Abstract
Background:
The ability to encode and consolidate motor memories is essential for persons with Parkinson’s disease (PD), who usually experience a progressive loss of motor function. Deficits in memory encoding, usually expressed as poorer rates of skill improvement during motor practice, have been reported in these patients. Whether motor memory consolidation (i.e., motor skill retention) is also impaired is unknown.
Objective:
To determine whether motor memory consolidation is impaired in PD compared to neurologically intact individuals.
Methods:
We conducted a pre-registered systematic review (PROSPERO: CRD42020222433) following PRISMA guidelines that included 46 studies.
Results:
Meta-analyses revealed that persons with PD have deficits in retaining motor skills (SMD = –0.17; 95% CI = –0.32, –0.02; p = 0.0225). However, these deficits are task-specific, affecting sensory motor (SMD = –0.31; 95% CI –0.47, –0.15; p = 0.0002) and visuomotor adaptation (SMD = –1.55; 95% CI = –2.32, –0.79; p = 0.0001) tasks, but not sequential fine motor (SMD = 0.17; 95% CI = –0.05, 0.39; p = 0.1292) and gross motor tasks (SMD = 0.04; 95% CI = –0.25, 0.33; p = 0.7771). Importantly, deficits became non-significant when augmented feedback during practice was provided, and additional motor practice sessions reduced deficits in sensory motor tasks. Meta-regression analyses confirmed that deficits were independent of performance during encoding, as well as disease duration and severity.
Conclusion:
Our results align with the neurodegenerative models of PD progression and motor learning frameworks and emphasize the importance of developing targeted interventions to enhance motor memory consolidation in PD.
INTRODUCTION
Parkinson’s disease (PD) is one of the most common neurodegenerative disorders and the number of persons with this clinical condition is expected to double by 2040 [1]. PD is a complex, heterogeneous, and progressive disorder, characterized by several motor and non-motor symptoms but its diagnosis is based on the onset of the cardinal motor features of the disease [2]. Pharmacological treatments are the first line of action to manage the motor symptoms of the disease but with time they tend to progressively lose efficacy [3]. As a result, patients with PD experience a relentless deterioration, leading to major motor dysfunctions, and eventually loss of autonomy [4]. Implementing non-pharmacological interventions to maintain the functional independence of these patients is thus important [5].
Motor rehabilitation helps patients with PD maintain the motor skills needed to function independently [6]. Motor learning, defined as the ability to acquire, adapt, and retain long-term skilled movements [7] is the base of motor rehabilitation [8]. Motor learning comprises motor memory encoding (i.e., acquisition) and consolidation (i.e., retention). Encoding, the on-line process during which sensory and motor information is acquired through deliberate or structured motor practice, is usually characterized by fast gains in skill performance during the initial phases of practice, followed by slower improvements (i.e., automatization) in later phases [9, 10]. Consolidation, in contrast, is the result of an off-line process lasting from several minutes to days, during which the sensory and motor information is used to form more stable and long-lasting motor memories that became less susceptible to interference [11–13]. Motor memory consolidation is inferred through either change in skill performance measured during retention tests, by the ability to generalize the acquired skills to other tasks assessed with transfer tests, or through savings (i.e., a more rapid rate of relearning at retention). Retention is clinically important because it reflects the permanent ability of the patients to perform a motor skill and not transient improvements in skill performance [7, 14].
Previous studies investigating deficits in motor memory consolidation in PD have reported inconsistent results [15–17]. Furthermore, the extent to which the task nature, amount of practice, type of feedback provided during motor practice, progression of the disease, and effect of antiparkinsonian medications moderate the capacity to consolidate motor skills has yet to be determined. This review, conducted following PRISMA guidelines [18] and registered in PROSPERO (CRD42020222433) [19], aimed to summarize the evidence regarding deficits in motor memory consolidation in people with PD relative to neurologically intact (NI) individuals. Determining to what extent people with PD have deficits in consolidation could stimulate the design of more targeted and effective motor rehabilitation therapies.
Task nature classification
CoM, center of mass; CoP, center of pressure.
METHODS
Eligibility criteria
The eligibility criteria of the studies included in the review were operationalized with the PECOS (population, exposure, comparator, outcomes, study design) framework [20, 21].
Search strategy
Two authors performed independently the electronic search on electronic databases (Web of Science, PubMed, Scopus, PsycINFO, SPORT Discuss) and screened the reference lists of relevant reviews [15–17, 22–32] as well as articles reviewed at the full-text level (see study selection section). The electronic search was neither language nor date restricted, but it was limited to peer-reviewed articles. The primary search was performed using the following three main terms and their variations: “Parkinson’s disease” (population), “healthy control” (comparator), and “motor learning” (outcome), combined with Boolean operators, and can be found in Supplementary Material 1. The final search was completed on December 2, 2021.
Study selection and data extraction
Two authors screened the list of titles and abstracts of articles retrieved in the search and selected potentially relevant articles for a more detailed review at full-text level. Following the screening of the articles, both authors held a meeting to compare their results. Disagreements were resolved by discussion including a third author.
