Abstract
The contribution of the microbiota to induce gastrointestinal inflammation is hypothesized to be a key component of alpha-synuclein (aSyn) aggregation within the gastrointestinal (GI) tract in the pathological progression of Parkinson’s disease (PD). The function of the GI tract is governed by a system of neurons that form part of the enteric nervous system (ENS). The ENS hosts 100–500 million nerve cells within two thin layers lining the GI tract. The gut-brain axis (GBA) is the major communication pathway between the ENS and the central nervous system. It has become increasingly clear that the microbiota in the gut are key regulators of GBA function and help to maintain homeostasis in the immune and endocrine systems. The GBA may act as a possible etiological launching pad for the pathogenesis of age-related neurodegenerative diseases, such as PD, because of an imbalance in the gut microbiota. PD is a multi-faceted illness with multiple biological, immunological, and environmental factors contributing to its pathological progression. Interestingly, individuals with PD have an altered gut microbiota compared to healthy individuals. However, there is a lack of literature describing the relationship between microbiota composition in the gut and symptom progression in PD patients. This review article examines how the pathology and symptomology of PD may originate from dysregulated signaling in the ENS. We then discuss by targeting the imbalance within the gut microbiota such as prebiotics and probiotics, some of the prodromal symptoms might be alleviated, possibly curtailing the pathological spread of aSyn and ensuing debilitating motor symptoms.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disorder affecting approximately 2–3% of people worldwide above the age of 65. With an increase in life expectancy and an aging population, PD has been determined to be the fastest growing neurological disorder [1]. PD affects males 1.5 times more than females with most cases arising in higher-income nations [1, 2]. Classified as a multi-faceted illness, there are several biological, immunological, and environmental factors that contribute to the pathological progression of this disease. Depending on these risk factors, the etiology of PD has been classically categorized as familial/genetic or sporadic/idiopathic. In this regard, the possibility that the factors contributing to PD exist on a spectrum with familial links that result in PD diagnosis at one end and purely environmental impacts at the other. Familial PD has a strong genetic component and is caused by specific gene mutations. However, since not all genes associated with PD give rise to Parkinsonian symptoms speaks to the role of environmental factors in the provocation of the illness. Further suggesting that sporadic/idiopathic PD may have multiple unknown contributing factors, or interactive effects of genetic vulnerability and environmental factors that make up the 95% majority of confirmed PD cases [3].
Whether the etiology of PD is familial or sporadic, cardinal symptoms among PD patients include resting tremor, bradykinesia, rigidity, postural instability, and stooped posture [4]. A diagnosis of PD is normally made only once these motor symptoms become apparent in an individual. Along with the emergence of the cardinal motor symptoms, it is also at this time that around 60–80% of dopaminergic neurons have degenerated in the substantia nigra (SN) and approximately 80% of dopamine levels are depleted in the nigrostriatal terminals [5, 6]. A key pathological hallmark associated with dopaminergic cell death is the intraneuronal misfolding and aggregation of the neuronal protein, alpha-synuclein (aSyn) [7].
aSyn is a 140 amino acid (aa) polypeptide encoded by the
Initially, aSyn aggregation begins slowly as monomeric aSyn starts to misfold and clump into oligomeric structures. Protein degradation systems like the ubiquitin-proteasome system and autophagy-lysosomal pathways are present to combat the formation of aggregated aSyn [14]. Over time, the ability to degrade abnormal protein structures declines, causing increased levels of misfolded aSyn. This creates an imbalance between monomer and oligomer forms of aSyn, resulting in misfolded protein accumulation. As the aggregation continues to develop, oligomeric forms of aSyn develop into stable insoluble fibrils (Fig. 1). It is hypothesized that the fibrils may spread to neighboring cells in a prion-like manner and act as a template for monomeric aSyn proteins to misfold, propagating aSyn-associated neurodegeneration [15]. Altogether, examining the ability of pathogenic aSyn to spread to neighboring cells and induce aSyn-associated cellular death may help to uncover the trajectory of tissue-level damage and determine its relationship to the various symptom profiles associated with PD pathology.
