Abstract
Family studies have linked several rare genetic variants to hereditary forms of Parkinson’s disease (PD). In addition to these monogenic forms, many PD cases are associated with genetic risk factors. Asymptomatic individuals carrying pathogenic variants linked to PD are at risk of developing the disease later in life, thereby providing a unique opportunity for the detection of the earliest pathophysiological and later clinical changes and, importantly, also of protective and compensatory features and mechanisms. However, the rarity of monogenic PD-causing variants is a major challenge of this approach. In this review, we discuss recent advances in the search for biomarkers in the prodromal/earliest phase of genetically linked PD.
Plain Language Summary
While the cause of most cases of Parkinson’s disease (PD) is still unknown, and age is considered the greatest risk factor, a combination of environmental influences and genetics are thought to affect disease risk and progression. The identification of carriers of pathogenic genetic changes, who have not yet developed motor symptoms of PD, offers the chance to closely monitor developing signs of PD. Some of these signs may be suitable biomarkers and could be used to predict early stages of the disease. In this review, we discuss recent advances in the search for biomarkers in the prodromal/earliest phase of genetically linked PD.
Keywords
INTRODUCTION
Genetic variants of the germline are inherited or arise de novo and are present from conception. There are several hereditary forms of Parkinson’s disease (PD).1,2, 1,2 When including
The pathology of PD—this largely applies to both “idiopathic” and genetic PD—is characterized in most cases by the presence of aggregated α-synuclein in the form of Lewy bodies and Lewy neurites in the brain. Current evidence suggests that the deposition and spread of pathological forms of misfolded α-synuclein drive the development and progression of the disease. 4 By the time a clinical diagnosis is confirmed, it is estimated that 40–60% of dopaminergic neurons have degenerated and synaptic function has reduced by up to 80%. 5 Therefore, neuroprotective interventions are required early on i.e. during the prodromal phase of PD. In this period, a clinical diagnosis based on fully evolved motor parkinsonism is not yet possible.6–8 This latent phase can vary from 5 to more than 20 years and is characterized by a variety of non-motor symptoms, in isolation or combined, including autonomic dysfunction, rapid eye movement sleep behavior disorder, olfaction, and depression.9–12 As indicated above, the prodromal phase is an important stage of the course of PD for investigating the pathophysiological cascade and, when neuroprotective treatment becomes available, to possibly slow or prevent the onset of motor symptoms in PD. 6 In order to identify individuals during this phase and thus provide an early diagnosis of PD, biomarkers are urgently needed. However, the identification of suitable biomarkers is challenging as the type of symptoms, prevalence, time of occurrence, and rate of progression of prodromal symptoms vary considerably between patients. 13
While the cause of most cases of PD is still unknown, and age is considered the greatest risk factor, a combination of environmental influences and genetics are thought to affect disease risk and progression. The identification of carriers of pathogenic variants or polygenic risk scores, who have not yet developed motor symptoms of PD, offers the chance to closely monitor developing signs of PD that may be suitable as biomarkers. In this review, we discuss recent advances in the search for biomarkers in the prodromal/earliest phase of genetically linked PD.
GENETICS OF PARKINSON’S DISEASE
Family studies of PD reported rare variants in more than 20 genes.
2
However, the relevance of some of these genes and variants is unclear, and replication or functional validation studies are missing. Over the past three decades, seven genes have unequivocally been linked to monogenic PD, including four autosomal dominantly inherited ones (α
In addition to the monogenic forms, many PD cases are associated with genetic risk factors. Seventy-eight independent genome-wide significant loci were identified in a large-scale multi-ancestry meta-analysis of PD including 49,049 cases, 18,785 proxy cases, and 2,458,063 controls.
17
Variants at the
COMBINING BIOMARKERS AND GENETICS IN PRODROMAL PARKINSON’S DISEASE
PD is a complex disorder comprised of various clinical phenotypes, pathological findings, and genetic predispositions. 20 Recent cohort studies have confirmed the high phenotypic variability and the numerous genetic variants associated with PD risk contribute to the heterogeneity of multiple motor and nonmotor aspects.21,22, 21,22 For example, the timescale over which PD with dementia may develop ranges from months to decades and neurodegeneration is not limited to the nigrostriatal dopaminergic denervation.23,24, 23,24 This heterogeneity in PD makes it challenging to find reliable biomarkers and intensive research efforts have been performed to detect biomarkers in cerebrospinal fluid (CSF), blood, and through imaging modalities. Within the realm of biofluidic biomarkers, α-synuclein, glial fibrillary acidic protein (GFAP), amyloid-beta, tau, p-tau, as well as inflammatory markers, mitochondrial DNA (mtDNA), and microRNAs have been discussed.25,26, 25,26 Of these, α-synuclein seed amplification assays (SAAs) have shown promise for the biochemical diagnosis of PD.27–29 GFAP could be of use mainly in tracking PD progression, the Alzheimer’s disease biomarkers amyloid-beta and (p)-tau are relevant to evaluating cognitive decline and motor function in PD.30,31, 30,31 Significantly increased levels of inflammatory markers (blood: IL-6, TNF-α, IL-1β, STNFR1, CRP, CCL2, CX3CL1, and CXCL12; CSF: IL-6, TNF-α, IL-1β, CRP and CCL2) were recently verified in a meta-analysis of PD patients and control groups confirming that neuroinflammation plays a role in PD. 32 The use of microRNAs and mtDNA as biomarkers for PD is new and promising but still in a developing phase.26,33, 26,33 Imaging modalities to test for PD include magnetic resonance imaging (MRI), dopamine transporter (DAT) single-photon emission computed tomography (SPECT), fluoro-3,4-dihydroxyphenylalanine (FDOPA) positron emission tomography (PET), and [18F]fluorodeoxyglucose (FDG) PET.25,34, 25,34
Based on the numerous studies of neuropathological findings, genetic contributions, and biomarker research, a novel biologically based classification of PD has been proposed. This classification takes the complexity and heterogeneity of the disease into account and defines disease status by the presence or absence of pathological α-synuclein in tissue or CSF, by the evidence of underlying neurodegeneration described by neuroimaging procedures, and by the occurrence of gene variants that cause or strongly predispose to PD.
