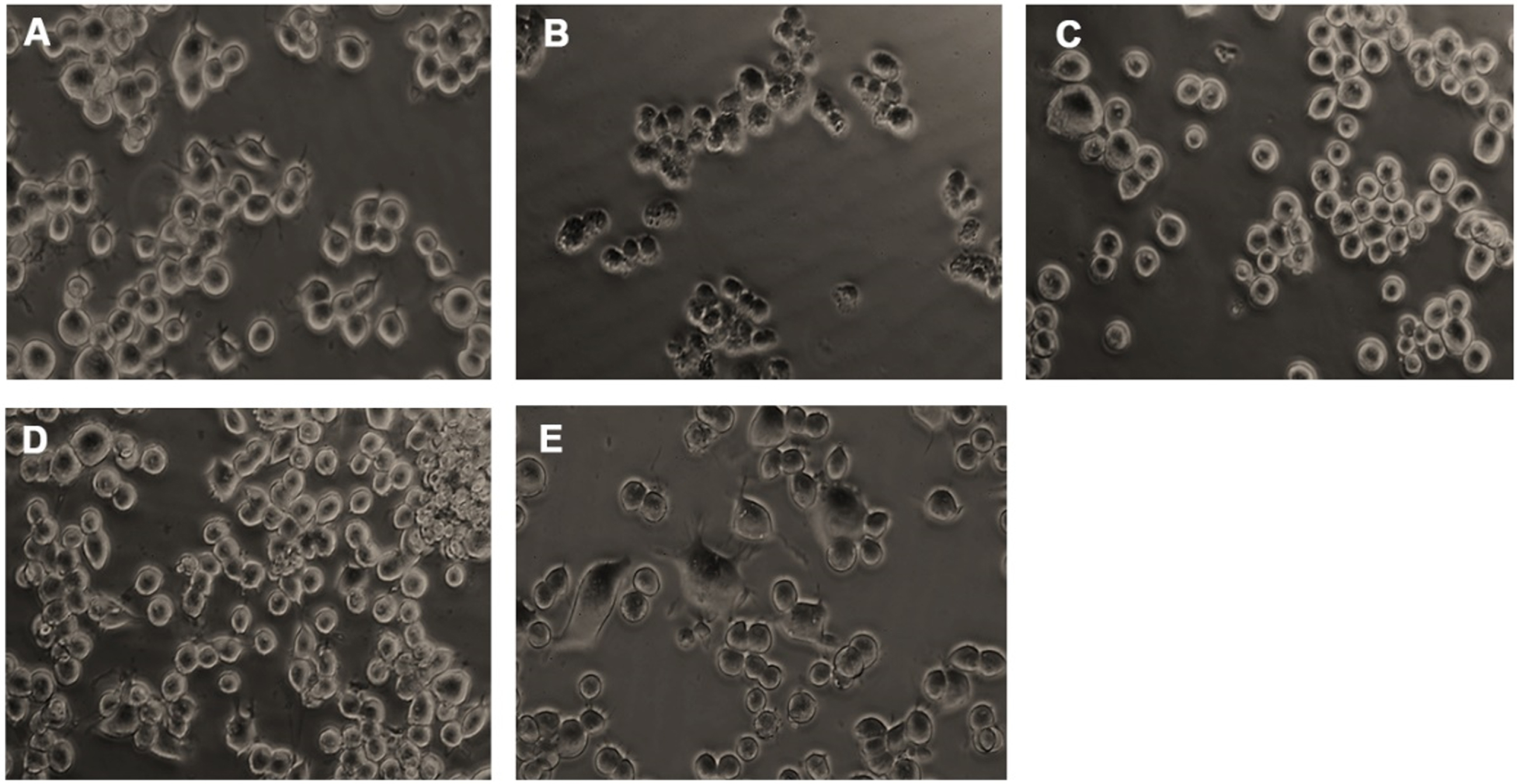
Microscopic images representing restoration from glucose-induced toxicity by HS. (A) Control (B) High glucose (175 mM) (C) HS-7.5 (pre-treatment 2 h) + high glucose (D) HS-10 (pre-treatment 2 h) + high glucose (E) HS-10
Previous reports suggest that high glucose-induced ROS generation is one of the critical events in making the biomolecules susceptible to harmful events and contributing to toxicity. Accordingly, exposure to high glucose significantly increased ROS generation (159.9±1.5%) compared to the control, which can also be observed from the increase in fluorescence intensity in confocal microscopic images (Fig. 4B, F). Nevertheless, pre-treatment (2 h) with HS (10
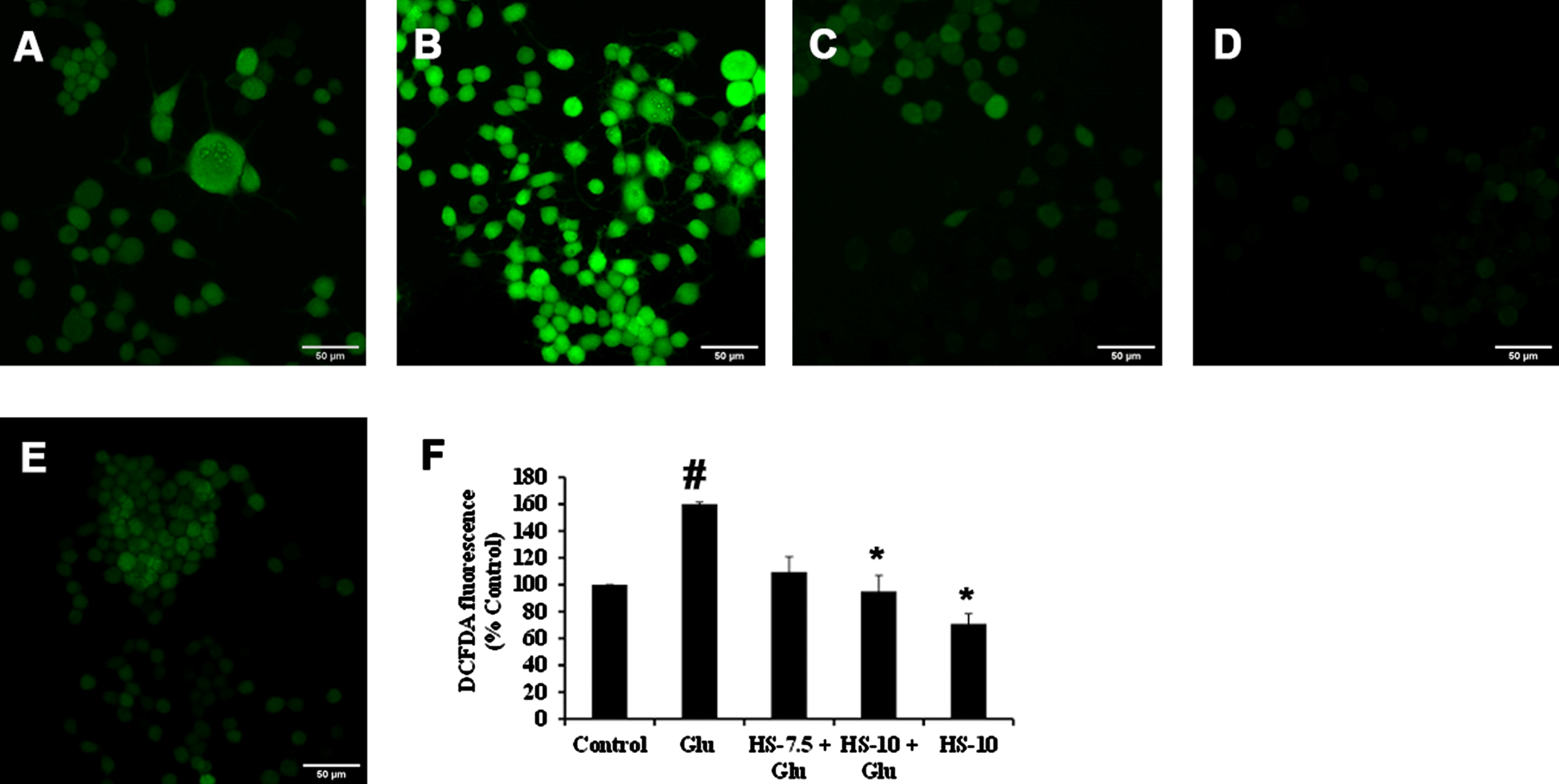
Confocal microscopic images representing attenuation of ROS formation by HS. (A) Control (B) high glucose (175 mM) (C) HS-7.5 (pre-treatment 2 h) + high glucose (D) HS-10 (pre-treatment 2 h) + high glucose (E) HS-10 (F) Quantification of ROS (Significance at
With the signifying role of ROS formation in high glucose-mediated toxicity observed, the role of antioxidant response element signaling pathway upon toxicity and the effect of HS to counterattack the response were studied using western blot (Nrf-2) and gene (NQO-1, HO-1) expression analysis. Protein expression analysis showed that upon high glucose treatment, Nrf-2 (0.89±0.08 fold) levels were reduced compared to the control. At the same time, a reversal in the effect was observed upon pre-treatment with HS extract. Moreover, HS alone (10

Effect of HS on stress response elements against glucose induced toxicity (A) Western blot expression of Nrf-2 (Lane: 1- Control; 2- High glucose; 3- HS-7.5 (pre-treatment 2 h) + high glucose; 4- HS-10 (pre-treatment 2 h) + high glucose; 5- HS-10) (B) Quantification of Nrf-2 expression (C) Transcriptional regulation of stress response genes (
In response to ROS generation, changes in