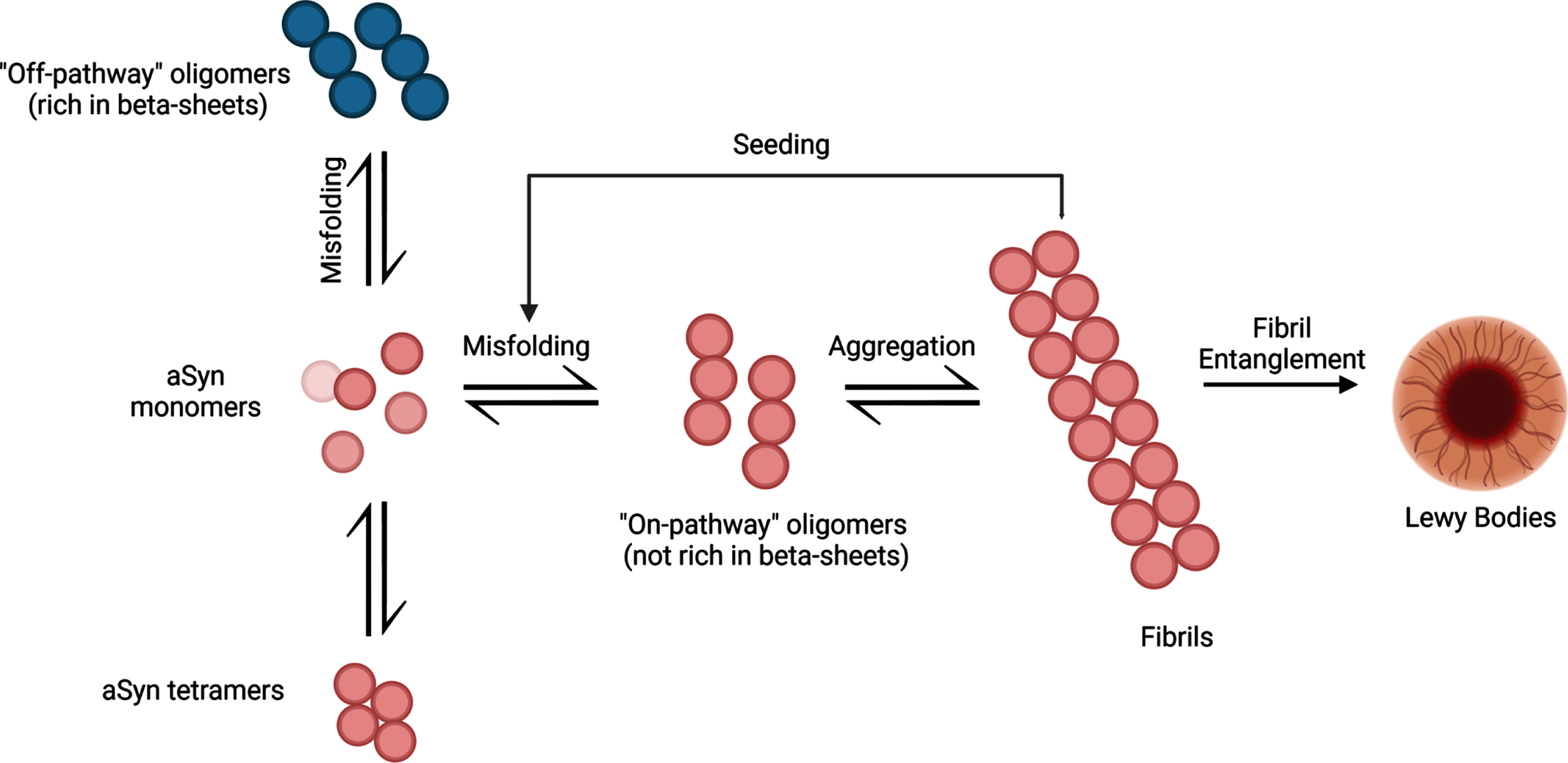
The aggregation process of aSyn protein. aSyn protein is normally found in natively unfolded monomeric structures but can also be found in α-helical tetramers when binding to lipid compounds. Aggregation of misfolded aSyn protein can either form oligomers that are rich in β-sheet formations (off-pathway) or oligomers that are not rich in β-sheet formations (on-pathway). Oligomers that are not rich in β-sheet conformations aggregate into fibrils, ultimately leading to the formation of LBs. These pathologic aSyn fibrils can act as a template for aSyn monomers and induce misfolding, creating a cycle of misfolding and aggregation of aSyn. Created with BioRender.com.
