Abstract
Background:
There is currently insufficient long-term data on costs of treatment in patients with Parkinson’s disease (PD), which is chronic and progressive, and associated with substantial healthcare costs. Identifying patterns in healthcare utilization and cost may illuminate further discussion on early intervention.
Objective:
To characterize long-term healthcare utilization and costs of PD in newly diagnosed patients managed by movement disorder specialists.
Methods:
Using a longitudinal matched-cohort study of linked data from the National Neuroscience Institute Parkinson’s disease and Movement Disorder and healthcare administrative databases in Singapore from 2008–2017, we compared healthcare utilization and costs between patients and controls matched on age, sex, race, and Charlson Comorbidity Index score.
Results:
1,162 patients met study inclusion criteria and 1,157 matched controls were identified. The total mean annual healthcare cost (at 2017 costs) was significantly increased in patients compared to controls from years 1–9 post-diagnosis. The increased cost was observed 2 years before diagnosis (USD2322 vs. 2052; p < 0.001). Mean annual cost attributable to PD increased from USD1854 at 1-year post-diagnosis to USD2652 at 9 years. Over 9 years, average costs were significantly higher across all domains of healthcare utilization except primary care—cost of intermediate and long-term care was increased by a factor of 2.5, specialist care by 2.3, emergency department visits by 1.6, and hospital admissions by 1.3.
Conclusion:
PD results in higher healthcare utilization and costs. Pre-diagnosis increase in healthcare utilization observed in patients supports the presence of prodromal PD symptoms and may present an opportunity for early diagnosis.
INTRODUCTION
Neurological disorders are the leading cause of disability, and amongst them Parkinson’s disease (PD) is growing at the fastest rate. There are presently 6.2 million individuals living with the disease, with the number projected to grow to 12.9 million by 2040 [1]. PD is a neuro-degenerative disorder that is associated with increased age. Thus, it can be reasonably postulated that global prevalence will increase with an aging population [2, 3]. The economic burden of PD is expected to grow exponentially due to the chronic and progressive nature of the disease with the development of motor and non-motor complications [4–10] as well as the increasing prevalence.
At present, there are few studies that explore long-term cost of illness in PD and fewer yet that combine both clinical and administrative databases [9–15]. The key limitations of existing studies are short duration of follow-up, recall bias associated with patient-reported costs, small sample sizes, and cross-sectional designs that do not allow the analysis of utilization and costs trends with disease progression [5–7, 15–20]. We therefore undertook this study to examine and compare longitudinal patterns of healthcare utilization and cost amongst newly diagnosed PD patients managed by movement disorder specialists and those without the disease, and to 1) estimate the incremental cost of care in PD patients and 2) determine the main domain drivers of healthcare cost, to inform future policy and fiscal planning for the long-term care of PD.
METHODS
Study design and data sources
This is a cohort study that utilizes two databases: a clinical database from the National Neuroscience Institute’s Parkinson’s Disease and Movement Disorder (PDMD) database and an administrative medical claims and subvention dataset from the Ministry of Health (MOH), Singapore. The PDMD database collects prospectively, from 2002, sociodemographic and medical information of all patients diagnosed with PD based on the National Institute of Neurological Disorders and Stroke (NINDS) criteria [21] at a tertiary referral center in Singapore and who are followed up by a movement disorder specialist. The MOH’s national administrative database captures claims from all public and private hospital inpatient, day surgery and outpatient visits covered under MediSave (a national medical savings scheme that all Singaporean citizens and permanent residents are enrolled in) and MediShield (a national health insurance scheme) from 2002. This database also captures subsidized primary care expenditures at polyclinics (public primary care clinics) and private general practitioners (GP) from 2007, and intermediate and long-term care (ILTC) services from 2014. Thus, in terms of patient population, the PDMD database forms an approximate subset of the national administrative database, as tertiary referral centers in Singapore fall under the umbrella of public or private hospitals. Insofar as patients at our center made claims for their hospital visits and/or specialist outpatient care services under MediSave or MediFund, their data are also correspondingly captured in the national administrative database. Our study only accounts for direct healthcare costs and does not include indirect costs such as informal care and loss of productivity.