Effect of HS on mitochondrial membrane potential. Confocal microscopic images representing alterations in
The effect of HS extract on various pathways and targets involved in hyperglycemia-induced cell death was studied by gene and protein expression analysis. Evidence suggests that endoplasmic reticulum stress (ER stress) occurs concomitantly with oxidative stress conditions under chronic hyperglycemia. The gene expression analysis shows significant upregulation of Grp78 (1.53±0.09), Gadd153/CHOP (1.40±0.14), Calpain-1 (2.82±0.14) and GSK-3β (1.42±0.12) genes indicating ER stress and activation of downstream mechanism to trigger apoptosis (Fig. 7A). In addition, the interactions between SIRT-1 and FOXO-1 play a vital role in high glucose associated cellular stress. The results show that upon high glucose treatment, the protein expression of SIRT-1 (0.84±0.07 fold) was decreased with a significant increase in the gene expression of FOXO-1 (1.28±0.05 fold) (Fig. 7A–C). Also, a significant (
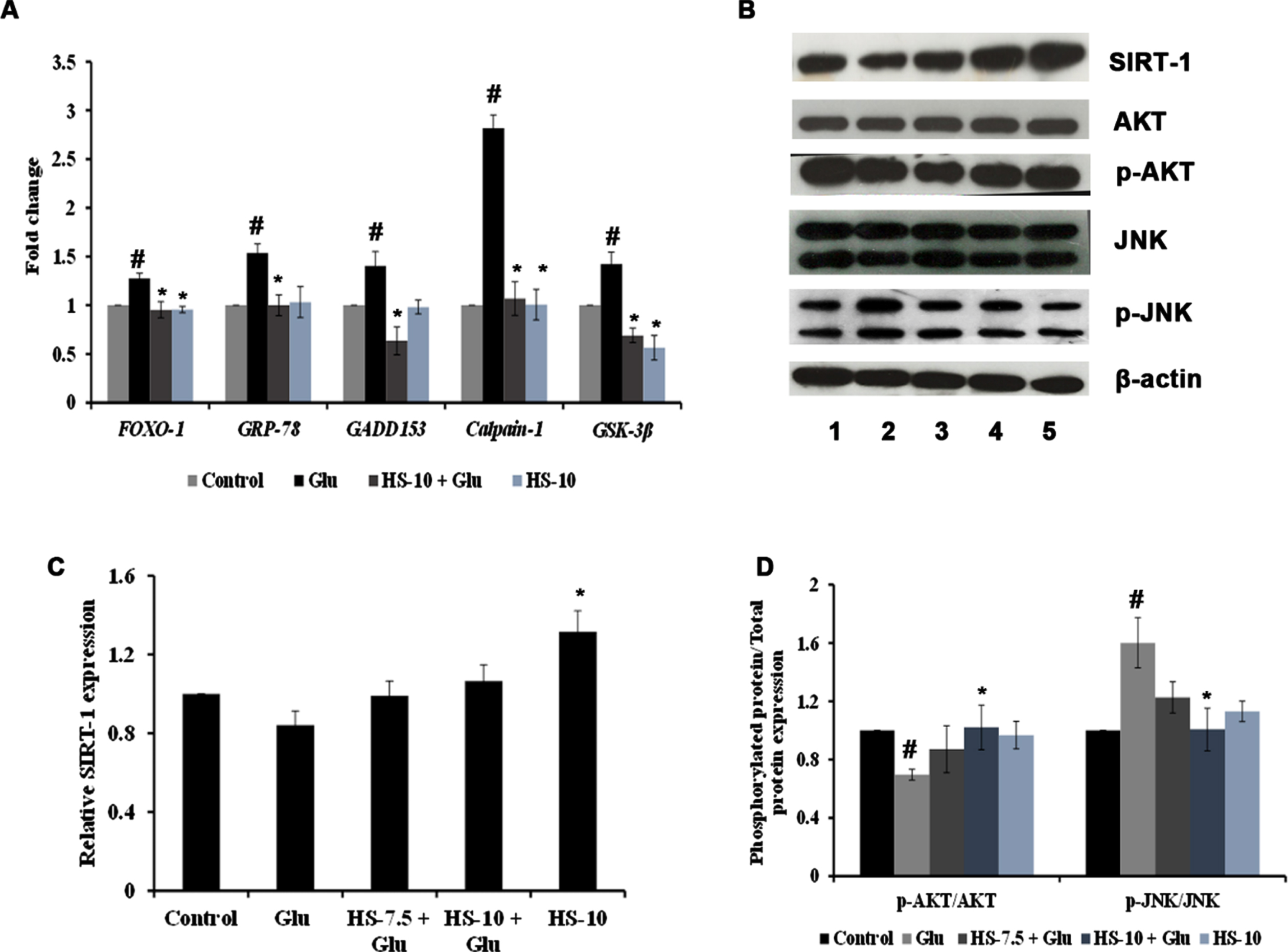
(A) Transcriptional regulation of genes involved in stress response by HS extract. (B) Representative blots showing the regulation of protein expression involved in cell signaling by HS extract (Lane: 1- Control; 2- High glucose; 3- HS-7.5 (pre-treatment 2 h) + high glucose; 4- HS-10 (pre-treatment 2 h) + high glucose; 5- HS-10) (C) Quantification of relative expression of SIRT-1 (D) Ratio of phosphorylated protein to total protein expression (p-AKT/AKT and P-JNK/JNK) (Significance at
In addition to

Molecular docking and mode of interaction of compounds (top 3 hits) from HS extract against FOXO-1 (A) Anisocoumarin H (B) Pelargonidin 3-rhamnoside 5-glucoside (C) allo-Aromadendrene (D) AS1842856 (Green indicates hydrogen bond, purple represents alkyl bond, pink represents
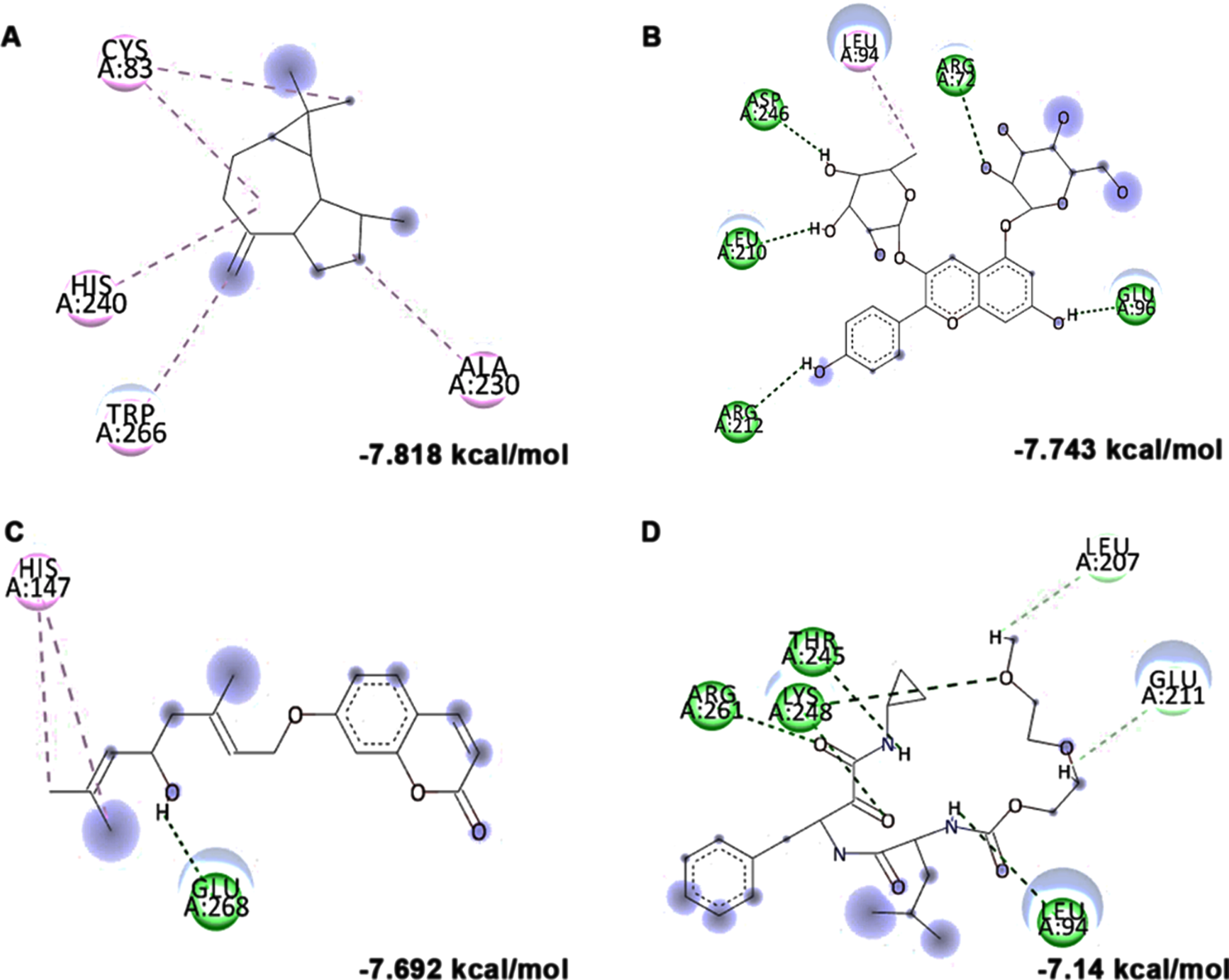