Although the diagnosis of PD is dependent on the emergence of motor symptoms, prodromal non-motor symptoms often start before motor symptoms emerge (Fig. 2) [16]. These non-motor symptoms range from cognitive and behavioral symptoms, to sleep disorders, and gastrointestinal issues, with gastrointestinal symptoms affecting 80% of PD [17]. Because gastrointestinal issues are common in PD and arise years prior to motor symptoms, viewing changes in gastrointestinal function may prove to be a biomarker in the early identification and treatment of PD [18, 19].
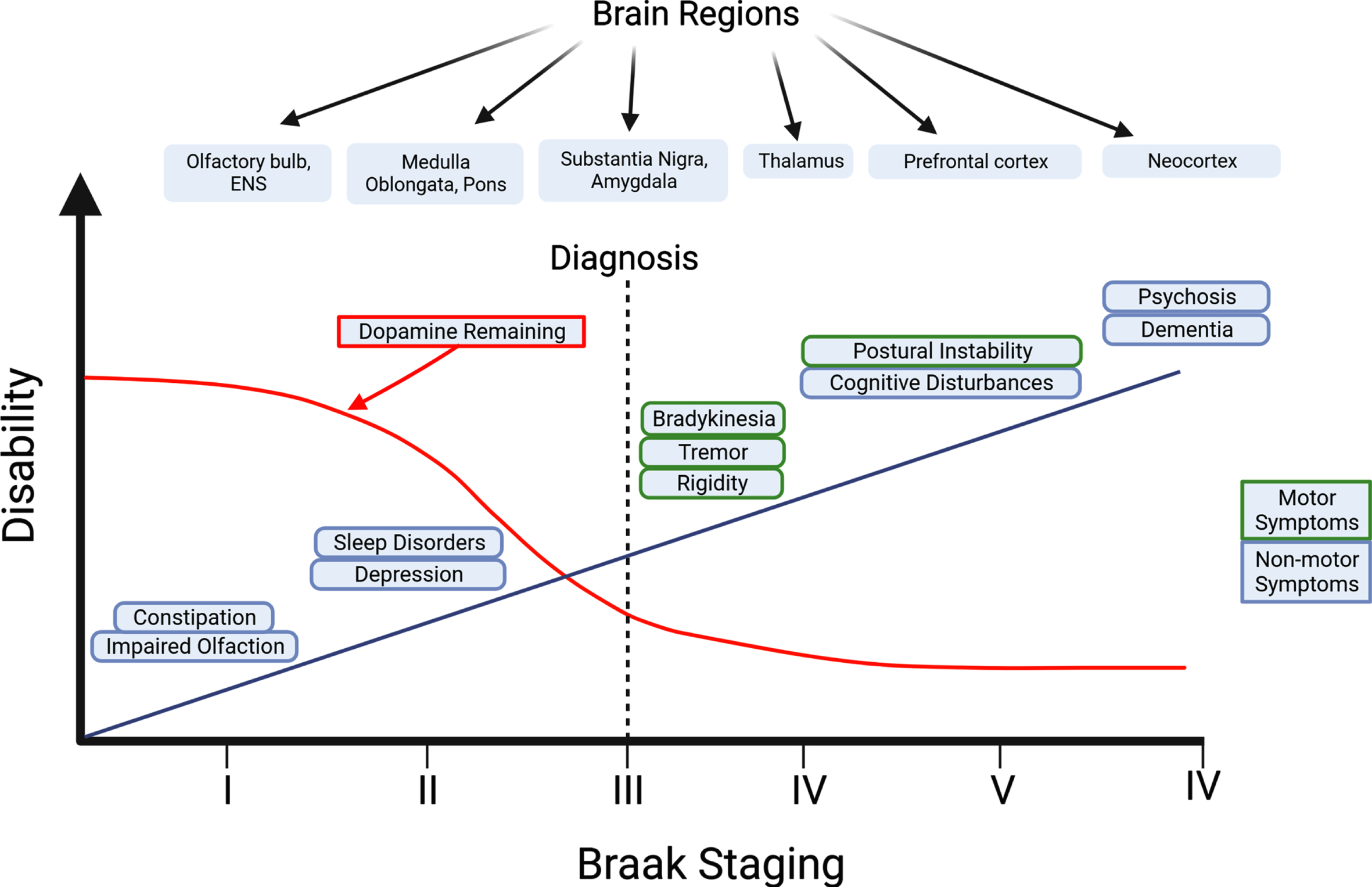
Progression of non-motor and motor symptoms in Parkinson’s disease. In the Braak Model of PD, it is hypothesized that the origin of PD originates in the PNS such as the olfactory bulbs and the ENS (Stage I). Non-motor symptoms can start to appear months or years prior to the onset of motor symptoms. Transmission of pathogenic forms of aSyn is thought to spread throughout the body in a prion-like manner, where aggregated aSyn can travel from the ENS to the brain via the vagus nerve. The diagnosis of PD is made once motor symptoms become apparent (Stage III). Long-term progression of PD causes a decrease in dopamine levels in the brain and an increase in the severity of motor and non-motor symptoms such as postural instability and cognitive disturbances, resulting in a dramatic decline in the quality of life of PD patients. Created with BioRender.com.
BRAAK STAGING
First described in 2003, the “Braak’s hypothesis” suggests PD pathology follows a six-stage sequence during disease development that may start in the peripheral nervous system (PNS) [20]. This model is further expanded by the “dual-hit” hypothesis, as it suggests that PD may originate from environmental toxins such as air pollutants and viruses that enter through the nasal and/or GI route before spreading towards the brain via the vagus nerve [21]. Alternatively, in