Abstract
Background
Epidemiologic evidence from different countries shows both increasing and decreasing incidence rates of Parkinson's disease over time without clear trends.
Objective
We aimed to investigate age-specific incidence rates of Parkinson's disease and possible explanations for the observed trends in Germany over a 10-year period.
Methods
Two different random samples of subjects aged ≥50 years included in 2004–2009 and 2014–2019, each consisting of 250,000 individuals, were drawn from Germany's largest health care insurance company followed up for new Parkinson's disease cases. We compared the age-specific incidence rates of Parkinson's disease for both cohorts and performed Cox regression models to calculate the hazard ratios (HR) of PD in the second period compared with the first period, adjusted for age, sex, and risk factors for Parkinson's disease.
Results
For most age groups in men and in women, we found lower age-specific Parkinson's disease incidence rates in the second period. Cox regression analysis showed an overall 18% risk reduction in Parkinson's disease incidence (HR = 0.82, 95% confidence interval [0.76–0.89]). Mean age at diagnosis increased in men (+1.9 years) and women (+0.8 years). After adjustment for risk factors, the HR was 0.78 [0.72–0.85]. Sensitivity analysis considering the competing event of death showed an HR of 0.79 [0.73–0.86]), demonstrating the independence of time trends from changes in death rates.
Conclusions
Our data show that the risk of Parkinson's disease has decreased over time and that this decrease is independent of factors such as changes in death rates, age structure, sex, and specific risk factors.
Plain language summary
Parkinson's disease is the second most prevalent neurodegenerative disorder, showing both increasing and decreasing incidence rates with no clear trend. Our group sought to investigate age-specific incidence rates of Parkinson's disease in Germany over a period of ten years. The data that were used for our analysis, were drawn from Germany's largest health care insurance company. Our study revealed that the risk of Parkinson's disease has decreased in the German population over the investigated time frame. However, we observed that this decrease does not depend on factors such as changes in death rates, age structure, sex, and specific risk factors.
Introduction
Parkinson's disease (PD) is a progressive degenerative disorder of the central nervous system, with multifactorial origins that predominantly affects older adults. Early symptoms of PD, such as constipation, sleep abnormalities, anemia, parosmia, skin disorders, and diabetes, can occur decades before the onset of the disease. 1 Overt disease is characterized by tremors, muscle rigidity, and slowness of movement. 2 In 2019, it was reported that approximately 8.5 million people may live with PD worldwide. The prevalence of the disease has doubled in the last 25 years, with further increase projected until the year 2030. 3
In Germany, a country with a rapidly aging society, PD disease burden is expected to increase substantially. In fact, one national study which examined insurance claims data from 2011 to 2019, found an increase in the absolute number and increased morbidity and mortality in PD patients as compared to persons without PD in a predominantly rural area of Germany. 4 Another recent cross-sectional study data from 2010 to 2019 reported a stable prevalence of PD patients and a slight decrease in new PD patients relative to all patients in general practitioners and specialist offices, indicating no relative increase alongside the absolute numbers. 5 These data were complemented by a recent nationwide assessment of data obtained from pseudonymized, cross-insurance claims data examining the years 2013 to 2019 yielding successive decreases in annual incidence rates between 25–30%. This decrease was observed in all age groups above 50 years, independent of sex, and was accompanied by an overall lower proportion of women suffering from PD over time. 6
Prompted by the inconclusive results, we assessed age-specific incidence time trends of PD in two cohorts using routine health claims data from Allgemeine Ortskrankenkasse (AOK), Germany's largest health insurer. We examined how changes in age and sex structure, prodromal and risk factor composition, and mortality could explain the observed time trends.
Methods
Data
We used two random samples of members of Germany's largest public health insurance fund, the AOK, each consisting of 250,000 people aged 50 years and over at the beginning of 2004 and 2014. The AOK provides health care services to approximately 30% of the German population and 50% at the highest age. The claims data were anonymized, and we did not have access to the primary data, hence no ethical review or patient consent was required. The individuals were followed until the end of 2009 and 2019, respectively. In addition to demographic information on sex, year and month of birth/death, and region of residence, our data include inpatient, and outpatient medical diagnoses coded according to the 10th revision of the International Statistical Classification of Diseases and Related Health Problems (ICD-10, https://apps.who.int/iris/handle/10665/246208). All information was available on a quarterly basis.
