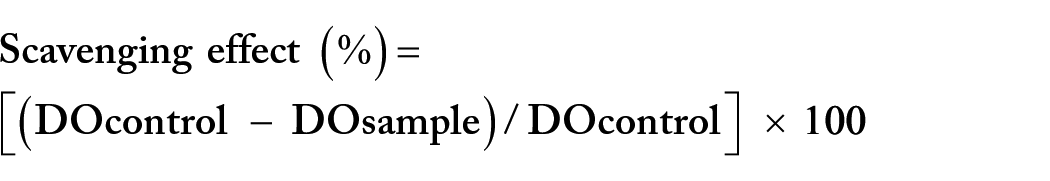Abstract
Oxidative stress and hyperglycemia are major disorders involved in the occurrence and severity not only of chronic non-communicable diseases but also of infectious pathologies. This study aimed to evaluate the in vitro antioxidant and antihyperglycemic properties of EcXaPu, EcXa, and EcPu. The antioxidant properties were evaluated using 3 mechanisms: radical scavenging; reducing property, and metal chelating. Finally, the antihyperglycemic properties were evaluated by 2 mechanisms: glucose adsorption and cellular glucose capture. The different formulations showed their ability to scavenge DPPH, ABTS, and NO radicals with SC50 ranging from 2.75 to 3.51 mg/ml, from 2.6 to 2.76 mg/ml, and from 2.59 to 3.3 mg/ml, respectively. All the formulations also reduced MoO42+ and Fe3+ and chelated Cu2+ and Fe2+. The different formulations adsorbed the glucose with glucose adsorption rates ranging from 72.83% to 87.01%. The different formulations also stimulated cellular glucose uptake, with uptake rates ranging from 31.9% to 50.71% in yeast cells and from 21.81% to 39.45% in muscle cells. These formulations could be potential agents to prevent and/or protect against biological disorders associated with oxidative stress and hyperglycemia.
Introduction
The first cause of death in the world are the chronic non-communicable diseases, which accounting for 71% of deaths and killing an estimated 41 million people each year. 1 Many metabolic risk factors are associated with these diseases, including oxidative stress and hyperglycemia, which are also responsible for the severity of these diseases. 2 The relationship between stress and hyperglycemia has also been demonstrated, as one can cause and/or aggravate the other and vice versa. Thus, hyperglycemia through numerous pathways such as activation of protein kinase C, the polyol, and hexosamine pathway, production of advanced glycation end products, and mitochondrial oxidative phosphorylation can generate a state of oxidative stress. 3 On the other hand, these 2 disorders are also associated with the occurrence of complications of infectious diseases.4,5 Numerous studies have shown the involvement of both oxidative stress and hyperglycemia in the severity of the global pandemic SARS CoV-2 infection.6 -8 In view of the strong involvement of oxidative stress and hyperglycemia in the occurrence of both chronic non-communicable diseases and infectious diseases, the management of these 2 disorders is essential and remains a public health challenge.
Numerous studies already showed the antioxidant and antihyperglycemiant properties of many plants extract from the Cameroonian pharmacopeia.9,10 These properties are most often attributed to the presence in these plants of secondary metabolites in these plants, such as polyphenols, which are known for their multiple biological properties.11,12 Herbal medicine is becoming more and more important with the diversity of natural resources and their abundance of bioactive compounds. Always in search of much more active substances, the combination of several plant extracts has long emerged in Asia. 13 However, it is only recently that this concept has been the subject of scientific publications in Cameroon. Indeed, in most traditional systems, non-communicable diseases such as diabetes are better managed by a combination of plants that can act through different mechanisms, than by individual plants. 14 According to a systematic review conducted in 2015, over the span of that year, work based on herbal formulations was mostly carried out on diabetes and oxidative stress. 15 Numerous antihyperglycaemic and antioxidant formulations have already been identified in India, such as the Mehani formulation 16 and ADPHF6. 17 In Cameroon, a study was also conducted on a plant formulation that is both an antioxidant and antihyperglycemiant. 18 Based on this observation, we were interested in a formulation based on the bark extracts of 3 plants from the Cameroonian pharmacopeia known for their antihyperglycaemic and/or antioxidant properties: Enantia chloranta stem bark, 19 Xylopia aethiopica fruit, 20 and Piper umbellatum leaves. 21 We aimed to evaluate the antioxidant and antihyperglycemiant properties of the polyherbal formulations based on these 3 plants.