other patients with PD, it also has been hypothesized to originate in the central nervous system (CNS) and affect the amygdala first before spreading to the SN and PNS [22]. In this “CNS-first” model, possible sites of LB pathology may originate in the amygdala or in off-regions such as the olfactory bulb [23]. In addition, there are key differences between the “PNS-first” and the “CNS-first” PD phenotypes. Primarily, in the “PNS-first” model there is a strong association in patients that experience REM sleep behavior disorders (RBD) in the prodromal phase, whereas in the “CNS-first” model, there is a lack of RBDs. Interestingly, despite nigrostriatal dopaminergic dysfunction occurring after PNS damage in the “PNS-first” model compared to dopaminergic dysfunction occurring before PNS involvement in the “CNS-first” model, motor symptom progression is faster in the “PNS-first” model [23]. While it is probable that there are different subtypes of PD as suggested by Borghammer & Van Den Berge [22], the “PNS-first” model continues to be used as the gold standard for PD progression since most PD-positive patients show this pattern of pathology [22]. When looking at the biopsies of PD patients, all patients showed signs of LB pathology in the esophagus, suggesting that LB formations could arise outside the CNS [24]. Further autopsy reports showed LB pathology in other ENS areas like the stomach, colon, and rectum [25]. To explore the consequences of LB pathology in the ENS, preformed aSyn fibrils (PFFs) were injected into the gastrointestinal tract of wild-type mice. Within days, phosphorylated-aSyn pathology was observed in both the ENS and the dorsal motor nucleus of the vagus nerve (DMV) before reaching the substantia nigra pars compacta (SNpc) [26, 27]. Taken together, these pieces of evidence help support Braak’s hypothesis of PD pathology originating in the PNS before reaching the brain via aSyn prion-like mechanisms.
