Abstract
Background:
The end-of-life period is associated with disproportionately higher health care utilization and cost at the population level but there is little data in Parkinson’s disease (PD).
Objective:
The goals of this study were to 1) compare health care use and associated cost in the last year of life between decedents with and without PD, and 2) identify factors associated with palliative care consultation and death in hospital.
Methods:
Using linked administrative datasets held at ICES, we conducted a retrospective, population-based cohort study of all Ontario, Canada decedents from 2015 to 2017. We examined demographic data, rate of utilization across healthcare sectors, and cost of health care services in the last year of life.
Results:
We identified 291,276 decedents of whom 12,440 (4.3%) had a diagnosis of PD. Compared to decedents without PD, decedents with PD were more likely to be admitted to long-term care (52% vs. 23%, p < 0.001) and received more home care (69.0 vs. 41.8 days, p < 0.001). Receipt of palliative homecare or physician palliative home consultation were associated with lower odds of dying in hospital (OR: 0.24, 95% CI: 0.19– 0.30, and OR: 0.38, 95% CI: 0.33– 0.43, respectively). Mean cost of care in the last year of life was greater for decedents with PD ($68,391 vs. $59,244, p < 0.001).
Conclusion:
Compared to individuals without PD, individuals with PD have higher rates of long-term care, home care and higher health care costs in the last year of life. Palliative care is associated with a lower rate of hospital death.
Keywords
INTRODUCTION
Worldwide, health care in the end-of-life period is increasingly being identified as an area of concern [1]. Often futile medical interventions are pursued while other determinants of a “good death”, such as dying in the preferred place (i.e., one that is appropriate given the individual’s medical complexity, is comfortable, and promotes contact with loved ones) and relief of physical pain, are neglected [2, 3]. In addition, the end-of life period is associated with disproportionately high health care costs [4]. Previous studies have demonstrated that patients with Parkinson’s disease (PD) have disproportionately high health care needs [5]; however, the end-of-life period has not been examined specifically. Currently there are over 6.2 million individuals with PD worldwide, and it is the fastest growing neurological disorder in the world [6], so end-of-life care of this patient population has important implications for health care planning.
Involvement of palliative care, the medical discipline focused on improving quality of life in the setting of life-limiting illness, is a proven strategy for reducing costs among those living with serious illness. In patients with other neurodegenerative conditions involvement of a palliative care specialist in the last year of life is associated with fewer deaths in hospital, and lower costs of care [7, 8]. PD experts have advocated for early palliative care to improve symptom management and promote advance care planning [9–11]. Although patients admitted to long-term care are expected to have palliative care services, including end-of-life care, provided by the staff responsible for their overall care, palliative care for home dwelling individuals with PD is not standard of care [12]. A better understanding of the current access to palliative care, the impact of palliative care on place of death, and factors associated with palliative care home visits is needed help inform strategies to improve the quality end-of-life care for patients with PD.
We sought to describe population level health care service utilization and direct health care costs among all decedents with PD in the last year of life, and to compare this with decedents without PD. In particular, we aimed to identify the frequency of home palliative care service use among those not admitted to long-term care, and the association with place of death.
METHODS
Study population
We conducted a retrospective population-based cohort study of Ontario decedents, capturing all deaths from January 1, 2015 to December 31, 2017. Our study population included decedents eligible for provincial health insurance in the last year of life. Ontario, a Canadian province with a population of more than 14 million individuals [13], consists of a largely single payer healthcare system, where all medical services deemed necessary are publicly funded. This program covers a large portion of hospital and ambulatory-based health care services for all residents. However, prescription drugs are only covered for those who are 65 years of age or older, those who require social assistance, and those receiving highly expensive medications that exceed pre-determined deductibles.