Molecular docking and mode of interaction of compounds (top 3 hits) from HS extract against Calpain-1 (A) allo-Aromadendrene (B) Pelargonidin 3-rhamnoside 5-glucoside (C) N-Feruloyltyramine (D) SNJ-1945 (Green indicates hydrogen bond, purple represents alkyl bond, pink represent a
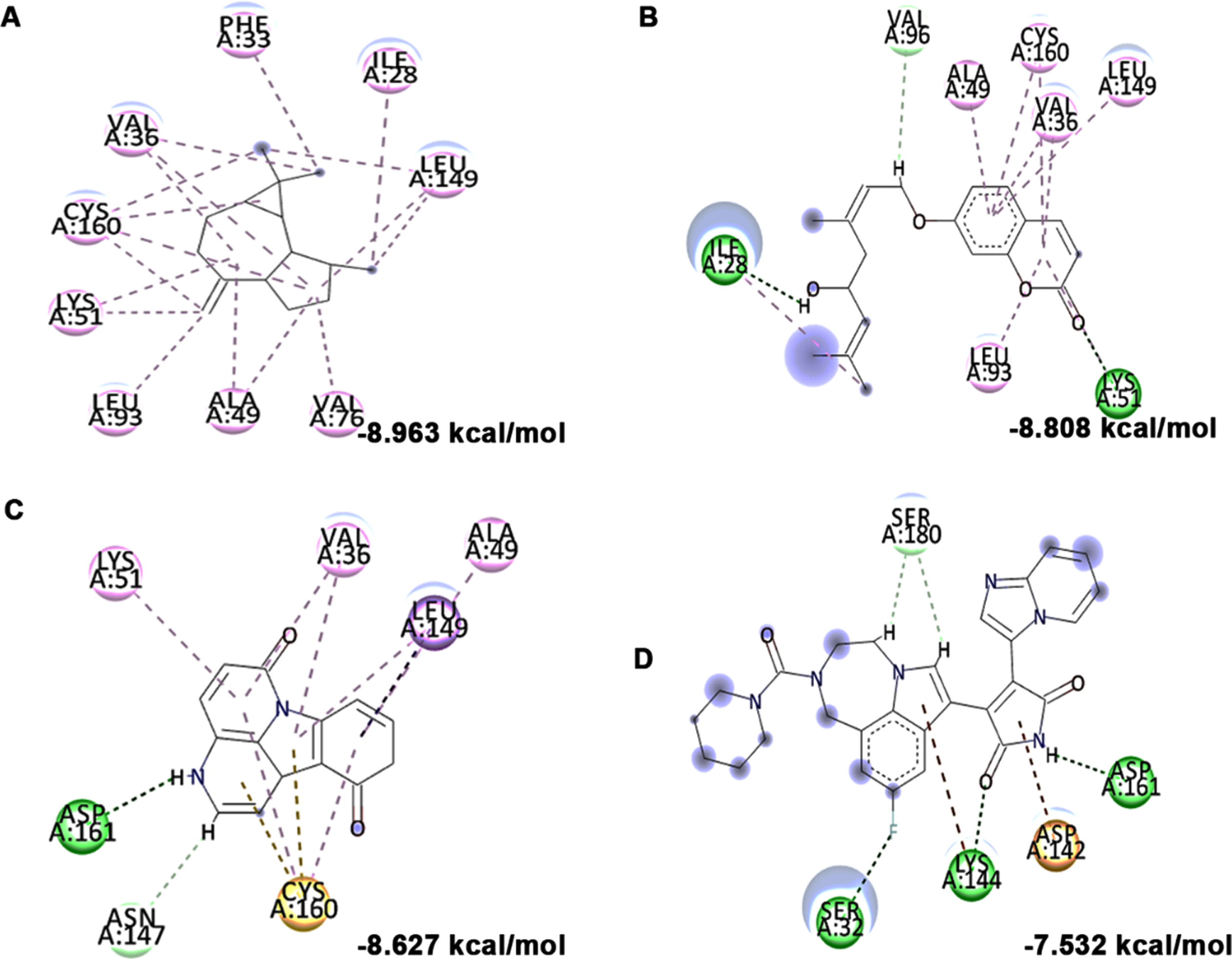
Molecular docking and mode of interaction of compounds (top 3 hits) from HS extract against GSK-3β (A) allo-Aromadendrene (B) Anisocoumarin H (C) 11-Hydroxycanthin-6-one (D) LY2090314 (Green indicates hydrogen bond, purple represents alkyl bond, pink represent a
High glucose-induced JNK activation causes abnormality in glucose metabolism and lipid accumulation leading to perturbations in lipid raft and the transcriptional regulation of candidate genes (LXR-α, LXR-β, Seladin-1, and ABCA-1) involved in maintaining homeostasis has been studied. Treatment with high glucose significantly downregulated both LXR-α and β (0.8±0.05 and 0.53±0.06 fold) and their downstream targets Seladin-1 (0.58±0.04) and ABCA-1 (0.66±0.06). However, a significant (
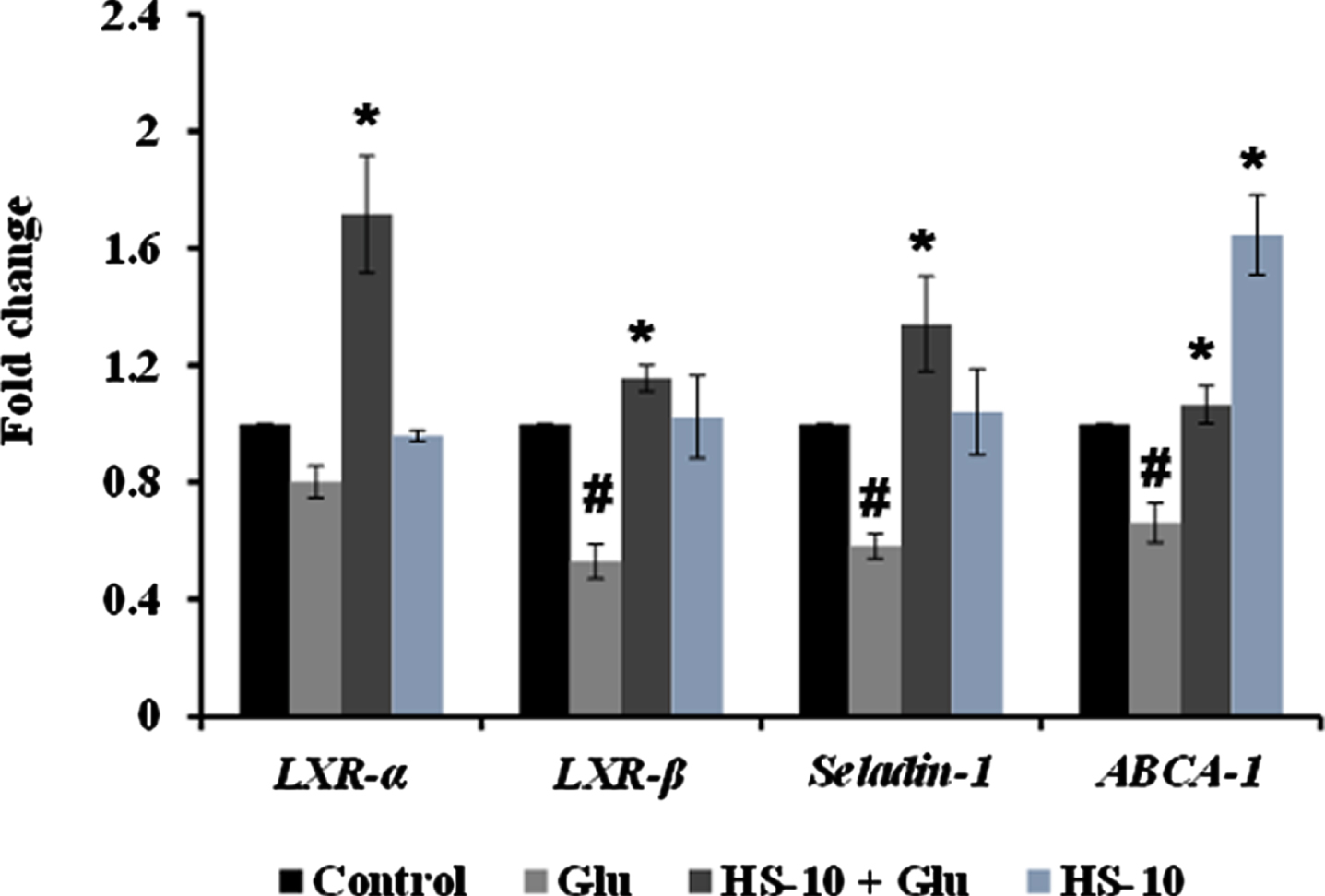
Transcriptional regulation of glucose and lipid metabolism associated genes (

Molecular docking and mode of interaction of compounds (top 3 hits) from HS extract against LXR-α (A) allo-Aromadendrene (B) 11-Hydroxycanthin-6-one (C) Sinapoyl aldehyde (D) GW3965 (Green indicates hydrogen bond, purple represents alkyl bond, pink represent
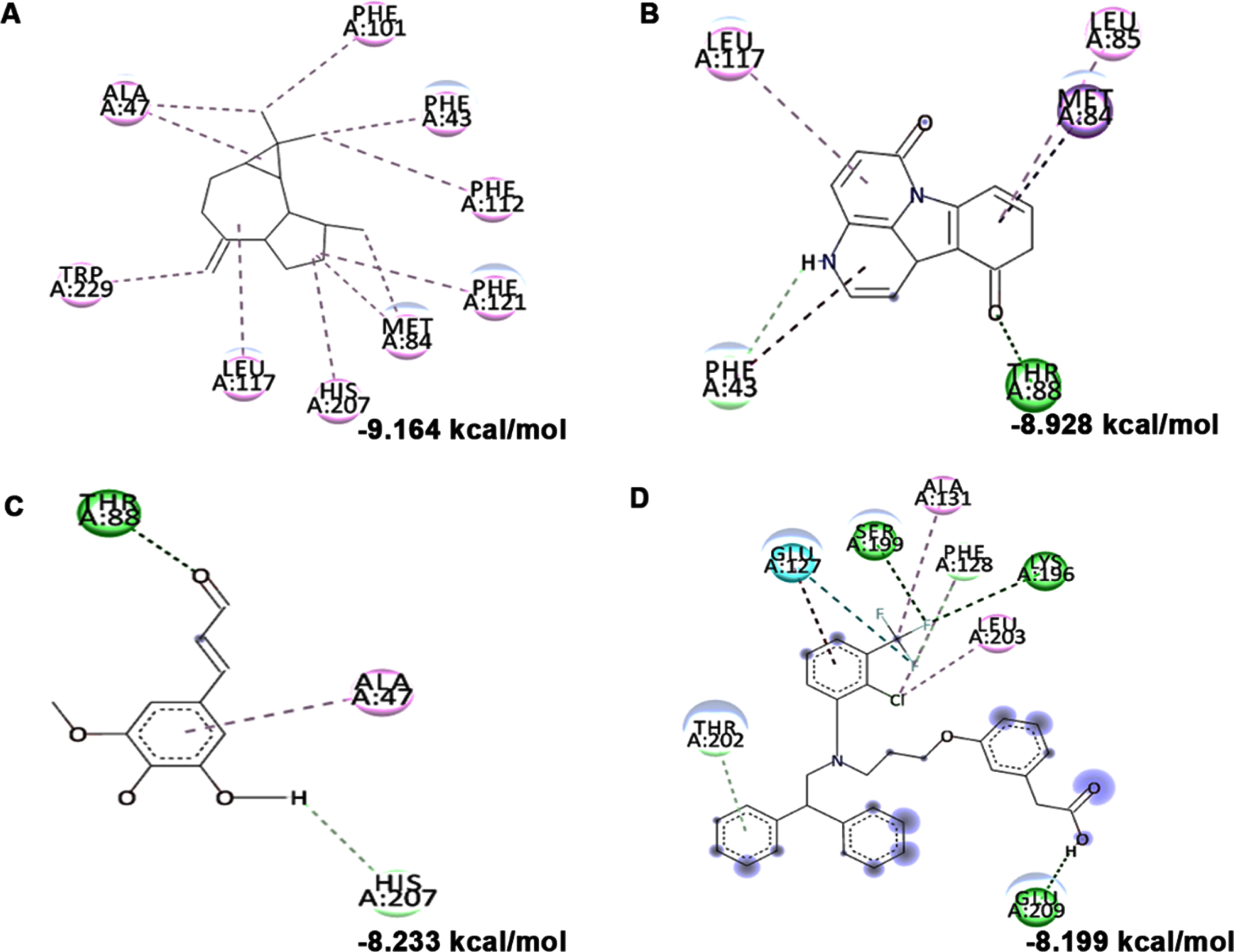
Molecular docking and mode of interaction of compounds (top 3 hits) from HS extract against LXR-β (A) allo-Aromadendrene (B) 11-Hydroxycanthin-6-one (C) Sinapoyl aldehyde (D) GW3965 (Green indicates hydrogen bond, purple represents alkyl bond, pink represents
The effect of HS extract on high glucose-induced cell death was analyzed by protein expression studies (cleaved Caspase-3, P53). The Neuro-2a cells on treatment with high glucose showed a significant (