THE ENTERIC NERVOUS SYSTEM’S ROLE IN PD
Referred to as the “second brain”, the enteric nervous system (ENS) contains 100–500 million neurons, making it the largest collection of neurons outside the CNS [28, 29]. These neurons are held within two thin layers lining the GI tract from the esophagus to the rectum and share similar chemical coding, structure, and function as neurons within the CNS [30]. The ENS consists of two networks of cells: the submucosal plexus, which regulates local functioning such as absorption and secretion, and the myenteric plexus, that is more involved in muscle movement including gut motility [28, 29]. Despite the ENS functioning independently from the CNS, frequent communication occurs between the two systems via the gut-brain axis (GBA). The GBA is a bidirectional communication pathway between the brain and the gut, and consists of crosstalk between the neuronal, hormonal, and immune systems [31]. The GBA is composed of multiple components including the CNS, the autonomic nervous system (ANS), the ENS, and the hypothalamic-pituitary-adrenal (HPA) axis [32]. Neuronal inputs from the ANS and the ENS innervate the vagus nerve and travel up to the lower brainstem regions. From here, information is passed on to higher order information processing neural networks (e.g., emotional, cognitive) of the brain [30]. While the GBA is a bidirectional pathway, most connections come from afferent projections from the gut and are heavily regulated by the gut microbiome [33].
The gut microbiome consists of a balance between bacteria, viruses, fungi, archaea, and many unicellular eukaryotes belonging to hundreds of different species [34, 35]. With a diverse microbiota profile, the gut microbiome influences the ENS in numerous physiological processes that include digestion, nutrient absorption, development, and metabolism [34, 36]. As microbiota are sensitive to changes in the local environment, their abundance levels and function can be altered by various factors such as vaginal birth, diet, genetics, environmental exposures, and antibiotic use [37]. Consequently, this may cause a shift in gut microbial composition towards an overabundance of pathogenic bacteria that are capable of releasing endotoxins that induce inflammation and compromise intestinal wall integrity, thereby leading to numerous diseases like IBS and inflammatory bowel disease (Fig. 3) [38–40].