Data sources and study definitions
Using unique encoded identifiers, we linked multiple health administrative datasets which were held and analyzed at ICES. ICES is an independent, non-profit research institute whose legal status under Ontario’s health information privacy law allows it to collect and analyze health care and demographic data, without consent, for health system evaluation and improvement. These datasets contained de-identified health information for all Ontario residents. All Canadian citizens and permanent residents (>99% of the population) receive complete coverage for medically necessary services from a provincial, single payer healthcare system. The databases record all medically necessary health services including physician, hospital, and demographic information, allowing for comprehensive tracking of healthcare encounters [14]. The following datasets were linked at the individual level: 1) Vital Statistics Database (Office of the Registrar General – Deaths) for capturing place, cause and date of death; 2) Registered Persons Database, for capturing all demographic information including age, sex, and postal code; 3) Ontario Health Insurance Plan Claims Database, for all claims data for physician-based services in inpatient and outpatient settings; 4) Home Care Database for publicly-funded homecare services; 5) Discharge Abstract Database, capturing all hospitalizations; 6) National Ambulatory Care Reporting System for all emergency department visits; 7) National Rehabilitation Reporting System for inpatient rehabilitation programs; and 8) Continuing Care Reporting System, for care provided in long-term care and complex continuing care (those in designated complex, non acute institutional care beds); 9) Statistics Canada Census data, for income quintile and rurality by postal codes; 10) Same Day Surgery database to capture information about same day procedures; and 11) the Ontario Drug Benefit claims database, for capturing prescription drug use for those aged 65 and older (Supplementary Table 1).
We categorized decedents according to diagnosis of PD (including parkinsonism (PKM) such as vascular parkinsonism, or multisystem atrophy) vs. no PD. Based on a validation study to identify patients with PD using the same administrative databases, we defined PD diagnosis as the earliest of the following: 1) a hospitalization with a PD or PKM diagnosis code (sensitivity 26%, specificity 100%), or 2) two physician claims with the same diagnosis codes billed within 1 year, with a minimum 30-day interval between each claim (sensitivity 71%, specificity 100%) [15]. For patients in the Ontario Drug Benefit database, PD or PKM was diagnosed at the earliest date of prescription for PD medication, along with one physician claim with a PD/PKM diagnosis within 6 months (sensitivity 66%, specificity 100%) [15]. The codes used to identify individuals with PD/PKM were ICD-10 codes G20, G21.0-G21.4, G21.8-9, G22, and F02.3 and the ICD-9 code 332. Individuals who did not meet any of these criteria were assigned to the non-PD cohort.
Outcomes
PD decedents were compared to non-PD decedents on the following measures in the last year of life: 1) place of death (acute care, subacute care, long-term care, other (i.e., home); 2) receipt of home-based palliative care, delivered by physicians and/or allied health providers; 3) major healthcare service utilization trends— including acute care, continuing care, outpatient care, home care, and use of in-hospital interventions (i.e., feeding tube placement); 4) and total and sector-specific direct healthcare costs, including costs associated with specific healthcare services (Supplementary Table 1). Place of death was determined by examining records across settings of care (Supplementary Table 1). Receipt of palliative care in the home was captured based on previously defined physician billing codes and service codes from home care [16] (Supplementary Table 1). In Ontario, palliative care in long-term care is provided by physicians associated with those institutions and is often not captured with palliative care billing codes. Therefore, palliative care consultation was only reported for home dwelling patients with PD.
We report costs by sector in the last year of life, adopting a payer (Ministry of Health and Long-Term Care, MOHLTC) perspective at an individual level. We assigned costs of all health care records paid for by the MOHLTC using previously described methods [17]. Briefly, cost information for sectors (e.g., hospitalizations, complex continuing care, and inpatient rehabilitation) that have global budgets (e.g., by institution or health region) is determined through a top-down approach through case-mix methodology. On the other hand, sectors that have fee payments directly associated with each use (e.g., drug costs or physician fees) have costs estimated directly. All costs were expressed in inflation adjusted 2020 Canadian dollars; we inflated past costs using health care-specific yearly consumer index reported by Statistics Canada. Total healthcare cost for the population was the sum of all costs among decedents captured within each respective sector.
