Abstract
Several dietary patterns and nutritional supplements have been linked to the development, progression, and symptomatic treatment of Parkinson’s disease (PD). Most of the evidence, at this point, is preliminary and based largely on observational studies. Interventional studies are scarce, so the evidence on effectiveness remains inconclusive. Dietary interventions could, analogous to exercise, potentially have a beneficial effect on disease symptoms as well as on the progression of the disease and should therefore be researched in high quality studies. Further work is also needed to study whether dietary interventions, when applied to an at-risk population, have any potential to postpone the onset of manifest PD. In this paper, we summarize all ongoing clinical trials on dietary interventions in PD. We found 10 ongoing studies, all aimed at a different intervention. These studies are mostly exploratory in nature or represent phase I or phase II trials focusing on safety, biological responses, and symptomatic effects. Taken together, we conclude that research on dietary interventions in persons with PD is still in its early days. The results of the various ongoing trials are expected to generate new hypotheses and will help to shape the agenda for future research on this important topic.
INTRODUCTION
Parkinson’s disease (PD) is a complex neurodegenerative disorder characterized neuropathologically by degeneration of dopaminergic neurons, as well as lesions outside the nigrostriatal circuitry [1]. These neurodegenerative changes lead to a wide range of motor- and non-motor symptoms, some of which are responsive to dopaminergic treatment. While the underlying disease mechanisms causing this neurodegeneration have not been fully clarified, treatment of the disease remains merely symptomatic by offering dopamine replacement therapy (medication for almost all individuals, and deep brain surgery for selected cases) [1]. Several potentially disease modifying drugs have been studied, but none of these have thus far been shown to alter the progressive course of the disease []. In the absence of disease modifying pharmacological approaches, increasing attention is being paid to non-pharmacological management of the disease [5]. Most work has so far focused on exercise, which can have positive effects on a wide range of symptoms such as gait, balance, and mood []. Exercise can also alleviate a range of non-motor symptoms, including, e.g., sleep [8] or depression [9]. At this moment, high-intensity aerobic exercise is hypothesized to be one of the very few intervention that could potentially impact the progression of PD []. The evidence for this assumption is based on a combination of animal studies [], exploratory randomized controlled trials in humans [15], neuroimaging studies showing evidence for exercise-induced cerebral plasticity in persons with PD [15] and longitudinal cohort studies [].
Longitudinal cohort studies have also identified other lifestyle factors that are related to the development, progression, or symptomatic treatment of PD, including diet and strategies to manage stress [18]. In this present paper, we focus on dietary patterns and nutritional supplements (which we will refer to as dietary interventions). One of the reasons why dietary interventions are generating such enormous interest is that, in recent years, the relationship between the gut microbiome and PD development has become clearer []. Another reason is the possible link between exposure to pesticides on the one hand, and the risk of developing PD on the other hand []. Humans can be exposed to pesticides in various ways, and exposure via the food chain is one of them. The latter notion actually directly links pesticides again to changes in gut microbiome, as is highlighted in an elegant recent review in this journal [24]. Indeed, in clinical practice, in patient forums and on social media, we see many people with PD (PwPD) who are deeply interested in the subject of nutrition, and they have many questions on what diet they should follow, or what supplements they should take.
Taken together, nutrition is potentially important in PD for at least three reasons. First, dietary factors can play a role in the preclinical phase by determining the risk of developing PD, either in a negative way or in a positive way (because of protective elements in food). Indeed, several dietary products have been associated with a decreased risk of future development of PD, such as adherence to the Mediterranean diet and the regular consumption of coffee or flavonoids; conversely, other dietary products such as dairy products have been associated with an increased risk []. Metabolic syndrome is another risk factor associated with a higher risk of developing PD [27], and this is of course also linked to diet.
