Abstract
The course of patients with multiple system atrophy (MSA) who undergo deep brain stimulation (DBS) is unclear. In a retrospective review of 1,496 patients with MSA evaluated at our institutions from 1998–2021, 12 patients underwent DBS; 9 had a diagnosis of Parkinson’s disease at the time of surgery. Nine patients reported initial improvement in at least one symptom and 7 experienced overall worsening following DBS. All patients had at least one red flag sign or symptom suggesting atypical parkinsonism prior to surgery. Considering overall poor outcomes of DBS in MSA, we recommend careful consideration of red flags in patient selection.
INTRODUCTION
Multiple system atrophy (MSA) is an atypical parkinsonian syndrome characterized by autonomic failure [1, 2]. MSA diagnosis is based on clinical signs and symptoms which may contribute to misdiagnosis such as Parkinson’s disease (PD) [3]. Deep brain stimulation (DBS) is used to treat patients with PD who suffer from motor fluctuations, dyskinesia, or tremor [4]. DBS is typically not recommended in patients with atypical parkinsonism, as previous case reports and small case series have shown less favorable effects. Due to the complexity of MSA diagnosis, some patients may be misdiagnosed with PD, particularly early in the disease, which may lead to patients with MSA undergoing DBS [4, 5]. We sought to report our experience with MSA patients who underwent DBS.
MATERIALS AND METHODS
We retrospectively reviewed medical records of all patients with a diagnosis of probable or possible MSA based on the second consensus criteria for MSA [2] that were evaluated at Mayo Clinic in Rochester, MN, Phoenix, AZ, and Jacksonville, FL between January 1998 to December 2021. We included patients with MSA who received DBS during their disease course. Patients were excluded if an alternative diagnosis to MSA was more likely. We recorded demographic, clinical, surgical, and neurophysiologic information. We considered the age at disease onset as the first time that patient reported symptoms of motor involvement or autonomic dysfunction. We reported levodopa equivalent daily dose (LEDD) [6] when available. We calculated the Composite Autonomic Severity Score (CASS) from the autonomic reflex screen (ARS), when available [7]. We evaluated the location of DBS leads in patients with available neuroimaging studies.
We reported categorical variables using number and frequency and interval variables using mean and standard deviation (SD). We used IBM SPSS version 28 (Armonk, NY) to conduct statistical analyses.
The Mayo Clinic institutional review board approved the study.
RESULTS
Baseline characteristics
