Abstract
Background
Tremor is prevalent among older adults and is suggested to be associated with the subsequent development of Parkinson's disease (PD). However, prospective evidence for the association is scarce.
Objective
This population-based prospective study aimed to estimate PD risk and to explore predictive factors for PD conversion in community-dwelling older people with tremor.
Methods
The study was based on the Beijing Longitudinal Study on Aging (BLSA) II. Tremor was assessed using a validated self-administered method. Participants without parkinsonism and with any tremor were followed for an average of 7.7 years. The standardized incidence ratio (SIR) was used to estimate the risk of developing PD in older people with tremor, with age- and sex-specific incidence rates of PD in the general Chinese population as a reference. Risk factors, prodromal symptoms, and tremor features were assessed to identify predictors for incident PD.
Results
Seventeen (8.4%) of 202 participants with tremor developed parkinsonism, including 9 (4.4%) diagnosed with PD. The presence of any tremor was associated with an increased risk of developing PD (SIR: 3.12, 95% confidence interval [CI]: 1.52–5.73). Sensitivity analyses yielded similar results. Probable REM sleep behavior disorder (RBD), identified using the RBD Questionnaire-Hong Kong, and self-report of new-onset rest tremor predicted PD conversion.
Conclusions
Tremor is associated with an elevated risk of PD in the general older population. Screening for RBD and monitoring the evolution of tremor characteristics using simple questionnaires may help identify individuals at high risk for PD among community-dwelling older adults with tremor.
Plain language summary
Tremor is common among older adults. Some people with tremor are concerned about whether they are more likely to develop Parkinson's disease (PD). However, current research hasn’t provided clear answers yet. To better understand this, the researchers compared the risk of developing PD in older adults with tremor to that of the general older population. They also looked for specific factors that might increase the risk of PD in older people with tremor. The study included 202 older people with any kind of tremor from a community in Beijing, China. None of the participants had PD at the start of the study. After an average of 7.7 years, 4.4% of these participants were diagnosed with PD. The results showed that older adults with tremor could be three times more likely to develop PD than the general older population. The study also found that tremulous older adults who had a sleep disorder called REM sleep behavior disorder were at an even higher risk of developing PD. In addition, if older people have tremor and notice it happening when the limbs are relaxed (called rest tremor), it is important to consult a neurologist. This type of tremor could be a sign that they might develop PD.
Introduction
Tremor is an involuntary oscillatory movement of a body part. 1 It is a common movement disorder in the older population, affecting approximately 10% of people older than 50 years. 2 Among various causes of tremor, Parkinson's disease (PD), along with other neurodegenerative parkinsonism, is particularly concerning due to its progressive nature and debilitating impact on daily functioning. Nevertheless, parkinsonian tremor, typically a rest tremor affecting the limbs, accounts for only a small proportion of tremor cases in the general older population. 2 In contrast, tremor in older adults generally presents as an action tremor, primarily involving the upper limbs and occasionally other body parts like the head. This tremor often lacks identifiable pathological causes2,3 and is suggested to be associated with aging,4,5 since it is increasingly prevalent with advancing age and associated with aging-related conditions and adverse outcomes.4,6–8
The association between tremor and aging raises the question of whether tremor is linked to the development of PD, which is a major age-related neurodegenerative disorder. 9 Earlier clinical studies have observed that idiopathic tremor, such as isolated unilateral action tremor, may precede parkinsonian motor manifestations by several years.10–12 Many studies suggest an association between essential tremor (ET) and the development of PD.13,14 Recent large registry-based case-control studies show that tremor substantially increases in PD cases compared to controls as early as 5 to 10 years before an established PD diagnosis when parkinsonian rest tremor typically occurs.15–17
The development of PD among older adults with tremor is clinically significant, given the increasing prevalence of tremor with aging and the debilitating impact of PD. It is essential to estimate the risk of developing PD in older adults with tremor and to identify individuals at high risk for PD within the general tremor population to enhance early diagnosis and intervention of PD. Although suggesting a potential link between tremor and PD, previous clinic- and registry-based studies can be susceptible to selection bias, as community-dwelling older adults are not inclined to seek medical care for tremor. 18 So far, few population-based prospective studies have quantified PD risk in community-dwelling older adults with tremor,14,19 and none have explored strategies to identify individuals at high risk for PD within the tremor population.
In this study, we estimated the risk of incident PD in a representative community-based cohort of older people with tremor compared to the general older population. Additionally, we explored risk factors that may contribute to the link between tremor and PD, as well as clinical predictors for incident PD and parkinsonism in older people with tremor.