Two authors extracted the following data from the studies: study design, number and characteristics of participants, characteristics of the motor task, as well as the outcomes and the endpoints used to assess motor learning. Means and standard deviations (SDs) of motor skill acquisition (i.e., encoding) and retention/transfer (i.e., consolidation) test scores were extracted. Subsequently, both authors compared their data to confirm that they were entered correctly.
When an article did not provide means and SDs and this information could be inferred from figures, data were extracted using a web-based tool (https://WebPlotDigitizer/). When this was not possible, the authors of the study were contacted. If data could still not be obtained, the study was not included in the quantitative meta-analysis and results were reported qualitatively.
Methodological quality assessment
Risk of bias at the study level was assessed by two authors that used the NIH Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies [33]. This tool, which comprises 14 items, has shown good validity to assess the methodological quality of observational studies [34]. Given the design of the studies included in the review, items 10 (exposure repeatedly measured over time) and 12 (assessors blinded to the exposure) were scored as “not applicable” and not considered for evaluating study quality [33] (Supplementary Material 2). The two authors rated each item as “yes”, “no”, or “not reported”. All responses other than “yes” indicate a risk of bias. The number of “yes” responses was used to calculate a percentage score (i.e., number of “yes”/12 * 100) and categorize studies as “good” (≥90%), “fair” (≥70, but < 90), or “poor” (<70%) [33]. Sources of bias and heterogeneity were investigated with funnel plots and Egger’s regression tests [35–37].
Main analysis and influence of moderators
We grouped studies that assessed motor memory consolidation (i.e., skill retention) following a single session of practice or after extended practice (≥2 sessions) because retention tests following single and extended practice reflect different stages of the motor memory formation process [11]. Subgroup meta-analyses investigated whether the task nature influenced motor memory consolidation. To this end, studies were classified following well-established motor learning classifications [11, 38–47] as sensory motor (SMT), sequential fine motor (SQT), visuomotor adaptation (VAT), gross motor (GMT), and speech motor (SPT) tasks (Table 1). The influence of augmented feedback [14, 15] was assessed by grouping studies based on whether extrinsic feedback was provided or not, and if so, which type: knowledge of results, performance, or both [14].
When at least 10 effect sizes were available [61], meta-regression was conducted to investigate the influence of different moderators and the interactions among them (e.g., disease duration * disease severity). Moderators included different features of PD [15] such as duration (i.e., years since diagnosis), severity of the disease (i.e., Hoehn and Yahr Score [62] and the motor score of the Unified Parkinson’s Disease Rating Scale (UPDRS) Part III Motor Examination) [63–65]. The moderating effect of the methodological quality of the studies (Supplementary Material 3) and, when possible, the effect of anti-parkinsonian medication (“on” vs. “off”), were also investigated (Supplementary Material 4).
Considering that differences in motor memory encoding (i.e., acquisition) between PD and NI groups [15, 32] could influence consolidation (i.e., retention), we also investigated its potential moderating effect using meta-regression and grouping studies that showed a significant improvement in skill performance during practice in favor of either NI individuals or PD patients, or that showed no difference between groups. We also explored deficits in skill transfer, which are reported separately in Supplementary Material 5.
When a study used different variations of the same motor task that still required similar motor and cognitive demands and it was possible to calculate multiple effect sizes, we pooled them together by creating a composite Z-score [66]. By contrast, when variations in the experimental conditions (e.g., blocked vs. random practice) were substantial, and thus potentially affecting the rate of encoding and/or consolidation [14, 67], we did not create a composite Z-score and treated the different conditions separately (Supplementary Material 6). All analyses were conducted using the primary outcome of the motor tasks.
Data analysis
Analyses were performed with R (https://www.r-project.org; version 4.0.3) using the packages meta, metafor, ggplot2, and robvis [61]. Data entered for each group included mean differences and pooled SD (SDpooled) for different endpoints, as well as the number of participants in each group. Data were analyzed as continuous variables using a random-effects model, the restricted maximum likelihood (REML), and the “Hedges” procedure method, to calculate the standardized mean difference (SMD) with a 95% confidence interval (CI) [61, 68].
To assess consolidation, we calculated the group mean difference in skill retention using end of practice and retention test scores (retention score –end of practice score) [14]. If end of practice scores could not be obtained, we used the scores of the retention test performed immediately after the end of acquisition [69]. Positive scores in the group mean difference in skill retention reflect successful consolidation. Similarly, skill acquisition (i.e., encoding) was calculated using the mean difference between scores obtained at baseline and the end of practice, either on a single session or multiple sessions (i.e., extended practice).
A p-value of less than 0.05 indicated statistical significance for an overall effect. Negative values represented worse skill consolidation for persons with PD in comparison with NI individuals. Heterogeneity was assessed with the I2 index, which was categorized as: low (0% to < 40%), moderate (≥40% to < 60%), substantial (≥60% to < 75%), or large (≥75%) [66] and its statistical significance was assessed with the Cochran’s Q test [61].