Second, diet can be important in people who have already developed symptomatic PD. One important factor to consider in the management of PD is the relationship between protein intake and absorption of levodopa in the gut (gastrointestinal absorption) and the brain (blood-brain barrier). Because dietary proteins split into amino acids after digestion, they compete with the absorption of levodopa when taken together. This may lead to reduced effectiveness of the dopaminergic medication and response fluctuations [28]. People with PD are therefore advised not to take proteins (e.g., dairy products) around their medication intake. This can, however, lead to insufficient protein intake which then impacts negatively on muscle quality and function [29]. Also, diet can affect the rate of gastric emptying, which is an increasingly recognized cause of unpredictable response fluctuations, such as dose failures or delayed “on” periods [30]. Another common and vexing problem for people with PD is constipation [], which is often one of the earliest symptoms, and which often even antedates the manifestations of motor parkinsonism. Constipation also impacts the absorption and efficacy of levodopa, in part because of secondary small intestinal bacterial overgrowth (SIBO) that may lead to production of enzymes that convert levodopa into dopamine within the lumen of the gut, thereby hampering its entry into the brain [33], Conversely, appropriate nutrition (i.e., sufficient fiber and liquid consumption) plays an important role in the management of constipation and the prevention of SIBO []. But many people with PD do not eat well enough, e.g., because the food does not taste well due to hyposmia, or because constipated patients avoid meals as these may cause gastrointestinal complaints such as bloating, abdominal pain, or winds. Inadequate diet then also impacts on the nutritional status, potentially leading to underweight and malnutrition [].
Finally, nutrition may potentially have a disease modifying effect because of its impact on mitochondrial function, inflammation, and immune responses [37]. Moreover, recent research suggests that dysregulation of the gut microbiome may play a role in the pathogenesis of the disease, for example by enhancing inflammation systemically and possibly also in the central nervous system []. This raises the exciting possibility that adequate nutrition could potentially impact on the development and progression of PD, but this proactive idea has thus far never been tested in controlled studies.
