Abstract
The ability to identify individuals in the prodromal phase of Parkinson’s disease has improved in recent years, raising the question of whether and how those affected should be informed about the risk of future disease. Several studies investigated prognostic counselling for individuals with isolated REM sleep behavior disorder and have shown that most patients want to receive information about prognosis, but autonomy and individual preferences must be respected. However, there are still many unanswered questions about risk disclosure or early diagnosis of PD, including the impact on personal circumstances, cultural preferences and specific challenges associated with different profiles of prodromal symptoms, genetic testing or biomarker assessments. This narrative review aims to summarize the current literature on prognostic counselling and risk disclosure in PD, as well as highlight future perspectives that may emerge with the development of new biomarkers and their anticipated impact on the definition of PD.
Plain Language Summary
An important goal of Parkinson’s disease research is to diagnose the disease at an earlier stage, even before the typical motor symptoms appear, in the so-called ‘prodromal phase’. Currently, there are no treatments available that can slow down or prevent disease progression in this early phase, even though many of the early symptoms are treatable. This raises ethical questions about whether people want to know their future risk of Parkinson’s and, if so, how this information should be given. This article summarizes the current state of knowledge, but also open questions about risk disclosure in the prodromal phase of Parkinson’s. Previous studies have shown that many people with early symptoms of Parkinson’s would like to know their risk, but that the individual’s wish to know (or not to know) must first be ascertained and respected. Future studies need to find out whether very early diagnosis of Parkinson’s might have an impact on people affected, for example in terms of psychological stress or anxiety, and whether cultural background might influence attitudes to risk disclosure. Furthermore, it is expected that in the future it will be possible to make an early diagnosis of Parkinson’s using specific new techniques, e.g., by testing spinal fluid. It is of utmost importance to find out if and how test results of these new techniques should be communicated to patients, even if they do not lead to direct medical treatment.
INTRODUCTION
In recent years, converging findings from neuropathological and clinical studies indicate that the clinical diagnosis of Parkinson’s disease (PD) is preceded by years of neurodegeneration. These early years and in particular the prodromal phase of the disease have increasingly been the subject of focus, as they present an opportunity for early treatment with anticipated disease-modifying therapies. Previously defined
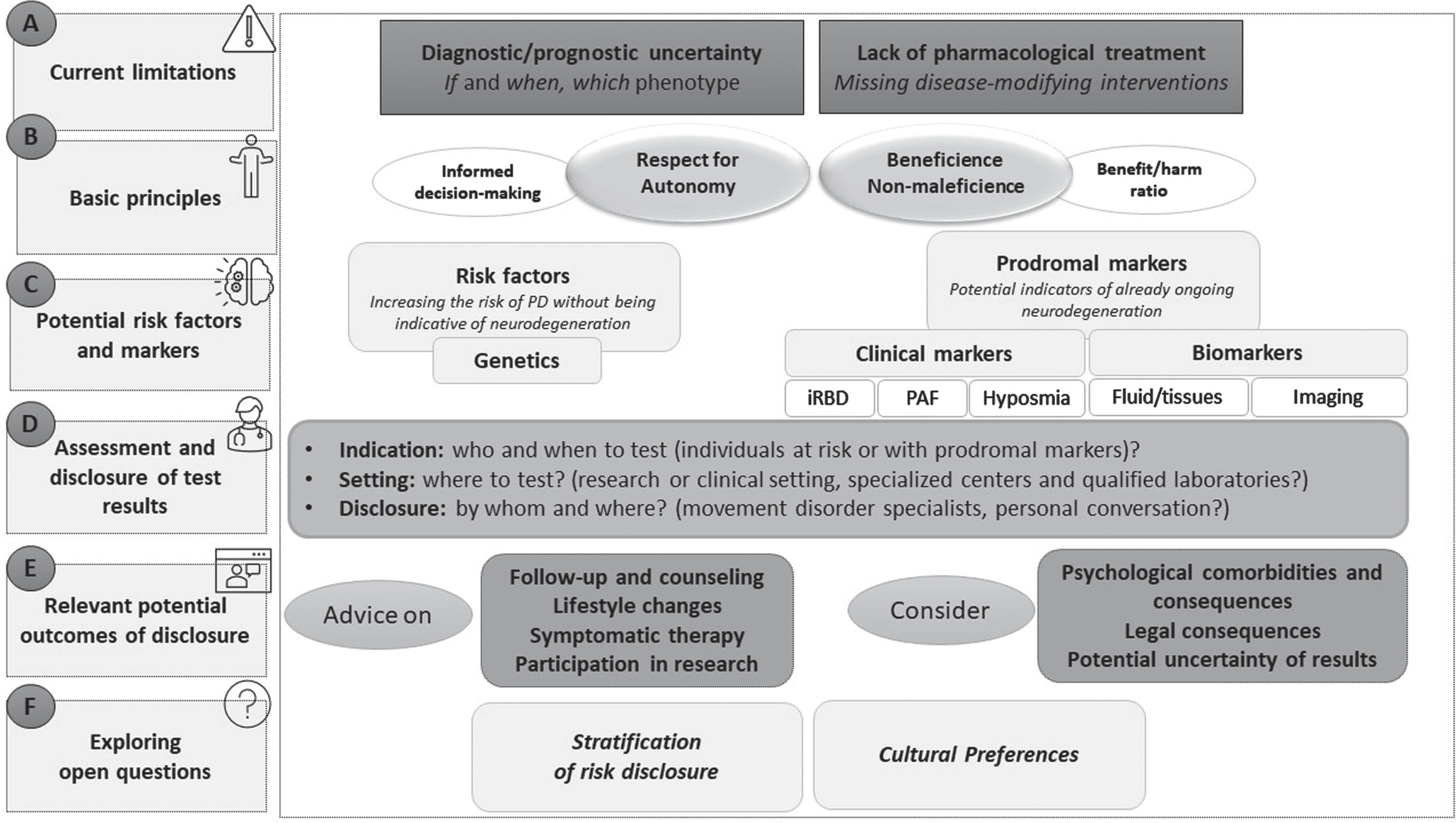
Pillars of risk disclosure and early diagnosis in Parkinson’s disease. The points mentioned in this figure were developed based on the review of the literature of Parkinson’s disease and other neurodegenerative diseases (in particular Alzheimer’s disease) and derived from empirically relevant factors.
(1) Uncertainty: although knowledge on the diagnostic value of clinical markers and biomarkers is constantly increasing, there is still prognostic uncertainty regarding
