Abstract
Background
The role of dietary factors as risk or protective factors for Parkinson's disease (PD) remains debated.
Objective
This retrospective case-control study aimed to evaluate the associations between foods identified through a data-driven analysis and PD, and to compare the relevance of dietary versus non-dietary factors as contributors to PD risk.
Methods
The study included 680 PD patients and 612 matched controls recruited from six Italian neurology centers. Dietary data were collected using a validated 77-item food frequency questionnaire, and factor analysis was conducted to identify groups of correlated foods (i.e., factors). Logistic regression models were used to assess the associations between these factors and PD, while non-dietary factors were subsequently included in the model for comparison.
Results
Seven factors were identified, four of which were significantly associated with PD. High consumption of sweets (Factor 1), red meat (Factor 3), and processed meats (Factor 6) was associated with an increased PD risk, whereas a high fruit intake (Factor 2) was protective. These associations remained significant after adjusting for other known non-dietary risk/protective factors. While the increased PD risk associated with dietary factors was weaker than that of non-dietary factors, protective dietary and non-dietary factors showed comparable effects in reducing PD risk.
Conclusions
Data-driven analysis identified foods potentially influencing PD risk, although non-dietary factors demonstrated a greater impact on PD risk. These findings highlight the need to integrate both diet and lifestyle habits into future PD research and prevention strategies.
Plain language summary
Parkinson's disease (PD) is a neurodegenerative disorder that affects millions of people worldwide. Scientists are still studying what causes PD, and some think that diet might play a role. In our study, we looked at how different eating habits might be linked to PD. We asked 680 people with PD and 612 people without PD about their food consumption habits. Then, we grouped these foods to see if certain eating habits were more common or uncommon in people with PD. We found that people who often ate sweets, red meat, and processed meats had a higher chance of having PD. On the other hand, people who ate a lot of fruit seemed to have a lower risk. However, we also found that other factors—such as family history, exposure to pesticides, oils, metals, and general anesthesia—had a bigger effect on PD risk than diet alone. This study suggests that eating habits might have some impact on PD, but they are not the main cause. Future research should look at both diet and other lifestyle habits to better understand how to prevent PD.
Keywords
Introduction
Parkinson's disease (PD) is considered a multifactorial disease with numerous risk and protective factors contributing to its pathogenesis.1,2 Given the evolving recognition of gastrointestinal involvement in PD and the potential interaction between the gut and brain, 3 several controlled studies have examined the effect of dietary factors on PD risk. Studies investigating dietary factors in PD can be classified into two categories: (i) studies assessing the effect of individual foods,4–7 and (ii) studies assessing the combined effects of several foods.8–11 However, investigations into the effects of single foods have yielded variable results across studies, likely due to differences in dietary habits among populations. Examining food associations rather than individual foods offers a more comprehensive approach. Most studies following this approach have relied on measuring adherence to pre-defined dietary patterns, such as the Mediterranean diet, the Mediterranean-DASH Diet Intervention for Neurodegenerative Delay (MIND) and the Dietary Approaches to Stop Hypertension DIET (DASH).9–11 Notably, these studies did not identify specific foods as potential risk factors.
An alternative approach to identifying food associations relevant to PD is the use of a data-driven methodology. To our knowledge, only a few controlled studies (two longitudinal cohort studies and two retrospective case-control studies)12–15 have examined data-driven dietary patterns potentially linked to PD. Three of these four studies failed to identify any data-driven dietary pattern significantly associated with PD risk,12,14,15 while one cohort study reported an association between a dietary pattern characterized by high intakes of fruit, vegetables, whole grains, legumes, poultry, and fish and a lower PD risk. 13 Several factors, including relatively small sample sizes, recruitment of unmatched controls, and the lack of simultaneous assessment of a wide range of potential confounding variables, may explain the inconsistency in results.