(A) Representative blots showing the regulation of protein expression involved in apoptosis by HS extract (Lane: 1- Control; 2- High glucose; 3- HS-7.5 (pre-treatment 2 h) + high glucose; 4- HS-10 (pre-treatment 2 h) + high glucose; 5- HS-10) (B) Quantification of relative expression of P53 and Caspase-3 (Significance at
Poor absorption, distribution, metabolism and excretion (ADME) properties of a molecule have a severe impact on the drug discovery pipeline. Therefore, it is important for a small molecule to meet the ADME requirements in order to be a successful drug. Among the 26 compounds identified from HS extract, 6 compounds, which showed activity through in silico studies were taken for ADMET predictions. A molecules bioavailability is determined by how it is absorbed and how it is processed by the liver. Based on Lipinski’s rule of five (RO5), the results were analyzed for each phytocompound (Table 3). Out of 6 compounds, Epigallocatechin 3-O-(3-O-methyl)gallate showed ‘n’ violation value of 2, while all the other compounds fit within the acceptable limits. Further, toxicity predictions (Table 4) indicate that all the analyzed compounds are inactive to carcinogenicity, hepatotoxicity, and cytotoxicity while Epigallocatechin 3-O-(3-O-methyl)gallate and N-feruloyltyramine showed immunotoxicity and 11-Hydroxycanthin-6-one showed mutagenicity.
Prediction of drug-likeness of selected compounds from HS by Lipinski’s rule of five
Prediction of drug-likeness of selected compounds from HS by Lipinski’s rule of five
Toxicity prediction of phytochemicals from HS
Hyperglycemia associated changes and the protective effect of HS extract were studied in the model system C. elegans (both wild type (N2) and transgenic strain expressing Aβ (CL2006)). In the current study, the HS extract dosage of 30, 40
To check the neuroprotective efficacy CL2006 strain was used and HS extract (30, 40