Methods
Study setting and population
The study was embedded in the Beijing Longitudinal Study on Aging (BLSA) II 20 –a population-based prospective study designed to investigate risk factors for major geriatric conditions, including PD. A multistage cluster random sampling method was employed to select a representative sample of 10,039 individuals aged 55 years and older from three urban districts (Xicheng, Dongcheng, and Xuanwu) and one rural county (Shunyi) of Beijing. The baseline visit was conducted in 2009. Two follow-up visits were conducted during 2010–2011 and 2013–2014. During each visit, medical history related to PD and other parkinsonism, along with data on risk factors and symptoms of PD, was collected. Each participant provided informed consent. The study was approved by the Research Ethics Committee of Xuanwu Hospital of Capital Medical University.
The current study included the BLSA II participants who reported any tremor, identified by a validated screening question as detailed below, during at least one visit from 2009 to 2014. The earliest visit during which the participants reported tremor was defined as their baseline for this study. Participants diagnosed with PD or parkinsonism at baseline or within two years after baseline were excluded to rule out prevalent PD cases. The eligible tremor cases were followed during 2017–2018 to identify incident cases of parkinsonism and PD.
The study was ultimately completed in two urban districts (Dongcheng and Xuanwu), where the 2017–2018 follow-up was conducted. Among the 4980 participants from the two districts, 399 reported tremor. After excluding 24 without data on baseline parkinsonism diagnosis, 18 with prevalent parkinsonism, 75 deceased, 76 lost to follow-up, and 4 refusing to participate, 202 participants with tremor attending the 2017–2018 follow-up were included in the final sample (Figure 1).
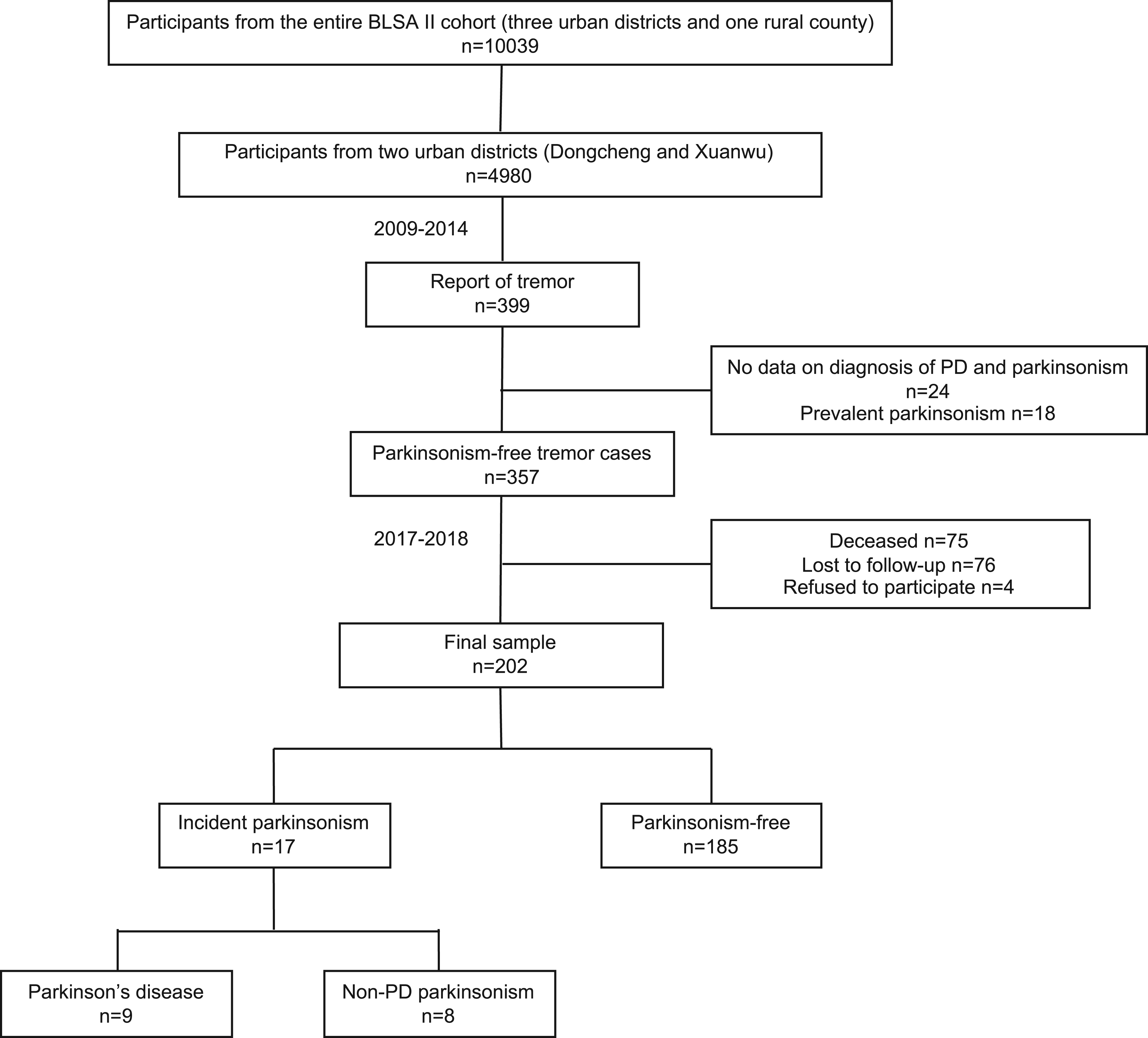
Flowchart of the study.