For meta-regression, we implemented the steps outlined by Harrer et al. (2019) [61] and followed the guidelines provided by Veroniki and colleagues [70]. Multicollinearity among predictors (r≥0.8) [61] was investigated using correlation matrices. To confirm the robustness of the meta-regression results and verify their true significance, we conducted permutation tests as described by Harrer et al. (2019) [61]. Permutation tests are a resampling method used to adjust the p-value of the meta-regression and thus control for type I error, which can be inflated when heterogeneity is present [71].
RESULTS
Articles retrieved
The stages of the search and review processes with the main reasons for exclusion can be found in Supplementary Material 7. The electronic search yielded 9,427 records but 59 additional studies from previous reviews were added. After removing duplicates, 4,237 abstracts were screened with 184 studies reviewed at full-text level. Ninety-five studies were excluded because they did not have a retention test, or the latter was assessed less than one hour after the end of practice (see eligibility criteria). Nineteen studies were excluded because they did not use an appropriate motor task/method to assess motor learning and thirteen studies used a study design that did not meet the inclusion criteria. Two studies used deep brain stimulation, one study included a control group with neurological conditions, and another study had a control group with an age that differed significantly from the PD group. Four studies were excluded due to the lack of a control/PD group and four studies were excluded for other reasons (e.g., duplicated data). After identifying 45 studies, one additional study [72] found in the reference list of studies reviewed was added. The review included a total of 46 studies but since it was not possible to obtain means and SDs from six studies [72–77], whose results are reported qualitatively, the meta-analyses included 40 studies [78–117].
Characteristics of the studies
A detailed summary of the 46 studies included in the review is reported in Table 2. Overall, data from 652 persons living with PD and 620 NI individuals acting as control were included. Of these participants, 550 were males and 423 were females, while the sex of 299 participants was not reported. The studies investigated mainly older adults with mean ages ranging from 52 to 74. Disease severity, which was not reported in eight studies [72, 113], ranged from I to IV on the Hoehn & Yahr scale. The mean overall scores of the motor evaluation conducted with the UPDRS part III, which was reported in 22 studies [75, 117], ranged from 8 to 33.4, indicating that the severity of motor symptoms of patients ranged from mild to moderate [65, 118]. Disease duration, which was reported in all but seven studies [81, 113], ranged from 1.3 to 11.1 years. Only three investigations [83, 88] manipulated (e.g., compared “on” vs. “off”) medication status, while two studies [87, 104] tested patients “off” medication (Table 2). Six studies did not report and/or evaluate cognitive functioning [72, 116]. In all, except five studies [74, 119] that reported small differences in cognition between groups, PD and NI participants had similar cognitive status. None of the studies that explored associations between cognitive scores and skill acquisition or retention found significant correlations [74, 107].
Forty studies [73–78, 113–117] assessed consolidation after a single session of practice, while 14 studies [72, 112] investigated consolidation after extended practice (≥2 sessions). Regarding the nature of the task, 25 studies used SMTs [72, 119]; 10 studies SQTs [73, 115]; three studies VATs [75, 94]; eight studies GMTs [81, 116], and one study SPTs [117]. Nineteen studies [75, 109–115] provided feedback in the form of knowledge of results (e.g., numeric score), three [87, 106] as knowledge of performance (e.g., movement trajectory), and four [81, 112] combined these two types of feedback. Of the remaining studies, five studies [73, 108] did not provide feedback and 16 [72, 117] did not explicitly state if feedback was provided or not.
Methodological quality
Details of the methodological quality assessment can be found in Supplementary Material 8. The percentage score and quality rating of each study are reported in Table 2. The mean±SD percentage score was 77.5±11.6% with 31 studies rated as “fair”, five as “good” and 10 as “poor”. The most common methodological flaws were the lack of both a sample size justification and information regarding the number of participants who were excluded from participation because they did not meet the inclusion criteria. The visual inspection of the funnel plots (Supplementary Material 9) and the results from Egger’s regressions (Supplementary Material 10) including studies of the meta-analyses, suggested no, or minimal, presence of heterogeneity and risk of biases at the study level. Finally, meta-regressions using the methodological quality as a covariate showed non-significant results, suggesting that low quality studies did not inflate between-group differences in skill retention after single (p = 0.1750) (Supplementary Material 3) or extended practice (p = 0.6850).
Consolidation after a single practice session
Overall, 17 [75, 117] of the 40 studies that investigated motor memory consolidation after a single practice session (42.5%), reported that persons with PD had poorer skill retention than the control group. These 17 studies employed SMTs, VATs, and SPTs (Table 2). The remaining 23 studies, most of which implemented SQTs and GMTs, revealed no significant differences in consolidation between groups [73, 116].