Selection of cases and controls
The study period was from 2008 to 2017 as data for matching cases to controls were available from 2008 onwards and claims data for primary care visits were available up till 2017. Cases were selected from the PDMD database according to the following criteria: 1) diagnosed with PD according to the NINDS criteria and verified by a movement disorder specialist, 2) aged 30 and above at time of diagnosis, 3) seen within 24 months of diagnosis by a movement disorder specialist, and 4) had a minimum of 2 follow-up visits to the neurologist or movement disorder specialist after the first clinic visit so as to allow for a minimum of 12 months of follow-up data to calculate annualized costs following index diagnosis. This yielded 1,162 cases.
The cases were then matched with patients without PD from the MOH database. Each case was matched to a control who was alive in the index year (year of diagnosis). Pairs were matched based on the following criteria: 1) age at diagnosis (in 3-year age bands), 2) race, 3) sex, and 4) modified Charlson Co-morbidity index [28] (CCI). The calculation of the modified CCI included all diseases in the CCI except dementia: acquired immunodeficiency syndrome, acute myocardial infarction, cancer, cerebrovascular disease, chronic pulmonary disease, congestive heart failure, diabetes, liver disease, peptic ulcer disease, peripheral vascular disease, moderate or severe renal disease, and connective tissue disease. Dementia was excluded as a comorbidity because a significant proportion of PD patients have cognitive impairment at diagnosis or develop cognitive impairment during the course of illness [22–27].
For the 1,162 cases, a 1:1 matching with replacement [29] was performed, and 1157 cases (99.6%) were successfully matched to controls. Controls who were matched to a case were excluded from the matching pool for 1 calendar year and re-introduced into the matching pool for subsequent years, such that 1 control could not be matched to more than 1 case within a year. Of all 1,157 controls, 1,155 were unique, after accounting for replacement.
Healthcare utilization and cost
Utilization (number of events) and total medical cost data (including consultation, medications, procedures and ancillary healthcare services) were extracted for the following healthcare domains—emergency department (ED) visits, hospital admissions, specialist outpatient care (SOC), primary care, and ILTC. ILTC services are heavily subsidized community healthcare services provided by government-linked/approved agencies or institutions; one branch of ILTC services encompasses center-based or residential services, which include the provision of physiotherapy, occupational, and speech therapy for the purpose of community rehabilitation.
Utilization and costs were calculated for 365-day periods from the index date for each domain. Results were obtained for 1–2 years prior to index date and 1–10 years post-index date, but as the sample size (n = 62) for 10-years post-index date was too small, it was excluded from the analysis. The mean annual utilization and cost for each patient was estimated for each available year of follow-up [15]. A detailed description of each domain is provided in the Supplementary Material.
Analysis of cost
This study extracted resource utilization and total medical cost incurred and employed the matched control cost-of-illness approach to study cost attributable to PD [15, 31]. In addition to demographic profile, cases and controls were also matched on CCI score, thus allowing us to approximate the burden of comorbidities. The cost attributable to PD was calculated by subtracting the mean cost incurred by controls from the mean cost incurred by cases. A 3% per annum inflation rate was applied to all costs except for SOC cost to account for inflation and cost variations; all costs presented are in 2017 costs. For SOC cost, a standard unit cost of SGD200 was multiplied to all recorded events. The standard rate is the calculated mean cost of one specialist care visit (regardless of medical or surgical discipline), and is inclusive of consultation, medication, and ancillary care. Results are presented in Singapore dollars (SGD) while costs are converted to and presented in US dollars (USD) in the discussion for ease of comparison with other international studies. A rate of SGD1.3360 to USD1 was employed, based on the 2017 yearly average exchange rate published by the US Treasury Department.
All analyses were conducted using Stata software (StataCorp. 2015. Stata Statistical Software: Release 14. College Station, TX, USA: StataCorp LP). Differences in utilization and costs variables between the matched groups were evaluated using Wilcoxon signed-rank tests and the results are presented as mean and standard deviation. Statistical significance was set at p < 0.05.
Ethics
The study was approved by our local Institutional Research Board.