To address the role of foods in the development of PD more effectively, we designed a large case-control study examining a wide range of foods in an Italian population. In this study, factor analysis was used to determine whether specific food associations are present in the Italian PD population. The identified food associations were compared between PD patients and control subjects using a logistic regression model, accounting for several known non-dietary risk/protective factors for PD as potential confounders. Finally, the simultaneous assessment of dietary and non-dietary risk/protective factors allowed us to measure and compare their relative effect sizes. We hypothesized that distinct foods may be associated with PD risk, and that their relative contribution may differ from that of established non-dietary risk/protective factors.
Methods
Study subjects
This is a retrospective case-control study conducted across six Italian neurology centers from September 2018 to September 2019. The methodology used to consecutively enroll patients and controls has been described previously. 16 In brief, idiopathic PD was diagnosed by senior neurologists with expertise in movement disorders using published standard criteria.17,18 Patients with monogenic PD and other forms of parkinsonism were excluded. Healthy controls were identified among relatives of neurologic outpatients without PD who visited the neurology departments during the study period. Controls were frequency-matched to cases by 5-year age stratum, sex, and referral center. Potential controls were excluded if they had received treatment with drugs known to induce parkinsonism or were related to a case in the study. Both cases and controls were unaware of the study hypothesis. The case-control ratio was approximately 1:1.
Diet questionnaire
Dietary data were collected using a validated 77-item food-frequency questionnaire (FFQ) developed for a multicenter case-control study on diet and cancer in Italy.19,20 We used this questionnaire because it was designed and validated for the Italian population: the questionnaire considered food items that explained the largest amount of variance in nutrient intake among Italian control subjects, as well as those foods that made a large contribution to total absolute nutrient and caloric intake in Italian control subjects.19,20 To account for the seasonal variation of food consumption, the frequency of use of some foods was asked for the season in which the food is mainly available (e.g., tomatoes in summer, oranges in winter, etc.). The FFQ assessed food consumption frequencies prior to the onset of motor signs for cases, and for the same pre-exposure period for matched controls. The pre-exposure period was defined by asking controls to report their dietary habits during the same calendar period as their matched PD case's pre-onset phase. Participants specified the frequency of consumption of each food item, categorized as: more than once a week (score: 1.3), once a week (score: 1), less than once a week but more than once a month (score: 0.7), less than once a month (score: 0.3), or never/less than once a year (score: 0). Vitamin D supplementation was recorded. The questionnaire was administered in person by trained medical interviewers to ensure consistency and reliability.
Non-dietary risk/protective factors
The non-dietary factors we considered in this study were from a large case-control study allowing the simultaneous evaluation of 31 putative risk/protective factors of PD, including environmental and lifestyle habits, comorbid conditions, and drugs. 16 In the present study, we considered only those risk/protective factors that yielded significant independent association with PD, i.e., coffee consumption, smoking, physical activity, family history of PD, dyspepsia, and exposure to pesticides, petroleum oils, metals, and general anesthesia. 16 Coffee consumption was included among the non-dietary risk/protective factors because it did not load on any of the factors identified in the factor analysis and is often considered a lifestyle habit, similar to smoking. Non-dietary risk factors previously associated with PD were assessed using a semi-structured questionnaire administered alongside the diet questionnaire. 16 First-degree relatives were considered affected by parkinsonism only if additional family members provided medical documentation or if an investigator confirmed the diagnosis. Participants were also asked about their occupation using questions from a prior population-based case-control study. Occupational exposure to pesticides, oils, metals, solvents, and paints was evaluated. 21 Smoking and coffee habits were assessed using a semi-structured questionnaire previously employed in dystonia studies. 22 Questions referred to the period before the onset of motor signs, and participants were classified as never-smokers or non-drinkers accordingly. The instrument was not specific for dystonia but only allowed information to be collected in a standardized manner. Physical activity prior to the onset of motor signs was evaluated by converting 17 selected activities into metabolic equivalents of task using the Pate model.23,24
Statistical analysis
Data cleaning included range and consistency checks. Analyses were performed using STATA 11 (StataCorp, College Station, TX). Data were expressed as percentages or mean ± SD, and groups were compared using χ2 tests, t-tests, or one-way analysis of variance with post hoc tests, as appropriate.
