Abstract
Parkinson’s disease (PD) by its common understanding is a late-onset sporadic movement disorder. However, there is a need to recognize not only the fact that PD pathogenesis expands beyond (or perhaps to) the brain but also that many early-onset patients develop motor signs before the age of 50 years. Indeed, studies have shown that it is likely the protein aggregation observed in the brains of patients with PD precedes the motor symptoms by perhaps a decade. Studies on early-onset forms of PD have shown it to be a heterogeneous disease with multiple genetic and environmental factors determining risk of different forms of disease. Genetic and neuropathological evidence suggests that there are α-synuclein centric forms (e.g.,
INTRODUCTION
Classically, Parkinson’s disease (PD) is considered a progressive, age-related neurodegenerative disorder of the elderly [1]. While this dogma is driven by the onset of motor symptoms, well-characterized prodromal clinical manifestations can precede motor deficits, and neuropathological protein aggregation likely begins decades before the onset of clinical symptoms given recent studies in Alzheimer’s disease (AD) and PD [2–4]. With the average age-at-onset of approximately 65 years being based on the appearance of motor symptoms, this raises the interesting question of whether prodromal clinical manifestations and early underlying molecular etiology justify the reclassification of PD as a ‘early-onset’ disease.
Defining early-onset Parkinson’s disease
Early-onset PD (EOPD), sometimes referred to as ‘young-onset Parkinson’s disease’, lacks a consensus definition, but generally refers to patients who develop the classical PD motor symptoms after 21 years old and before 50 years of age [5, 6]. Patients presenting with PD motor symptoms earlier than 21 years old are classified as ‘juvenile PD’ and usually present with atypical and more severe clinical features [7]. EOPD and juvenile PD cases together comprise between 5-15% of all patients with PD, and are more frequent in males than females (1.5-1.7 : 1) [8, 9]. Age of onset is not the only differential feature between EOPD and the more typical patients with late-onset PD (LOPD; age of onset > 60-65 years) as there are significant differences in disease progression, clinical features, response to medication, and postmortem pathology. Historically, EOPD cases have been reported to be more likely familial (∼20-30% of cases are familial) than sporadic LOPD (where 10-15% report family history) [10]. The discovery of novel EOPD genetic and environmental causes and their potential interplay may also still help to further elucidate the pathological mechanisms of the more common late-onset patients with PD. Like typical LOPD, EOPD is clinically and pathobiologically heterogeneous and the most current genetic and environmental etiologies of EOPD will be discussed herein (Fig. 1).
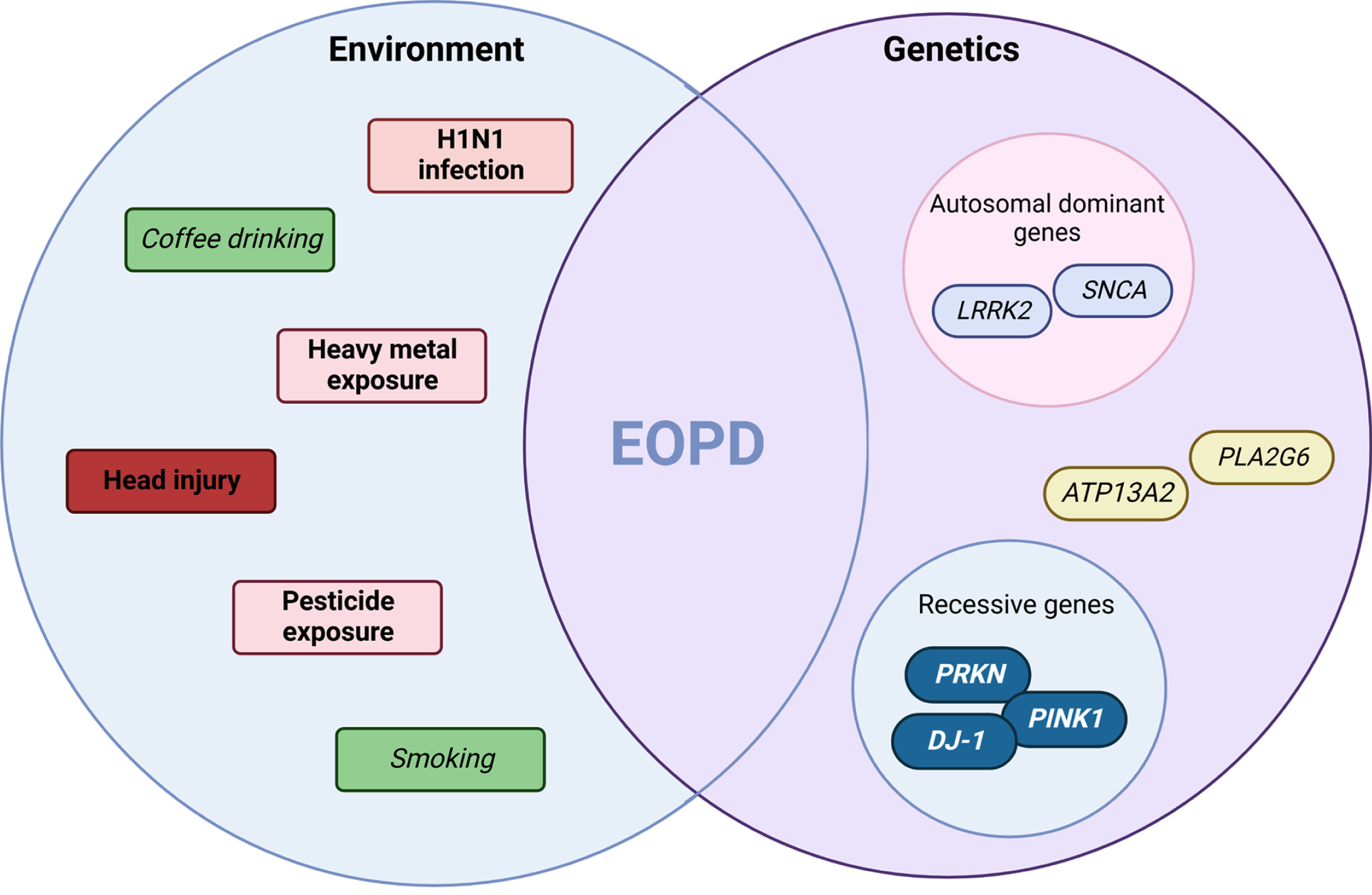
Clinical characteristics and neuropathology of EOPD