Besides some small pilot studies on different kinds of dietary patterns, i.e., the ketogenic diet [38] or Mediterranean diet [39], and studies of supplements, i.e., vitamins and antioxidants [37], the current evidence on nutrition in PD is mostly observational in nature []. So, overall, the evidence on the merits of any PD-specific diet(s) remains inconclusive [40]. Following the example of exercise, the time is now ripe to take nutrition to the test, just as one would do when evaluating a new drug, and to do this in high quality randomized clinical trials. In this review, we will summarize all ongoing clinical trials on a nutritional intervention (dietary pattern or nutritional supplement) in PD. In doing so, we hope to offer more insights into where this field is headed and which gaps need to be filled with solid scientific research.
OVERVIEW OF RANDOMIZED CONTROLLED TRIALS ON NUTRITION
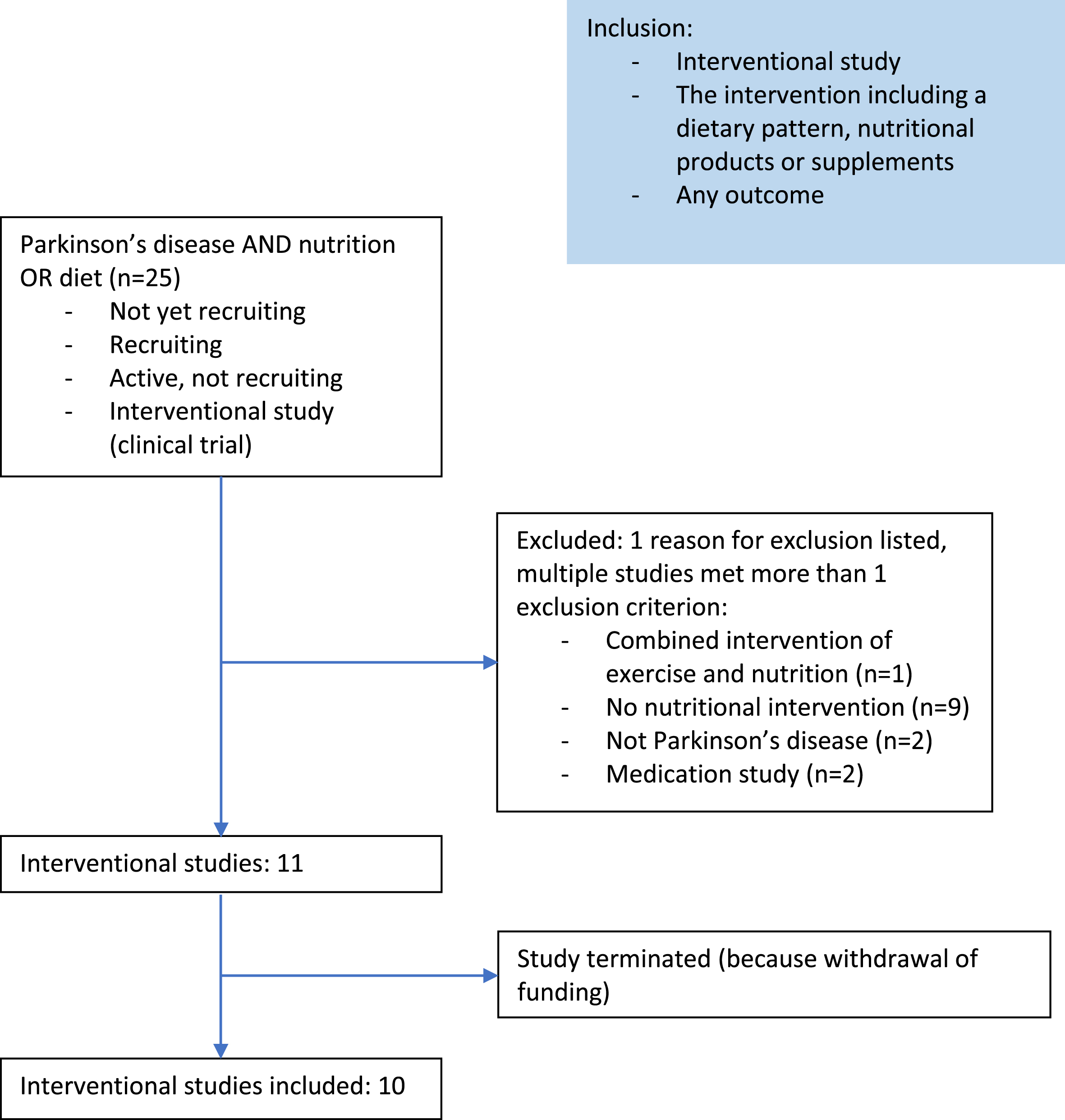
We searched Clinicaltrials.gov for interventional studies in PD with the terms “NUTRITION’’ or “DIET” and “Parkinson’s disease”. Studies were only included if their status was “not yet recruiting”, “recruiting”, or “active, not recruiting”. Studies were excluded if it was not an interventional study, a combined intervention with exercise, or including another population than people with PD (Fig. 1). This resulted in the inclusion of 11 studies. After the start of the review process, 1 study was terminated because of withdrawal of funding. This open label pilot study on the biological responses of supplementing short-chain fatty acid (BUTTER study) was therefore excluded from this paper. The general characteristics of the included 10 studies are described in Table 1.
Baseline demographics of the first 108 PwPs enrolled on the HBC pathway
Population
Samples sizes vary between 20 and 180. Most studies include people with PD older than 40 years and in Hoehn and Yahr stage 1-3. Two studies are not only including people with PD, but also other neurological conditions, such as Multiple System Atrophy and Lewy Body Dementia. One study even includes people with a condition that is not neurological: Rheumatoid Arthritis.
Interventions
Three of the ongoing trials are studying a dietary pattern: the Mediterranean diet versus an adapted Mediterranean – ketogenic diet (study 1), consistent protein intake versus redistributed protein intake and a gluten free diet versus a gluten containing diet. Five different supplements are being studied (fiber, vitamin B3, ketones, vitamin E, and carbohydrate), all in early stages focusing on safety, biological responses, and preliminary clinical effects. Finally, one study evaluates the clinical effects of modified food textures for people with dysphagia and one study evaluates the biological response of fasting.
Primary and secondary outcome measures
A great variety of outcomes is included in the different studies. We see a mix of outcomes related to metabolism and the biological response to a supplement and clinical outcomes (i.e., disease symptoms and quality of life). Also, the microbiome is included as an outcome in some studies. None of the studies explicitly aims to study disease progression.
Follow-up duration
The duration of the interventions varies from 1 week to 12 months.