Assessment of tremor
Each participant in the BLSA II cohort was asked a tremor screening question: “Do you experience any tremor in the limbs or other body parts?”. A similar question has exhibited good sensitivity in previous epidemiological studies. 21 Participants who answered “yes” to the screening question completed a structured questionnaire of tremor features, including tremor distribution (head, unilateral limb, or bilateral limbs), activation conditions (rest tremor = tremor occurring when the limbs are relaxed, or action tremor = tremor occurring during movement or while holding objects), and tremor severity (being progressive, stable, or improved). Based on the answers to the questionnaire, tremor characteristics were categorized into four practical types: rest tremor in the limbs, unilateral action tremor, bilateral action tremor, and isolated head tremor. Tremor-producing conditions, such as medications and comorbidities (Supplementary Material), were reviewed for each participant. In addition, participants who screened positive for tremor underwent standard physical examinations conducted by trained community physicians or nurses. The participants were instructed to extend their arms forward to assess postural tremor in the upper limbs.
Since tremor assessment relied primarily on self-reports in the BLSA II cohort, we tested the agreement between self-report and clinical assessment of tremor (Supplementary Material). Clinical tremor assessment was conducted by movement disorder specialists following a standardized protocol. Substantial agreement was observed between the tremor screening question and clinical examination (Cohen's κ = 0.71, 95% confidence interval [CI]: 0.59–0.83) (Supplemental Table 1). Furthermore, the tremor screening question identified individuals with occasional tremor that was not apparent during a single clinical examination. No other movement disorders were observed in subjects with self-reported tremor.
Therefore, we conducted the study in the participants with self-report of any tremor, indicated by a positive response to the tremor screening question. We also repeated the study analysis in a subset of participants with clinically-confirmed postural tremor, as assessed by community physicians and nurses.
Identification of PD cases
During the three visits from 2009 to 2014, cases of PD and other parkinsonism were identified through structured interviews by trained investigators. Participants or their proxies were asked about medical history of PD or other forms of parkinsonism, including neurodegenerative conditions such as multiple system atrophy and progressive supranuclear palsy, as well as secondary parkinsonism, such as vascular and drug-induced parkinsonism. Information on the time of diagnosis and anti-parkinsonian treatments was also collected. Medical records were reviewed if accessible.
During the 2017–2018 follow-up, diagnoses were made by research neurologists specialized in movement disorders according to the MDS Clinical Diagnostic Criteria for Parkinson's Disease, 22 based on comprehensive neurological examinations. Participants unable to attend clinic examinations underwent a two-phase process to confirm the outcome. First, one of the research neurologists conducted a telephone interview to obtain information on parkinsonian symptoms and previous diagnoses. Proxies were contacted for the participants who were unable to communicate. Suspected parkinsonism was defined as having a prior diagnosis or presenting any of the following: tremor at rest, slowness, or two other parkinsonian motor symptoms (listed below). In the second phase, research neurologists conducted home visits to examine the suspected parkinsonism cases. For the deceased participants with prior parkinsonism diagnoses, research neurologists interviewed proxies and reviewed available medical records to validate the diagnoses.
Risk factors and clinical predictors of PD
Risk factors were selected based on the hypothesis that factors predisposing to both tremor and PD, such as aging, familial susceptibility, and environmental exposures, might contribute to their potential association.5,23,24 Information on demographics, lifestyle factors, family history, comorbidities, history of brain trauma, and exposure to toxins was collected using structured questionnaires. Serum lipid and urate levels were measured. Aging parameters included intrinsic capacity, which encompasses five domains reflecting overall physical and mental abilities, 25 and frailty, which represents vulnerability due to diminished physiological reserve. 20
Candidate clinical predictors included motor and non-motor symptoms of prodromal PD, as well as tremor features. The motor symptoms, including smaller handwriting, softer voice, inexpressive face, slowness/stiffness, reduced arm swing, falling, and shuffling gait, were assessed using a questionnaire adapted from a well-validated one. 26 Probable REM sleep behavior disorder (RBD) was determined using the RBD Questionnaire-Hong Kong (RBDQ-HK). 27 Depression was evaluated with the 15-item Geriatric Depression Scale (GDS-15). 28 Constipation and hyposmia were assessed using the questions: “Do you have constipation (fewer than three bowel movements per week)?” and “Do you have an impaired sense of smell?”. Tremor features at baseline were assessed by the aforementioned questionnaire. The evolution of tremor features was evaluated through repeated assessments of tremor features over time.