To create our analysis sample, we identified new PD cases in both cohorts, excluding individuals with at least one diagnosis of progressive supranuclear ophthalmoplegia (G23.1), multisystem atrophy (G23.2-G23.3), dementia with Lewy bodies (G31.82), corticobasal degeneration (G31.0), or diagnoses of atypical and secondary parkinsonism (G21 and G22) as well as patients with valid PD diagnoses in 2004/05 and 2014/15. The first valid PD diagnosis between 2006–2008 or 2016–2018 was considered, while 2009 and 2019 were used for diagnosis validation only. PD was defined using ICD-10 code G20. Our internal validation strategy involved inpatient discharge or secondary diagnoses and verified outpatient diagnoses. Valid diagnoses required further concurrent inpatient and outpatient diagnoses or a second occurrence over time. In the case of death in the quarter of the first diagnosis, it was considered valid even without a confirmative diagnosis.
Statistical analysis
To compare the incidence rates of PD between the two periods, we calculated age-specific PD incidence rates by dividing the number of PD cases per 1000 person-years at risk by sex and period. We smoothed the frequency distribution of age at first PD diagnosis using the Stata smoother “4253eh, twice” and performed Cox proportional hazard regression models and calculated the PD hazard ratios (HRs). At first, we performed a regression model with period as an explanatory variable to quantify the time trend effect (Model 1), adjusted only for sex and age, modelled by a second-degree polynomial function (age + age2). Secondly, we ran a regression model additionally adjusted for risk factors associated with PD (type 1 diabetes E10; type 2 diabetes E11; hypertension I10; hypercholesterolemia E78; obesity E66; gastritis K29; Crohn's disease K50; traumatic brain injury S06.0, S06.1, S06.2, S06.3; alcohol misuse F10.1, F10.2; nicotine abuse F17.1, F17.2; drug abuse F11-F16, F18-19, Z71.5, Z72.2). In the third model, we performed Fine and Gray competing risk regression analysis 7 of model 2, considering death as a competing event (Model 3).
The analysis time started on 1st January 2006/2016 and was measured in months. The time of PD diagnosis was set in the middle of each quarter. The time of death was defined as the middle of the month of death. Individuals were censored at the time of change in public health insurance or at the end of the follow-up period (end of 2008/2018). The proportionality assumption was tested using Cox models. All analyses were performed using Stata/MP 16.1.
Results
Descriptive results
Our analysis sample consisted of 446,796 individuals, documented free of PD at the beginning of the observation period, in 2006 (n = 222,147) and 2016 (n = 224,649). The sex and age distributions of both PD-free cohorts are presented in Table 1. From these people, 1,295 individuals developed PD between 2006 and 2008 and 1,128 individuals developed PD between 2016 and 2018. With the exception of men aged 50–54 and women aged 50–64, the age-specific PD incidence rates observed in the second period, were consistently lower than those identified 10 years earlier (Figure 1a, b). A statistically significant decrease was observed in the 70–74 age group in men, as indicated by non-overlapping confidence intervals (Figure 1a). The incidence rate of PD for the two time periods studied increased with age as expected; this observation was documented for the age groups starting from 50–54 and up to 85–89 years. The highest age group (>90 years) had a slightly lower incidence rate (Figure 1a, b). A detailed table with the incidence rates is provided in the Supplemental Table 1.