Material and Methods
Plant materials
Enantia chlorata stem bark, Xylopia aethiopica fruit, and Piper umbellatum leaves were harvested in January 2019 at Ondodo (East-Cameroon), January 2019 at Bafoussam (West-Cameroon), and February 2019 at Bazou (West-Cameroon) respectively. They were isolated, cut into small pieces and then dried in the sun till a constant weight was attained. After grounding, a powder was obtained.
Preparation of extracts
A mass of 100 g of each powder was mace red in 400 ml of mixture of water/ethanol (95%) in a ratio of 1:1 (v/v). After 48 hours of maceration, the extracts were filtered using Whatman #2 filter paper and concentrated by a rotavapor before drying in an oven at 50°C.
Preparation of herbal formulations
The polyherbal formulations were prepared as follows:
- F1 (EcXaPu): Enantia chloranta stem bark + Xylopia aethiopica fruit + Piper umbellatum leaves (1:1:1 w/w);
- F2 (EcXa): Enantia chloranta stem bark + Xylopia aethiopica fruit (1:1 w/w);
- F3 (EcPu): Enantia chloranta stem bark + Piper umbellatum leaves (1:1 w/w).
Quantitative determination of polyphenolic compounds
Determination of total polyphenol content
The polyphenol content was evaluated using the method described by Singleton and Rossi and Rossi. 22 To 30 µl of the formulation, 1 ml of Folin Ciocalteu was added. Thirty minutes after the incubation at 25°C, the absorbance was read at 750 nm using a spectrophotometer. Catechin was used as standard. The total polyphenol was expressed in microgram equivalence of gallic acid/g of the formulation.
Determination of flavonoid content
The flavonoid content was evaluated using the method described by Aiyegoro and Okoh. 23 A volume of 1 ml of formulation (1 mg/ml prepared in an ethanol solution) was added to 1 ml of aluminum chloride, 1 ml of potassium acetate, and 5.6 ml of distilled water. The mixture was incubated at 25°C for 30 minutes. The absorbance of the reaction mixture was read at 420 nm with a spectrophotometer. Quercetin was used as the standard. The flavonoid content was expressed in milligram equivalence of catechin/g of formulation.
Antioxidant properties
Scavenging radicals
DPPH assay
The DPPH scavenging properties of the formulations was evaluated by the method of Katalinić et al. 24 In total, 0.975 ml of a methanolic solution of DPPH (0.3 mM) was added to 25 µl of formulations (1; 2; 3; 4; and 5 mg/ml). The mixture was kept in the dark and incubated at 25°C for 30 minutes. The DPPH methanolic solution without the formulations was used as a control. The absorbance of the mixture was read at 517 nm.
ABTS assay
The ABTS+ scavenging properties of the formulations was evaluated by the method of Re et al. 25 The ABTS+ solution (8 mM of ABTS, 3 mM of potassium persulfate in 25 ml of distilled water) was conserved at 25°C for 16 hours in the darkness. After 1:10 dilution of the ABTS+ solution with ethanol (95%), a volume of 0.5 ml was added to 10 µl of formulations (1; 2; 3; 4; and 5 mg/ml). The mixture was incubated for 30 minutes at 25°C. The ABTS+ solution without the formulations was used as a control. The absorbance of the mixture was read at 734 nm.