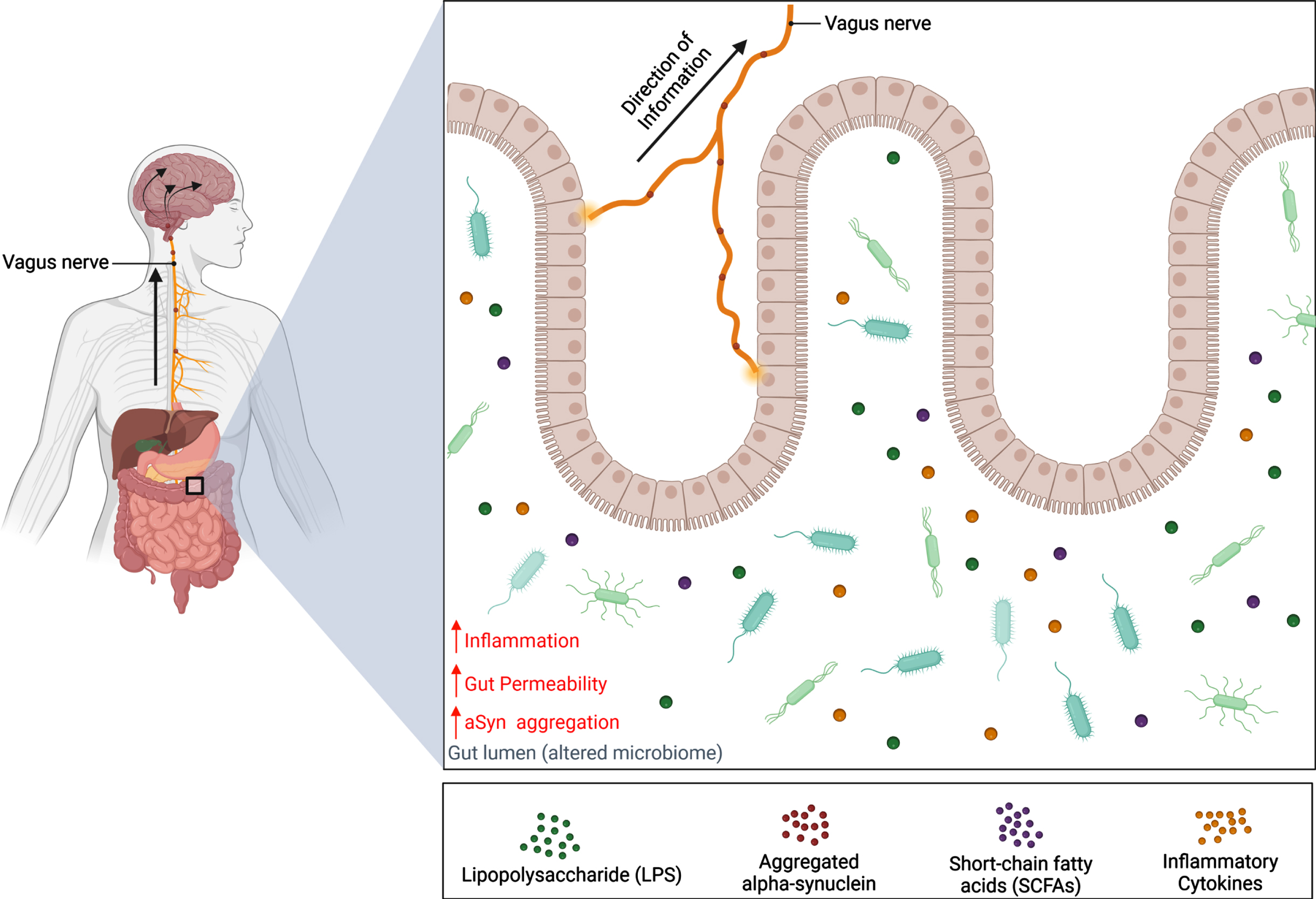
Schematic representation of the gut-brain axis in Parkinson’s disease. Alternations of the microbiota composition can be caused by various factors such as genetics and environmental risks. Dysbiosis of the microbiome can contribute to PD pathology in the gut before spreading to the brain via the vagus nerve. This results in a decrease in abundance of beneficial bacteria and a reduction of their metabolites, SCFAs, which have been shown to lower inflammation and gut permeability. Meanwhile, the abundance of harmful bacteria increases, along with the release of endotoxins like LPS. It is hypothesized that these endotoxins can increase inflammatory cytokine expression and cause the gut to become permeable for bacteria and endotoxins to enter the CNS and induce aSyn misfolding. Consequently, aSyn pathology can develop in the gut and travel up the vagus nerve where pathogenic aSyn can spread to brain regions in a prion-like manner. Created with BioRender.com.
GUT MICROBIOTA ALTERATIONS IN PD
The GI symptoms that PD patients experience may be caused by changes in the composition of the GI microbiome. In studies that investigate the composition of a patient’s microbiome, there are two techniques available: 16 S rRNA gene-based amplicon sequencing and whole-genome shotgun metagenomic sequencing. The former technique extracts the DNA from all cells in the area of interest and targets the 16 S rRNA gene portion of the bacterial genome and amplifies it using PCR for microbial composition. Whereas whole-genome shotgun sequencing takes the DNA from cells and breaks it up into small fragments and amplifies by means of PCR. Next, a DNA library is created from these DNA fragments. By using computer software, these fragments are joined together from their overlapping sequences to create a larger strand of DNA, eventually forming a whole genome sequence [41]. While both techniques identify the composition of microbial communities, inconsistent findings may arise when comparing studies that use different sequencing approaches [42]. Given that 16 S rRNA sequencing is more affordable it is more commonly used, although output using this technique limits its findings to genus-level. In contrast, WGS is known for being more expensive, yet it can provide more details pertaining to the species-specific microbial compositions [41].