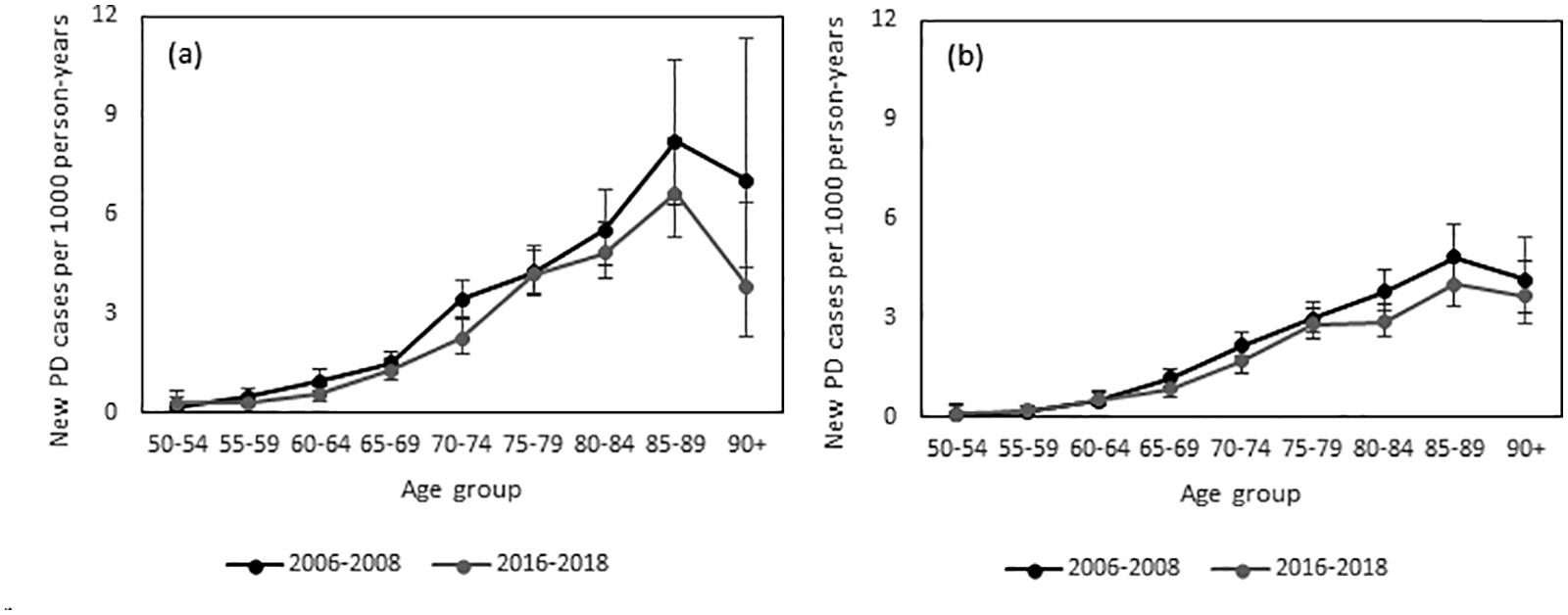
Age-specific Parkinson's disease (PD) incidence rates per 1000 person-years for the two periods 2006–2008 and 2016–2018, in men (a) and women (b). Source: AOK 2004–2009, AOK 2014–2019.
Sex and age distribution of both PD-free cohorts in 2006 and 2016. Source: AOK 2004–2009, AOK 2014–2019.
Over the 10 years, the mean age of the first PD diagnosis increased from 74.70 years [95% CI:74.03–75.37] to 76.64 years [75.95–77.33] in men and from 78.34 years [77.74–78.94] to 79.09 years [78.39–79.79] in women. Figure 2 shows the observed and smoothed distribution of age of the first PD diagnosis in men (Figure 2(a)) and women (Figure 2(b)) in both periods. For both sexes, there was a marked decrease in new cases of PD between the ages of 65 and 75. This observation cannot only be attributed to a possible lower disease burden, but also to a cohort effect, as these age groups of the second cohort come from smaller birth cohorts (see also Table 1 for the number of individuals in each age group). For men, the curve in the second period shows a significant shift to higher ages, whereas for women, the distribution is narrower in the second period.