NO assay
The NO scavenging properties of the formulations was evaluated by Griess’ reaction. 26 A volume of 2 ml of sodium nitroprusside (10 mM) dissolved in 0.5 ml phosphate buffer saline (0.025 M; pH 7.4) is mixed with 0.5 ml of different formulation (1; 2; 3; 4; and 5 mg/ml). The mixture was incubated at 25°C for 150 minutes. Then, 0.5 ml of incubation mixture was diluted with 0.5 ml of Griess’ reagent (1% sulphanilamide, 2% Ophosphoric acid, and 0.1% napthyl ethylenediamine dihydrochloride). The control was done without the formulations. The absorbance of the mixture was read at 546 nm.
For each antiradical assay, the scavenging percentages were calculated as follows:
The scavenging Concentration (SC50) parameter was used for the interpretation of the results. 27
Reducing property
This property was evaluated by the capacity of the formulations to reduce the MoO42− and Fe3+.
Phosphomolybdenum method (total antioxidant capacity)
Total antioxidant capacity was measured by the method of Prieto et al. 28 A volume of 0.1 ml of formulations (5; 6.25; 7.5; 8.75; and 10 mg/ml) was mixed to 1 ml of reagent work (sulfuric acid [0.6 M], sodium phosphate [28 mM], and ammonium molybdate [4 mM]). The mixture was incubated at 95°C for 90 minutes. After cooling, the absorbance was read at 695 nm. The total antioxidant capacity was expressed in mg Equivalence of Trolox/mg of the formulation.
Reducing power method
The reducing power method was evaluated by the method of Oyaizu. 29 In total 0.5 ml of the formulation was added to 0.5 ml of sodium phosphate buffer (pH 6.6; 200 mM) and 0.5 ml of potassium ferricyanide (1%). The mixture was incubated at 50°C for 20 minutes and then 0.5 ml of trichloroacetate (10%) was added. After centrifugation at 650 rpm for 10 minutes, 1 ml of the supernatant was mixed to 1 ml of distilled water and 0.2 ml of ferric chloride (0.1%). The control was done by replacing formulations with distilled water. The absorbance was read at 700 nm. The ion reducing power was expressed as percentages and calculated as follows:
Metal chelating
The metal chelating properties were assessed by the Cu2+ and Fe2+ chelating through the lipoperoxidation and hemolysis assays respectively.
Lipoperoxidation assay
The lipoperoxidation assay was evaluated by the method of Okhawa et al. 30 A volume of 150 µl of each formulations (1.25; 2.5; 3.75; and 5 mg/ml) was added to 50 µl of an emulsion of olive oil (olive oil [10], phosphate buffer [10 mM, pH 7], and tween [20%]). A volume of 50 µl of FeSO4 (40 µM in a phosphate buffer) was added to initiate lipoperoxidation. The mixture was incubated at 37°C for 16 hours in the dark. The reaction was stopped by cooling and addition 50 µl of EDTA (20 mM) and 50 µl of vitamin C (4 mM). Then, 0.5 ml of trichloroacetic acid (20%) and 0.5 ml of thiobarbituric acid (0.78%) were added and the final mixture was incubated at 95°C for 45 minutes. After cooling at 25°C and centrifugation, the absorbance was read at 532 nm. The control was done by replacing formulations with phosphate buffer. The lipoperoxidation inhibition was calculated as follows:
Hemolysis assay
The hemolysis assay was evaluated by the method of Arbos et al. 31 A volume of 0.1 ml of formulations (5; 6.25; 7.5; 8.75; and 10 mg/ml) was added to 0.5 ml of NaCl (0.9%) and 50 µl of red blood cell suspension. The mixture was incubated at 37°C for 30 minutes. After incubation, 50 µl of CuSO4 (0.1 M) was added and the mixture was reincubated at 37°C for 30 minutes. The absorbance of the supernatant was read at 540 nm. The control was done without the formulations. The hemolysis inhibition was calculated as follows:
For the 2 metal chelating assays, the Inhibition Concentration (IC50) was calculated.
Antihyperglycemiant properties
The antihyperglycemiant properties of the formulations were assessed by 3 mechanisms: glucophagic effects, cellular glucose uptake stimulating effects, and inhibition of carbohydrate digestion.
Glucophagic effects
The glucophagic effects were evaluated through the glucose adsorption assay.