It is clear that for PD in general the clinical manifestation of symptoms is heterogeneous, and a number of clinical subtypes have been described [11, 12]. With this in mind, a diagnosis of EOPD is based on the presence of clinical manifestations that define parkinsonism before a predefined age, and it remains unclear whether using age at onset as a cut-off produces a more homogeneous patient group [13, 14]. A number of studies confirm no significant difference for tremor and bradykinesia between EOPD and LOPD; however, EOPD patients are less likely to present with gait disturbances [15], possibly due to a lower burden of comorbidities affecting gait either due to the younger age of onset, or a slower disease progression. Dystonic posture and post-exercise gait disorders have been reported frequently EOPD as the first symptom [16]. As EOPD and LOPD progress, differences can become more apparent with rigidity, dystonia, dyskinesia, painful cramps, and sexual dysfunctions being more frequent in EOPD than LOPD [17]. By contrast, EOPD patients have a lower risk of developing dementia compared to LOPD patients [17, 18], perhaps again reflecting the general overall health of the individual, e.g., better vascular health and potentially different mechanism of neuropathology progression between EOPD and LOPD. In addition, early studies suggested preservation of cardiac sympathetic innervation in patients with
The reasons for these clinical differences are not entirely clear, but age, environment, or underlying genetic variants likely play important roles. Additionally, diverse distribution of α-synuclein-positive Lewy body (LB) pathology may result in different clinical phenotypes. In LOPD, although remaining controversial, α-synuclein pathology is believed to play an important role in dopaminergic neuronal loss and the subsequent onset and progression of motor symptoms. However, patients diagnosed with EOPD can demonstrate heterogeneous neuropathology, which may or may not include α-synuclein deposits. Specifically, some patients with genetic forms of EOPD harboring familial mutations in
While PD is pathologically defined by the presence of LBs, the biological origin, mechanisms of formation, and role(s) in disease remain unclear to date. Interestingly, recent accumulating data suggest that membranous organelles together with α-synuclein are the main components of LB and that the process of LB formation itself could be one of the main drivers of neurodegeneration, rather than simple α-synuclein fibril formation [38–40]. Of note, alterations in lipid metabolism and trafficking as well as mitochondrial and autophagic-lysosomal function, emerge as common biological pathways that can contribute to LB formation [41], and may provide further links between the various genetic and environmental factors in EOPD and/or LOPD. These studies have raised more questions than answers in regard to whether there are specific subtypes of disease, and whether EOPD represents a distinct entity compared to LOPD.