When pooled together in the meta-analysis, data from the 35 studies [78, 113–117] (47 effect sizes and 1187 participants) investigating motor memory consolidation after a single practice session showed a small significant effect in favor of NI individuals (SMD = –0.17; 95% CI = –0.32, –0.02; p = 0.0225; N = 47; I2 = 39.6%). Heterogeneity was low but statistically significant (Q-test: p = 0.0034). Sub-group analyses revealed a significant moderating effect of the task nature (p < 0.0001) (Fig. 1) and type of feedback (p = 0.0238) (Table 3). Specifically, people with PD showed worse poorer consolidation in both SMTs (SMD = –0.31; 95% CI –0.47, –0.15; N = 25; I2 = 27.4%; p = 0.0002) and VATs (SMD = –1.55; 95% CI = –2.32, –0.79; N = 3; I2 = 0%; p = 0.0001) but not in SQTs (SMD = 0.17; 95% CI = –0.05, 0.39; N = 10; I2 = 0%; p = 0.1292) or GMTs (SMD = 0.04; 95% CI = –0.25, 0.33; N = 8; I2 = 0%; p = 0.7771). The only study that investigated SPTs showed a large effect in favor of the NI group (SMD = –1.28; 95% CI = –2.07, –0.50; p = 0.0013). Sub-group analyses pertaining to feedback are reported in Table 3.
The results of the meta-regression exploring the influence of disease duration and severity, as well as their interactions are reported in Supplementary Material 3. None of these analyses yielded significant results, suggesting that disease duration and severity did not affect motor memory consolidation following a single session of practice. Similarly, consolidation did not seem to be moderated by improvements in skill performance during acquisition (Supplementary Material 3). Due to the small number of studies, we could not establish direct comparisons to investigate potential moderating effects of medication status (“on” vs. “off”). However, sensitivity analyses revealed that studies that manipulated medication status did not influence the results of the meta-analyses (Supplementary Material 4).
Study characteristics
*Primary outcome; ßretention following extended practice; Δ subsample used in the analyses; Qdata for meta-analysis unavailable; A, age; B, both FB; CoM, center of mass; CoP, center of pressure; DD, disease duration (years); EMG, electromyography; FB, feedback; GMT, gross motor task; H&Y, Hoehn and Yahr scale; KP, knowledge of performance; KR, knowledge of result; M/F, male/female; Med, medication status during practice: on/off; N, number; NI, neurologically intact; NIH, study quality; NP, not provided; NR, not reported; PD, Parkinson’s disease; Ret. Int., retention interval; SMT, sensory motor task; SPT, speech motor task; SQT, sequential fine motor task; UPDRS, Unified Parkinson’s Disease Rating Scale part III; UPDRSMDS, Movement Disorder Society-UPDRS part III; VAT, visuomotor adaptation task.
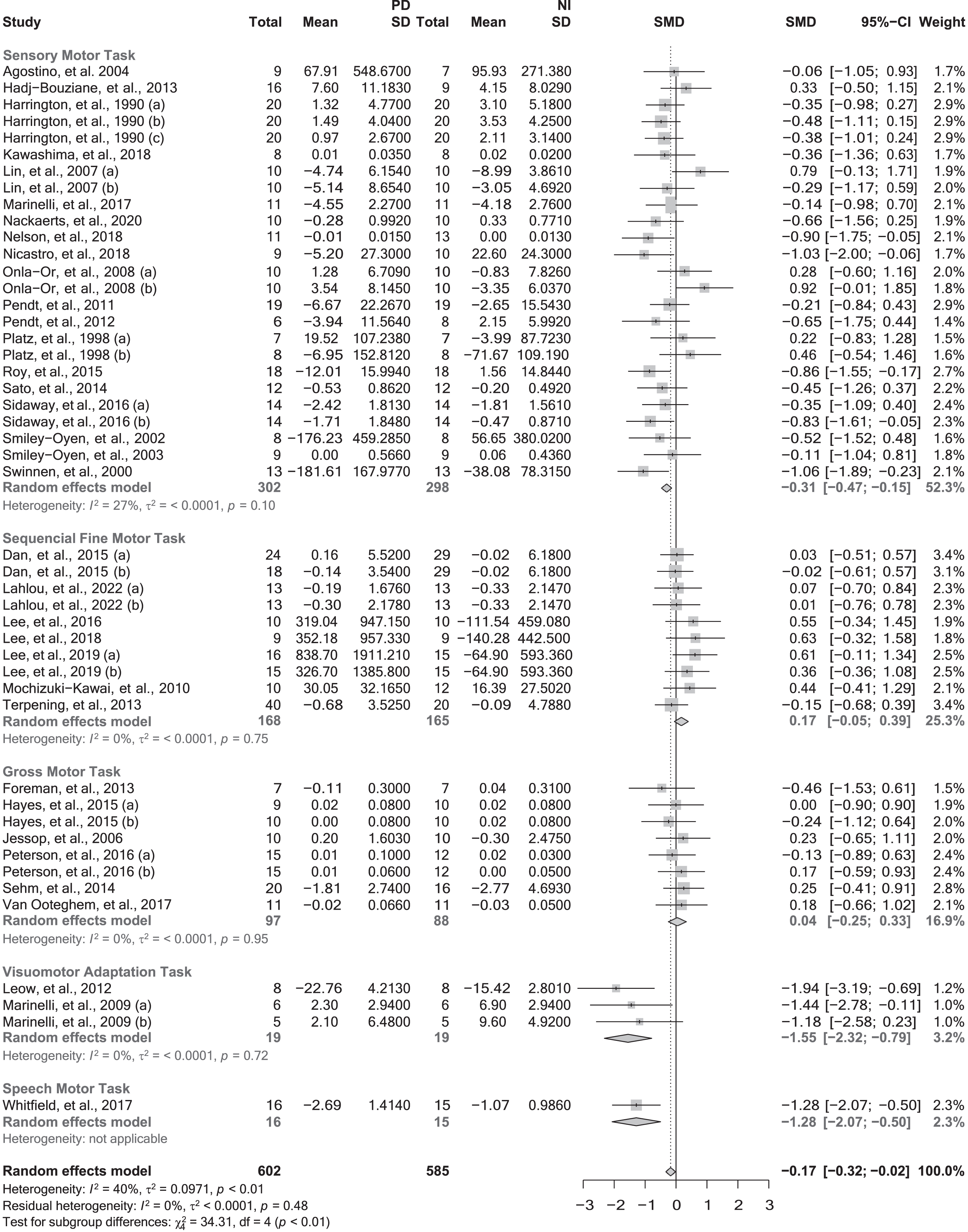