Given the large number of foods we investigated, we used factor analysis, an established statistical method, to identify food associations. The advantages of this methodology include reducing data complexity, identifying hidden factors, simplifying the interpretation of data by grouping variables together under a single factor 25 ; disadvantages include subjectivity in making decisions about the number of factors to extract, and overfitting which occurs when the model fits the data too closely and is unable to generalize to new data. Factor analysis included principal component analysis (PCA) and exploratory factor analysis (EFA). PCA conducted on the 77 dietary variables (expressed as food intake frequency) classified food items based on correlation and relevance before extracting the food patterns. The retained factors were made orthogonal using varimax rotation. Factors with eigenvalues greater than 1.0 were retained, and variables with factor loadings <0.3 were excluded.
To assess associations between foods and PD, non-conditional logistic regression models adjusted for potential confounders were computed for cases and controls. Multivariate analysis began with all study variables in the initial model. Variables were iteratively removed based on p-value significance and changes in odds ratios (ORs), resulting in a main-effects model. Estimates were adjusted for age, sex, education level, and referral center. Collinearity was assessed using correlation coefficients, ensuring no values approached +1.
For dietary factors significantly associated with PD in multivariate modeling, correlations between exposure and disease duration (measured by motor symptom onset) were evaluated among cases. Spearman's rank correlation analysis was also used to assess relationships between age and selected dietary factors. All statistical tests were two-tailed, and significance was set at p < 0.05.
Ethical approval and consent
The study received approval from the local ethics committee on human experimentation (Sapienza University of Rome Ethics Committee, No. 4734). Written informed consent for research participation was obtained from all patients.
Results
A total of 1292 participants (680 PD patients and 612 healthy controls) met the eligibility criteria during the study period, agreed to participate, and completed both the study and risk factor questionnaires. The demographic and clinical characteristics of PD patients and control subjects, including non-dietary risk factors previously identified in this population, are summarized in Table 1. The median time between the onset of motor symptoms in PD cases and questionnaire administration was 5 years (IQR: 3–8 years). Cases and controls were similar in sex distribution and body mass index but differed in age and years of education. Factor analysis identified seven major factors among PD patients. Rotated factor loadings with absolute values greater than 0.30 for individual food items are presented in Table 2. The retained food variables were categorized as follows: Factor 1 (six types of sweets), Factor 2 (six types of fruits), Factor 3 (six types of red meat), Factor 4 (five types of salad), Factor 5 (cruciferous vegetables, peas, and beans), Factor 6 (cured meats), and Factor 7 (alcoholic beverages including wine, beer, and grappa). Unadjusted comparisons between cases and controls showed significant associations for Factors 1, 3, 5, and 6 with PD (Table 3). When analyzing individual foods within these factors, most foods in Factors 1 (sweets), 3 (red meat), and 6 (cured meat) were directly associated with increased PD risk. Conversely, citrus fruits within Factor 2 contributed significantly to the inverse association with PD (Supplemental Table 1).
Demographic and clinical features and non-dietary risk factor profile of Parkinson's disease (PD) case patients and healthy control subjects.