Environmental determinants
Exposure to environmental agents may modify the risk of developing PD with the same environmental variables affecting LOPD believed to also play a role in EOPD. Studies specifically investigating the association between environmental exposure and EOPD found that only a positive history of head injury seemed to increase the risk of EOPD compared to LOPD [42]. Interestingly, smoking was found to be protective against PD in general with a stronger association with a younger age at onset [43]. Coffee consumption has been reported as possible protective factors in the development of PD; however, the role of coffee intake has not been extensively explored in EOPD yet.
Historically, the H1N1 influenza pandemic of 1917-1918 has been considered a possible environmental cause of parkinsonism. In particular, the development of von Economo encephalitis was directly caused by H1N1 [44]. In addition, results of a study on trends of incidence and birth cohorts of PD reported a slight but significant increase of future risk of PD in men with a date of birth between 1915-1924 [4]. Recently, exposure to H5N1 has been considered a possible PD causative infection in experimental model of PD [45]. It is possible that systemic viral (or potentially bacterial) infection represents the first “hit” of the dual-hit theory of PD [46]. Additional definitive studies will be needed in the future to explore the role of the current SARS-CoV-2 (COVID-19) pandemic on PD risk although studies have shown that neurodegenerative processes are likely[47–49].
The discovery of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) induced parkinsonism dramatically advanced the understanding of the pathophysiology of parkinsonism and increased the understanding of possible risk factors for EOPD [50]. MPTP-induced mitochondrial damage raises the possibility that cases of non-drug induced EOPD have mitochondrial dysfunction as the underlying pathogenesis [51]. On the other hand, in the genetic and non-genetic forms of EOPD, the involvement of environmental agents seems to cause less acute and more long-lasting effect later in life. Paraquat and a number of pesticides have been identified as a risk for PD either as a direct exposure or via polluted water [52, 53]; however, the information on EOPD is limited. Certainly, the duration and amount of exposure has an important weight in the development of PD, together with an individual (genetic) predisposition.
Therefore, in EOPD the environmental risk factors (alone or acting in cluster) may need a stronger individual genetic predisposition to contribute to EOPD. In fact, a study reported an increase risk of developing PD and having a more severe disease in farmers that had head trauma, exposure to pesticides and a genetic susceptibility allele of the
ENVIRONMENT-GENE INTERACTIONS
The majority of PD cases are idiopathic, with inherited genetic forms accounting for approximately 10% of PD cases [56]. Many have postulated that PD arises from an interaction between genetic susceptibility and environmental exposures [57]. One of the most studied interactions in PD is between smoking and monoamine oxidase B (MAO-B) enzyme. As noted earlier, smoking has been shown to be protective against PD; however, different polymorphisms in the gene encoding MAO-B may have opposite effects on PD risk in smokers [58]. Another example of gene-environment interaction comes from a genetically predetermined inability to metabolize organophosphates and pesticides that in turn may result in an increased risk of PD [59]. Chronic exposure to rotenone, a mitochondrial complex I inhibitor and pesticide, may alter the genetic expression and production of α-synuclein and DJ-1, leading to PD pathogenesis [60]. In addition, chronic exposure to heavy metals and pesticides is associated with a younger onset of PD [61]. It is also possible that genetic factors only become relevant when placed in the context of an environmental agent and these genes may be more complicated to identify than those that have been identified as monogenic drivers of disease.