HS extract extends lifespan in wild type nematodes against high glucose-induced toxicity (A) Glucose treatment in a dose-dependent manner reduced the average lifespan of N2 nematodes (B) Plots representing a reduction in life span in N2 nematodes upon treatment with glucose (100–600 mM) (C) HS extract increased the average lifespan of N2 worms against high glucose treatment (500 mM) (D) Plots showing lifespan extension of N2 worms by HS against high glucose (significance level at
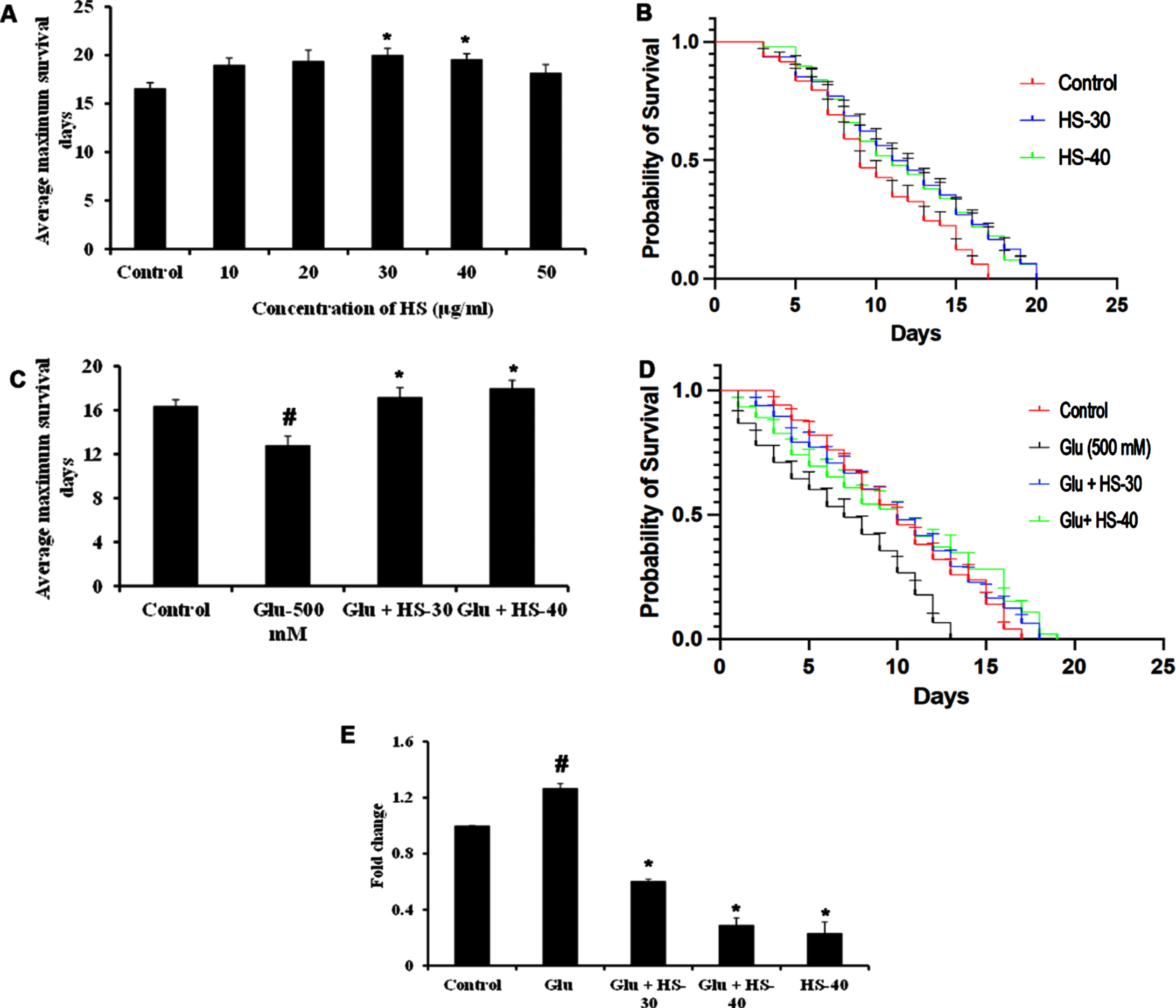
HS extract extends lifespan in CL2006 transgenic strain against high glucose-induced toxicity (A) HS treatment in a dose-dependent manner increased the average lifespan of CL2006 strain (B) Plots showing life span extension by HS extract (30, 40
Diabetes mellitus has been constantly reported to activate or suppress several cellular pathways in the brain and is associated with cognitive dysfunction and neurodegeneration [50]. The current study is aimed to investigate the efficacy of ethanolic extract of H. sabdariffa calyxes, a plant with both reported anti-diabetic and neuroprotective potential against high glucose-induced neurotoxicity. Hyperglycemia-induced memory impairment has been associated with a decline in cholinergic functions and increases acetylcholinesterase enzyme (AChE) activity, cleaving the neurotransmitter acetylcholine and inhibiting neurotransmission [51]. Our study shows that HS extract could significantly inhibit the cholinesterase enzyme dose-dependent, indicating the neuroprotective potential.
Pioneering studies have reported high glucose-mediated neurotoxicity in several neuronal cells [52]. Concomitantly, glucose exhibited neurotoxicity in Neuro-2a cells in a dose-dependent manner, with 175 mM showing a 50% reduction in viability at 24 h. The incidence of cell death was associated with increased ROS generation, induction of oxidative stress and inhibition of Nrf-2 expression. The results align with the previous reports stating that high glucose can evoke