Overall, several studies have suggested that distinct differences in gut microbiota exist among PD patients compared to healthy controls [43–48]. For example, in a recent meta-analysis, the microbial genera
Altered microbial composition in the gut can also modify the number of by-products that bacteria produce. Bacterial metabolites known as short-chain fatty acids (SCFAs) are created by bacteria that break down fiber and resistant starch from the diet. The main SCFAs produced are acetate, propionate, and butyrate [49]. Although SCFAs levels are reduced in PD patients, the role of SCFA in PD remains controversial [50, 51]. Numerous beneficial functions have been associated with SCFAs as they help regulate intestinal wall integrity, curb inflammation, and reduce blood-brain barrier (BBB) permeability [51, 52]. However, in an animal study conducted by Sampson et al. [50], oral administration of SCFAs to germ-free mice accelerated aSyn pathology and exacerbated inflammation mediated by activated microglia, suggesting that an extreme imbalance in SCFAs could have negative outcomes for disease.
Due to the microbiome in the gut being extremely heterogeneous, it may be possible that any alterations in the gut may affect the abundance of several bacterial populations. These changes may cascade to extreme boundaries such as changes in SCFA production. Thus, this review seeks to provide insight for how altered gut microbiota composition may influence various mechanisms that propagate PD symptomatology.
THE IMPACT OF ALTERED GUT MICROBIOTA COMPOSITION ON PD SYMPTOMOLOGY
Inflammation
One contributing factor to gut dysfunction is inflammation, which is also a major component of PD pathogenesis [53]. Recent studies suggest that systemic inflammation in both the CNS and PNS contributes to promoting PD pathology via the chronic release of pro-inflammatory cytokines and the proliferation of immune cells stemming from alterations in gut functioning [54]. As described above, the local environment that surrounds the gut is heavily influenced by the microbiota. Due to alterations in gut composition, the number of by-products that the bacteria produce and release into the gut’s local environment may affect PD pathogenesis. A meta-analysis determined that bacteria belonging to the families
Other bacteria, such as
Mood
Of the non-motor symptoms that arise in PD, depression and anxiety have been commonly observed. While PD patients report a significant decrease in the quality of life (QoL) after being diagnosed [69], it was also reported that anxiety and depression-like symptoms can be experienced before motor symptoms [70]. While the mechanisms for these mood disorders are not fully established, several factors, such as inflammation and neurotransmitter imbalance, have been shown to contribute to the onset of these non-motor symptoms [71, 72].
Anxiety is a severe type of mental illness and is the most prominent psychiatric feature of PD patients as it affects more than half of those with this disease [69]. While anxiety is characterized by chronic overactivation of the HPA axis, human studies have suggested that acute stress can increase intestinal permeability via mast cell activation [73]. Paired with an inflammatory state caused by altered gut microbiota composition, the intestinal and BBB permeability becomes severely weakened. Accordingly, LPS released from gram-negative bacteria such as
Although it is not surprising that individuals with PD report a decrease in QoL, it may be that altered gut microbiota composition could play a major role in modulating one aspect of QoL like depressive behaviors via impaired neurotransmission. While changes in monoamine activity are not the sole cause of depression, they are a critical component that underlies depressive-like symptoms. As previously mentioned, the ENS shares similar chemical signaling as the CNS. More than 90% of serotonin and other neurotransmitters such as gamma-aminobutyric acid (GABA) and dopamine are produced by gut microbes [80–82]. While serotonin that is synthesized outside the CNS cannot cross the BBB, the serotonin precursor, tryptophan, can. Considered an essential amino acid, tryptophan cannot be produced within the body and therefore must be consumed through the diet. Once in the gut, tryptophan can be delivered to the brain for serotonin production. However, there are multiple mechanisms in the GI tract that may take up the tryptophan supply and metabolize it into other metabolites such as indole metabolites and metabolites in the kynurenine pathway [83]. With an abundance of gram-negative bacteria such as
In addition to disturbances in serotonin production, altered gut microbiota composition may also impact dopamine production. As gram-negative bacteria abundance increases, beneficial bacteria, such as
Alpha-synuclein protein aggregation
Based on the “PNS-first” hypothesis, aSyn aggregation has been hypothesized to originate in the gut before traveling to the brain via prion-like transmission [15]. Studies have suggested that levels of aSyn aggregation can be influenced indirectly by the gut microbiome [94]. Kim et al. [67] induced human recombinant aSyn into forming fibrils in the presence or absence of LPS. After 7 days of fibrillization, the LPS-free aSyn fibrils or LPS-aSyn fibrils were injected into the striatum of mice. Six months after injection, it was observed that LPS-treated fibrils promoted the misfolding of endogenous aSyn into oligomeric forms. These LPS-treated aSyn aggregates had different conformations compared to the wild-type aSyn fibrils, suggesting that LPS-treated aSyn could form distinct configurations based on different environmental factors such as those within the gut and could present different clinical features or lead to different aSyn-related pathological trajectories.