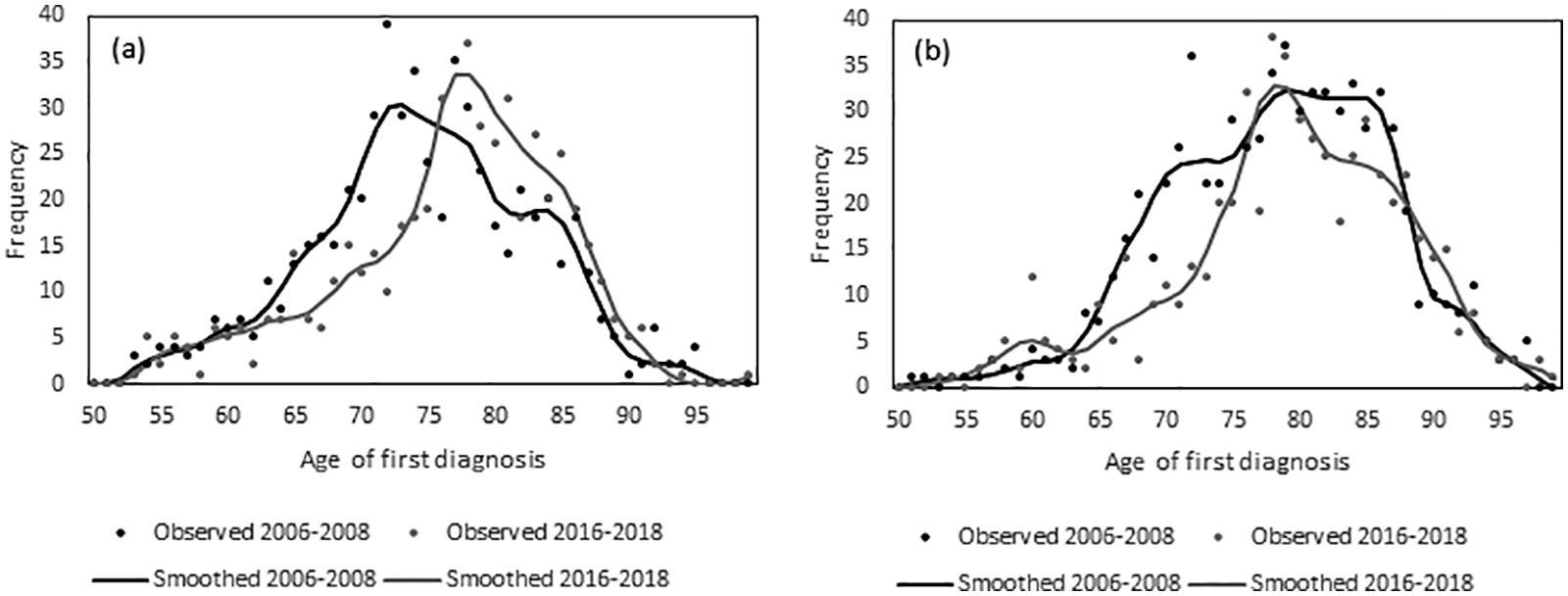
Observed and smoothed frequency distribution of age at first diagnosis of Parkinson's disease (PD) for the two periods 2006–2008 and 2016–2018, in men (a) and women (b). Source: AOK 2004–2009, AOK 2014–2019.
Model results
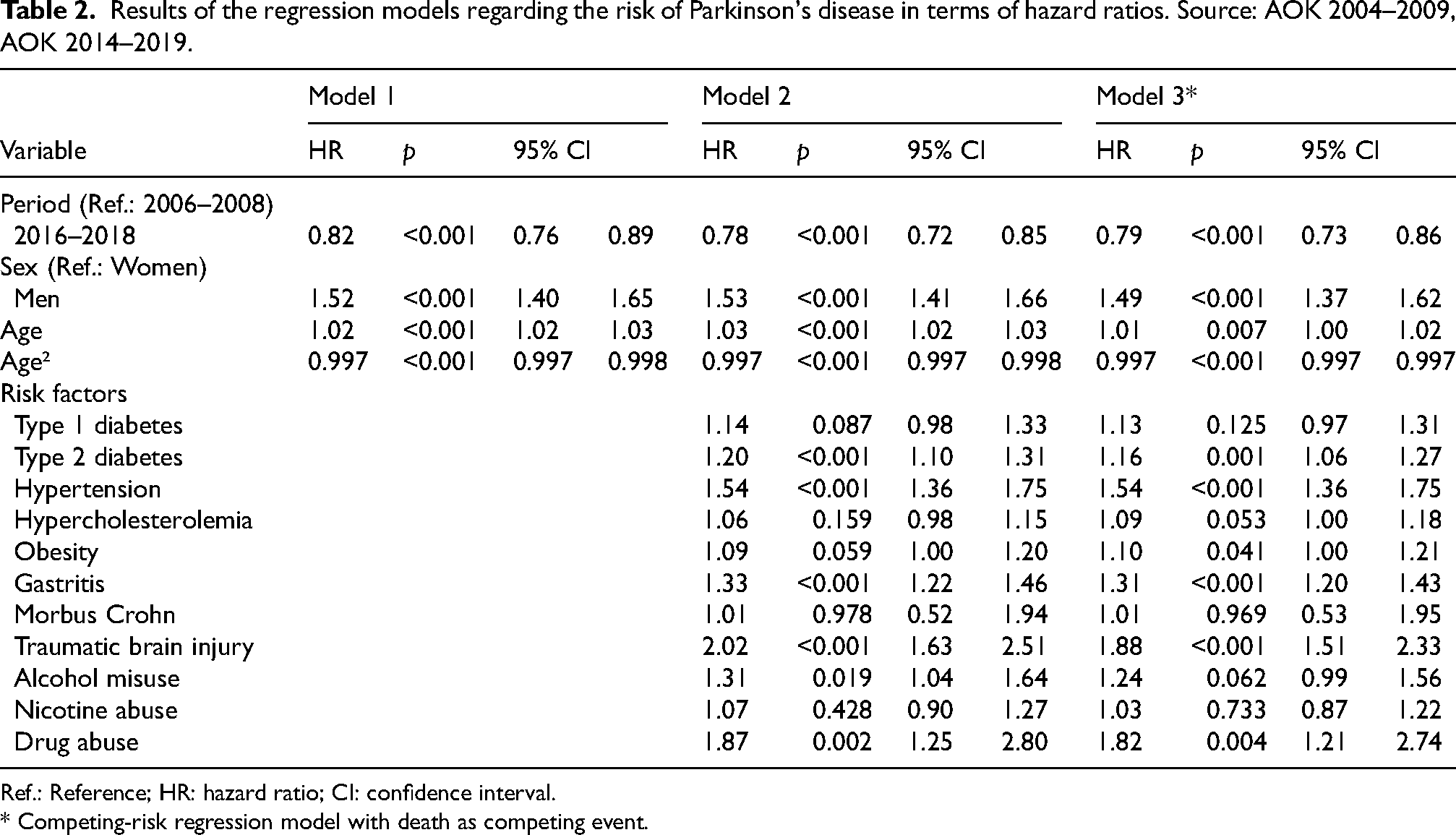
Taking into consideration age and sex, Cox regression revealed a 18% reduced risk of PD (HR = 0.82 [0.76–0.89]) in the 2016–2018 period as compared to the first period ten years earlier (Table 2, Model 1). Men had a significantly increased risk of PD (HR = 1.54 [1.40–1.65]) compared to women. The estimators for age showed a positive association with the linear term (HR = 1.02 [1.02–1.03]) and a negative association with the quadratic term (HR = 0.997 [0.997–0.998]), representing the leveling-off of PD incidence in the highest ages.
Results of the regression models regarding the risk of Parkinson's disease in terms of hazard ratios. Source: AOK 2004–2009, AOK 2014–2019.
Ref.: Reference; HR: hazard ratio; CI: confidence interval.
* Competing-risk regression model with death as competing event.
When adjusting for risk factors of PD, the risk of PD was 22% (HR = 0.78 [0.72–0.85]) lower in the second period than in the first period (Table 2, Model 2). The following risk factors were significantly positively associated with PD: type 2 diabetes (HR = 1.20 [1.10–1.31]), hypertension (HR = 1.54 [1.36–1.75]), gastritis (HR = 1.33 [1.22–1.46], traumatic brain injury (HR = 2.02 [1.63–2.51]), alcohol misuse (HR = 1.31 [1.04–1.64]), and drug abuse (HR = 1.87 [1.25–2.80]).
The competing-risk model accounting for the competing event of death revealed a 21% (HR = 0.79 [0.73–0.86]) reduction in the risk of PD in the second period as compared to the first period (Table 2, Model 3), suggesting that potential changes in mortality within the 10 years did not affect the Cox model results.