(2) Consequence of disclosure: So far early risk disclosure is not clearly advantageous from the perspective of treatment. Although non-motor symptoms occurring in the prodromal phase of PD often require symptomatic treatment, which may justify early diagnosis in some way17,18, 17,18, an evidence-based intervention that might prevent or slow down the disease in the prodromal phase does not yet exist.
However, as research on the prodromal phase of PD is crucial for further developments to counter the disease, it is of utmost importance to navigate these ethical issues and define clear guidelines for research and clinical practice. Within this review we first summarize current discussion points and results of completed studies on ethical issues around prognostic counseling in prodromal PD, with the objective to provide a framework for risk disclosure in early/prodromal PD that can be applied in both research and clinical settings. Following the rapid growth in knowledge of the last few years, this review additionally aims to address unanswered questions and future perspectives of early diagnosis of PD, that must be addressed in future studies as a basis for advanced risk disclosure guidelines.
STATE OF THE ART
Basic principles of early risk disclosure in PD
Autonomy
Previous publications on risk disclosure in prodromal PD, including experts’ and patients’ opinions, clearly emphasized that respect for patient autonomy must be a central component of prognostic counseling7,8,19–23, 7,8,19–23 (Fig. 1B). That implies that the patient’s
The respect for autonomy is challenged by unresolved issues on how to proceed with incidental information of prodromal symptoms or risk factors, including for example the diagnosis of iRBD being discovered during evaluation for a primary presentation of obstructive sleep apnea9,24,25, 9,24,25 or the finding of a genetic risk variant for PD, e.g., within direct-to-consumer genetic testing (DTC-GT). If patients seek medical advice due to burdensome symptoms or a positive family history for PD, they generally have time beforehand to inform themselves on potential outcomes of assessments and actively sought clarification, while patients who receive information in above scenarios do so without preparation. So far, there is no consensus for (prodromal) PD if incidental diagnosis should be conveyed in the same way as for primary diagnosis. The most appropriate approach in clinical practice at present is to inform patients in advance of the possibility that the planned examinations (e.g., polysomnography) may generate incidental findings.7,12,26,27, 7,12,26,27
Finally, the above-mentioned ethical guidelines to respect patient autonomy have been mainly discussed for a clinical setting, while they are not directly comparable with research settings. A particular challenge is the recruitment of individuals from the general population, where the screening process for studies on prodromal PD sometimes actively point out specific prodromal signs or risk factors, which were not previously considered relevant by the participant. In this scenario, ethicists should be involved early in the study design. 7 In general, disclosure of results and transparency regarding reasons for enrollment are recommended, if preceded by a shared decision making and patient education. 7
Beneficence and nonmaleficence
The second and third key ethical principles for prognostic counseling are the consideration of beneficence and nonmaleficence to promote patient benefit and avoid harm7–10,12,23,28,29, 7–10,12,23,28,29 (Fig. 1B).