RESULTS
Cohort profile
In total, 1157 PD patients were matched to controls based on age, sex, race, and CCI score at baseline. The number of PD patients and controls with utilization and cost data dropped year-on-year; there were 132 PD patients and controls at year 9. The demographic profile of PD patients and controls are summarized in Table 1. The mean (SD) age of the cohort was 66.1 (9.8) years, and there were slightly more males (55.7%). Majority of case and controls were Chinese (88.5%). Most PD patients (82.6%) were at Hoehn and Yahr (HY) stages 1–2 at baseline, reflecting correctly that this was a newly diagnosed cohort of patients with early disease. The cohort was followed up for a mean (SD) duration of 5.4 (2.6) years.
Demographic profile of PD patients and controls
CCI, Charlson Comorbidity Index; H&Y, Hoehn & Yahr.
Average healthcare utilization and cost
PD patients recorded significantly greater average utilization of healthcare services across all domains in comparison to matched controls (Table 2). Compared to controls, PD patients had more hospital admissions (0.4 vs. 0.3) and primary care visits (4.2 vs. 3.8), an almost 2-fold increase in ED visits (0.5 vs. 0.3) and specialist visits (5.3 vs. 2.3), and a 3-fold increase in ILTC use (7.1 vs. 2.5) per year. Primary care utilization was comparable between PD patients and controls (4.1 vs. 3.8).
Average utilization and cost of healthcare per year of follow-up post-diagnosis
Wilcoxon signed-rank test is performed to compare utilization and cost variables between PD patients and matched controls. All differences are significant at p < 0.05, except for the domain primary care (p = 0.15). PD, Parkinson’s disease; ED, emergency department; ILTC, intermediate-and-long-term care facilities.