Discussion
We found an overall decrease in the age-specific incidence rates of PD over a ten-year period in both males and females, even when controlling for sex, PD risk factors, and changes in mortality as a competing risk. While more than half of all considered risk factors were associated with an increased risk of PD, they did not change the significant decline in PD risk. Thus, typical PD risk factors cannot explain the declining time trend in PD incidence rate. In Western aging societies, epidemiologists have observed an increase in the incidence and prevalence of non-communicable diseases.8,9 Hence, one could assume that the prevalence and incidence of PD should increase over time in Western aging societies, mainly with increasing life expectancy, now reaching 80 years in the EU in 2020 according to Eurostat (www.ec.europa.eu). In fact, the absolute number of new PD cases is increasing globally with an aging population and in males, with some regional variations. 10 In alignment with existing studies, we also found an augmentation of PD incidence rates with increasing age in the German population that was studied.
However, data from two other national-level studies in Germany confirmed the decrease in the annual incidence of idiopathic PD by 25–30% for ages 50 and above in both sexes within the same observation period from to 2013 to 2019.5,6 Additional studies conducted in other countries have partly reported similar trends while the overall number of subjects suffering from PD stays high. A study in Estonia reported that the overall PD incidence has remained comparatively stable over the last 20 years. 11 Another study conducted in Rotterdam, showed an overall decreased incidence of PD between 1990 and 2011. 12 Similar results have been reported in South Korea demonstrating that between 2012 and 2015, the incidence of PD gradually decreased. 13 In a large cohort study from Ontario, Canada, the incidence of mid/late-onset Parkinsonism decreased by 13% between 1996 and 2014. 14 Several concerns regarding the missing mechanistic explanation and the pure epidemiological nature of the data have been raised in follow-up debates. 3
The pathophysiological reason for the decreased incidence of PD observed in Germany is not clear, as most Westernized countries report an increase,3,10 and only a few others show a decrease in PD incidence.12,13 Variations in the prevalence and incidence of PD are influenced by several factors. For instance, there is suspicion of similar geographical patterns of specific pesticide use and PD incidence. This refers to toxins, including paraquat, which has been banned in many countries but is still in use in greater quantities in the U.S.,15,16 a country that reports continuously increased PD rates, at least in a regional fashion. 17 Moreover, nationwide study conducted in France aimed to examine the association of PD incidence with pesticide expenditures. 18 A 16% higher PD incidence was identified in cantons with the highest pesticide expenditure for vineyards without designation of origin, characterized by high fungicide use. The industrial solvent chlorinated halocarbon trichloroethylene (TCE) is another neurotoxic chemical linked to PD. Trichloroethylene can contaminate the air, water, and soil, and although its usage has been banned by the European Union and two U.S. states, it is still permitted for certain purposes 19 and is detected in drinking water. 20 Several studies in rodents have shown that TCE induces dopaminergic neurodegeneration.21,22 TCE is also able to induce alterations in the gut microbiome in a manner that reflects microbial signatures observed in idiopathic PD, confirming that exposure to this chemical may influence PD etiology via the gut-brain axis. 23 An epidemiological study conducted on twins confirmed a significant association between TCE exposure and PD risk.24,25 However, according to the abovementioned study, the interval from the time of exposure to PD diagnosis varies from 10 to 40 years. This delay complicates the identification of a direct incidental link between TCE and PD although new evidence further corroborates this association. 25 So far, the varied levels of pesticide use and the interplay with other risk factors do not allow a correlation between pesticide use and PD incidence. 26
Another dynamic factor may be the continuous industrialization of countries such as China, where the prevalence has doubled from 1999 to 2016. 3 Asian countries had previously lower incidence rates due to the lower prevalence of certain pathogenic risk factors and the absence of age-related changes in nigrostriatal dopaminergic neurons.17,27 Additionally, socioeconomic factors may interact with genetic factors, considering for example that differences in LRRK2 mutations may contribute to variations across ethnic groups, even in sporadic PD.28,29 This fact is further complicated by the occurrence of age-related conditions in subjects with migration backgrounds in Western countries.