oxidative stress while causing alterations in the stress response mechanisms [53]. The Nrf-2 is one of the critical regulators maintaining redox homeostasis and is involved in the ROS clearance mechanisms by activating several genes involved in antioxidant defense mechanisms [54, 55]. Although it is expected to suppress downstream effectors (NQO-1 and HO-1) of Nrf-2, gene expression studies showed that upon high glucose treatment, a significant increase in the expression of both the genes. Previous studies have reported on the increase in expression of HO-1 upon high glucose treatment in diabetic mice retina and astrocytes through NADPH oxidase (Nox)/ NF-κB and AP-1 mediated mechanism resulting in apoptosis [56, 57] indicating NQO-1 and HO-1 can also be activated through Nrf-2-independent mechanism upon high glucose treatment. Further, increased generation of ROS under high glucose conditions caused a loss in
Sirtuin-1 (SIRT-1) is involved in several cellular signaling, including ageing, stress response, and apoptosis. A noticeable reduction in SIRT-1 expression has been observed in cellular and experimental animals in response to diabetic and neurodegenerative conditions, implicating the protein’s importance [62, 63]. The Forkhead family of transcription factors, especially FOXO-1, is one of the downstream targets of SIRT-1, and the SIRT-1/FOXO-1 axis is reported to protect against oxidative stress. Previous studies have reported both the activation and suppression of FOXO-1 under high glucose-induced oxidative stress [63–65]. Although contradicting results have been observed under hyperglycemic conditions, it should be noted that short-term activation of FOXO-1 results in the increased expression of antioxidant defense enzymes to cope with the cellular stress, while long-term activation leads to cellular apoptotic pathways [66, 67]. In the current study, HS extract treatment increased the expression of SIRT-1 and downregulated FOXO-1 expression and protected Neuro-2a cells from high glucose-induced toxicity.