Glucose adsorption assay
The glucose adsorption assay was done by the method of Ou et al. 32 A volume of 0.5 ml of formulations (5; 10; 15; and 20 mg/ml) was added to 0.5 ml of glucose solution (12.5, 25, 37.5, and 50 mM). The mixture was incubated at 37°C for 60 minutes. After centrifugation at 4000g for 20 minutes, the supernatant was collected and the glucose content was determined by the method of Trinder. 33 The glucose bound was calculated as follows:
Stimulation of cellular glucose uptake
The cellular glucose uptake stimulating effects were evaluated through glucose uptake by yeast and muscle cell assays.
Glucose uptake by yeast cells
Yeast cells suspension was prepared by the method of Cirillo. 34 A volume of 0.5 ml of formulations (2.5; 5; 7.5; and 10 mg/ml) was added to 0.5 ml of glucose solution (25 mmol/l). The mixture was incubated at 37°C for 10 minutes. Then 50 μl of yeast suspension was added to start the reaction. The mixture was vortexed, and incubated at 37°C for 60 minutes. After incubation, the mixture was centrifuged at 3000g for 5 minutes. The supernatant was collected and the glucose content was determined by the method of Trinder. 33 The control was done without the formulation. The glucose uptake was calculated as follows:
Glucose uptake by muscle cells
Glucose uptake of the formulation by muscle cells was evaluated by the method of Al-Awadi et al. 35 Muscle tissue obtained from psoas muscle of adult rats was cut into pieces of 0.25 g, and preincubated in a Krebs solution containing glucose (11.1 mM) for 5 minutes in the CO2 incubator. Three mediums were done including muscle tissue alone (Control), muscle tissue with insulin (50 mU/l), and muscle tissue with both insulin and formulation (5, 7.5, and 10 mg/ml). Each medium was incubated for 2.5 hours in a CO2 incubator. Aliquots of 2 ml were removed from each medium after 30 minutes of incubation, and glucose content was determined by the method of Trinder. 33 Glucose uptake was calculated as follows:
Statistical analysis
Statistical analysis was done by the Statistical Package for Social Science software, version 20.0 for Windows. One-way Analysis of variance between groups was done with Least Significant Difference. A Post hoc test was realized to compare values 2 by 2. A significativity level was retained at .05. The SC50 and IC50 values were Oed by linear regression.
Results
Polyphenolic compounds content of formulations
The polyphenolic compounds content of formulations is shown in Table 1. Total polyphenols content was varied to 351.11 at 487.11 µg EGA/mg of Formulation, while the flavonoids content was varied to 150.57 at 280.86 µg EC/mg of Formulation. F1 contains the most total polyphenols and flavonoids, while F2 contains the least of both.
Phytochemicals content of formulations.

Abbreviations: EC, equivalence of Catechin; EGA, equivalence of Gallic acid; F, formulation.
Values assigned with different letter sin the same line are significantly different at P < .05 according to LSD.
Antioxidant properties
Scavenging radicals
The DPPH•, ABTS•, and NO• scavenging properties of different formulations are shown in Table 2 through their SC50. All formulations showed scavenging activities on the 3 radicals. The SC50 for the DPPH• radical ranges from 2.75 mg/ml (F1) to 3.51 mg/ml (F3), for the ABTS• radical from 2.6 mg/ml (F1) to 2.76 mg/ml (F3), and for the NO• radical from 2.59 mg/ml (F2) to 3.3 mg/ml (F1). F1 has the highest DPPH• and ABTS• scavenging activities and F2 has the highest NO• scavenging activity.
SC50 of different radicals.

Abbreviation: F, formulation; SC, scavenging concentrations.
Values assigned with different letters in the same line are significantly different at P < .05 according to LSD.
Reducing property
The abilities of formulations to reduce the MoO42− and Fe3+ are shown in Table 3. All formulations showed a total antioxidant capacity (TAC) in a manner proportional to their concentrations. TAC varied between 1.75 and 2.72 mg ET/mg of formulation for F1, 1.58 and 2.15 mg ET/mg of formulation for F2, and 2.5 and 3.55 mg ET/mg of formulation for F3. F3 has the highest TAC, while F2 has the lowest.