Statistical analyses
We used the standardized incidence ratio (SIR), defined as the ratio of observed to expected incident cases, to estimate the relative risk of developing PD in older people with tremor. The 95% confidence interval (CI) of SIR was determined using the mid-P exact test. We used age-stratified, sex-specific data on incidence rates of PD in the general Chinese population to determine the expected number of incident PD cases. 29 We referenced the incidence rates of PD in 2018, which were the highest between 2009 and 2018. Person-years were calculated from baseline to the earliest event: (1) last follow-up, (2) parkinsonism diagnosis, or (3) death.
Baseline data of risk factors and candidate predictors were compared between the subjects who developed PD and those who remained free of parkinsonism using the Student's t-test or Mann-Whitney U test for continuous variables, and the chi-square test or Fisher's exact test for categorical variables. Group comparisons of motor and non-motor symptoms were adjusted for age and sex, which are major covariates, 30 using logistic regression analyses. For the variables demonstrating significant differences between groups, Cox proportional-hazards models were used to estimate hazard ratios (HR) and 95% CIs controlling for age and sex. The proportional hazards assumption was tested using Schoenfeld residuals. Statistical significance was set at p < 0.05. Analyses were performed using STATA 14.0 and OpenEpi online software version 3 (www.OpenEpi.com).
Sensitivity analyses
When estimating the risk of incident PD, our main analysis was conducted in the participants with self-report of any tremor. We performed a sensitivity analysis in the participants with clinically-confirmed postural tremor, and another analysis in those without medical conditions known to cause tremor. To minimize misdiagnosis of PD, we only included incident cases examined in person in a sensitivity analysis, excluding those identified through medical review. We also performed an analysis restricted to the participants without rest tremor or any other parkinsonian motor symptom at baseline to address the potential misidentification of prevalent PD as incident cases. Additionally, to mitigate the dilution effect caused by including tremor cases in the reference population, we repeated the analyses using the adjusted PD incidence rates in the general population by excluding the contribution of tremor cases.
Results
Participants and outcome
The study sample comprised 202 participants with tremor. Baseline characteristics are summarized in Table 1. The tremor cases not followed were older and had more parkinsonian motor symptoms at baseline (Supplemental Table 2).
Baseline characteristics of participants reporting tremor.

Data are presented as numbers (percentages) for categorical variables and means (SDs) for continuous variables.
Data are available for 200 participants.
Data are available for 64 participants.
Including isolated rest tremor in the limbs and mixed tremor (rest tremor in the limbs + any other type of tremor)
After a mean (SD) follow-up of 7.7 (1.7) years, 17 of the 202 participants with tremor developed parkinsonism, including 9 diagnosed with PD. The mean (SD) interval from baseline to PD diagnosis was 6.4 (2.3) years, ranging from 4 to 9 years. The risk of PD was elevated among older people reporting any tremor compared with the general population (SIR = 3.12, 95% CI: 1.52–5.73). Sensitivity analyses revealed similar results (Table 2 and Supplemental Table 3).
Risk of PD in older people with tremor compared with the general population.

SIR: standardized incidence ratio; CI: confidence interval
Motor symptoms included rest tremor, smaller handwriting, softer voice, inexpressive face, slowness/stiffness, reduced arm swing, falling, and shuffling gait.
Risk factors and clinical predictors
Participants who developed PD and parkinsonism were older and more likely to be male compared with those who remained free of parkinsonism, while the differences were not statistically significant. Risk factors at baseline were comparable between the groups (Table 3).
Risk factors of PD at baseline.