Factors that potentially influence an individual‘s reaction to disclosure
Factors mentioned in this Table are based on the literature in the fields of cancer, Huntington’s disease, and Alzheimer’s disease.
Recommendations for risk disclosure for future Parkinson’s disease
Recommendations mentioned in this Table are based on review of the literature on neurodegenerative diseases and bad-news conversations and derived from empirically relevant factors for building a disclosure strategy.
But even if all measures are taken to consider predictable and unpredictable factors, a negative reaction is still possible. In this context, it is positive that studies in individuals with prodromal (iRBD) and clinical PD have shown that those affected are generally in favor of risk disclosure.11,21,23,42, 11,21,23,42 Moreover, follow-up studies in AD reported no significant increase in depression or anxiety following risk disclosure.114,115, 114,115 However, the available data for PD so far are cross-sectional and limited in terms of cultural and diagnostic diversity. Future studies should investigate the attitudes of underrepresented populations and the long-term psychological consequences of risk disclosure in PD in more depth.
Most importantly, prognostic counseling should be adapted to current knowledge about the prognostic significance of specific prodromal and risk markers. The following sections therefore discuss specific markers that are currently of particular importance for prodromal research, as they are most commonly used to recruit prodromal cohorts worldwide (Fig. 1C).4,5,44, 4,5,44
Specific risk factors, prodromal markers, and biomarkers
Risk factors
Additionally, it possible to calculate polygenic risk scores (PRS) from the number of risk alleles for each GWAS-significant variant associated with PD. Although PRS estimates individual genetic liability towards PD due to common variation, it still lacks acceptable diagnostic or predictive capability. PRS alone is therefore not particularly useful for early detection and are not clinically meaningful to warrant disclosure to individuals currently.45,63,64, 45,63,64
In conclusion, there remains much work to be done to determine which genetic results are sufficiently important to feed back to individuals and the circumstances under which this should be done. Most importantly, affected individuals should be included in further studies and longitudinal data are needed to assess long-term effects of risk disclosure.
Clinical prodromal markers
There is a lack of consensus about what information should be imparted to patients with iRBD, both at the point of diagnosis and during follow-up, given the above limitations and basic principles of autonomy, beneficence and nonmaleficence. In recent studies that sought the views of patients with iRBD, ∼90% wanted to receive information about prognosis.21,23, 21,23 Of those, most wanted to receive this information at the point of diagnosis and from their clinician, citing the importance of maintaining trust between doctor and patient. Given significant research interest in iRBD, there is a large volume of material online that patients can access.23,44, 23,44 Failure to explore whether a patient wishes to receive this information risks erosion of trust between clinician and patient. Patients value honesty and several recommended delivering information in a stepwise manner, with the authors endorsing careful communication to impart facts without use of unnecessarily ‘triggering’ phrases (e.g., ‘neurodegenerative’ or volunteering exact phenoconversion rates). While these were the first two studies to assess the views of patients with iRBD, similar surveys have been conducted with patients with PD and with clinicians who see patients with iRBD.6,11,22,29,67, 6,11,22,29,67 These broadly found agreement with one another, that disclosure is situation-specific and heavily nuanced. All found that disclosure was predicated on establishing whether the patient wants to know or not (autonomy) and that risk disclosure should not be foisted on a patient (nonmaleficence). However, although the respect for autonomy is paramount, previous studies have shown that in clinical settings, less than 50% of patients diagnosed with iRBD were asked about their preferences.10,23, 10,23 Probing the patient’s wishes by asking them whether they wish to know about prognostic risk, when in terms of timing, and in how much detail, should be the starting point when discussing the diagnosis of iRBD.
Biomarkers
In recent years, there have been rapid technical advances in prognostic and diagnostic biomarkers for PD, including fluid/tissue-based biomarkers and imaging. One of the most promising techniques is the amplification of pathological
Advances in the assessment of fluid biomarkers is comparable to developments in AD, where CSF and blood have been intensively investigated to diagnose prodromal and even preclinical stages of AD. In contrast to the lack of debate in PD, the technical advances in biomarkers
100
and the steps toward a biological definition of AD
101
have led to a vivid discussion how biomarker results should be disclosed.102,103, 102,103 So far, several studies in AD investigated the potential impact of disclosing (liquid) biomarkers and found no severe harm following disclosure, while some positive effects, including lifestyle changes, have been observed.
104
However, it should be noted that individual views and perceptions may vary considerably, and although consequences such as anxiety or depression have rarely been reported, they are not non-existent.