Statistical analysis
Descriptive statistics were used to compare characteristics between PD and non-PD decedents. Characteristics include: sex, age, area-level income quintile, rurality, time from PD diagnosis to death, chronic diseases, number ofcomorbidities. Descriptive statistics were also used to describe the receipt of healthcare services in the last year of life, and place of death. Bivariate comparisons between PD and non-PD decedents were conducted using one-way ANOVA tests (for mean values), Kruskal-Wallis tests (for median values), and chi-squared tests (for categorical variables). Statistical significance was defined as a p-value of < 0.05 based on two-tailed hypothesis testing.
Multivariable logistic regression analyses were performed to determine the association between home-dwelling PD decedent characteristics and: 1) the likelihood of dying in an acute care hospital, adjusted for sex, age, income quintile, rurality, number of comorbidities, receipt of a palliative physician home visit or homecare service in the last year of life; 2) the likelihood of receiving a palliative physician home visit adjusted for sex, age, income quintile, rurality, number of comorbidities, and neurology consultation in the last year of life. Multivariable logistic regression analyses were not performed for those residing in long-term care as physician billing codes from the administrative databases may undercount the provision of physician-based palliative care in this setting (with many residents receiving palliative care services from nurses and personal support workers instead). In addition, supporting patients even at the time of death, rather than transferring patients to hospital, is part of the mandate for long-term care. Residents of long-term care were therefore excluded from the analysis of dying in an acute care hospital. All analyses were conducted using SAS Enterprise Guide 7.1 (SAS Institute Inc., Cary, NC).
Data availability
The dataset from this study is held securely in coded form at ICES. Data sharing agreements prohibit ICES from making the dataset publicly available. However, access may be granted to those who meet prespecified criteria for confidential access, available at https://www.ices.on.ca/DAS.
Ethics approval
The use of the data in this project is authorized under section 45 of Ontario’s Personal Health Information Protection Act (PHIPA) and does not require review by a Research Ethics Board.
RESULTS
Cohort characteristics
We identified 291,276 decedents of whom 12,440 (4.3%) had a diagnosis of PD. Cohort characteristics at the time of death are displayed in Supplementary Table 2. Comparing baseline characteristics between decedents with and without PD, those with PD were older (mean age 83 vs. 77 years, p < 0.001), had fewer comorbidities (median 2 vs. 3 p < 0.001) but had a higher prevalence of mood disorders (9% vs. 5%, p < 0.001) and dementia (32% vs. 15%, p < 0.001). For decedents with PD, the mean age at the time of diagnosis was 75 years (SD±10) and the mean time between PD diagnosis and death was 7 years (SD±6).
Services received in the last year of life
Health care service utilization in the last year of life and the place of death are shown in Supplementary Table 3 and Fig. 1. Notably, decedents with PD spent fewer days in the emergency department (ED) (mean 2.0 vs. 2.6, p < 0.001) and were less frequently admitted to the intensive care unit (ICU) (16% vs. 25%) in the last year of life compared with those without PD. Among patients admitted to hospital mechanical ventilation and tracheostomy was less common in the PD group (8.0 % vs. 13.7 %, p < 0.001 and 0.7% vs. 1.2%, p < 0.001); however, feeding tube placement was higher (2.7% vs. 1.6%, p < 0.001). Just over half (52%) of all decedents with PD were admitted to long-term care, compared to less than a quarter (23%) of decedents without PD. Decedents with PD spent over twice as many days in long-term care (mean 149.4 vs. 63.5, p < 0.001) and more days in complex continuing care (i.e., sub-acute care hospitals) (mean 8.0 vs. 5.4, p < 0.001). They also received more non-physician homecare (mean days 69.0 vs. 41.8, p < 0.001). In the last year of life, the proportion of PD decedents receiving at least 1 consultation from a neurologist was three-fold greater than those without PD (36% vs. 11%, p < 0.001). The proportion of PD decedents receiving a consultation from a gerontologist was two-fold greater compared to the non-PD decedents (22% vs. 13%, p < 0.001).