14
This biological classification of PD could improve objective preclinical and clinical diagnosis, disease staging, and enable the accurate subdivision of PD according to disease mechanisms. Moreover, it could also advance research in multiple fields, such as epidemiology, biomarker discovery, and patient-specific development of disease-modifying therapies. To assess biomarkers of prodromal PD in a longitudinal and multimodal fashion, a homogeneous group of non-manifesting carriers of gene variants, which cause or strongly predispose to PD, need to be recruited. However, the rarity of monogenic PD-causing variants is a major challenge of this approach. Therefore, only a few studies combining biomarker analysis and genetic PD have been performed so far. These studies focused mainly on
α-SYNUCLEIN SEED AMPLIFICATION ASSAYS
α-synuclein-SAAs are based on the self-replicative nature of misfolded α-synuclein aggregates (seeds). In these assays, α-synuclein seeds in biological fluids are amplified by a cyclical process and detected by fluorescent dyes specific for amyloids. 37
α-synuclein-SAAs have been performed using CSF and blood samples from patients with genetic PD (Table 1). Analysis of CSF samples revealed that
α-synuclein seed amplification assays in patients with genetic Parkinson’s disease
NMC, non-manifesting carrier.
Analysis of blood samples from patients with genetic PD has only been performed for biallelic
In summary, while data suggest that α-synuclein-SAAs represent very promising biomarkers for PD diagnosis, it remains unclear whether this method will be suitable for detecting prodromal stages of genetically linked PD. Notably, in patients with rapid eye movement sleep behavior disorder, α-synuclein positivity detected by SAA in CSF was associated with an increased risk of subsequent diagnosis of PD or dementia with Lewy bodies. 42
NEUROIMAGING MARKERS
DAT-SPECT has been established as a method to detect the presence of nigrostriatal deficit not only in clinical PD but also in the early or pre-symptomatic stages of the disease and to monitor DAT loss with the progression of neurodegeneration.
43
Therefore, DAT-SPECT has also been preferentially used to study genetic PD. Overall,
Decreased DAT binding was shown in 43.7% of 49 asymptomatic G2019S carriers.
44
In contrast, in a large cross-sectional study investigating non-manifesting
In an attempt to assess whether a combination of systematic clinical testing and different imaging techniques in familial PD patients could detect subclinical signs in the preclinical and prodromal stages of PD, a retrospective evaluation was reported of a cohort of 39 participants, which included
CONCLUSIONS AND PERSPECTIVES
α-synuclein SAAs show clear potential to detect idiopathic PD and to distinguish patients from healthy controls. However, genetic PD has currently been studied only in a very limited number of patients and genetic conditions. DAT-SPECT has been established as a reliable biomarker in idiopathic and genetic PD, but the findings for prodromal PD of genetic subgroups are not yet conclusive. We are still at the beginning of our ability to understand and diagnose the early stages of the disease, mainly due to the lack of data that reflect these stages at the molecular level.
However, there are promising findings, especially in carriers of the GBA1 N409S variant, who show evidence of abnormal CSF α-synuclein aggregation prior to other detectable clinical or biomarker changes, including alterations in DAT imaging.
29
Nevertheless, further studies on more easily accessible biomarkers, such as a blood-based diagnosis are needed. Notably, increased blood-derived mtDNA damage was found not only in
For future studies, it will be important to identify genetically defined subgroups to establish at-risk cohorts (Fig. 1). Patients should be stratified according to genetic variant severity and those carrying severe mutations to be preferentially included. Longitudinal research is needed to examine the predictive value of α-synuclein SAA, mtDNA damage, and DAT-SPECT over time. In this context, repeated collections of biomaterials, starting at the prodromal disease stage, are required, and samples will have to be analyzed in a multimodal fashion, including multi-omics as well as patient-derived experimental disease models to detect new biomarkers specific for PD. In a recent approach, mass spectrometry-based proteomic phenotyping was used to identify a panel of blood biomarkers in early PD. 47 Furthermore, a combination of systems biology and data integration methods was employed to identify differences in transcriptomic and metabolic signatures between PD patient and healthy individual-derived midbrain neural precursor cells. 48 These and other approaches will provide new single biomarkers or biomarker panels that form a distinct signature of PD. Moreover, performing timely studies during the progression from health to disease might inform us about biomarkers that reflect the underlying pathological processes and present possible read-outs for target engagement.