105
Moreover, risks of stigmatization and discrimination have been discussed.103,106, 103,106 To date, ethical recommendations for AD support the disclosure of biomarker results in research,107,109, 107,109 provided, of course, that the wishes of the recipient have been explored beforehand. However, use of preclinical/prodromal biomarkers in clinical practice
110
will bring new ethical challenges, including potential legal and social implications, e.g., insurance policy issues.
32
Therefore, standardized methods for the assessment of biomarkers and disclosure of results in a clinical setting need to be prepared in advance. The following requirements and key issues for biomarker disclosure have been proposed for AD and can be derived for PD43,104,111, 43,104,111 (Fig. 1D): i) A clear distinction should be made between the use of biomarkers in research and in clinical setting. Diagnostic validity must be clearly established before transition to clinical practice, which is still pending for many
In addition to fluid and tissue-based biomarkers, dopaminergic imaging (FDG-PET and DAT-SPECT) is currently the biomarker with the highest predictive value for PD. 1 . Although dopaminergic imaging has been used for years in studies of prodromal cohorts, again in contrast to AD and Amyloid-PETs,106,112,113, 106,112,113 no guidelines for disclosure of imaging results have yet been established. To date, it can only be inferred from AD studies that disclosure of imaging results in the context of clinical trials is most likely safe and therefore feasible if desired by participants.113–115 However, in clinical practice, if a person shows prodromal symptoms such as iRBD or hyposmia, there is no consensus on the use of dopaminergic imaging, which is handled very differently. It is therefore of utmost importance to establish clear guidelines for clinical practice as to when dopaminergic imaging should be used when a prodromal phase is suspected.
OPEN QUESTIONS AND FUTURE PERSPECTIVES
While the above-mentioned previous studies and ethical debates already form a usable basis to address ethical implications of risk disclosure in PD, the following important questions remain unanswered so far and need to be addressed in future studies (Fig. 1F).
Stratification of risk disclosure
Given the differences in life expectancy, disease severity, caregiver burden and treatment options,116–118 it seems to be of great importance to affected persons
Several studies suggest that certain clinical symptoms may indicate the development of specific
The second uncertainty faced by people with expected prodromal PD and physicians who disclose risk is
Cultural preferences
One acknowledged aspect of risk disclosure for future PD is that it should be tailored according to personal preferences and values of the individual. This requires consideration of the cultural, economic, social, and educational background of the individual, which varies immensely. Similar concerns were raised in other fields, where delivering “bad news” is frequent. For instance, while in Western populations a complete and straightforward disclosure of cancer diagnosis or prognosis is preferred, this may not be the case in certain in African, Asian, or Hispanic communities. 125 So far, the available data on disclosure of future PD indicates that the attitudes of German and Turkish PD patients towards risk disclosure are similar.11,67, 11,67 Likewise, results of studies investigating the psychological impact of disclosing imaging or CSF AD biomarker status seem to be parallel in Western and Japanese societies.114,126,127, 114,126,127 While these reports are encouraging when it comes to mitigating the effect of culture on disclosure, they are by no means sufficient. The current literature on risk disclosure is derived mainly from European and North American populations, which constitute only 17% of the global population, and they are biased by culture/geography.
An all-encompassing, worldwide guideline for risk disclosure in PD is impossible, as it is not feasible to take account of all the values of different cultures. Thus, data from underrepresented populations are critically needed to understand whether and how cultural values and attitudes play a role in disclosure and counseling, a caveat that has also been highlighted in the field of AD.32,102, 32,102 This would allow local institutions to develop culture-specific guidelines for risk disclosure and prognostic counseling that would embrace both universal ethical values and cultural sensitivities.
CONCLUSION
Disclosing the risk of future PD signifies the beginning of a new period for the individual, which has several negative and positive facets. The period starting from the moment of disclosure until the clinically manifest disease will be long and subject to many unanswered questions and unsolved issues that have yet to be discovered and discussed. Recent advances in biomarker studies will change our understanding of PD and their currently defined phases, ushering in the era of a biologically defined disease rather than a clinical diagnosis.78,128–130, 78,128–130 Although the regular use of biomarkers for early diagnosis of PD is to be expected, the discussion on the ethical implications of these scientific advances is still lacking and lags far behind the progress made in AD research. In addition, long-term psychological consequences of risk disclosure, cultural and personal preferences and potential challenges coming with risk stratification have not been addressed sufficiently so far. However, the worldwide recruitment of prodromal cohorts offers a great opportunity to involve those affected in the process at an early stage. It should therefore be an important goal to determine the perspectives and wishes of patients to address these challenges and open questions in the future.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
Alastair Noyce is an Editorial Board Member of this journal, but was not involved in the peer-review process nor had access to any information regarding its peer-review.