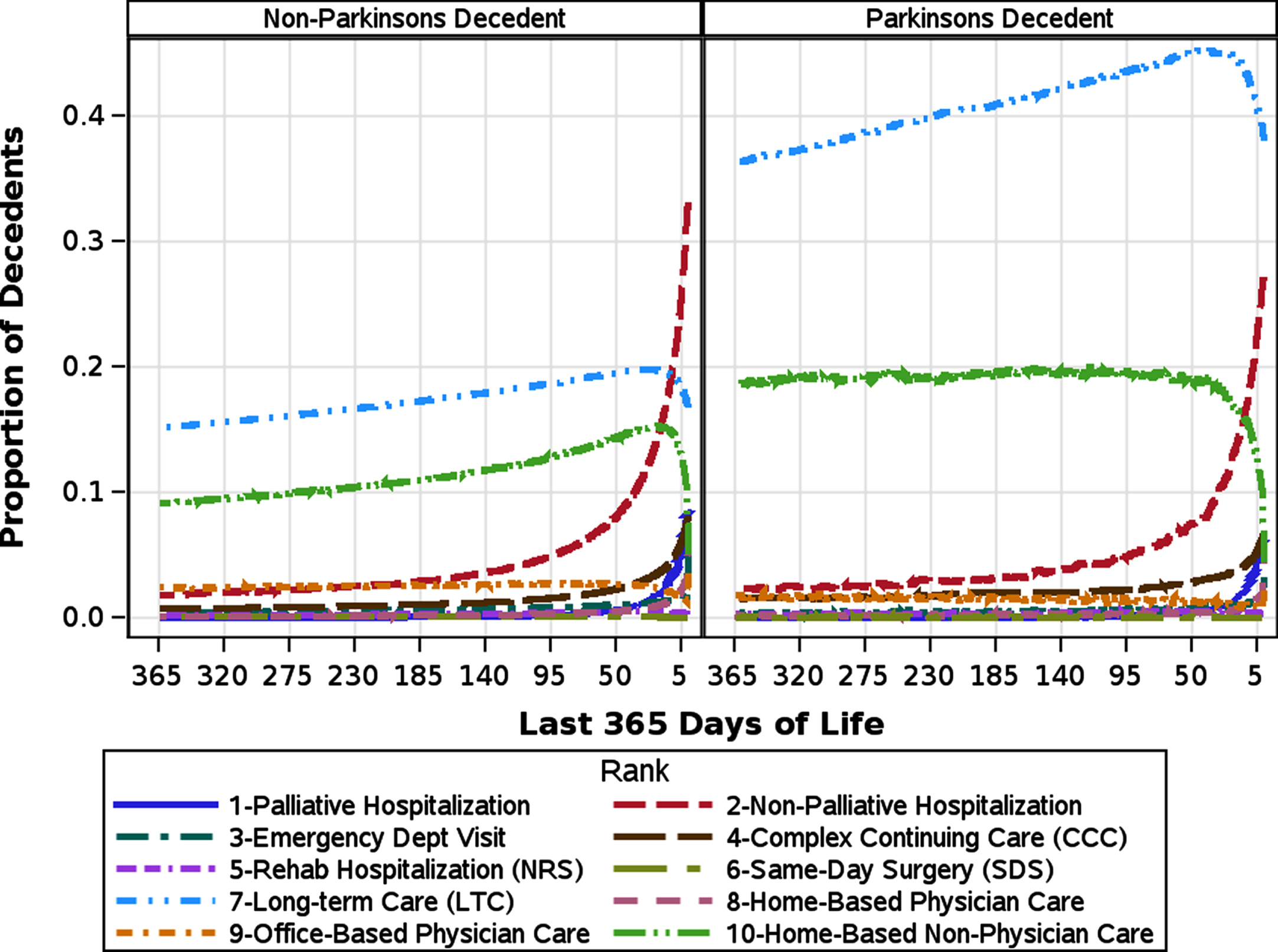
Health care utilization by sector stratified by Parkinson’s disease status.
In the sub-group of decedents who were not admitted to long-term care in the last year of life, decedents with PD received more home-based non-physician care (114.5 vs. 47.4 days, p < 0.0001) compared to decedents without PD (Table 1).
Places of death and places of care in the last year of life by Parkinson’s disease status (excluding individuals who spent≥1 day in long-term care)
aidentical p value for mean and median values unless otherwise specified.