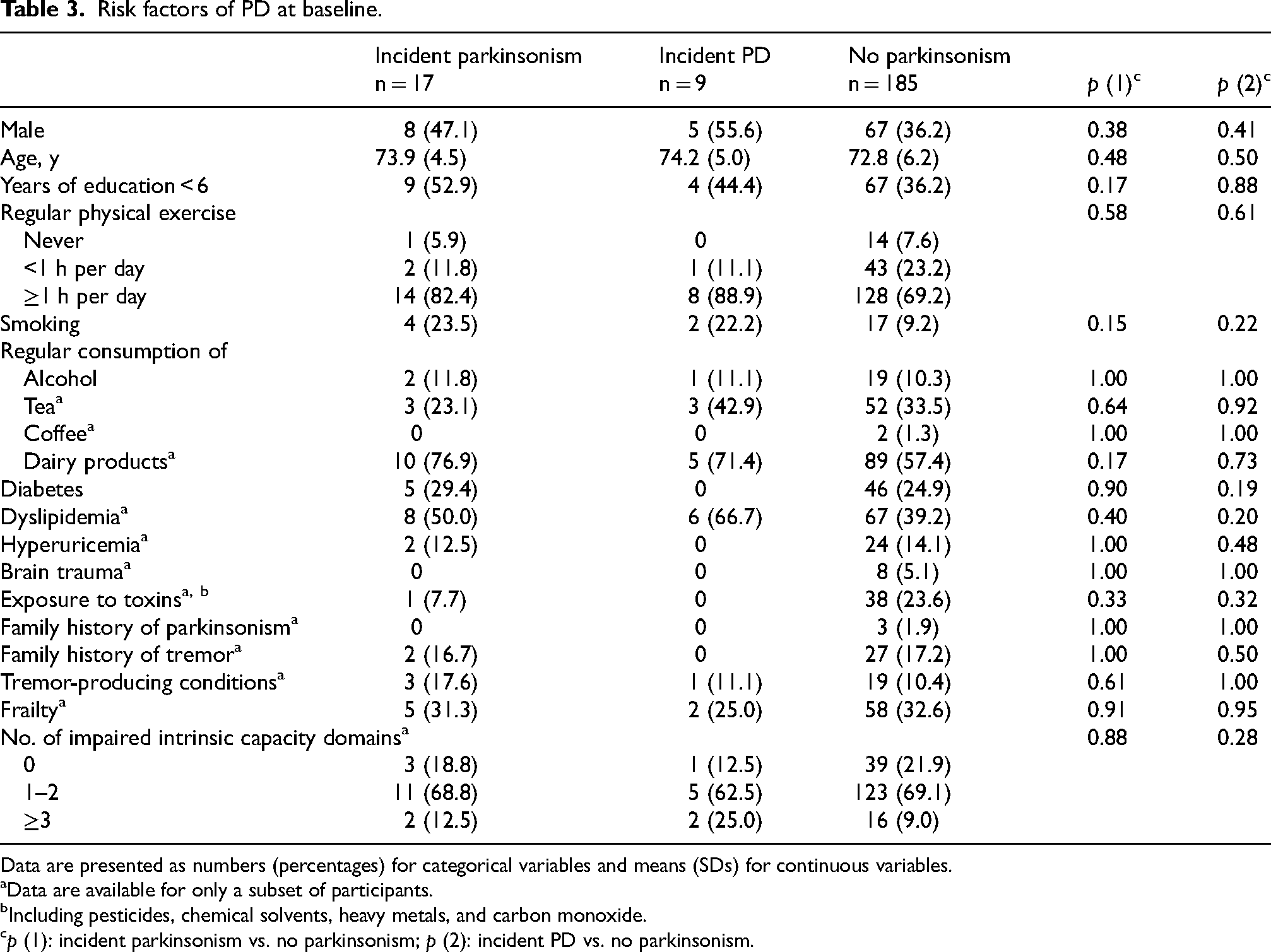
Data are presented as numbers (percentages) for categorical variables and means (SDs) for continuous variables.
Data are available for only a subset of participants.
Including pesticides, chemical solvents, heavy metals, and carbon monoxide.
p (1): incident parkinsonism vs. no parkinsonism; p (2): incident PD vs. no parkinsonism.
Over 60% of older adults with tremor reported at least one motor symptom at baseline. However, none of the motor symptoms were associated with incident parkinsonism or PD diagnosed at least 4 years later (Table 4). Probable RBD was more prevalent at baseline in the participants who developed PD compared with those who remained free of parkinsonism (25.0% vs. 5.0%, p = 0.03), and significantly associated with an increased risk of incident PD after adjusting for age and sex (HR = 6.60, 95% CI: 1.30–33.52, p = 0.02).
Motor and nonmotor symptoms at baseline.