A similar trend was observed when comparing healthcare costs between PD patients and controls: PD patients had significantly higher cost across all healthcare domains except primary care; the cost of ILTC was increased by a factor of 2.5, specialist care by 2.3, ED visits by 1.6, and hospitalizations by 1.3. Across 9 years of follow-up, the average yearly cost of healthcare in PD patients was SGD6143±9401 compared to SGD3822±9026 in controls. Overall, hospitalizations accounted for the majority of total cost for PD patients (48.6%), followed by ILTC (26.3%).
Time trends in healthcare costs
Total healthcare cost
Mean total annualized healthcare cost was consistently higher amongst PD patients compared to controls through years 1–9 post-diagnosis (Fig. 1). The increase in cost was observed 2 years before diagnosis (SGD3102 vs. 2742; p < 0.001).
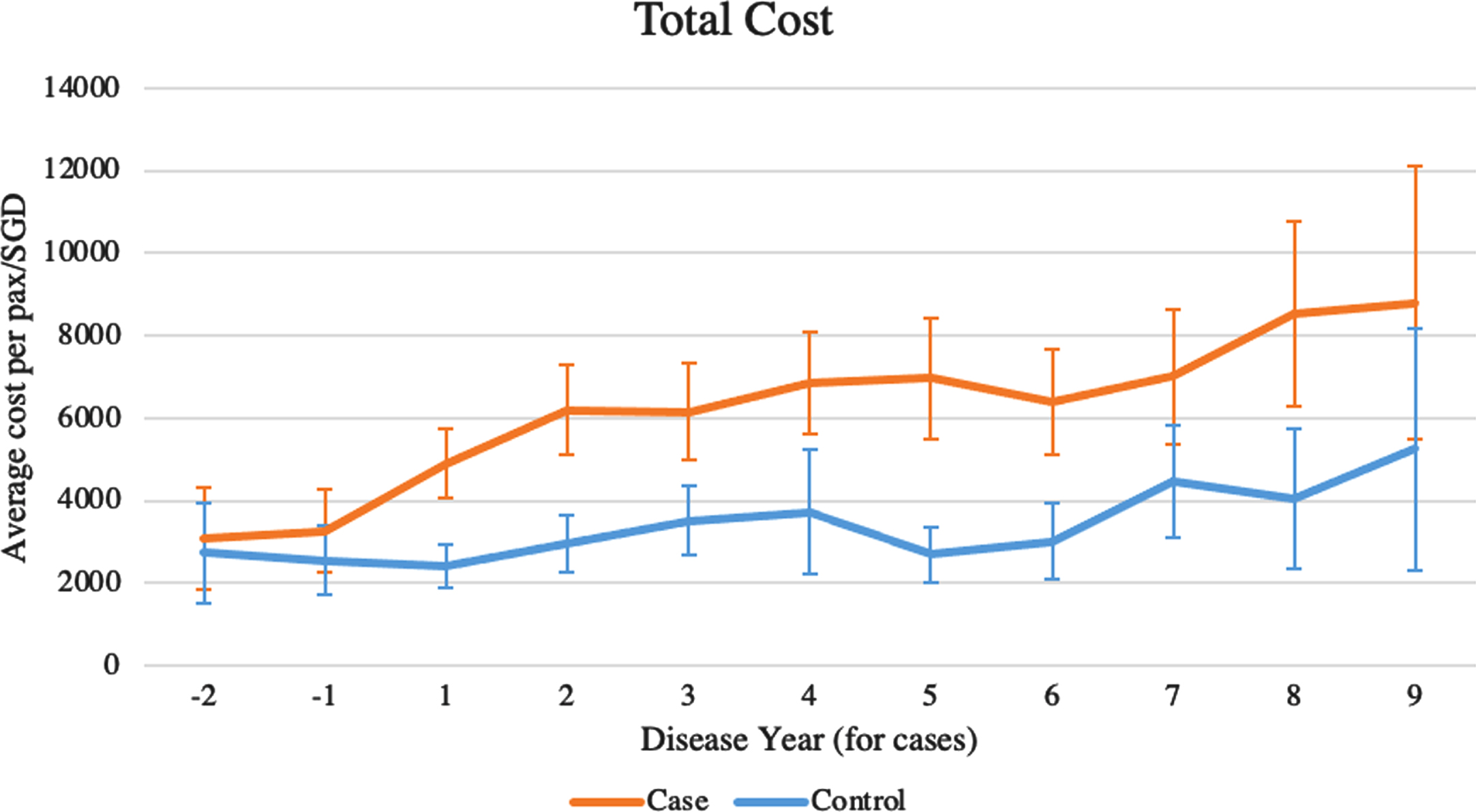
Average annual cost, total. Wilcoxon signed-rank test is performed to compare total annual healthcare cost incurred by PD patients and matched controls respectively. All differences are significant at p < 0.05. Each bar represents 95% confidence interval.
Domain specific healthcare cost
Mean annualized cost was higher amongst PD patients across all healthcare domains, except for primary care (Fig. 2). Costs of ED visits, hospitalizations, SOC, and ILTC increased steadily over 11 years in both PD patients and controls. Notably, the cost of specialist care amongst PD patients increased sharply by 91.4% between the years immediately before and after diagnosis, from SGD587 one year prior to index date to SGD1124 one year after index date. After the index date, PD patients incurred almost double the cost of specialist care compared to controls yearly (Fig. 2d).