Genetics determinants
PD is a genetically heterogeneous disease with family members who carry the same genetic mutation presenting with varying symptomatic manifestation (e.g., age of onset, severity of symptoms, and disease progression rate), suggesting that even less complex Mendelian and monogenic cases of the disease rely on environmental factors or additional genetic modifiers/risks.
Currently, pathogenic variants of variable penetrance have been identified in a handful of genes widely accepted as major determinants or strong risk factors for inherited forms of parkinsonism and PD including
Some genetic mutations are associated with atypical presentations of EOPD, with extrapyramidal symptoms as part of a more complex phenotype. Various forms of variable dystonia-parkinsonism syndromes associated with early onset parkinsonian features have been described; mutations in the
The identification of genes related to PD has provided useful insights into specific biological pathways involved in PD pathobiology and progression. Mutations in confirmed PD genes mainly alter the dopaminergic neuron maintenance/homeostasis through various mechanisms including, synaptic vesicle protein defects, alterations of mitochondrial quality control, and endosome-autophagy-lysosome dysfunctions [74–76]. For EOPD, two genetic drivers of disease are currently considered: 1) the three recessive genes
SNCA and LRRK2: from early to late, from familial to sporadic
As the presence of LBs is considered the defining pathological confirmation of a PD diagnosis [77], the most relevant genetic cause of EOPD is the highly penetrant mutations observed in the
The normal function of α-synuclein remains unclear but it is thought to play several roles in synaptic regulation and neurotransmitter release [81]. Pathologically, overexpression of α-synuclein or folding defects/toxic gain of function mutations lead to the accumulation and aggregation of this protein in the pre-synaptic termini of neurons. This leads to a cascade of events that includes neurotransmission inhibition, inhibition of exocytosis, cell stress, and cell death, which then spreads in a prion-like manner through the synaptically connected brain regions at variable rates consistent with Braak staging [82]. However recent links to other genes related to PD also link α-synuclein levels to mitochondrial function (
Leucine-rich repeat kinase 2 (LRRK2) is a large protein with multiple domains and GTPase and kinase activity that is widely expressed in different tissues [83–85]. Mutations in