Blinding
Of the 10 studies included, three are performed double blind, and four single blind (1 participant blinded, 3 assessor blinded). The remaining three studies are open label studies. Two of them do not include a control group.
Compliance
Very little attention is being paid to compliance. Only two studies (on dietary patterns) mention that participants will be guided by a professional in adhering to the study regimen. Only one study mentions monitoring compliance.
IPD sharing statement
None of the studies indicated to be sharing Individual participant data (IPD).
Participants in the MeDi-KD group will receive a modified Mediterranean-ketogenic diet. The ketogenic component of the diet includes limiting the intake of carbohydrates to about 10% of all calories consumed per day. The ketogenic ratio (the ratio of fat to carbohydrates) will be gradually increased during the first week from 1:1 to 3:1. The Mediterranean component of this dietary pattern includes consuming green leafy vegetables, nuts, and olive oil, while limiting the consumption of processed or fried food, red meat, full-fat dairy, and sweets.
Secondary: Retention and adherence. Retention will be measured by the number of participants who complete the study. Adherence is quantified by the percentage of time spent in clinically relevant levels of ketosis (>0.5 mmol/L) by each participant throughout each intervention, as measured using daily breath ketone analyses.
Other outcomes: Changes from baseline after each intervention in: motor and non-motor PD symptoms (MDS-UPDRS; Movement Disorders Society-Unified Parkinson’s Disease Rating Scale), fatigue (Fatigue Severity Scale), apathy (Starkstein Apathy Scale), depressive symptoms (Beck Depression Inventory II), anxiety (Parkinson’s Anxiety Scale (PAS)), quality of life (Parkinson’s disease questionnaire-39), capacity for performing physical activities (Physical Activity Scale for Individuals with Physical Disabilities), stool consistency (Bristol Stool Chart), constipation and irritable bowel symptoms (Rome III module).
Dietary habits before the study are measured using the Canadian version of the Diet History questionnaire. Cognitive function is measured using the National Health Institute Toolbox-Cognitive battery (NIHTB-CB)).
Secondary: Change from baseline to 5-week follow-up in physical activity (actigraphy) and total PD symptoms (total MDS-UPDRS score).
Secondary: Digestive health (Gastrointestinal Symptom Rating Scale), laxative use, stool frequency, stool consistency (Bristol Stool Form Scale), body composition (bioelectrical impedance spectroscopy), appetite (Council on Nutrition Appetite Questionnaire (CNAQ), nutrition risk (Patient Generated- Subjective Global Assessment (PG-SGA)), constipation-related Quality of Life (Patient Assessment of Constipation- Quality of Life questionnaire), non-motor symptoms (Non-Motor Symptom Scale for Parkinson’s Disease (NMSS)), Parkinson’s disease related Quality of Life (Parkinson’s disease Questionnaire 39 (PDQ-39)), handgrip strength (dynamometer).
Other outcomes: changes in quality of life related to digestion (Digestion-associated quality of life questionnaire), stress (10-point severity Likert scale), physical activity (International Physical Activity Questionnaire).
Secondary: Frequency and severity of adverse events, changes in health-related quality of life (EuroQol 5L), NAD-metabolites in whole blood and CSF (HPLC-MS and the NADmed method), Non-motor symptoms (MDS-NMS), cognition (MoCA), gastrointestinal dysfunction (modified GIDS-PD), PD symptoms (MDS-UPDRS).
Other outcomes: Change in gene and protein expression levels related to lysosomal and proteasomal function (RNA sequencing), proteomics (LC-MS)), levels of one carbon metabolism metabolites (HPLC-MS metabolomics in whole blood and CSF), levels of monoamine neurotransmitters in CSF, genomic distribution of DNA methylation and levels of DNA methylation (Illumina Infinium MethylationEPIC Kit), levels of histone acetylation and genomic distribution of histone acetylation (immunoblotting and chromatin immunoprecipitation sequencing (ChIPseq)), gut microbiome composition (assessed by metagenomics in fecal samples), fecal metabolomics and levels of inflammatory cytokines in serum and CSF (ELISA method).