In the decedents with PD not admitted to long-term care, neurology consultations were much more frequent compared to the overall PD group (50% vs. 36%), but the rate of geriatrician consultations was similar (26% vs. 22%). Overall, home dwelling decedents with PD received less office-based physician care when compared to decedents without PD (9.8 vs. 11.5 days, p < 0.001).
Palliative care
The PD cohort were less likely to receive physician palliative home visits and non-physician palliative homecare visits (20% vs. 22% p < 0.001; 13% vs. 22, p < 0.001 respectively) when compared to those without PD (Supplementary Table 3). In the subgroup of decedents who were not admitted to long-term care in the last year of life; however, more decedents with PD received a physician palliative home visit (35% vs. 27%) compared to decedents without PD, although fewer received non-physician palliative care (24% vs. 28%, p < 0.001) (Table 1). In both PD and non-PD decedents, the initiation of palliative care services occurred almost exclusively in the last 3 months of life with most occurring in the last few weeks (Fig. 2).
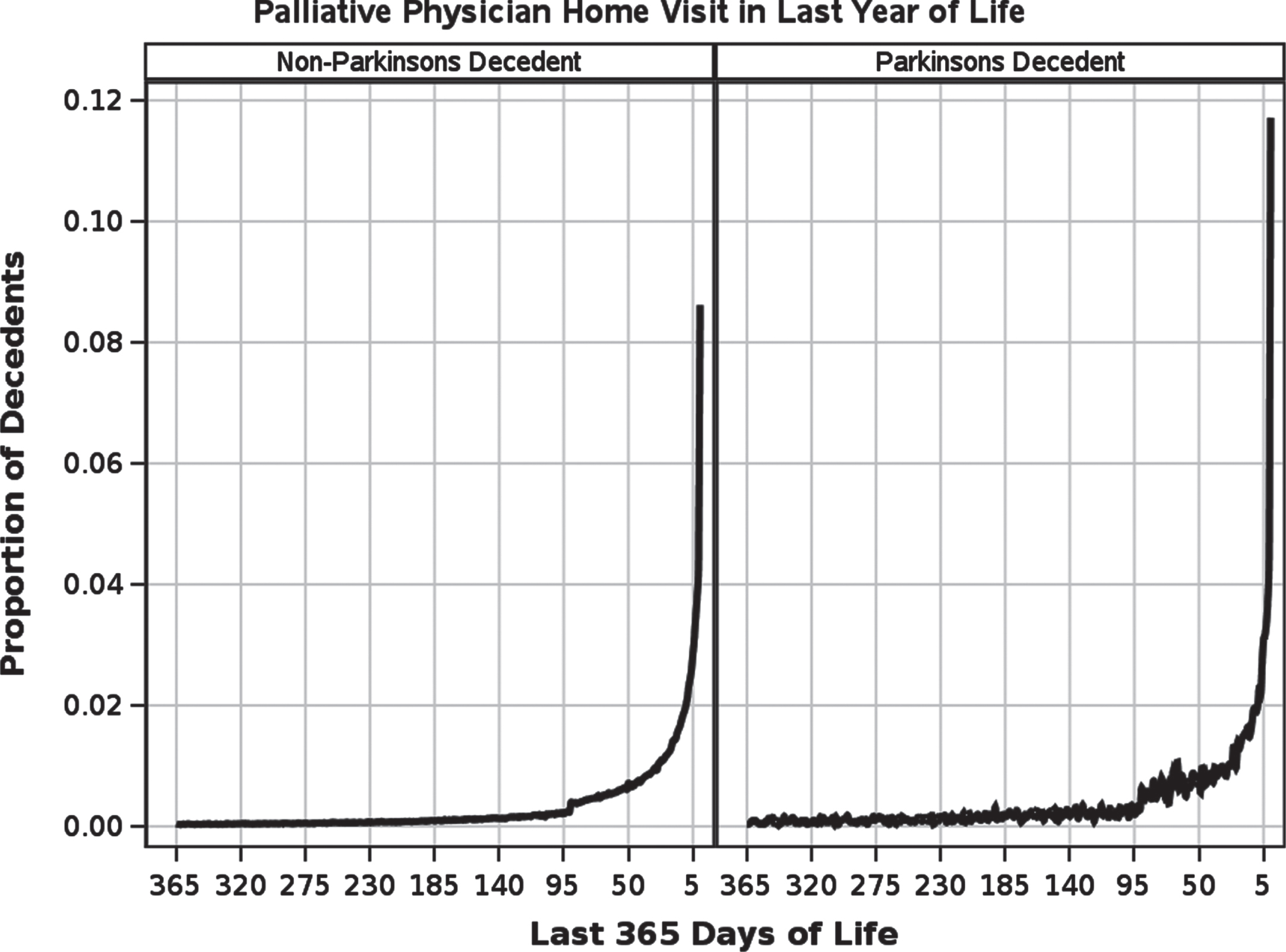
Proportion of decedents with a palliative physician home visit in last year of life stratified by Parkinson’s disease status.