Reducing capacities of formulations.

Abbreviations: ET, equivalent trolox; F, formulation.
Values assigned with different letters in the same column are significantly different at P < .05 according to LSD.
The Fe3+ reducing capacity is expressed as a percentages of iron reducing and varied from 5.53% to 22.81%. F1 has the highest iron reducing capacity, while F3 has the lowest.
Metals chelating
The abilities of formulations to inhibit lipoperoxidation by chelating Fe2+ and hemolysis by chelating Cu2+ are shown in Table 4 through their IC50. All formulations showed inhibiting activities on lipoperoxidation and hemolysis. The IC50 varies from 2.53 mg/ml (F1) to 3.17 mg/ml (F2) for lipoperoxidation and from 2.57 mg/ml (F2) to 4.08 mg/ml (F3) for hemolysis. F1 has the highest lipoperoxidation inhibitory activity and F2 has the highest hemolysis inhibiting activity, while F2 and F3 have respectively the lowest lipoperoxidation and hemolysis inhibiting activities.
IC50 of lipoperoxidation and hemolysis.

Abbreviations: F: formulation; IC, inhibition concentrations.
Values assigned with different letters are significantly different at P < .05 according to LSD.
Antihyperghycemiant properties
Glucophagic properties
The glucophagic properties of formulations were evaluated through the glucose adsorption capacity (Figure 1). The different formulations adsorbed glucose in a dose-dependent manner. The percentages of glucose adsorption varied from 77.93% to 82.43% for F1, 76.43% to 87.01% for F2, and 73.83% to 83.62% for F3. F2 showed the best glucophagic potential.

Glucose adsorption capacities of formulations.
Glucose uptake by yeast cells
The effects of formulations on glucose uptake by yeast cells are shown in Figure 2. The different formulations stimulated glucose uptake by the yeast cells in a concentration-dependent manner. The percentages of glucose uptake for F1 ranged from 31.9%, 45.18%, 35.51%, and 50.71% for F2, and 36.69% to 50.45% for F3. F3 and F2 showed the best glucose uptake by the yeast cells.

Glucose uptake by yeast cells.
Glucose uptake by muscle cells
The effects of formulations on glucose uptake by yeast cells are shown in Table 5. Insulin alone resulted in a muscle glucose uptake of 11.89%. However, the different formulations at all concentrations significantly increased muscle glucose uptake compared to insulin alone, with uptake rates ranging from 33.28% to 39.45% for F1, from 32.26% to 35.52% for F2, and from 21.81% to 23.43% for F3. F1 was the most efficient in glucose uptake by muscle cells.
Glucose uptake by muscle cells.

Values assigned with different letters in the same column are significantly different at P < .05 according to LSD.
P < .05: significantly different in comparison to MT + insulin.
Discussion
One of the ways to combat chronic non-communicable diseases and many infectious diseases would be to act on the metabolic disorders common to these different diseases, including oxidative stress and hyperglycemia. This study aimed to evaluate the antioxidant and antihyperglycemiant properties of EcXaPu, EcXa, and EcPu.