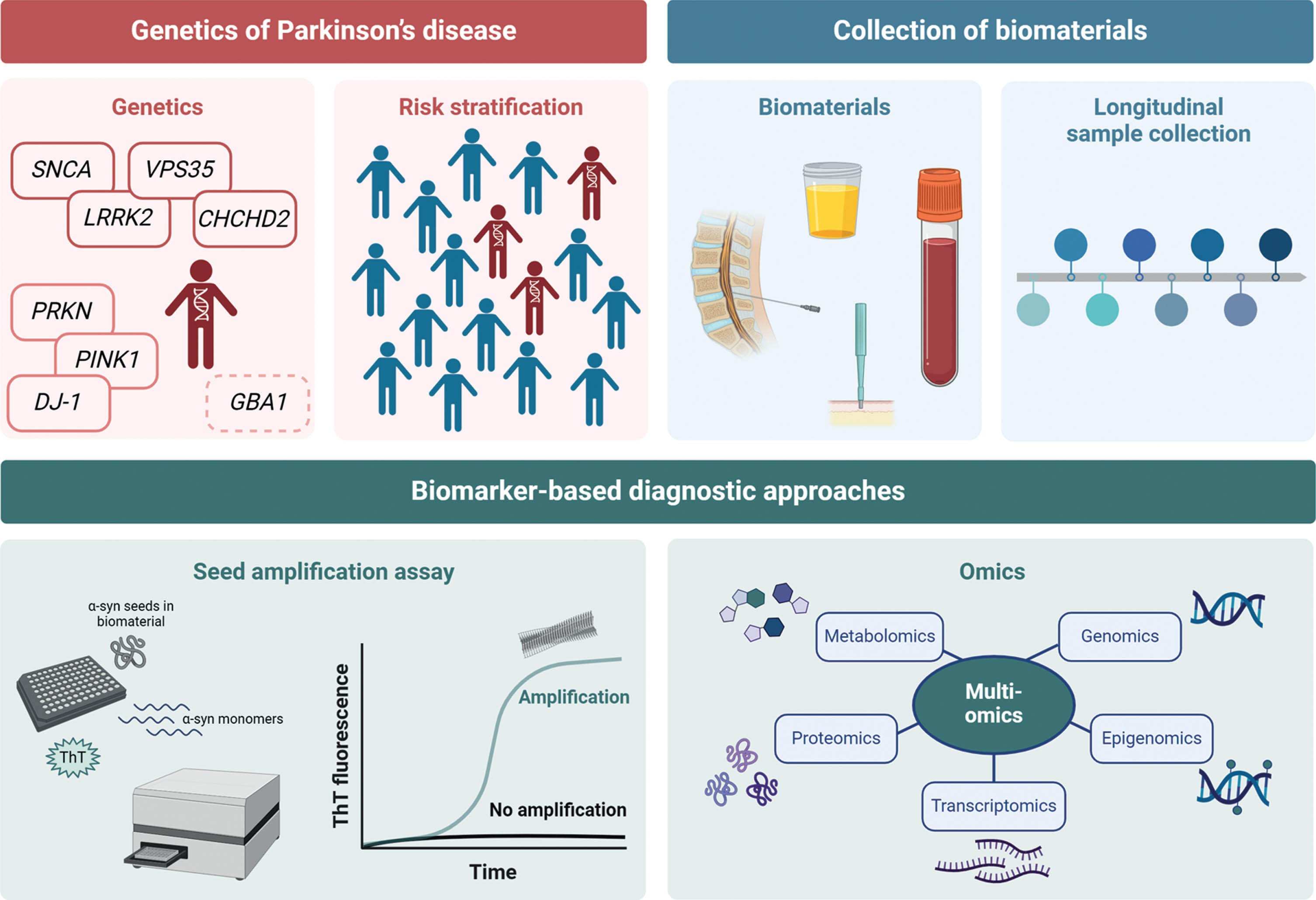
Scheme on the search for new biomarkers. (i) Stratification of risk groups of asymptomatic mutation carriers, (ii) longitudinal collection of biomaterials, (iii) verification of biomarker search by using the established α-synuclein seed amplification assays, and (iv) search for new biomarkers using a multi-omics approach. Created with BioRender.com.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
C.K. serves as a medical advisor to Centogene, Takeda, Retromer Therapeutics, and Lundbeck, and received speakers’ honoraria from Bial and Desitin and royalties from Oxford University Press.