Evidence suggests that endoplasmic reticulum stress (ER stress) followed by unfolded protein response (UPR) occurs concomitantly with oxidative stress conditions under chronic hyperglycemia [68]. The primary regulator of the UPR mechanism, Glucose-regulated protein 78 (GRP78), is upregulated in response to high glucose and evokes the expression of a series of transcription factors to cope with the cellular changes. The failure of this adaptive mechanism would result in the activation of the cell death pathway through the upregulation of C/EBP homologous protein/ growth arrest- and DNA damage-inducible gene 153 (CHOP/GADD153) [69]. The downregulation of Grp78 and CHOP has been reported to inhibit ER stress-induced cell death in diabetic rats and neurodegenerative conditions [40, 70]. In agreement with the previous reports, high glucose treatment induced the expression of Grp-78 and CHOP and increased Calpain-1 and GSK-3β expression, indicating ER stress. Calpain-1 activation has been associated with hyperglycemia-mediated apoptosis in various cellular models [71, 72]. In addition, calpain-1 activates GSK-3β and further contributes to insulin resistance and tau phosphorylation under diabetic and Alzheimer’s conditions, respectively [73, 74]. GSK-3β is also regulated by PI3K/AKT-mediated phosphorylation. Overactivation of GSK-3β under disease conditions occurs with repression of AKT phosphorylation [75]. Downregulation of the AKT signaling pathway has been observed under hyperglycemic conditions in cellular models [76, 77]. In the current study, it can also be observed that there is a significant reduction in p-AKT/AKT under high glucose treatment, which could be due to the increase in ROS levels corroborating with the earlier reports and pre-treatment with HS extract reversed the effect to offer neuroprotection.