Motor memory consolidation after a single session of practice grouped by the task nature. CI, confidence interval; NI, neurologically intact individuals; PD, people with Parkinson’s disease; SD, standard deviation; SMD, standard mean difference. Negative values represented worse skill consolidation for persons with PD in comparison with NI individuals. The solid vertical black line represents an SMD of 0. The dashed vertical grey line represents the overall Random Effect Model.
Sub-group analysis for motor memory consolidation after a single session of practice
CI, confidence interval; I2, heterogeneity; K, knowledge; N, number of effect sizes; SMD, standardized mean difference.
Consolidation after extended practice
Only studies employing SMTs and GMTs investigated the effect of extensive practice on motor memory consolidation in PD. Therefore, the results pertaining to extended practice cannot be generalized to other types of motor tasks. Only two [76, 107] of the 14 studies that investigated consolidation following extended practice (14.3%) found that persons with PD had poorer skill retention than the control group. Except for one study [108] that observed better retention in persons with PD using a GMT, the remaining studies reported no significant differences in consolidation between groups, regardless of whether SMTs [72, 112] or GMTs [81, 112] were implemented.
When pooled together in the meta-analysis, the data of 12 studies [78, 112] (17 effect sizes and 403 participants) that investigated motor memory consolidation following extended practice using SMTs and GMTs revealed no significant differences between groups (SMD = –0.04; 95% CI = –0.24, 0.15; p = 0.6565; N = 17; I2 = 0%). Similarly, subgroup analyses did not reveal any significant moderating effect regarding the task nature (p = 0.0745) (Fig. 2), or feedback provided (p = 0.9511).
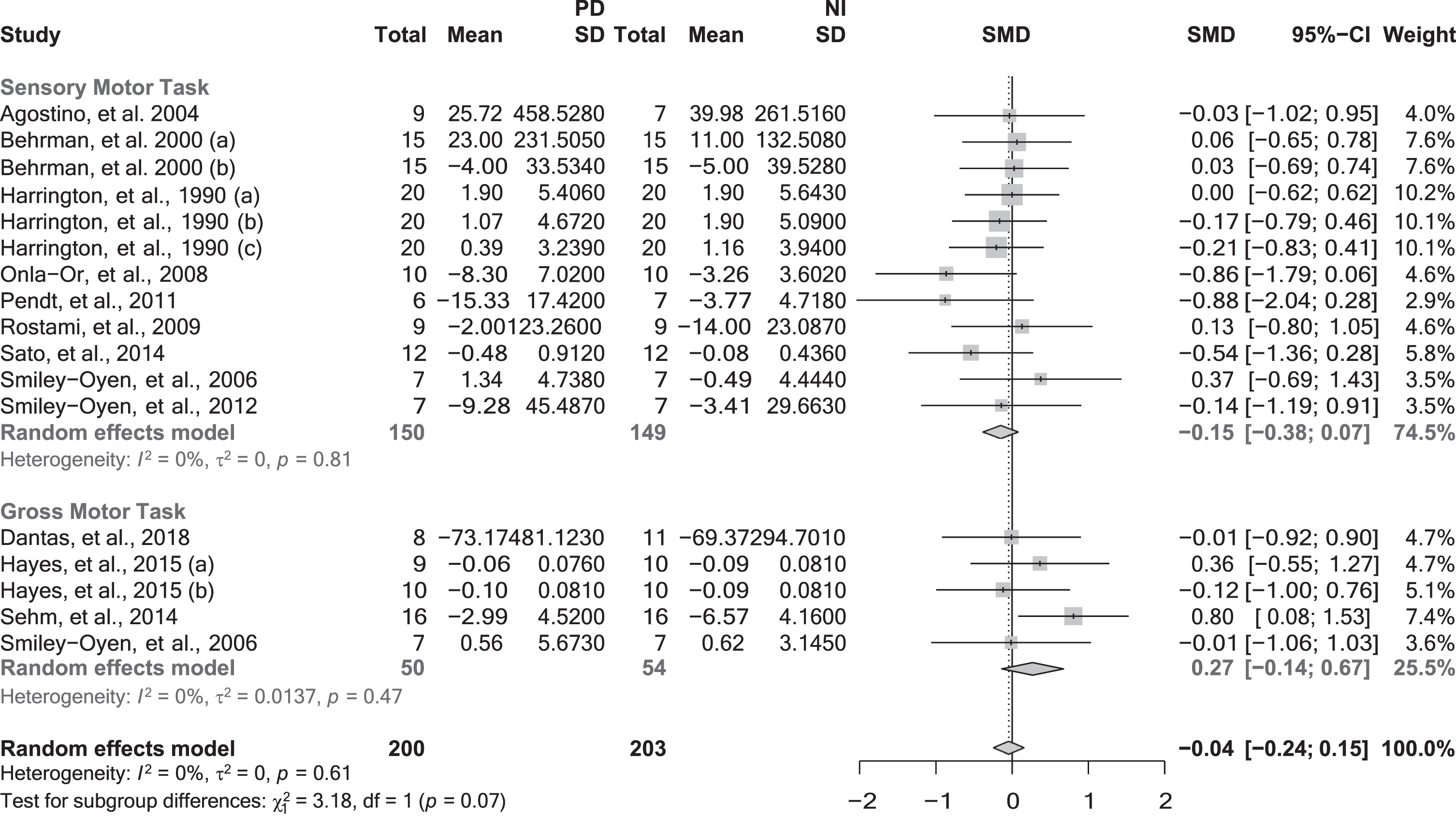
Motor memory consolidation after extended practice grouped by the task nature. CI, confidence interval; NI, neurologically intact individuals; PD, people with Parkinson’s disease; SD, standard deviation; SMD, standard mean difference. Negative values represented worse skill consolidation for persons with PD in comparison with NI individuals. The solid vertical black line represents an SMD of 0. The dashed vertical grey line represents the overall Random Effect Model.
The results of meta-regression analyses exploring the influence of disease duration (p = 0.1980) and severity (p = 0.7730), as well as their interaction (p = 0.2600), were non-significant, indicating that these factors did not moderate motor memory consolidation after extended practice. Importantly, performance during skill acquisition (i.e., encoding) did not appear to moderate retention either (p = 0.7162). Consistent with the results of single practice studies, sensitivity analyses showed that medication status did not influence the results of the meta-analyses including extended practice studies (Supplementary Material 4).