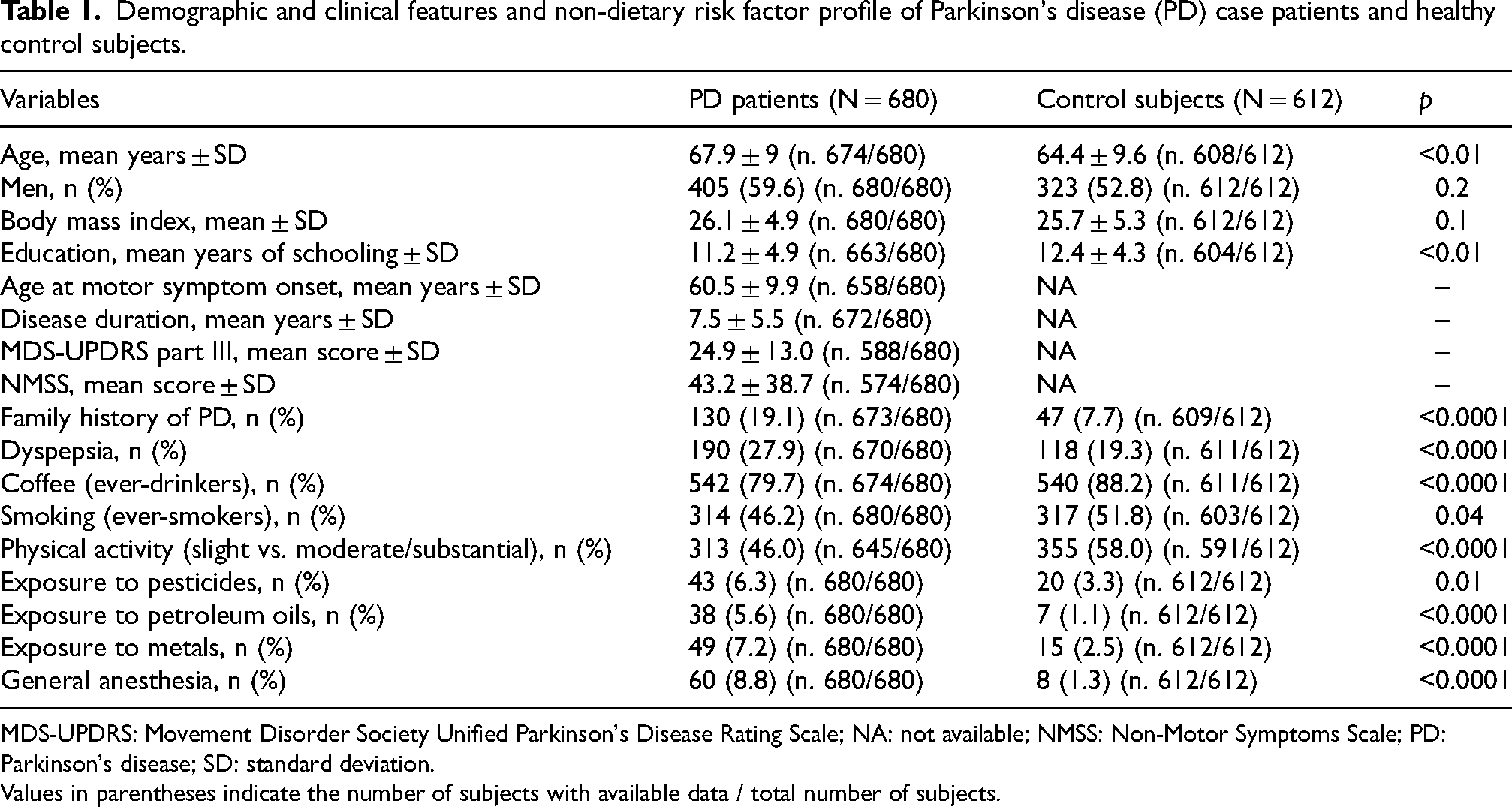
MDS-UPDRS: Movement Disorder Society Unified Parkinson's Disease Rating Scale; NA: not available; NMSS: Non-Motor Symptoms Scale; PD: Parkinson's disease; SD: standard deviation.
Values in parentheses indicate the number of subjects with available data / total number of subjects.
Results of factor analysis. Rotated factor loadings (pattern matrix) and unique variances.

Comparison of the overall food frequency in Factors 1 to 7 between cases and controls.