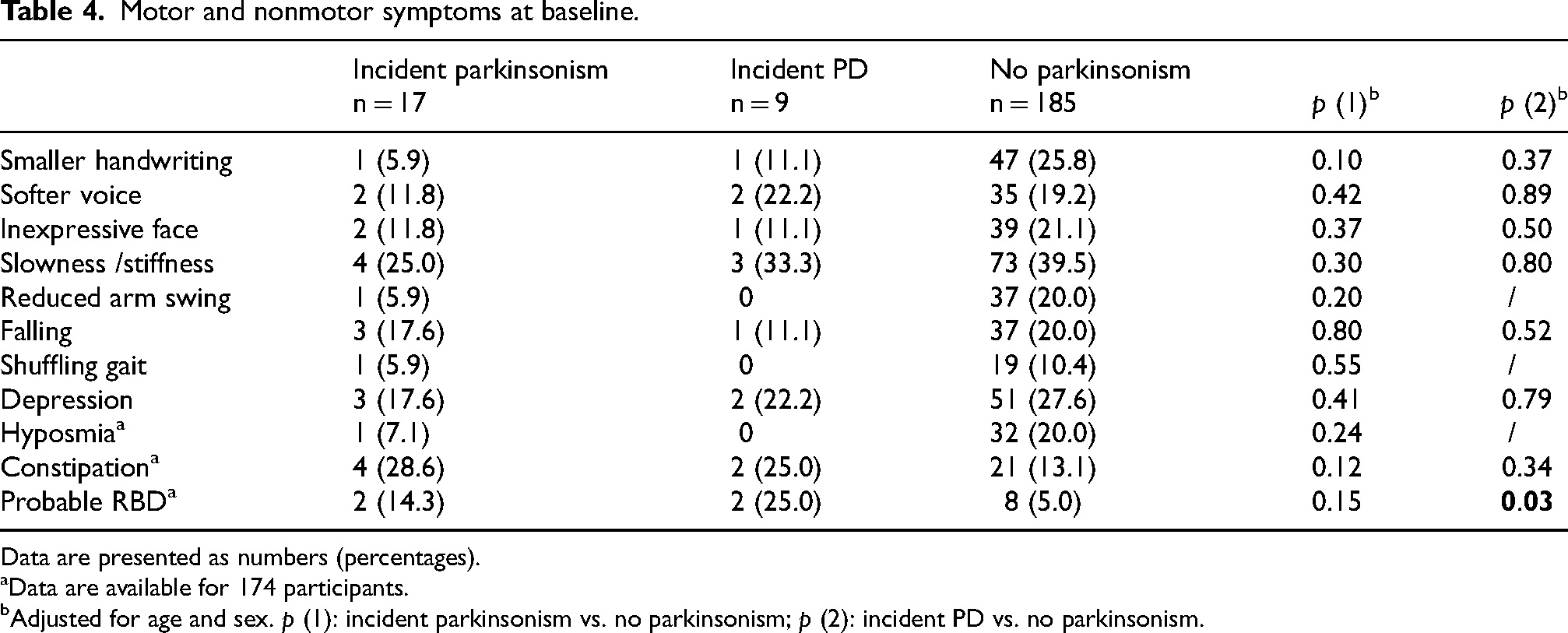
Data are presented as numbers (percentages).
Data are available for 174 participants.
Adjusted for age and sex. p (1): incident parkinsonism vs. no parkinsonism; p (2): incident PD vs. no parkinsonism.
A subset of 61 participants provided information on tremor features both at baseline and during at least one follow-up examination. Baseline tremor features did not differ between the participants who developed parkinsonism and those who did not (Table 5). When considering the changes of tremor features over time, incident parkinsonism cases were more likely to report the emergence of rest tremor during follow-up compared with those free of parkinsonism (80.0% vs. 1.8%, p < 0.001). Among these incident cases, all reported rest tremor onset within 1 year before parkinsonism diagnosis, except one case with dementia who was unable to provide a reliable onset time. New-onset rest tremor during follow-up was significantly associated with conversion to any parkinsonism (HR = 38.48, 95% CI: 3.94–375.75, p = 0.002) and PD (HR = 32.14, 95% CI: 2.13–484.48, p = 0.01).
Tremor features at baseline and changes during follow-up.
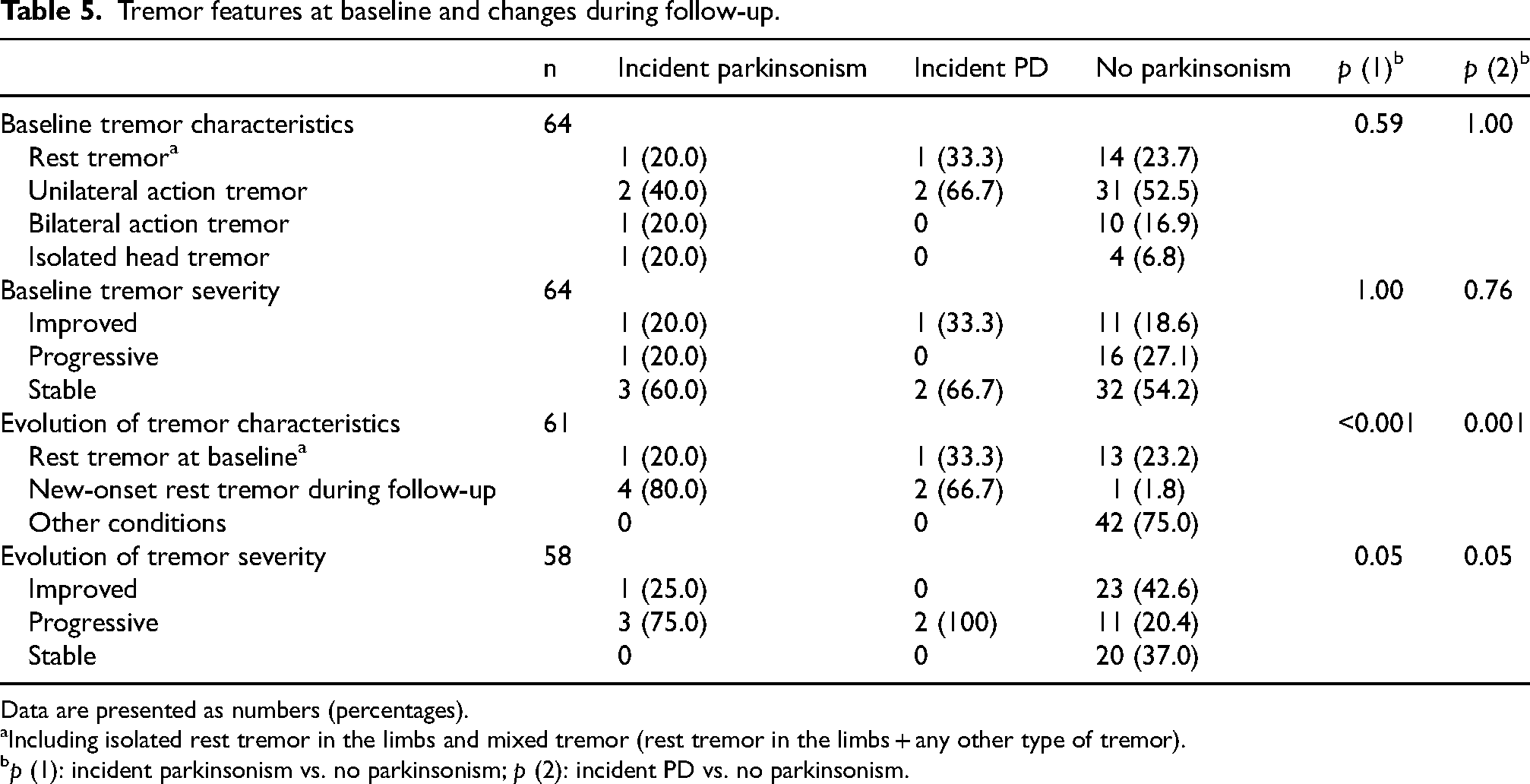
Data are presented as numbers (percentages).
Including isolated rest tremor in the limbs and mixed tremor (rest tremor in the limbs + any other type of tremor).
p (1): incident parkinsonism vs. no parkinsonism; p (2): incident PD vs. no parkinsonism.
Discussion
In this population-based prospective study, older people with tremor exhibited an increased risk of developing PD over 4 to 9 years of follow-up. Probable RBD, identified using the RBDQ-HK questionnaire, preceded PD diagnosis by at least 4 years, suggesting it as an early marker of tremor cases at risk for PD. New-onset rest tremor during follow-up indicated an impending conversion to parkinsonism, regardless of baseline tremor features.