Place of death
Overall, decedents with PD were less likely to die in an acute care setting compared to decedents without PD (36% vs. 46%, p < 0.001) (Supplementary Table 3) but more likely to die in long-term care (39% vs. 17%, p < 0.001). However, when considering only home-dwelling decedents with PD, the rate of death in an acute care setting was higher (52%), similar for decedents without PD (54%,<0.001) (Table 1).
Multivariable analyses
When examining the likelihood of dying in an acute care hospital among home dwelling PD decedents, we found that receipt of palliative homecare and receipt of at least one physician palliative home visit were both associated with significantly lower odds of dying in hospital when compared to no palliative care involvement (Odds Ratio [OR] 0.24, 95% confidence interval [CI]: 0.19–0.30, and OR: 0.38, 95% CI 0.33–0.43 respectively), controlling for covariates (Table 2). Living in a rural residence was associated with greater likelihood of dying in acute care (OR 1.42, CI:1.17–1.72). When examining the likelihood of receiving a physician palliative home visit among community dwelling PD decedents, we found that the odds increased with increasing age (age 45–55 OR:.32, CI: 0.16–0.66 vs. age 85–95: OR: 0.72, CI:0.55–0.95), as well as increasing income (highest income quintile: OR: 1.28, CI: 1.08–1.51). The odds of receiving a physician palliative home visit was lower for decedents living in a rural setting (OR 0.77, CI: 0.65–0.92) and patients who had visited a neurologist in the last 3–12 months before dying (OR 0.8, CI 0.72–0.89).
Multivariable logistic regression modelling the odds of dying in an acute care hospital for individuals with Parkinson’s disease
Health care costs in the last year of life
Table 3 illustrates the total and mean cost of care per decedent in the last year of life for individual sectors of the health care system. The total mean cost was greater in the PD cohort. The greatest difference in costs stem from long-term care expenditures, which were more than two-fold greater for the PD cohort ($21, 955.75 vs. $9,003.21, p < 0.001). Costs pertaining to homecare and complex continuing care were also greater for the PD cohort ($6,858.68 vs. $4,370.12, p < 0.001; $4, 910.10 vs. $3, 253.76, p < 0.001 respectively). By contrast, costs pertaining to inpatient care, emergency department visits, outpatient physician billings were all lower in the PD cohort.
Mean costs (Canadian dollars adjusted to 2020) per decedent in the last year of life by health care sector
ODB, Ontario Drug Benefit; SD, standard deviation.
DISCUSSION
This study uses population-based data from a universal health care system to provide a robust overview of end-of-life health care service utilization and costs across a wide range of healthcare sectors for decedents with PD. Our study demonstrates that patients with PD require extensive, chronic support in the last year of life.
Half of patients with PD are admitted to a long-term care facility in their last year of life, while home-dwelling individuals with PD are receiving 3 times as many days of homecare compared to their non-PD counterparts. As the population of PD is expected to increase substantially in the next 10 years [18] funding for long-term care and homecare support services as well as training health care workers (such as nurses and personal support workers) will be essential to support this patient population in the coming years.