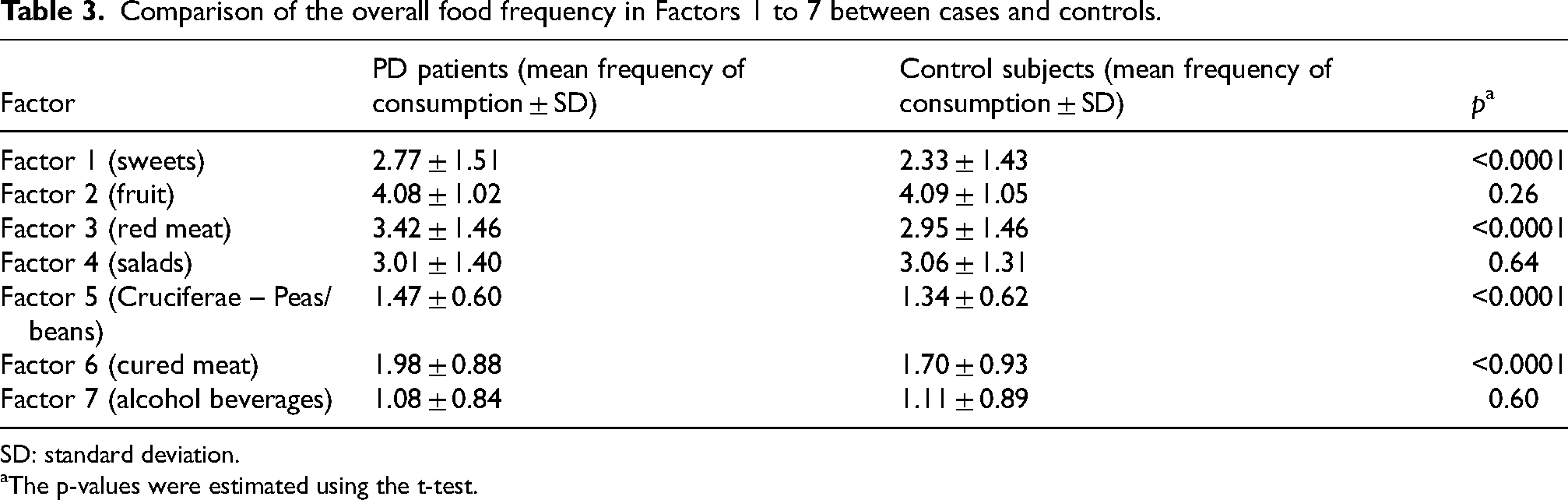
SD: standard deviation.
The p-values were estimated using the t-test.
The initial multivariate analysis model included age, sex, years of education, referral center, dietary Factors 1 to 7, and known non-dietary risk/protective factors for PD (family history of PD, coffee consumption, smoking, physical activity, dyspepsia, and exposure to pesticides, petroleum oils, metals, and general anesthesia). The final main-effects model identified the following independent risk factors: dietary Factors 1 (sweets), 3 (red meat), and 6 (cured meat), family history of PD, dyspepsia, and exposure to pesticides, petroleum oils, metals and general anesthesia. Protective factors included dietary Factor 2 (fruit), coffee consumption, and cigarette smoking. Physical activity was excluded from the final model due to confounding by dietary Factors 1, 3, and 6. Table 4 presents the strength of the estimates from the main-effects model, indicating that non-dietary factors provided a potential greater contribution to PD risk than dietary factors. A Directed Acyclic Graph illustrating factor inclusion/exclusion rationale is provided in the Supplemental Figure 1.
Results of multivariable logistic regression analysis in 680 cases with Parkinson's disease and 612 controls (main-effects model).