Some methodological considerations and limitations should be noted when interpreting the excess risk of PD in older people with tremor. First, despite substantial agreement with clinical assessment, self-reports of tremor may still introduce misclassification bias. However, we noticed that self-reports could be more effective in detecting occasional tremors that might be missed during brief clinical examinations. Occasional tremor should not be overlooked, as it accounts for a significant proportion of tremor cases in the community setting 31 and may be a prodrome for PD. 32 In future community-based studies, combining clinical examinations with continuous home-based tremor monitoring could provide more precise data.
Second, due to the complexity of tremor classification, we were unable to categorize each tremor case in this epidemiological study accurately. As we assessed the overall PD risk associated with any tremor without distinguishing between specific tremor syndromes, the most prevalent tremor syndromes, such as essential tremor and enhanced physiological tremor, may have significantly influenced the estimated PD risk. In contrast, less common tremor syndromes (e.g., functional tremor) were likely present in the study sample, but their contribution to PD risk may not have been fully reflected. Nevertheless, our sample was derived from a well-designed population-based cohort, making it representative of the general tremor population and enhancing the generalizability of our findings. By assessing overall tremor-related PD risk in a representative sample, we provide results that are applicable to the general population, where precise tremor classification is often impractical. Future studies should categorize tremor types and distinguish syndromic diagnoses to clarify their differential associations with PD risk.