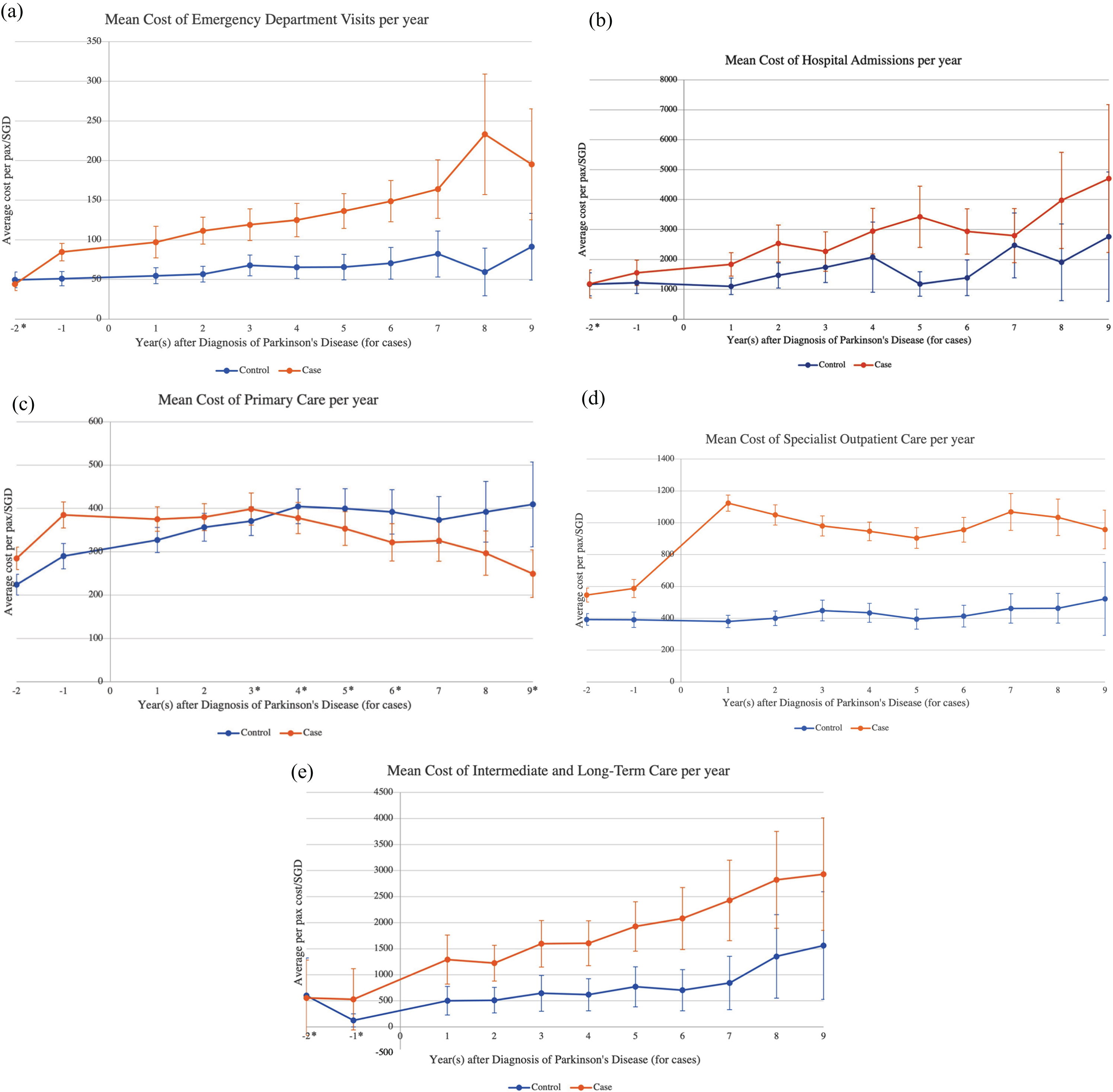
Average annual cost, by healthcare domains. Wilcoxon signed-rank test is performed to compare annual cost incurred by PD patients and matched controls respectively in each healthcare domain. All differences are significant at p < 0.05 except for years indicated with an asterisk (*). Each bar represents 95% confidence interval.
Primary care cost for patients with PD increased from SGD284.78 at 2 years prior to index date to SGD399 at 3 years after index date, after which it decreased steadily at an average rate of 7.81% per year to SGD249 at year 9. In contrast, primary care cost for controls increased from SGD224 at 2 years prior to index date to SGD409 at year 9.
Cost attributable to Parkinson’s disease
The cost attributable to PD increased from SGD360 at 2 years prior to index date to SGD2477 in the first year of diagnosis to SGD3543 at year 9 (Fig. 3). Costs of ILTC, hospitalizations, and specialist care account for majority of the incremental cost, with each domain contributing an average of 41.7%, 29.9%, and 25.9% respectively to the total attributable cost per year, post index date.