The activation of the JNK signaling pathway also follows the ER stress in cells. It is associated with cell death, and inhibition of JNK phosphorylation has been reported as a mechanism to protect cells against high glucose-induced toxicity [78, 79]. Further, glucose-induced JNK activation causes abnormality in glucose metabolism and lipid accumulation through the repression of Liver X receptors (LXRs). The LXRs belong to the nuclear receptor superfamily and regulate the activation of several transcription factors involved in cholesterol and lipid metabolism [80]. In addition, LXRs activation was reported to regulate the abundance of lipid rafts within the plasma membrane enriched with cholesterol and several cell signaling proteins [81, 82]. High glucose-mediated hampering of cholesterol biosynthesis has been found to disrupt lipid raft stability in INS-1 β-cells causing diabetic complications and inducing β-secretase-mediated Aβ production through the reorganization of lipid raft coupled with LXR downregulation in SK-N-MC cells, causing neurodegeneration [83–85]. Experimental evidence shows an inverse correlation between JNK phosphorylation, LXR activation, and LXR agonist TO901317 to suppress ROS-mediated JNK phosphorylation and activate the AKT pathway in diabetic mice and SK-N-MC cells treated with high glucose [85, 86]. Further, the downstream targets of LXRs, including ATP-binding cassette transporter A1 (ABCA-1), Seladin-1 that are involved in cholesterol export and maintaining lipid raft integrity, were also downregulated under hyperglycemic conditions causing perturbations in cholesterol homeostasis and lipid raft integrity [87, 88]. Akt can also activate Seladin-1 under stress stimuli and exhibits pro-survival effects to offer neuroprotection [89, 90]. In the current study, high glucose treatment induced JNK phosphorylation followed by repression of LXRs and the downstream targets Seladin-1, ABCA-1 to disrupt cholesterol homeostasis. However, HS pre-treatment significantly reversed the effect and protected the cells from neurodegeneration.
Further, JNK activation can also induce the expression of the tumor suppressor protein to induce apoptosis against high glucose conditions [91, 92]. In addition to JNK, ER-stress associated GSK-3β activation also induces the expression of p53 and caspase-3 to promote apoptosis [93–95]. Corroborating the previous reports, high glucose treatment in Neuro-2a cells induced apoptosis through the upregulation of p53 and cleaved caspase-3. However, HS pre-treatment protected the cells by significantly downregulating the expression of apoptotic proteins and offered neuroprotection.
In addition to the
LC-MS/MS analysis of the ethanol extract of HS revealed several bioactive compounds, including Hibiscus acid, Hibiscus acid hydroxyethylester, Epigallocatechin 3-O-(3-O-methylgallate), 3’,4’,7-Trihydroxyisoflavan, 11-Hydroxycanthin-6-one, Pelargonidin 3-rhamnoside 5-glucoside, Glucovanillin, Anisocoumarin H, Isobetanin, allo-Aromadendrene, 3-dehydroquinate and so on. Previous studies have reported on the antioxidant, anti-diabetic and neuroprotective effects of some of the phytochemicals identified from HS. In silico docking approach, which is one of the powerful tools in drug design, was employed with the identified compounds against potential protein targets to substantiate the obtained results. Herein, blind docking was performed to identify potential binding sites through several trials and energy calculations by searching the entire surface of the protein target. Pioneering studies have indicated that apart from the primary binding sites, there occurs biologically relevant secondary binding sites and the addition of small molecules to such sites could modulate the function of the protein. In this regard, in our current study we have employed blind docking, which is considered to be unbiased as it scans the entire structure of the protein for finding out the putative binding site of ligand [99–101]. Among the identified compounds, allo-Aromadendrene, 11-Hydroxycanthin-6-one, Sinapoyl aldehyde, Pelargonidin 3-rhamnoside 5-glucoside, Anisocoumarin H and N-Feruloyltyramine have been repeatedly found to have a high binding affinity with the various target proteins through covalent hydrogen bond, alkyl bonds,
Moreover, Pelargonidin has been reported to ameliorate oxidative stress in HepG2 cells, improve memory deficit in the AD rat model, and ameliorate diabetic neuropathy in rats [104–106]. The red-beet betalain pigments containing isobetanin inhibit Aβ aggregation and toxicity in the AD transgenic C. elegans model [107]. Various
Conclusion
Overall, the ethanol extract of the red calyxes of HS exhibits a neuroprotective effect against high glucose-induced through regulating genes and proteins involved in stress response and cellular signaling. Further, high glucose-mediated reduction in lifespan was reversed through the inhibition of Aβ expression. Our results propose that HS with its active constituents, including allo-Aromadendrene, Anisocoumarin H, and N-Feruloyltyramine can be considered promising therapeutic agents to treat hyperglycemia associated neurodegenerative diseases. As, HS has been already reported to be safe to consume in human, further studies on individual compounds identified, as well as the formulation of extract and testing in higher model system or the elderly individuals with AD might prove beneficial in the treatment/management of neurodegenerative diseases.
Footnotes
Acknowledgments
DSM wishes to thank the Second Century Fund (C2F) for Postdoctoral Fellowship, Chulalongkorn University, Thailand, for the support. MIP and JMB wish to thank the Ratchadaphiseksomphot Endowment Fund for Postdoctoral Fellowship, Chulalongkorn University.
Funding
The present work was supported by Natural Products for Neuroprotection and Anti-ageing Research Unit.
Conflicts of interest
The authors declare no conflict of interest.