Since polyphenols in general and flavonoids, in particular, are strongly involved in the antihyperglycemic and antioxidant properties of plants,36,37 we started this work by quantifying their contents in the 3 formulations. The different formulations had a considerable levels of total polyphenols and flavonoids. However, there were very large differences between the different formulations, which could be explained by their composition, the constituent plants, or even the proportions of plant extracts used. Subsequently, we evaluated the antioxidant properties of the formulations by 3 mechanisms: free radical scavenging, reducing, and metal chelating. The radicals DPPH•, ABTS•, and NO• were trapped by the formulations. These results can be due to the flavonoids content in our formulations. The redox potential of the OH groups of flavonoids gives them the ability to transfer a proton and/or an electron, thus trapping the free radicals. The antiradical strength of our formulations corroborates with the studies of Deepak and Anurekha and Anurekha, 38 who had already shown that the combination of 4 extracts of plants had antiradical activities against DPPH. Kabilan et al 39 also showed the same effects with their polyherbal formulation, while Chanthasri et al 40 showed the antiradical activities against DPPH and ABTS of 20 polyherbal formulations. Prety and Surech 41 also showed that HP-4, a formulation made from 4 medicinal plants, had antiradical activities against NO•. This ability to transfer electrons was confirmed in the second mechanism, which consisted of evaluating reducing power through the ability of the formulations to reduce the MoO42− and Fe3+ ions. The various formulations resulted in a reduction of these 2 ions. These results can still be attributed to the flavonoids content of the formulations. Khan et al 42 have already demonstrated the capacity of flavonoids to reduce MoO42−. On the other hand, numerous formulations of medicinal plants have already proved their reducing properties, as in Gupta et al 43 and Kajaria et al 44 which exhibited an iron reducing power. All formulations also chelated Fe2+ and Cu2+, thus inhibiting lipoperoxidation and hemolysis, respectively. These results would be due to the ability of the phenolic compounds contained in these formulations to chelate iron, inhibiting the formation of the ferryl-perferryl complex, initiating the lipoperoxidation or the blocking of the EOR, thus preventing the oxidative action of the membrane lipids. 45 Indeed, flavonoids are renowned for their ability to chelate Fe2+ and Cu2+, thus preventing lipid peroxidation. 46 This ability to protect from lipoperoxidation could account for the protection of the erythrocyte membrane, which also consists essentially of lipids. In the same sense, the N-Miracle formulation has already shown its ability to inhibit lipid oxidation due to its content in flavonoids. 47 Shanmugasundaram et al 17 showed the ability of polyherbal formulation ADPHF6 to chelate iron ions. Chanthasri et al 40 also showed the same activity with their 20 polyherbal formulations.
In the last part of our study, we evaluated the antihyperglycemic properties of the 3 formulations. Two antihyperglycemic mechanisms were investigated: glucophagic potency and cellular glucose uptake. The different formulations showed glucophagic power, materialized by the glucose adsorption rates obtained (Figure 1). This activity is thought to be due to the formation of osidic bonds following the complexation of glucose by the hydroxyl groups of flavonoids to form glycosyl flavonoids. 48 Sapadipa et al 49 have also shown the ability of a polyherbal formulation called Mehon to adsorb glucose. All 3 formulations also stimulated peripheral glucose uptake by both yeast cells (Figure 2) and muscle cells (Table 5). This would account for the ability of the polyphenols contained in our formulations to mimic the effects of insulin or to stimulate non-insulin-dependent transporters with respect to the effects observed at the level of yeast cells, which make an insulin-independent cellular uptake. Whereas the effects observed at the muscle level would account for the ability to stimulate insulin sensitivity and GLUT 4 translocation, thus increasing muscle glucose uptake. Gaikwad et al 12 had already noted the ability of polyphenols to decrease hyperglycemia by cellular glucose uptake following an increase in insulin sensitivity. Riaz et al 50 have shown the ability of a polyherbal formulation to stimulate glucose uptake by yeast cells. While Paddy et al 51 showed the stimulation of muscle glucose uptake by a polyherbal formulation.
Conclusion
The 3 polyherbal formulations (EcXaPu, EcXa, and EcPu) showed both antioxidant and antihyperglycaemic properties and acts through different mechanisms. This demonstrates the value of combining plant extracts to achieve better effects for certain diseases.
Footnotes
Acknowledgements
The authors would like to express their thanks to the Department of Biochemistry of the University of Yaounde, for providing all the necessary lab facilities.
Author Contributions
Essola, Takuissu, and Youovop carried out the study and wrote the manuscript; Fonkoua and Mandob reviewed, Ngondi contributed to the conception and analysis of data, and Gouado assisted with and supervised the manuscript writing. All authors have read and approved the final manuscript.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.