Despite lower use of acute care services, homecare and long-term care costs result in higher overall healthcare costs for PD decedents. To date, there are no comparable studies examining end of life health care costs in patients with PD however, our findings support other studies which have shown higher costs of care for the PD population compared to the general population [5, 19]. In this Canadian study, the annual total direct medical costs associated with PD decedents were $6,826 United States dollars [USD] [adjusted to 2020] greater than the costs associated with non-PD decedents. A previous study from the US showed that there was an even higher cost burden, with the annual total medical costs for PD patients being $24,423 USD [adjusted to 2017] greater than the general population [5].
Our findings suggest an opportunity to reduce costly acute care deaths and improve quality of end-of-life care by increasing palliative care at the end of life. In our study, less than one-third of community-dwelling individuals with PD received palliative homecare; however, palliative care was associated with reduced risk of dying in hospital. Most individuals report they would prefer to die at home [20]; however, in our study, half of home-dwelling decedents with PD died in acute care setting. This is similar to rate of acute care deaths seen in PD patients in the United Kingdom (UK) (43%) [21]. It is in stark contrast to the US where 9% of patients with PD die in hospital [22]. In the US, between 2003 and 2017, there was an absolute decrease in the proportion of hospital and long-term care deaths in individuals with PD by 9% and 10% respectively and an increase of home deaths and hospice deaths by 11% and 8% [22]. Reductions of the proportion of acute care deaths of the general population in the US and in the Netherlands have been attributed to palliative care including home-based care and hospices [23].
There are several reasons palliative care is not routinely part of the multidisciplinary team treating those with PD. Palliative care has traditionally been organized to provide care to patients in the terminal phase; however, individuals with PD may develop progressive disability over 10–15 years and the terminal phase is harder to define. This slow deterioration and uncertainty with respect to prognostication may be part of the reason that patients did not access palliative care until the last few weeks of life. Late access to palliative care limits the opportunity to provide symptomatic care, have advance care planning discussions and set up supports for end-of-life care. Earlier introduction of palliative care may help to maximize quality of life for patients and families, while reducing use of unnecessary hospital services and associated expenditures [24]. Although personalized algorithms predicting life expectancy exist for patients with amyotrophic lateral sclerosis [25] and for elderly individuals requiring homecare [26], to date, there is no such algorithm for individuals with PD. A personalized tool to predict mortality in patients with PD could act as a red flag for palliative care consultation. Other studies have demonstrated that an algorithm-based alert to clinicians of a high mortality risk was associated with increased rates of goals of care discussions and palliative care consultation [27, 28]. Although some indicators of mortality are shared by other groups for which personalized algorithms have already been developed— such as recent weight loss, comorbid illness, dependence for activities of daily living which also predict mortality in community dwelling elderly [26]— other indicators that are more specific for PD could further refine predictive models of mortality for the PD patient population (for instance, by including indicators such as postural instability, dysphagia or dementia) [29]. In 2021, Akbar et al. suggested PD specific hospice referral criteria independent of mortality including 1) critical nutritional impairment, a life-threatening complication in the prior year, or motor symptoms impairing the ability to perform self-care; 2) rapid or accelerating disease progression and disability; or 3) advanced dementia meeting the hospice referral criteria [29]. Whether these recommendations will improve access to hospice care remains to be seen.
In this study, only half of community dwelling patients with PD had seen a neurologist. This is a concern as PD care by a neurologist has been previously shown to be associated with fewer adverse events, less acute care utilization and fewer hospitalizations and rehospitalizations [30, 31]. The low rate of neurologist consultations may reflect difficulty coming to the clinic due to high disability as patients admitted to long-term care had the lowest rates of visits with a neurologist (21%). The association of outpatient neurologist consultations with lower rates of palliative care consultation may reflect that neurologists are seeing the healthier, more mobile population of patients with PD. Recent widespread use of virtual visits may improve access to neurologists. Our study data supports the need for neurologists to have conversations regarding advance care planning and prognosis early in the disease, when patients and caregivers are still coming to clinic.