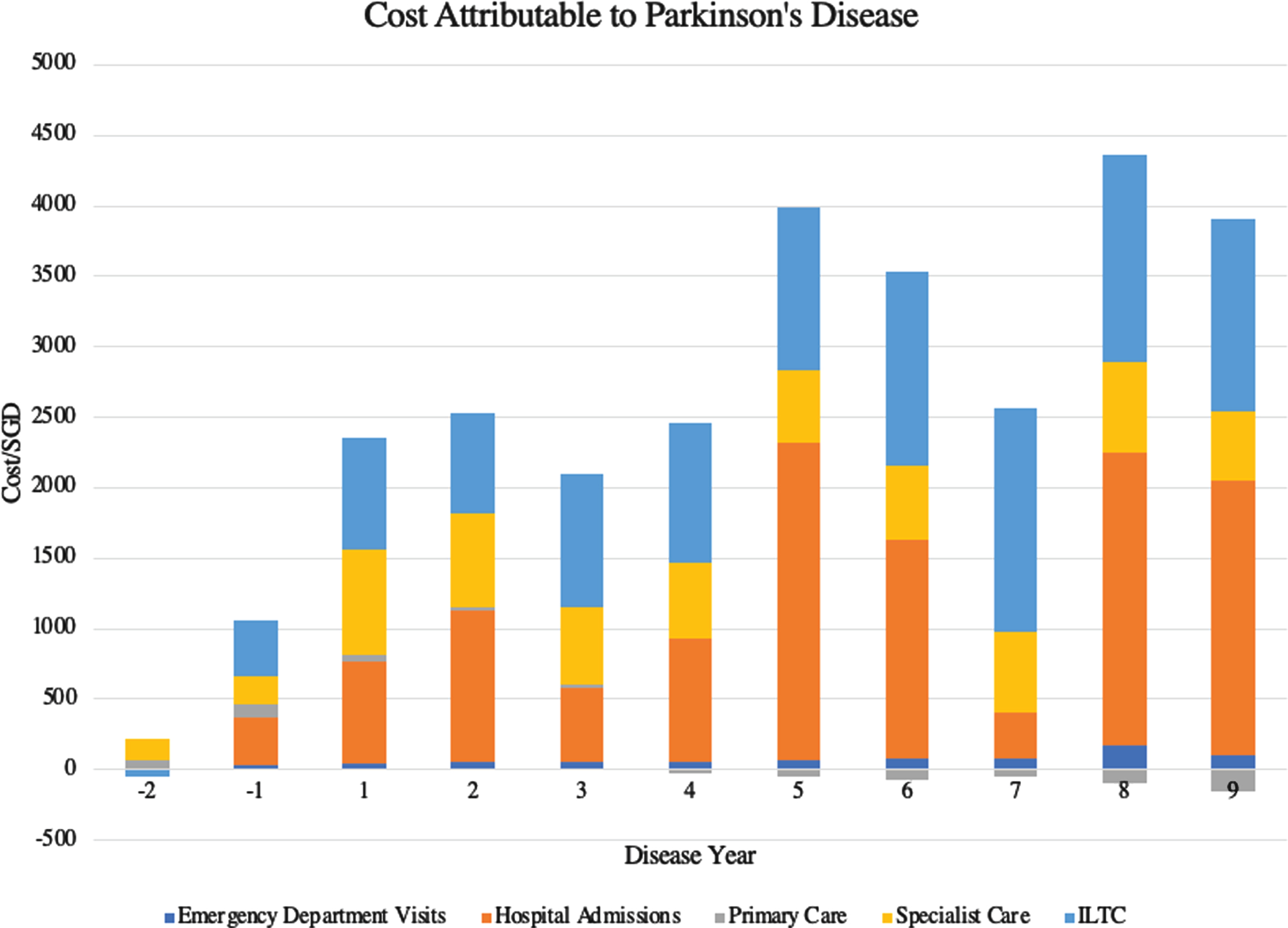
Cost attributable to Parkinson’s Disease.
DISCUSSION
This study evaluated the longitudinal cost of care in PD patients managed by movement disorder specialists by determining the incremental healthcare utilization and cost incurred by PD patients compared with controls matched on age, sex, race, and comorbidities. It adopted a net cost approach to take into consideration all medical costs that made be directly or indirectly associated with PD. The average annual utilization rates and costs incurred by PD patients post-diagnosis, compared to controls, were higher across all healthcare domains except primary care. Attributable cost to PD rose from SGD2477 (USD1854) to SGD3543 (USD2652) over 9 years after diagnosis. Higher costs were observed up to 2 years prior to PD diagnosis (SGD3102 vs. 2742; USD2322 vs. 2052).