Secondary: Change in cognition (Montreal Cognitive Assessment), severity of autonomic symptoms (Autonomic Scale for Outcomes in Parkinson’s Disease), spatio-temporal parameters of the gait (investigated by GaitRite system), quality of sleep (Pittsburgh Sleep Quality Index), mood (Beck depression inventory), change in quality of life (Quality of life questionnaire).
Secondary: Root mean square of center of pressure (COP) during the instrumented Sway test (iSWAY) eyes open condition, cognition (Montreal Cognitive Assessment), mobility (Timed Up and Go Test), balance (Mini Balance Evaluation Systems test), gait speed (Protokinetics Zeno™ Walkway), cognition (Wechsler Adult Intelligence Scale III and IV (WAIS-III, WAIS IV) Digit Symbol Coding test, Parkinson’s Disease (PD)-Cognitive Rating Scale, Boston Naming Test, California Verbal Learning Test-II, Stroop Color Word Interference Test, Delis-Kaplan Executive Function System (DKEFS) Trail Making Test, DKEFS Sorting, Digit Span test, Matrix Reasoning test, Benton Judgement of Line Orientation scores, the Controlled Oral Word Association or “FAS” test, reaction time test.
Secondary: Mean change from baseline to week 104 in disease severity, individual cognitive domain z scores on comprehensive neuropsychological testing, MDS-UPDRS total score, quality of life (Parkinson’s Disease Questionnaire-39), MDS-UPDRS part II and III, mean change in levels of blood-based biomarkers (including total antioxidant status TAS, oxidative stress biomarkers and αsynuclein), difference of type and incidence of Adverse Events (AEs) and Serious AEs, Schwab and England Activities of Daily Living (SE-ADL) scale.
Secondary: Gut microbial remodeling (change in fecal butyrate-producing bacteria based on high-throughput amplicon sequencing of the V4 variable region of the microbial 16s ribosomal ribonucleic acid (RNA).
Secondary: Suction and ab-ingestis events related to textured food change (Novel Non-Motor Symptoms Scale for Parkinson’s Disease - Domain 6: Gastrointestinal tract).
Other: Functional parameters related to malnutrition, gait speed, grip strength, muscle mass, Body Mass Index (BMI), arm circumference, biomarkers (plasma cholinesterase concentration (U/ml), plasma transferrin concentration (mg/dL), plasma albumin concentration (g/dL), lymphocyte count in 1 microliter (µL) of blood), nutritional status evaluation (measured by phase angle and derived body composition data and Mini Nutritional Assessment Score (MNA) score) and bolus transit time evaluation.
Secondary: Resting blood pressure, heart rate, abdominal circumference, Waist to Hip Ratio, Body Mass Index (kg/m2), Disease Activity Score 28, Health Assessment Questionnaire (HAQ), Simplified Disease Activity Index Score (SDAI), Hannover Functional Ability Questionnaire (Funktionsfragebogen Hannover, FFbH-R), MDS-UPDRS, Parkinson’s Disease Sleep Scale-2 (PDSS-2), Parkinson’s Disease Questionnaire-39 (PDQ-39), Non-motor symptoms questionnaire (NMSQ), Non Motor Symptoms Scale (NMSS), Stress questionnaire (Cohen Perceived Stress Scale, CPSS), Quality of Life questionnaire (WHO-5), Hospital Anxiety and Depression Scale (HADS), Mood questionnaire (Profile of Mood States, POMS). [Change over baseline to 12 months] Sociodemographic Measurements. [Baseline] Behavioral Factors, such as: Physical inactivity, coffee, health promoting activities, alcohol consumption and smoking status. Dietary behavior, expectation questions, differential blood count, Hepatic transaminases (GPT, GOT) and Gamma glutamyl transpeptidase (y-GT), Bilirubine (total, direct, indirect in mg/dL), Total protein in grams per liter (g/L), Albumine in grams per liter (g/L), Creatinine in µmol per liter (µmol/L), estimated glomerular filtration rate (eGFR) in milliliter per minute (mL/min), Alkaline Phosphatase in units per liter (U/L), Urea in milligrams per deciliter (mg/dL), blood lipids and fasting glucose (triglycerides (mmol/L), total cholesterol (mmol/L), LDL (mmol/L), HDL (mmol/L), fasting glucose (mmol/L)), HbA1C (mmol/mol Hb, %), TSH (mU/L), IGF-1 (ng/mL), Insulin (mU/L), High sensitive CrP (mg/L), Rheumatoid factor (RF, IgM) (U/mL), Anti-cyclic citrullinated peptide (ACPA) (U/mL), Zonulin (ng/mL), Fatty acid binding protein 2 (FABP2) (pg/mL), Plasma Calprotectin (µg/g), Fecal Calprotectin (µg/g), Phenotyping of immune cells, Urine metabolomics (10 ml midstream urine) and oral microbiota analysis in saliva.