Another important concern is the misidentification of prevalent PD cases as incident ones. Prevalent PD cases were identified by reviewing medical history and previous diagnoses. Consequently, some PD patients undiagnosed at baseline might be misclassified as incident cases. However, the prevalence of parkinsonism in the BLSA II cohort is comparable to the published epidemiological data (approximately 2% in adults ≥ 55 years),33,34 indicating that this method of parkinsonism identification would not miss a significant number of prevalent cases. Furthermore, we excluded PD cases diagnosed within two years after baseline, according to previous work in China showing that the diagnostic delay of PD is typically around 15 months and that most PD patients initially presenting with tremor are diagnosed within 20 months. 35 In our study, the shortest latency between baseline and PD diagnosis was 4 years (mean: 6.4 years) for the incident cases, who were unlikely to be undiagnosed prevalent PD considering the long diagnostic delay. Moreover, sensitivity analyses showed that the excess risk of PD remained significant among the participants without complaints of any parkinsonian motor symptom. PD patients would be less likely to report only non-specific tremor without any typical motor symptom.
Fourth, not all participants with tremor were examined in person to confirm the outcome of incident parkinsonism. For those unable to attend the clinic, we employed a two-phase method and only examined positive screeners for parkinsonism. Consequently, some incident PD cases may have been missed, which could bias our results towards the null.
Fifth, loss to follow-up should be considered. Subjects not followed were older and had more parkinsonian symptoms at baseline, and thus may be more likely to develop PD. Consequently, loss to follow-up could have led to a conservative estimate of PD incidence in older people with tremor.
Strengths of our study included a well-designed population-based cohort to minimize selection bias and a longer follow-up period than in previous studies. Moreover, our study encompassed comprehensive data on risk factors and clinical markers of prodromal PD, which were not included in previous studies, allowing us to explore the underlying factors linking tremor to PD and identify clinical predictors for PD conversion in the tremor population.
To our knowledge, two population-based prospective studies have been published investigating the relationship between tremor and the risk of PD, both supporting our findings. A Spanish cohort study revealed a four-fold higher risk of PD in older people with ET. 14 While this study offers valuable evidence linking tremor with increased PD risk, ET represents just one facet of the complex profile of tremor in prodromal PD. Tremors other than ET have been observed to precede PD conversion.11,12,36,37 Comparable to our estimate, an earlier study demonstrated a two-fold higher risk of incident PD in older adults with self-reported tremor, although it did not specify how well the self-reports agree with clinical tremor assessment. 19 Consistent results were found in a subsequent nested case-control study conducted in the same cohort. 38
We identified probable RBD at baseline and new-onset rest tremor during follow-up as predictors of PD conversion in older people with tremor, though the limited number of PD converters restricted precise estimation of the hazard ratios. No significant associations were observed between incident PD and other less specific symptoms, such as constipation, olfactory impairment, depression, motor symptoms, and baseline tremor features. These findings are consistent with existing literature, highlighting RBD and rest tremor as robust predictors of PD conversion. 24 Our findings suggest that screening for probable RBD with a simple questionnaire and self-monitoring of new-onset rest tremor could help identify tremulous individuals at high risk for PD conversion, offering an accessible approach for PD screening in community-dwelling older adults with tremor.
The mechanisms underlying the elevated risk of PD in older adults with tremor remain unclear. The tremor population may either be at the prodromal stage of PD (similar to the idiopathic RBD population), or represent a high-risk group with increased susceptibility to PD. In our study, PD conversion occurred, on average, more than 6 years after baseline, which is significantly longer than the typical interval between the onset of parkinsonian tremor and PD diagnosis.15,39 The finding suggests that in our cohort, tremor may indicate increased susceptibility to PD rather than a prodromal motor symptom. Though we explored potential contributing factors such as clinical aging parameters, environmental exposures, and familial susceptibility, none were statistically significant. A larger sample size and a more refined assessment of risk factors are needed in future research.
In summary, our study shows that community-dwelling older people with tremor are at higher risk of developing PD, highlighting the need for PD screening in this population. Simple questionnaires for RBD and self-monitoring of tremor evolution may be feasible screening tools to identify high-risk individuals for PD among community-dwelling older people with tremor.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251327138 - Supplemental material for Risk of Parkinson’s disease in older people with tremor: A population-based prospective study
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251327138 for Risk of Parkinson’s disease in older people with tremor: A population-based prospective study by Xitong Xu, Jie Chen, Shaozhen Ji, MeiJie Chen, Shuying Liu and Piu Chan in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We appreciate all the participants and medical staff for their contributions to the study.
Ethical considerations
The study was approved by the Research Ethics Committee of Xuanwu Hospital of Capital Medical University.
Consent to participate
Each participant provided informed consent.
Consent for publication
Not applicable
Funding statement
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China [No. 2021YFC2501200, 2018YFC1312001, 2017YFC0840105, 2017YFC1310200] and the Key Realm R&D Program of Guangdong Province [No. 2018B030337001].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data are not publicly available due to privacy regulations and ethical restrictions. The data supporting the findings of this study are available on request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