DISCUSSION
The results of this review confirm that, in comparison with NI individuals of similar age, persons with mild to moderate PD have deficits in the capacity to consolidate motor skills after a single practice session. These findings, which have important clinical implications, suggest that to maximize the long-term retention of motor skills, motor rehabilitation should not only focus on optimizing skill acquisition and thus encoding but also on ensuring an effective motor memory consolidation process [15, 120]. Deficits in motor memory consolidation, however, do not affect all types of motor tasks to the same extent. Differences between PD and NI individuals were found to be statistically significant only in the consolidation of motor skills acquired during STMs, VATs, and SPTs, although only one study investigated SPTs and thus these results should be interpreted cautiously. Patients with PD, in contrast, appear to have a more preserved capacity for efficiently consolidating, and thus retaining, skills acquired during SQTs and GMTs. Motor rehabilitation programs aimed at maintaining motor function in PD should target a broad range of tasks to ensure functional mobility in multiple activities of daily living [16, 121]. However, identifying task-specific deficits could inform clinicians to design more targeted interventions to optimize motor memory consolidation [6].
During the first symptomatic stages of PD [122, 123], neurodegenerative changes are localized within the basal ganglia and cortico-striatal motor networks while neocortical areas are less affected [124–126]. Subsequently, structural and functional signs of deterioration appear in the cerebellum [127, 128] and, to a lesser extent, in cortical areas such as parietal [129] and primary motor (M1) cortices [130, 131]. As the disease progresses, neurodegeneration changes within the basal ganglia spread across the striatum [126, 132], affecting non-motor cortico-striatal circuits [124, 133] and the less affected contralateral side [123, 134]. It will be in the late stages of PD when neurodegeneration will eventually reach neocortical areas [122, 135]. Whereas the encoding and consolidation of SMTs involve mostly basal ganglia, cerebellum and cortical areas such as M1 [13, 41], VATs engage primarily the striatum [26, 136], cerebellum, fronto-parietal lobules and M1 [13, 137–139] (Table 1). Neuroimaging studies have demonstrated that deficits in the consolidation of these types of motor tasks in PD are associated with alterations in parietal and cortico-striatal connectivity and dopamine uptake [75, 140]. Deficits in the consolidation of motor skills acquired during the practice of SMTs and VATs in persons with mild to moderate PD could therefore be explained by the deterioration that these specific areas of the brain experience during the early symptomatic stages of the disease.