While this cost difference was also observed in the United Kingdom (£3021 in 2013 costs) [15] and the United States (USD5362 in 2013 costs) [32] in similarly designed studies looking at direct medical costs, the absolute cost difference and percentage increase in PD patients found in this study are significantly lower. A cross-sectional study conducted in Singapore also found that the mean annual total cost of PD (direct and indirect costs) was much lower in Singapore (USD10,129 in 2008 costs), compared with other developed Western countries [20]. A more recent study of elderly Medicare beneficiaries with PD reported primary PD-related costs of USD3429 (adjusted to 2018 costs), but almost half the cohort was ≥80 years (compared to our patients’ mean age of 66.2 years) [33]. Age is a well-established predictor of cost-of-illness in PD [9, 33]. Aside from differences in study methodologies, these discrepancies in costs are likely to be accounted for by differences in healthcare financing, cost structure, as well as efficacy and efficiency of the healthcare system [20, 34].
Additionally, approaches to pharmacological and non-pharmacological treatment of PD patients also differ according to region. The relative lower cost of healthcare amongst PD patients in Singapore compared to their Western counterparts may be in part due to lower utilization of dopamine replacement therapy, measured in levodopa equivalent daily dose. In a study of pharmacological treatment in a newly diagnosed cohort of PD patients from our center, we reported mean (SD) LEDD of 204 (113) [35]. Another center in Singapore reported LEDD of 352.45 (305.36) in patients with average duration of disease of 5.80 years [36]. Overall, these levels are lower than those reported in Western countries [36–38]. The cost discrepancy may also be attributed to lower rates of advanced surgical treatment in Singapore compared to other developed Western countries. From 2007 to 2017, our center performed deep brain stimulation (DBS) on 67 patients (out of 3556 patients), resulting in a utilization rate of 1.88%. A similar rate of 1.8% was also reported in another cohort of PD patients in Singapore [35]. This is lower than the average of 5–10% in other high-income countries [39]. Not taking into consideration the difference in cost of DBS across the various high-income countries, the lower utilization of DBS in Singapore may contribute to the lower average cost of healthcare for PD patients. However, this will not account for differences that are already apparent in the initial years of disease when patients are not yet DBS candidates.
Our study demonstrated an increase in healthcare utilization and cost even before the diagnosis of PD, with significant differences in primary care (3.5 (6.1) vs. 2.6 (4.0)) and specialist care events (2.4 (3.8) vs. 1.7 (3.2)) and costs at 2 years pre-index date. This was not observed in 2 previous studies where direct medical costs did not differ between PD patients and controls in a year just before diagnosis [32, 41]. The majority of our patients (82.6%) had early-stage disease (HY stage ≤2) at diagnosis. While the results could be confounded by the minority of patients who already had more advanced disease at diagnosis, a better explanation is the phenomenon of prodromal PD. Subtle motor and non-motor symptoms have been reported in PD patients up to 10 years prior to PD diagnosis. In particular, balance impairment, hypotension, urinary dysfunction, dizziness, depression, and fatigue have been recorded up to 5 years pre-diagnosis while constipation and tremor were observed at higher incidence rates up to 10 years pre-diagnosis [42]. This increased attendance of PD patients at primary and specialist care facilities affords clinicians the opportunity of conducting screening in a suitable clinical setting so as to identify patients with prodromal PD symptoms and allow for timely intervention or clinical trials. The recently updated Movement Disorder Society web-based prodromal PD risk calculator can further facilitate the identification of this group of patients who will need closer monitoring [43].
PD patients utilized more healthcare resources across all domains except primary care and incurred more costs correspondingly, compared to matched controls. The sharp increase in specialist care utilization and cost in the immediate year post-diagnosis can be attributed to the cost of investigations and active management of PD. This is contrasted with the time trend of primary care cost in our PD patients, showing that cost fell steadily from year 3 to year 9 post-diagnosis. This model of PD care in Singapore, in which there is a transfer of care from the primary care setting to specialist care at the point of diagnosis is in keeping with the American Academy of Neurology’s quality measures recommendations that neurology care should be available throughout the course of illness [44]. While previous studies have shown that neurologist care in PD is associated with fewer hospitalizations and acute care utilization, the jury is still out on its cost-effectiveness [45, 46].