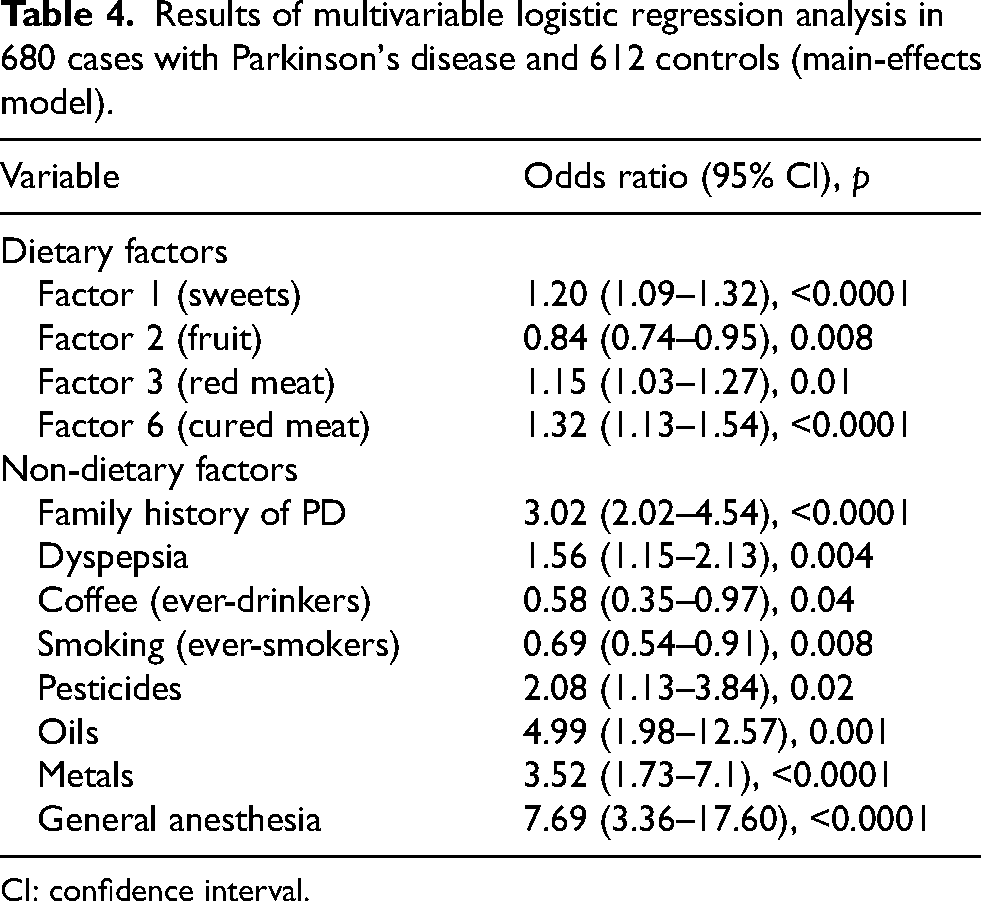
CI: confidence interval.
Spearman correlation analysis showed no significant correlation between PD duration and the frequency of Factors 2 (r = 0.04, p = 0.37), 3 (r = 0.02, p = 0.65), or 6 (r = −0.06, p = 0.13) in PD patients. However, a significant positive correlation was observed between PD duration and Factor 1 (r = 0.1, p = 0.03). In both PD patients and healthy controls, significant correlations were observed between age and Factor 1 (PD patients: r = −0.21, p < 0.0001; control subjects: r = 0.14, p = 0.001) and Factor 6 (PD patients: r = −0.1, p = 0.01; control subjects: r = −0.11, p = 0.008). No significant correlations were found between age and Factor 2 (PD patients: r = −0.01, p = 0.76; control subjects: r = 0.03, p = 0.51) or Factor 3 (PD patients: r = −0.03, p = 0.4; control subjects: r = −0.03, p = 0.53).
Discussion
Factor analysis identified seven factors, four of which were significantly associated with PD. Factor 1 (characterized by six types of sweets), Factor 3 (characterized by six types of red meat), and Factor 6 (characterized by cured meats) were associated with an increased PD risk. In contrast, Factor 2 (comprising six types of fruit) was associated with a reduced PD risk. For Factors 1, 3, and 6, most foods contributed similarly to increased PD risk, whereas the protective effect of Factor 2 was primarily driven by the presence of citrus fruits. The associations between PD and these foods remained statistically significant when included in a logistic regression model accounting for non-dietary risk factors identified in the same population. However, non-dietary factors had a markedly stronger influence on PD risk or protection than dietary factors.
An inherent limitation of the case-control design is the difficulty in establishing the temporal sequence between exposure and disease. Given the prolonged and often insidious onset of PD, diet may evolve following PD onset, leading to cause-and-effect bias. Despite this, stratification of cases based on disease duration revealed no increase in the relative frequency of consumption of dietary factors as the disease progressed, suggesting that the associations are unlikely secondary to the disease itself. However, the significant correlation between PD duration and Factor 1 raises the possibility of bias in this specific case. Notably, age showed opposite correlations with Factor 1 (sweets) in patients and controls, suggesting that age-related consumption patterns may have influenced the observed association. Although age was included as a covariate in the multivariate models, some residual confounding may persist and should be considered when interpreting this finding.