The major strength of this study is the inpatient and outpatient health care information it provides at a population-level. Furthermore, the diagnosis was established using a diagnostic algorithm that was validated using the same administrative database [15]. We also employed detailed case-costing methodology to assess the economic burden of PD [17]. Nevertheless, our study also has several limitations. One major limitation of this study is that the retrospective design may have introduced a source of selection bias which is responsible for the shorter than expected time from diagnosis to death (i.e., 7 years vs. 16 years [32]) and the greater mean age at diagnosis (77 years vs. 65–66 years [18, 32]). By selecting patients who had been diagnosed with PD within 20 years prior to death, we are including all of the oldest patients with PD and potentially excluding some patients with early onset PD. As a result, this would increase the average age at diagnosis. In addition, previous studies have shown that patients with late onset PD have shorter disease duration (11 years vs. 20 years) and survive longer than patients with early onset PD (mean age at death: 77 years vs. 65 years) [33, 34]. In our study, the older age at the time of death among PD decedents (compared to non-PD decedents) was likely related to the late age of diagnosis and the corresponding lack of early deaths in this group. For example, death occurred under the age of 65 years in 20% of non-PD decedents versus only 3% of PD decedents. Despite this selection bias, we feel that the end-of-life health care utilization trends reported in this population-based study— which uses administrative data collected within a single payer healthcare system— accurately reflects the experience of the vast majority of PD decedents in Ontario. As each health care system is unique, the use of medical services by patients in Ontario may not reflect the reality in other countries; however, it is likely generalizable to other high-income jurisdictions with similar public-payer health care systems. The costs reported in this study underestimate the true cost of care. We did not include medication costs for patients under 65 years of age or additional services paid for privately by the decedent such a home nursing care. The comparator group of decedents without PD is heterogenous however this study primarily provides descriptive information that is irrespective of the comparator group. The PD group is also somewhat heterogenous as it includes patients with secondary PD and other parkinsonian conditions that are not strictly idiopathic PD. In the validation study the PKM group accounted for only 17% of the entire PD/PKM cohort [15] so the main trends seen in the study are likely driven by the PD cohort. In addition, the PKM patients also suffer significant disability and tend to experience a more rapid deterioration in their condition, often resulting in greater needs throughout their disease course; this population is important to consider for health care planning [35]. Although we have demonstrated there is a clear association with palliative care and reduced risk of dying in hospital, we cannot make any causal inferences about whether palliative care itself led to this finding. As we are limited by the information that is routinely collected in the linked databases, there may be confounding factors affecting the access to palliative care or rate of death in acute care that were not detected with this study.
Conclusions
This study demonstrates the costly and chronic nature of health care needs of individuals with PD in the last year of life; half of all decedents with PD are either receiving homecare or residing in a long-term care facility. Although home-based palliative care is associated with a reduced risk of dying in hospital, only a minority of home dwelling patients with PD receive palliative care in the last year of life and half die in an acute care facility. Therefore, there is an opportunity to reduce costs while improving quality of life for patients with PD by providing more palliative homecare in the last year of life. Further research is required to identify individuals with PD at end-of-life in order to initiate palliative care in a timely fashion.
ACKNOWLEDGMENTS
This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). This study also received funding from the Canadian Institutes for Health Research Grant No. PJT-153251 and from the Bruyère Research Institute Center for Individualized Health. Tanuseputro is supported by a Physicians’ Services Incorporated (PSI) Graham Farquharson Knowledge Translation Fellowship.
This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). This study also received funding from the Canadian Institutes for Health Research Grant No. PJT-153251 and from the Bruyère Research Institute Center for Individualized Health.
Parts of this material are based on data and information compiled and provided by Canadian Institutes of Health Information (CIHI). The analyses, conclusions, opinions and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred. We thank IQVIA Solutions Canada Inc. for use of their Drug Information File.
ICES is an independent, non-profit research institute funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care (MLTC). As a prescribed entity under Ontario’s privacy legislation, ICES is authorized to collect and use health care data for the purposes of health system analysis, evaluation, and decision support. Secure access to these data is governed by policies and procedures that are approved by the Information and Privacy Commissioner of Ontario.
CONFLICT OF INTEREST
None of the authors have any disclosures or conflicts of interest to report.