DISCUSSION
This review shows that research on dietary interventions in PD is still in its early days. We found a limited number of 10 ongoing phase I, phase II, or even more exploratory trials (i.e., open label, no control group). These studies will not be able to definitively answer critical questions about effectiveness but are expected to further shape our ideas about which dietary patterns or supplements may be worth investigating in a more rigorous way. In addition, most of these studies will look into the working mechanisms and biological responses which will offer essential information on the potential of disease modification induced by nutritional supplements. Unfortunately, very little attention is being paid to compliance, which can be quite a challenge for nutritional interventions.
The ongoing trials all investigate a different dietary pattern or nutritional supplement. Considering the dietary patterns, the Mediterranean diet and ketogenic diet are popular and have been subject of previous studies, but without conclusive evidence []. Observational studies previously suggested that the Mediterranean diet is associated with a delay in disease onset [42] and a reduction in disease progression [37]. A previous randomized controlled trial showed that the Mediterranean diet positively affected executive functioning, attention, language, and memory [39]. The Mediterranean diet is further thought have an anti-inflammatory effect, to reduce oxidative stress, C reactive protein, and fasting insulin, and to improve gut microbiota [26]. The exact mechanisms have, however, not been elucidated. Current work will not help to unravel these potential neurobiological effects since only one study on the Mediterranean diet is currently being performed (study 1). This study primarily aims to assess the safety and adherence of this intervention. Moreover, this study combines the Mediterranean diet with a ketogenic diet. The ketogenic diet is based on a high percentage of lipids and a low percentage of carbohydrates and proteins. A concern of the ketogenic diet is that it reduces appetite and carries a risk of malnutrition. It is also difficult diet to adhere to in the long term because of the effort it requires to prepare meals and the consequences related to the social aspects of eating together. A pilot randomized controlled trial showed that it is safe for people with PD to follow a ketogenic diet for 8 weeks. The diet also improved non-motor symptoms [43]. A combination with the Mediterranean diet, as is currently being studied, may be a more attractive alternative on the long term. This new study (study 1, Table 1) will give insights into the safety, adherence, and retention of this combined dietary pattern. Other ways to reach a ketogenic state is by supplementation of ketones (ongoing study 11) or by fasting (ongoing study 6). As such, this ongoing work will give more insights into the biological mechanisms of these two options. None of these studies is primarily aiming to study the effectiveness of ketogenesis. The other dietary pattern currently being studied is a gluten free diet (study 5, Table 1), which has not been studied before in PD.
Among the supplements, we found studies supplementing vitamin B3, vitamin E, carbohydrate, and fiber. While the additional value of vitamin intake in PD remains controversial, some vitamins have been proposed to have a beneficial effect. Vitamin B3 is hypothesized to have a neuroprotective effect through multiple mechanisms, e.g., an anti-inflammatory effect, oxidative stress reduction, and perhaps dopamine suppletion [44]. In an open label effectiveness trial, 12 months of daily 250 mg vitamin B3 supplementation resulted in reduced PD motor symptoms (primary outcome), increased handwriting size, mood, and postural control and decreased fatigue [45]. The ongoing study included here (study 4, Table 1) is a double-blind placebo-controlled study, which uses a much higher dosage (of up to 3000 mg per day) than in the previous study aiming to investigate optimal dosage and safety. Vitamin E has also been shown to have some positive effects on disease symptoms [46] and on the inflammatory profile in small sample of people with PD [47]. The large DATATOP study in the 90’s, however, in which deprenyl and vitamin E were provided, did not show a potential for disease modification []. The study included here (study 7, Table 1) is a phase II study including a larger cohort and a longer follow-up period. Importantly, this study will evaluate clinical as well as mechanistic outcomes. There is little evidence on the potential role of macronutrients (carbohydrates and fats) in the development and progression of PD [26]. Study 8 will give some preliminary evidence on supplementing carbohydrates. Interestingly, we did not find any ongoing clinical trial on for example caffeine (despite its fairly consistent negative association with the risk of developing PD), minerals, proteins or flavonoids, for all of which associations with PD have been found previously [].