Correlation analysis highlighted two critical points. First, the frequency of intake for Factors 2 (fruits) and 3 (red meat) did not vary with age in either PD patients or controls. Second, the frequency of consumption for Factor 6 (cured meat) tended to increase with decreasing age in both cases and controls. These findings suggest that the associations between PD and Factors 2 (fruits), 3 (red meat), and 6 (cured meat) are relatively stable across age, supporting their potential role as risk modifiers for PD. These observations align with previous studies showing associations between fruits (Factor 2), red meat (Factor 3), and cured meat (Factor 6) and PD risk. The protective effect of fruits in this study was primarily due to the inverse association with citrus fruits, which may offer neuroprotective benefits through bioactive compounds such as flavonoids, vitamin C, and terpenes, which have antioxidant and anti-inflammatory properties.26–28 These mechanisms reduce oxidative stress and inflammation, key contributors to dopaminergic neuron degeneration in PD. 29 Further research is needed to confirm these findings in clinical settings.
The association between PD risk and red and processed meat also has strong biological plausibility. High levels of saturated fats, heme iron, and nitrites in these foods contribute to oxidative stress, inflammation, and neurodegeneration. 30 Advanced glycation end products (AGEs) formed during high-temperature cooking exacerbate oxidative damage and promote inflammatory pathways, further increasing PD risk. 31 These mechanisms strengthen the biological rationale for the observed associations.
Our analysis did not identify a risk-modifying role for certain foods (e.g., milk, animal-based fats, and vegetables) previously linked to PD risk in other populations.1,2,32–34 Although carbohydrates (excluding free sugars) are a major source of energy in the Italian diet, we could not identify any dairy or carbohydrate group in our PD sample. It is possible that this reflects the specific dietary habit of the Italian PD population. 35 Similarly, some dietary component previously described as protective factors, 34 such as vegetables (Factors 4 and 5), were not significantly associated with PD risk in our population. This does not necessarily imply a lack of biological plausibility or potential benefit, but rather reflects the relative weight of these foods within the overall dietary landscape captured by our analysis.
A novel finding from this study is that the identified foods were largely independent of widely accepted non-dietary risk/protective factors. Most non-dietary factors were also independent of the identified foods. Additionally, the significant protective association between physical activity and PD risk observed in prior studies 16 may result from confounding by dietary factors such as sweets, red meat, and processed meat. Given the protective role of physical activity against other neurodegenerative diseases like Alzheimer's disease, 36 this finding raises the possibility that the observed benefits of physical activity may reflect related dietary habits.
Another important contribution of this study is the simultaneous assessment of dietary and non-dietary risk/protective factors in the same sample. This allowed for a direct comparison of their relative effect sizes. Interestingly, the estimates of the associations between PD and dietary risk factors (Factors 3 and 6) were weaker than the estimates from non-dietary risk factors (e.g., family history, pesticide exposure). The strong association observed between exposure to general anesthesia and PD risk raises the question of whether anesthesia itself may contribute to PD development, possibly through neurotoxic or inflammatory mechanisms, or whether it reflects surgical procedures undertaken for prodromal conditions (e.g., chronic constipation, autonomic dysfunction, urinary disturbances) that precede PD diagnosis. Future prospective studies are warranted to clarify the nature of this association. In contrast, the protective effects of Factor 2 (fruits) and non-dietary factors such as coffee consumption and smoking were of similar strength.