Given the broad implication of cortico-striatal networks in the acquisition of SQTs [13, 141], the preserved capacity that patients with PD showed for successfully consolidating motor skills encoded during the practice of these motor tasks was unexpected. The reason for this preservation is unknown but it could be related to the capacity to activate neocortical areas that are consistently engaged during the practice of SQTs [40] and tend to be less affected by neurodegeneration in the early symptomatic stages of the disease [122]. Indeed, compared to SMTs and VATs, the performance of SQTs is characterized by greater activation of cortical areas such as the left dorsal premotor cortex, the supplementary motor cortex as well as the superior parietal cortex [40]. Increased participation of the cerebellum during motor practice could also contribute to the preservation of SQTs in people with PD [142–145], although this brain structure displays signs of structural and functional alterations already during the Hoehn & Yahr stage II–III of the disease [135, 146]. It is noteworthy that the activation of cortical and cerebellar regions is even more pronounced during the acquisition of explicit variants of the SQTs used in all the studies of the meta-analysis, in which participants are consciously aware of the repeating numerical sequence embedded in the motor task [40]. Importantly, these patterns of brain activation are not only present during acquisition but also during motor memory consolidation [147]. Clearly, more studies are needed to identify which mechanisms underlie the preserved capacity to retain SQTs shown by people with PD.
Deficits in the consolidation of SQTs could nevertheless become more pronounced when neurodegeneration progresses [148, 149] and thus alterations in motor automaticity mechanisms that are more relevant in the late phases of sequential motor learning start to emerge [150, 151]. In one study included in the review, participants performed a retention test of a SQT 10–18 months after initial practice [73] and only those patients whose disease severity worsened, transitioning from Hoehn and Yahr stage I to II [64] during the retention interval, showed significant deficits in skill retention, indicating poorer consolidation [73]. In addition, two studies of the review reported deficits only at the end of the second day of practice [80, 115], reinforcing the idea that deficits in motor skills practiced during SQTs become more pronounced in late phases of motor learning [9], when sequential finger movements should be performed automatically and with less attentional demand. The results of these studies suggest that while patients in the initial symptomatic stages of the disease can encode and consolidate sequential fine motor skills similarly to NI individuals, they can show deficits in the ability to automatize these motor skills [150, 151]. Cognitive strategies such as verbal instructions, cueing, and segmentation, could help those individuals to shift learning toward a more volitional mode of action to compensate for deficits in automaticity [121].
The performance of GMTs requires complex postural control strategies involving multiple brain structures such as the brainstem, cerebellum, basal ganglia, thalamus, and neocortical areas (e.g., sensory-motor cortex), as well as the integration of information from proprioceptive, vestibular, and visual systems [38, 152]. We found that people with mild to moderate PD can improve the performance of GMTs and retain these gains similarly to NI individuals. This finding is encouraging because patients with PD suffer from gait disorders and postural stability problems [153] that aggravate despite pharmacological treatment [154, 155] and that lead to an increased risk of falls [156–160]. GMT-based training could be a good strategy to slow down the deterioration of these complex motor skills [6] as this type of training can potentially mitigate alterations in neuroplasticity commonly observed in patients with PD [161]. For example, step training increases intra-cortical inhibition measured with transcranial magnetic stimulation [162], an indirect marker of γ-aminobutyric acid (GABA) activity, which tends to be suppressed in people with PD [130, 164]. Additionally, dynamic balance training has been shown to increase gray matter in the cerebellum, parietal and temporal lobes, as well as in the pre-motor cortex in patients with PD [108]. Taken together, these studies reinforce the importance of gait and postural control training in PD. Initiating these interventions as early as possible could potentially preserve neuroplasticity and slow down the progressive motor deterioration of the disease [6]. Nevertheless, additional research is needed to confirm whether our findings can be translated to different and more complex GTMs such as gait and can be generalized to real-world clinical scenarios [6, 165].
We used subgroup analyses and meta-regression techniques to explore factors moderating the capacity to consolidate motor skills in PD such as performance during acquisition, disease severity and duration, amount of practice and feedback provided. Although we cannot rule out the possibility that a ceiling effect in motor skill acquisition in NI individuals could have masked differences [14], the rate of skill improvement during acquisition was similar between groups. Regardless, acquisition performance and thus encoding did not appear to modulate the capacity to improve consolidation processes in patients with PD (Supplementary Material 3). Similarly, neither disease severity, disease duration, nor their interaction, had any significant influence on the observed deficits in motor memory consolidation, challenging previous studies indicating that these factors had detrimental effects on consolidation in people with PD [32, 167]. Discrepancies with previous studies could be explained by the fact that most studies included in our review recruited patients with mild-to-moderate PD. This possibly resulted in a very homogenous sample that limited the capacity of the meta-regression to capture moderating effects of disease severity or, to a lesser extent, disease duration on consolidation processes. Therefore, these results should be interpreted with caution, and further research is needed to determine whether the capacity to consolidate some specific motor skills is affected or worsens more rapidly as the disease progresses.