Along with LPS inducing aSyn aggregation, there are certain bacteria such as curli-producing
TYPES OF MICROBIAL THERAPY IN PD
Probiotics
Probiotics composed of beneficial bacteria have been shown to be a useful treatment to combat altered gut microbiota composition and alleviate PD-like symptoms. As described above, the local environment that surrounds the gut is heavily influenced by the microbiota. In an
Prebiotics
Like probiotics, prebiotics can be used to modify the gut bacteria composition within the gut and promote health benefits. In contrast to probiotics, prebiotics are not living organisms and are referred to as “selective fermented ingredients that result in specific changes, both in the composition and/or activity of the gastrointestinal microbiota that confers benefits” [106]. Prebiotics are composed of dietary fiber that cannot be processed in the upper GI tract and must travel down to the lower GI tract where they are fermented in the colon [107]. While prebiotics are considered dietary fibers, not all dietary fibers are prebiotics. Once in the colon, prebiotics can be broken down by
Fecal microbial transplantation
Fecal microbial transplantation (FMT) refers to the transfer of healthy gut microbiota from a healthy donor in a fecal suspension into the recipient’s gut to re-establish a healthy gut microbial community [115]. Currently, FMT treatment is generally considered safe with few short adverse effects (e.g., bloating, abdominal pain, nausea, and diarrhea) and long-term adverse effects (e.g., obesity, irritable bowel syndrome, and rheumatoid arthritis) [116]. Over the decade, FMT has gained interest in treating gastrointestinal diseases to which in 2013 the Food and Drug Administration (FDA) had issued a statement requiring physicians to submit an Investigational New Drug (IND) application for the use of FMT in patients. With feedback from the medical community, the FDA eased up regulations with FMT and allowed its use to patients who do not respond to standard therapy for
CONCLUSIONS
Although the presence of motor symptoms is necessary for the diagnosis of PD, it may be equally important to investigate non-motor symptoms as early indicators of this disease. Accordingly, emerging evidence has described that within the gut, dysbiosis of the microbiota can potentially increase the risk of inflammation, gut permeability, and induce aSyn aggregation. However, an important caveat to consider in human studies is that the beneficial outcomes that were described are self-reported. Hence, more research is needed to provide direct evidence that bacterial mechanisms can contribute to PD symptomatology. This leads to future research aimed at identifying specific bacterial strains that initiate PD pathology may be a viable therapeutic approach for the treatment of this disease. Taken together, by focusing on restoring a healthy microbiome composition, in addition to combating the microbial metabolites that interact with the environment that promote a pro-inflammatory state or induce aSyn misfolding, some of the symptoms like irritable bowel syndrome and constipation would likely be improved if not alleviated. This would mark the start to the improvement of QoL for the patients which has been a long-standing goal of the healthcare industry.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