This study has strengths and limitations. While selection bias due to the hospital-based design cannot be entirely ruled out, the demographic and clinical features of the study population closely resemble those of the general PD population in Italy. 34 The large sample size provided sufficient statistical power to examine all variables, 37 supporting the exclusion of non-significant factors like Factors 4 (salads), 5 (cruciferous vegetables, peas, and beans), and 7 (alcohol beverages), and physical activity. The recruited population was relatively homogeneous in terms of geographic origin and ethnicity, minimizing the potential for regional variability in risk factors. Adjustments for recruiting center further mitigated this concern. There was a small age difference between cases and controls (<5 years), but this was consistent with the study's matching criteria and age adjustments in the multivariate analysis further minimized potential confounding effects. Given the retrospective assessment, information from the FFQ may have been burdened by recall bias. However, stratification of cases based on disease duration revealed no change in the relative frequency of consumption of dietary factors as the disease duration increased; and a differential recall between case and control subjects was unlikely because food consumption frequencies were assessed prior to PD onset for cases, and for the same pre-exposure period for matched controls. Dietary habits may have been modified after disease onset, introducing a potential bias. Although we sought to minimize this risk by assessing the stability of the identified foods and evaluating correlations with disease progression, such bias cannot be completely ruled out. A further limitation is the absence of a nutrition researcher in the study team. Although we relied on a validated FFQ and structured administration procedures, the administration of the FFQ and the interpretation of food data might have benefited from the involvement of researchers with specific expertise in nutritional epidemiology. In addition, administration of the FFQ only in one time point may have limited the ascertainment of daily intake. The generalizability of our findings to non-Italian or non-Mediterranean populations may be limited due to cultural differences in dietary habits and lifestyle. Finally, our statistical approach used a combination of factor analysis and case-control comparison (by using a logistic regression model accounting for several known non-dietary risk/protective factors for PD as potential confounders) which allowed better performance and more accurate results.
In conclusion, this study identified associations suggesting that high consumption of meat and processed meat may be linked to increased PD risk, while fruit consumption may be associated with reduced risk. These associations were independent of established non-dietary risk factors, although food factors may exert some confounding effects on known non-dietary factors like physical activity. Comparisons of relative effect sizes suggest that food factors may have a smaller impact on PD risk than non-dietary factors, while the possible protective effects of fruit consumption are comparable to those of coffee and smoking. These findings provide valuable insights for designing future risk factor studies and prevention programs in PD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251388058 - Supplemental material for The impact of diet on Parkinson's disease risk: A data-driven analysis in a large Italian case-control population
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251388058 for The impact of diet on Parkinson's disease risk: A data-driven analysis in a large Italian case-control population by Angelo Fabio Gigante, Barbara Vitucci, Vittorio Velucci, Roberta Pellicciari, Nicola Modugno, Sara Pietracupa, Maria Ilenia De Bartolo, Matteo Costanzo, Claudio Terravecchia, Marcello Mario Mascia, Antonella Muroni, Tommaso Ercoli, Paolo Solla, Francesca Magrinelli, Antonella Conte, Giovanni Fabbrini, Alessandra Nicoletti, Michele Tinazzi, Alfredo Berardelli, Giovanni Defazio and Daniele Belvisi in Journal of Parkinson's Disease
Supplemental Material
sj-docx-2-pkn-10.1177_1877718X251388058 - Supplemental material for The impact of diet on Parkinson's disease risk: A data-driven analysis in a large Italian case-control population
Supplemental material, sj-docx-2-pkn-10.1177_1877718X251388058 for The impact of diet on Parkinson's disease risk: A data-driven analysis in a large Italian case-control population by Angelo Fabio Gigante, Barbara Vitucci, Vittorio Velucci, Roberta Pellicciari, Nicola Modugno, Sara Pietracupa, Maria Ilenia De Bartolo, Matteo Costanzo, Claudio Terravecchia, Marcello Mario Mascia, Antonella Muroni, Tommaso Ercoli, Paolo Solla, Francesca Magrinelli, Antonella Conte, Giovanni Fabbrini, Alessandra Nicoletti, Michele Tinazzi, Alfredo Berardelli, Giovanni Defazio and Daniele Belvisi in Journal of Parkinson's Disease
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
ORCID iDs
Ethical considerations
The study received approval from the local ethics committee on human experimentation (Sapienza University of Rome Ethics Committee, No. 4734).
Consent to participate
Written informed consent for research participation was obtained from all patients.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received funding from the Italian Ministry of Health “Progetto di Ricerca Corrente – IRCCS Neuromed”.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data are available upon reasonable request. Requests for data sharing can be sent to the corresponding author, Barbara Vitucci, at the email address brbrvitucci@gmail.com.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