At this point, no specific recommendations on diet or supplements can be given based on rigorous scientific research. However, healthy nutrition in general is important for everyone, including people with PD who improve their functioning by reducing obstipation (thereby increasing levodopa efficiency) and by preventing malnutrition, deficiencies, and weight loss. Two ongoing studies specifically aim at reducing obstipation (study 3) and preventing malnutrition by offering food with different textures to people with dysphagia (study 10). A study that would be of great interest is to evaluate the effect of offering personalized guidance by an expert dietician to people with PD, giving them individualized advice tailored to their usual food intake and diet-related symptoms or problems (e.g., obstipation, timing of protein intake). Although this type of intervention is not intended to alter the course of the disease, it could potentially have an enormous clinical effect because of the various symptomatic improvements [41].
The current overview shows that various different nutritional interventions are being studied, mainly with respect to their biological response, safety and preliminary clinical effects. These are important steps that must be taken before moving towards larger and more rigorously designed clinical trials, which come with great challenges because of numerous methodological considerations. A first challenge relates to how a dietary intervention is delivered. Especially when a dietary pattern is being studied, the options of ‘feeding’ (i.e., providing all foods or ingredients during the study period) or ‘dietary counseling’ both have advantages and disadvantages. Especially when counseling is performed, the exact dosing becomes more difficult to monitor and tracking compliance will be challenging. Feeding, on the other hand, is more expensive, certainly for the undoubtedly large-scale studies that will be necessary, and also not realistic when considering future implementation strategies in real life [52]. For studies evaluating a supplement, adherence is less complicated, but still challenging. It is also easier to design a placebo intervention when testing supplements, thus ascertaining blinding, while this is practically impossible when studying a dietary pattern. Only one of the studies included in this overview clearly indicated how adherence will be monitored (study 1). Surprisingly, none of the studies explicitly stated how they will register dietary intake which may obviously be a serious confounder in this type of study [52].
While there is increasing evidence that diet could play an important role in the development, progression and management of clinical manifestations of person with PD. Diet may also empower PwPD in self-management. The field clearly needs more high-quality research. This is not an easy task, given the great variety in potentially important diets and nutritional supplements, the great number of possible working mechanisms and the many methodological challenges related to studying nutrition. We expect that the ongoing studies reviewed here will result in valuable information that can help to design future clinical trials. Research on diet and nutrition in PD is still in its’ early days but holds great promise and opportunities for the future.
Footnotes
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The Center of Expertise for Parkinson & Movement Disorders was supported by a center of excellence grant of the Parkinson’s Foundation.
NmdV reports grants from The Netherlands Organisation for Health Research and Development (ZonMw), The Michael J Fox Foundation, and Verily Lifes Sciences.
CONFLICT OF INTEREST
BRB has received honoraria from serving on the scientific advisory board for AbbVie, Biogen, and UCB; has received fees for speaking at conferences from AbbVie, Zambon, Roche, GE Healthcare, and Bial; and has received research support from the Netherlands Organization for Scientific Research, The Michael J. Fox Foundation, UCB, AbbVie, the Stichting Parkinson Fonds, the Hersenstichting Nederland, the Parkinson’s Foundation, Verily Life Sciences, Horizon 2020, the Topsector Life Sciences and Health, the Gatsby Foundation, and the Parkinson Vereniging. BRB currently serves as co-Editor-in-Chief of this journal but was not involved in the peer-review process nor had access to any information regarding its peer review. He also serves on the editorial board of Practical Neurology and Digital Biomarkers.
NMdV is an Editorial Board member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer review.
All other authors have no conflict of interest to report.