Of 1,496 patients diagnosed with MSA, 12 patients underwent DBS (Table 1). Surgery was done at our institution in 1 patient (patient 7). Of the 12 patients, 11 were female. Five patients were deceased at the time of medical record review with a mean survival time from disease onset of 11.60 (±4.73) years and mean duration from DBS to death of 5.20 (±2.94) years. Disease course for all patients is shown in Fig. 1. The mean duration of symptom onset to DBS was 6.33 (±3.70) years. Three patients underwent DBS with a diagnosis of parkinsonism or atypical parkinsonism (Table 1). Excluding these 3 patients, the mean duration from the time of PD diagnosis to DBS was 5.22 (±3.59) years.
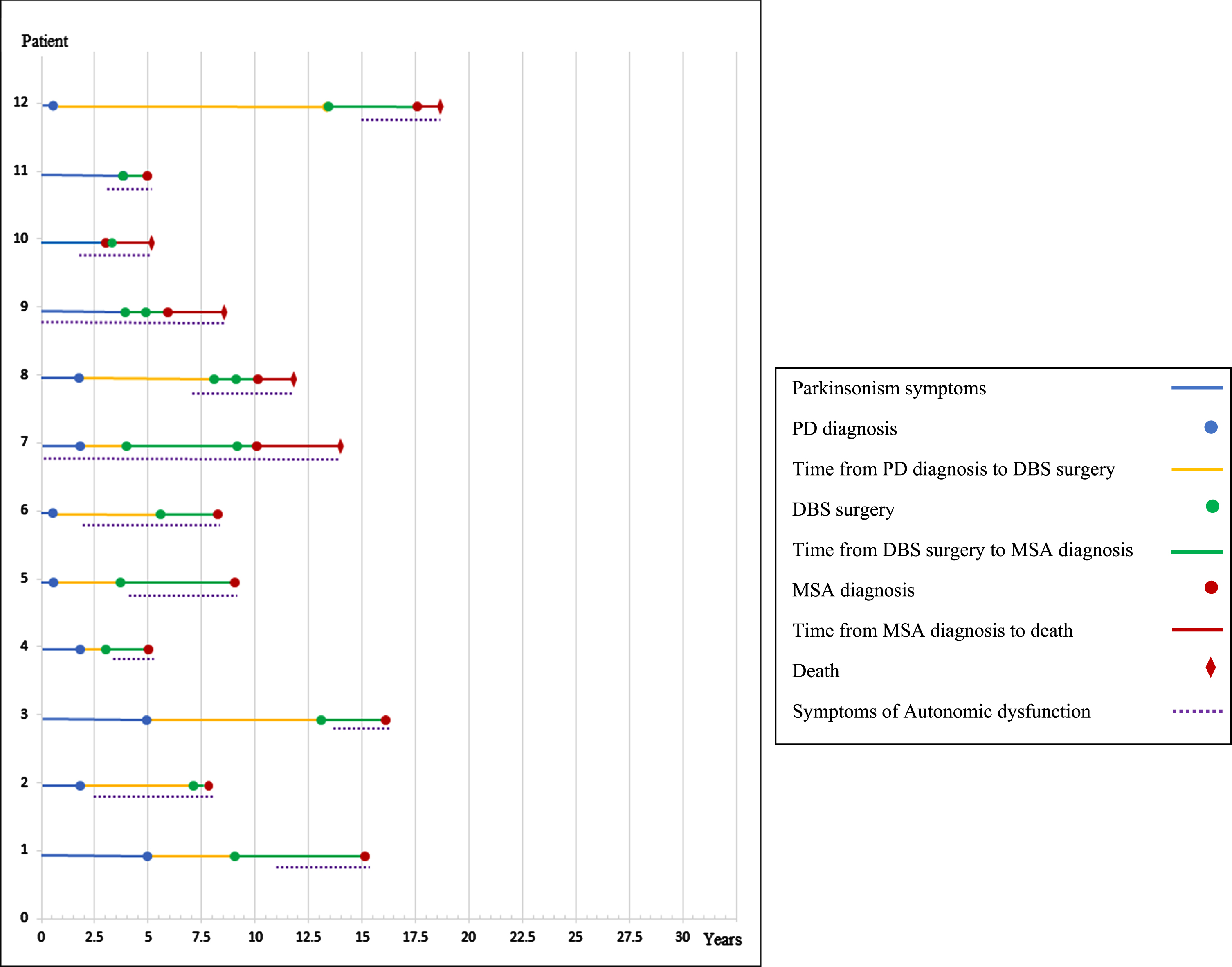
Patients’ disease course. PD, Parkinson’s disease; DBS, deep brain stimulation; MSA, multiple system atrophy.
Patients’ characteristics
PD, Parkinson’s disease; DBS, deep brain stimulation; STN, subthalamic nucleus; GPi, globus pallidus internus; Vim, ventral intermediate nucleus of the thalamus; MSA, multiple system atrophy; LEDD, L-dopa equivalent daily dose; yr, year; m, month; N/A, not available; LID, L-dopa-induced dyskinesia; MCP, middle cerebellar peduncle. *Early falls were defined as recurrent falls and early instability within 3 years of disease onset.
All patients had at least one symptom or sign suggestive of atypical parkinsonism prior to DBS (Table 1). Also, 3 patients had MRI findings supportive of MSA prior to DBS (Supplemental Table 1). In 6 patients, symptoms of autonomic dysfunction were present before DBS. Response to levodopa and LEDD before surgery is shown in Table 1.
Disease course after surgery
The bilateral subthalamic nucleus (STN) was the most common location for DBS leads (66.6%). Appropriateness of the location of DBS leads was able to be assessed in 8 patients (Supplementary Table 1), among them the locations were appropriate in 7 patients.
Overall, motor symptoms continue to progress after DBS with transient responses noted by some patients in severity of tremor and dystonia. Four patients reported partial to complete improvement in their tremor. Three patients reported onset of dystonia following DBS. Dyskinesia transiently improved in 4 patients, followed by worsening. Regarding gait and posture, 3 patients had transient improvement while two patients developed freezing or imbalance shortly after surgery.
Autonomic symptoms often surfaced or progressed following surgery (Supplementary Table 2). In 4 patients, urinary incontinence developed within a year after surgery.
Three patients developed cognitive or mood symptoms including 1 patient that reported hallucinations and delusions, and another reporting worsening of depression that further improved when DBS was turned off.
DBS was eventually turned off in 4 patients due to inefficiency or adverse effects.
MSA diagnosis
The mean duration of symptom onset to MSA diagnosis was 9.08 (±4.73) years and the mean duration of DBS to MSA diagnosis was 2.89 (±1.99) years.
Neuroimaging data were available in 8 patients, among them 7 had findings suggestive of MSA [8]. Brain autopsy was not performed in any of the patients.