Hospitalizations were the costliest events for PD patients and controls alike, contributing the most to total healthcare costs annually. The greatest domain driver of incremental cost when comparing PD patients to controls was ILTC, with PD patients using almost 3-fold the services and incurring a similar increase in cost. ILTC services comprise home-based (medical and nursing, meal delivery, personal care, and transport), center-based (rehabilitation, day care, and hospice care), and residential services (community hospitals and nursing homes). As PD patients are frequent users of these services, ILTC may be an appropriate setting to proactively screen them for potential disease complications and intervene to prevent costly hospitalizations. As observed in our study, PD patients encountered healthcare providers in ILTC settings more often than their specialist care providers (7.1 vs. 5.3 events/year). Thus, it is crucial that healthcare providers in the community are equipped to care for PD patients, to ensure continuity and completeness of their disease management. This may call for a multifaceted approach of restructuring of healthcare delivery systems (including fiscal policies), continuing medical education, training community-based healthcare professionals, and developing evidence-based guidelines [47, 48].
To our knowledge, this study is the first that takes a longitudinal view of cost-of-illness with a relatively large sample size and that combines objective administrative cost data with clinical information. A chief advantage of our study is the completeness of our direct medical cost estimates as the national MOH administrative covers at least 95% of citizens and permanent residents and captures long-term care costs, including institutionalization costs. The longitudinal nature of our data allows a better understanding of patterns and heterogeneity in healthcare utilization and costs over the natural history of PD, to facilitate intervention and policy planning. The diagnosis of PD in our cohort is robust as all patients were diagnosed and managed by a movement disorder specialist.
There are some limitations in this study. Firstly, this study did not take into account indirect costs in the form of informal caregiving or productivity losses. Thus, it is likely to underestimate true societal cost of disease as previous studies [8, 19] have shown that direct medical costs only account for 30–35% of total cost-of-illness. However, it was not the aim of our study to capture all cost-of-illness, but rather to identify patterns that can translate into areas for improvement in care and ultimately, reduction of cost to patients and the healthcare system. Drawing data from administrative databases ensures high fidelity and reduces recall bias inherent to financial burden questionnaires often used to capture indirect cost data. Secondly, the MOH database may underrepresent primary care utilization. Primary care in Singapore chiefly comprises polyclinics (public primary care institutions) and private GP clinics. While all polyclinics were captured in the database, only GP clinics participating in the national Community Health Assist Scheme were captured, resulting in a coverage of approximately 62% of all GP clinics. In addition, PD cases were drawn from the database of a tertiary healthcare institution where patients receive specialist care. The cost of specialist care may hence be overestimated amongst cases. Thirdly, we did not have information on yearly LEDD or utilization rates of advanced surgical therapies in our particular cohort of PD patients, to provide possible explanation for differences in direct healthcare costs in comparison to other high-income Western countries. Lastly, we did not take into consideration the impact of comorbidities or frailty on cost of healthcare amongst PD patients; indeed, we controlled for comorbidities by matching cases and controls on modified CCI score to approximate overall PD-specific disease burden. Frailty is increasingly recognized as an important factor for health outcomes in PD and future observational study analyses will have to adjust for it. [49]
This study has yielded new information on the incremental cost of care in PD and identified domains that drive this expenditure. These identified patterns can potentially inform efforts to improve PD care and reduce its economic burden, through suitable screening modalities, early medical intervention, and ancillary care to stymie disease progression and development of complications.
Footnotes
ACKNOWLEDGMENTS
This study was supported by the Singapore Ministry of Health National Medical Research Council under its Open Fund Large Collaborative Grant (MOH-OFLCG18May-0002). We would like to thank all participants and neurologists from the National Neuroscience Institute, Singapore for their support for the study.
CONFLICT OF INTEREST
Louis Chew Seng Tan has received honorarium for a speaking engagement from Lundback Singapore Pte. Ltd. on one occasion in the past year (unrelated to research project).
All other authors have no financial support or funding to disclose.