Importantly, subgroup analyses revealed that deficits in motor memory consolidation were reduced when more practice was afforded [16, 95]. With additional practice, patients with PD appear to be able to consolidate motor skills acquired during SMTs similarly to NI individuals [6, 168]. Extended motor practice possibly allows patients the time to encode sensory and motor information more effectively, compensating for some of the neural deficits that they display during motor learning [22, 98]. This is consistent with studies showing that, compared to NI individuals, persons in the early stages of PD require greater neural activity during practice to achieve similar skill performance [22, 169–172]. However, extended motor practice alone, while being effective during the early phases of sensory motor learning, may not be sufficient during late phases that involve automatization [9, 151].
Augmented feedback provided as knowledge of results, alone or in combination with knowledge of performance, also improved motor memory consolidation [6, 173]. Persons with PD experience proprioceptive [174, 175] and central processing integration impairments [127, 176] as well as attentional deficits [177]. Augmented feedback could therefore help these patients activate their cognitive reserve to use a more volitional mode of action [121] as well as to focus on the extrinsic information of the task during practice to enhance consolidation processes [173, 179].
Limitations
It is important to acknowledge that, given their design, the results of the studies investigating consolidation following single practice cannot be easily translated to specific rehabilitation interventions in PD. They can, however, provide important information to guide the general principles of clinical practice [7]. It is further important to emphasize that most studies of the review included participants in the early symptomatic stages of PD. More studies are thus needed to determine if the task-specific deficits in motor memory consolidation found in this study remain present or are augmented in later stages of the disease. Given the limited number of studies available, it was not possible to conduct additional analyses investigating interactions between augmented feedback and task nature. Future studies should determine which forms, frequency, timing, and focus (internal vs. external) [179] of augmented feedback is more effective to improve consolidation processes across different (simple vs. complex) motor tasks [180].
Since only three studies manipulated medication status [83, 88], we could not investigate the effects of antiparkinsonian medications on motor memory consolidation. Although our sensitivity analyses suggested that medication status did not affect consolidation (Supplementary Material 4), the limited data available suggests that assessing the influence of medication requires additional studies. The effects that antiparkinsonian medications can have on motor memory encoding and consolidation are complex and conflicting results have been reported [15, 181–183]. More investigations are needed to elucidate the effects of these medications on motor memory consolidation processes and whether their potential modulating effects are similar across different types of motor tasks at different stages of the disease. Finally, since in most studies, participants had similar cognitive levels, it was not possible to determine the influence of cognition deficits (or lack thereof) on consolidation [16] and whether certain motor tasks are more affected by cognitive deterioration in PD.
Conclusion
Persons with PD who perform sensory motor skills and sequential movements daily need to constantly adapt pre-existing motor routines to cope with the motor dysfunctions arising during the disease [8, 120]. Similarly, these individuals suffer from gait disorders and postural instability [156–160], as well as speech and voice disorders [184–186]. The results of this review confirm that people with mild to moderate PD have deficits in motor memory consolidation processes affecting primarily SMTs and VATs following a single practice session. Our results suggest that extended motor practice and augmented feedback might be valuable means to reduce deficits in the consolidation of functional skills, but more evidence is needed to confirm if these strategies are effective to improve different motor tasks in clinical practice. Overall, our findings underline the importance of developing targeted interventions to enhance motor memory processes to support long-term skill retention in PD.
Footnotes
ACKNOWLEDGMENTS
We would like to thank the authors of the papers included in the review who provided additional information: Dr. Bo Foreman, Dr. Dagmar Sternad, Dr. Lisa Maurer, Dr. Alfredo Berardelli, MSc Soraya Lahlou, Dr. Ya-Yun Alice Lee, Dr. Evelien Nackaerts, Dr. Lucio Marinelli, Dr. Ann Smiley-Oyen, Dr. David Wright, Dr. Julien Doyon, Dr. Nicolas Nicastro, Dr. Karen Van Ooteghem, Dr. Jason Whitfield, Dr. Carolee Winstein, and Dr. Leszek Kaczmarek. We would also like to thank Alina Andretzky for reviewing an early version of this manuscript.
FUNDING
The study has been supported by Parkinson’s Canada, Graduate Student Award Competition, and the Fonds de recherche du Québec - FRQS (doctoral scholarships). Marc Roig was supported with a Fonds de Recherche Santé Québec (FRQS) Salary Award (252967) and CIHR Projects Grant (02109PJT-468982-MOV-CFAA-244681). Simon Steib received funding from the German Foundation of Neurology. Jacopo Cristini received funding from Parkinson’s Canada, Graduate Student Award Competition (GSA-2021-0000000080), and FRQS (doctoral scholarship - 317490). Zohra Parwanta received funding from Graduate Student Award Competition, and FRQS (doctoral scholarship). Bernat De las Heras received funding from FRQS (doctoral scholarship). The funding sources did not have any involvement in the study (i.e., design, data analysis and interpretation) as well as in the decision to submit the manuscript for publication.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author upon reasonable request.