All patients developed symptoms of autonomic dysfunction during their disease course, and all reported bladder and urinary symptoms. TST results were available in 9 patients; in 8 patients it was supportive of MSA (Supplementary Table 1). CASS was able to be calculated in 6 patients with a mean CASS of 6.16 (±2.13).
DISCUSSION
We present a large case series reporting DBS outcomes in patients subsequently diagnosed with MSA. An extensive review of patients’ records showed that all patients had at least one red flag sign or symptom suggestive of atypical parkinsonism at the time of surgery.
While some patients reported beneficial effects in terms of tremor and levodopa-induced dyskinesia with DBS, which are both among the established beneficial effects of DBS in PD patients as well, most of the patients did not experience significant improvement and reported symptom recurrence/progression and, in some cases, new symptom development. Our observation supports findings from previous studies regarding questionable long-term efficacy of DBS in atypical parkinsonism including MSA [5, 10].
Although early DBS in patients with PD is associated with beneficial outcomes, implanting within five years from PD diagnosis may increase the chance of including patients with atypical parkinsonism [4]. This is supported by the mean duration from PD diagnosis to DBS surgery of 5.22 years among our cases.
In a recent systematic review, the authors concluded that although patients with MSA may experience some beneficial effects in their motor symptoms with DBS, the considerable frequency of cognitive adverse effects undermine the possible efficacy of surgery [10]. In the present study, we observed different adverse events including cognitive disturbances, gait difficulty, autonomic dysfunction, dystonia and bulbar symptoms among our patients following surgery. Excluding the possibility of adverse effects due to the suboptimal lead location in a subset of patients; for the majority it is yet unclear whether these symptoms were a direct adverse effect of surgery or occurred as part of the progressive course of the underlying disease. MSA has a progressive course with a median survival of 9.8 years from symptom onset [11]. Considering the median duration from symptom onset to surgery of 4.5 years in our study, most of the patients were in the middle stage of their disease at the time of surgery and symptom progression was expected.
One study investigated adverse events after DBS in a group of patients who underwent surgery for different indications (PD, dystonia, tremor, etc.). The authors categorized any worsening of axial symptoms during the first 6 months after surgery as probably related to DBS in patients with PD [12]. In our study, a third of patients experienced worsening and new development of axial symptoms, including freezing of gait and dysarthria, within 6 months of surgery which suggests that some of the worsening seen in our patient could be an adverse effect of surgery.
Nine out of 12 patients in our study reported an initial improvement in at least one symptom following surgery. This initial improvement was as brief as less than a month in 2 cases. Reasons for the initial symptom improvement followed by symptom worsening in MSA patients after DBS are unclear but may relate to a symptomatic effect that is short-lived due to the rapidly progressive course of MSA. It may also be due to the micro-lesion effect which conversely may also be responsible for subsequent clinical deterioration [13].
The indication for DBS surgery is of great importance. While satisfactory levodopa response is an important criteria for choosing patients with PD that may benefit from DBS [14], inadequate response to levodopa was the indication for surgery in some of our patients.
Assessing patients for the presence of red flags, including autonomic dysfunction, to uncover signs of atypical parkinsonism is recommended [9]. We suggest autonomic function testing before considering DBS in patients with PD in whom the physician is uncertain about the diagnosis. Autonomic failure is unusual early in the disease course among patients with PD, while it is the mainstay in MSA [1].
There are some limitations to the present study. First, it is a retrospective study, and recall bias regarding the symptoms timeline is possible. Second, patients underwent surgery at different centers, with different providers making the initial PD diagnosis. Third, there is a possibility that pathological diagnosis may not be MSA as none of the patients underwent brain autopsy [3]. Fourth, objective assessment of patients’ symptoms was not consistently performed.
In summary, our findings do not support DBS in patients with MSA. Careful consideration of atypical parkinsonism clinical and neuroimaging red flags before choosing patients with PD for DBS is necessary. Autonomic function testing may help with clarifying the diagnosis.
CONFLICT OF INTEREST
There is no support or financial issues from all authors relative to the research covered in the submitted manuscript.
Footnotes
ACKNOWLEDGMENTS
Supported in part by NIH (P01NS44233, U54NS065736, K23NS075141, R01 FD004789, R01 NS092625), Mayo CCaTS (UL1TR000135), Cure PSP Foundation, NIH (R01 NS092625, U19 AG71754, UL1 TR000135), FDA (FD-R-07290), Michael J. Fox Foundation, Bishop Dr. Karl Golser Foundation, Sturm Foundation, Mayo Center for Regenerative Medicine, and Mayo Funds. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NIH.