The association between smoking and PD incidence might represent a good example of how pathophysiology and socioeconomic factors interact. Some studies have suggested that nicotine and tobacco may have neuroprotective effects and may reduce the risk of PD.30,31 Against this background, the introduction of a state law for the protection of non-smokers in 2007, 32 which banned smoking in restaurants and public places, and the observed decrease in smoking prevalence, especially in the female population, 33 one might expect an increase in PD incidence rates in Germany. A similar association has been documented for caffeine consumption. 34 A further area of interest is the growing role of the microbiome in the pathophysiology of PD. This is a contributing factor that is considerably affected by the environment. 35 According to a recent systematic review, 36 PD patients present a unique microbiota composition in comparison with healthy individuals, and these distinctive gut microbiome differential patterns may have an association with PD pathogenesis.
Another interesting factor that has been identified, was the association between residential greenness and PD. In Metro Vancouver, Canada, a research study that revealed the protective effects of greenness on PD. 37 Similar results were obtained in a study sample from Korea that took place between 2007 and 2015, 38 as well as in China between 2015 and 2018. 39 These studies demonstrate how the presence of greenness in residential areas may have a protective role in neurodegenerative disorders, and the degree to which governments should consider such effects in their public health and more general strategies. For Germany, one study reported a slight increase of biocapacity between 1990 and 2015. 40
Despite the advantage of using routinely collected data (large sample sizes, low panel attrition, no self-selection into study, and no recall bias due to self-reported diseases and conditions), a thorough investigation of our data led us to identify several constraints to be considered. Although the database that was used for our study is large, it is not strictly representative of the German population. There are about 70 million people covered by over 100 health insurances and the AOK covers the largest part (30%) of the population. Data are routinely collected for reimbursement purposes and are subject to legal changes. Nevertheless, previous studies have shown that the data used can provide reasonable estimates of PD incidence rates. 41 Changes in the coding of diagnoses do not necessarily reflect changes at the epidemiological level. In our case, we do not assume declining awareness of PD across the two observation periods. In fact, the opposite would be expected given the introduction of chronic disease coding incentives in 2013, 42 which could lead to higher rates in the second period. In addition, we observed the time of the first diagnosis rather than the time of onset. Delayed diagnosis may partly explain lower rates in the second period. A sensitivity analysis revealed a slight increase from 10.0 to 12.6 months over the 10 years (results not shown) when the time between the diagnosis of at least two symptoms (tremor, gait impairment, anosmia, constipation, and dizziness) of possible PD and the first PD diagnosis was considered. A temporal shift in the health behavior preferences of the insured could also affect temporal trends in diagnoses. However, this can only be captured to a limited extent by health claims data, which require people to go to the doctor when they have an illness. The mean number of comorbidities (based on the comorbidities of the Elixhauser Comorbidity Index) rose from 3.3 to 3.9 diagnoses over the 10-year period, which indicates an increase rather than a decrease in the use of medical services. A bias in disease burden could also be introduced by changes in birth cohort sizes; therefore, we controlled all our models for the age structure of both cohorts.
In conclusion, our study showed a decrease in PD incidence rates in most age groups in Germany and a shift towards higher ages of first diagnosis. Single risk factors and comorbidities remained positively associated with PD incidence. However, no clear associations were identified to explain the observed patterns. Further research is needed to fully understand the relationship between PD, its risk factors, and comorbidities, and how these affect trends in the occurrence of the disease.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241306132 - Supplemental material for Declining trends in the incidence of Parkinson's disease: A cohort study in Germany
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241306132 for Declining trends in the incidence of Parkinson's disease: A cohort study in Germany by Anne Fink, Maria Angeliki S Pavlou, Kirsten Roomp and Jochen G Schneider in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We are grateful to the Scientific Research Institute of the AOK, WIdO, for providing the data. We thank Gabriele Doblhammer for helpful discussions about the study.
Funding
Anne Fink is employed by German Center for Neurodegenerative Diseases and holds a lectureship at the Europäische Fachhochschule (EUFH).
Jochen Schneider, Maria Pavlou and Kirsten Roomp are employed by the University of Luxembourg.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The scientific research institute of the AOK (WIdO) has strict rules regarding data sharing because of the fact that health claims data are a sensitive data source and have ethical restrictions imposed due to concerns regarding privacy. Anonymized data are available to all interested researchers upon request. Interested individuals or an institution who wish to request access to the health claims data of the AOK may contact the WIdO (webpage: ![]() , mail: wido@wido.bv.aok.de).
, mail: wido@wido.bv.aok.de).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