PINK1 and PRKN: regulation of mitochondria quality control
The high energy demand of dopaminergic neurons means a large amount of reactive oxygen species (ROS) is produced in these neurons. As ROS are toxic to mitochondria and to the cell, it is essential that the mitochondrial quality control is efficient in these neurons [75, 90]. Mitochondrial damage and clearance defects are relevant for neurodegenerative disorders in general, and EOPD in particular has been hypothesized to result from a breakdown of these quality control pathways in some cases [91]. This hypothesis is in part driven by the identification of recessive loss-of-function mutations in two genes,
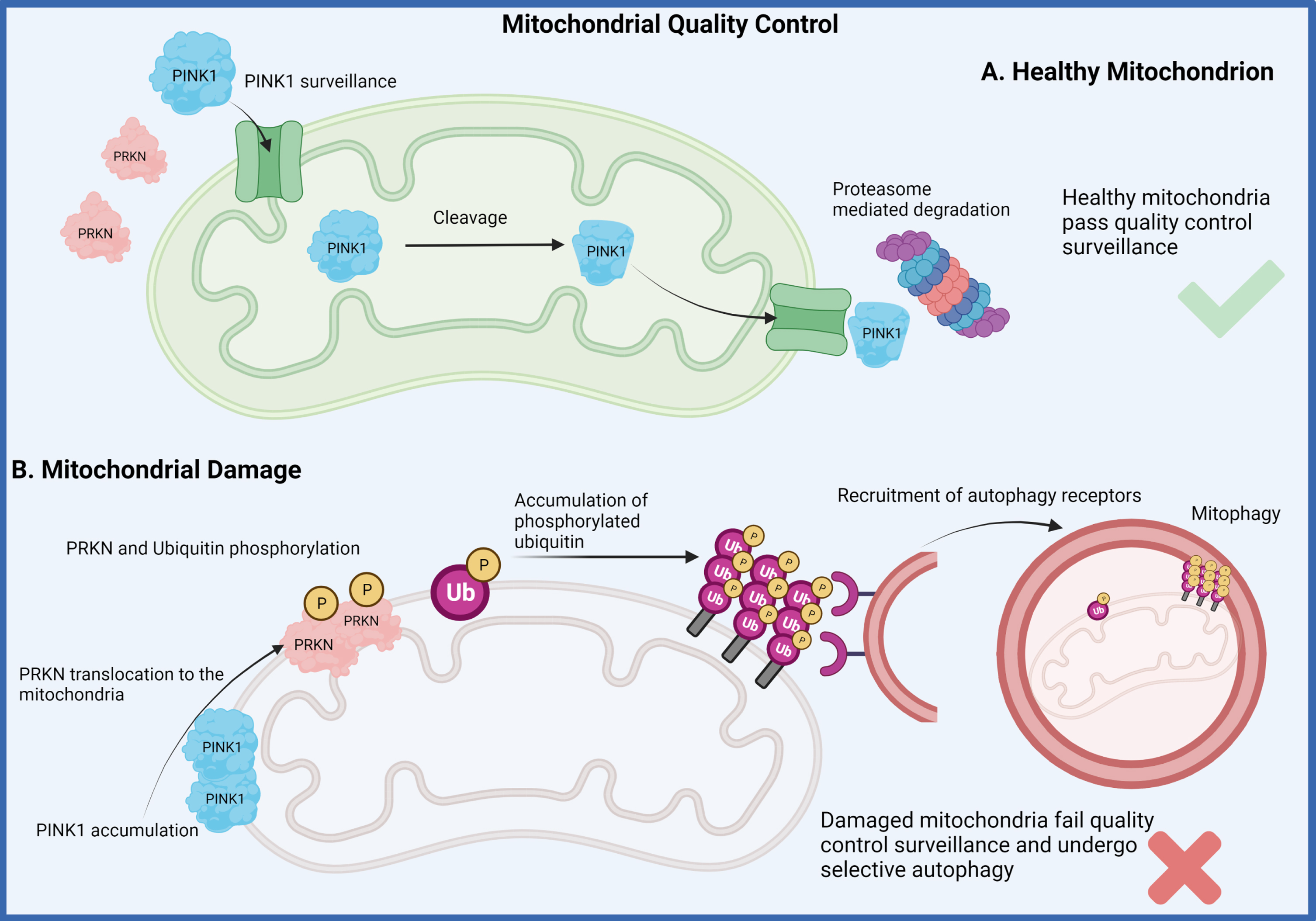
PINK1 is a mitochondrial ubiquitin (Ub) kinase that constantly surveilles organelle health through its regulated import into healthy organelles and rapid cleavage and degradation therefrom. When mitochondrial damage is present though, PINK1 is no longer imported but locally accumulates on the outer mitochondrial membrane where it recruits and activates the E3 Ub ligase PRKN from the cytosol to jointly label and target damaged mitochondria for degradation [75].
The mean age at onset of patients with
Interestingly, even though the function of
There is controversy whether
AGE-AT-ONSET MODIFIERS
Patients with EOPD have considerable age-at-onset (AAO) variation. Genetic variation and sex differences very likely contribute to AAO [116]. Multiple studies have attempted to identify genes or variants that modify the AAO of PD with very little success, likely due to the low sample size of their datasets. The largest aggregate of PD genome wide association studies (GWAS) to date includes a meta-analysis of 17 datasets, with 37,688 PD patients, 18,618 UK Biobank proxy-cases and 1.4 million controls. This study identified 90 independent common genetic risk factors in individuals of European ancestry [89]. Currently, there are efforts to generate a predictive genetic risk score (GRS) that is based on cumulative genetic risk, this may also help determine AAO prediction, as it has been shown that a subset of risk variants overlap with AAO modifiers [117]. A Mendelian randomization portal that uses thousands of GWAS data is now available for the research community in hopes to identify modifiers of disease progression (https://pdgenetics.shinyapps.io/MRportal/) [118].
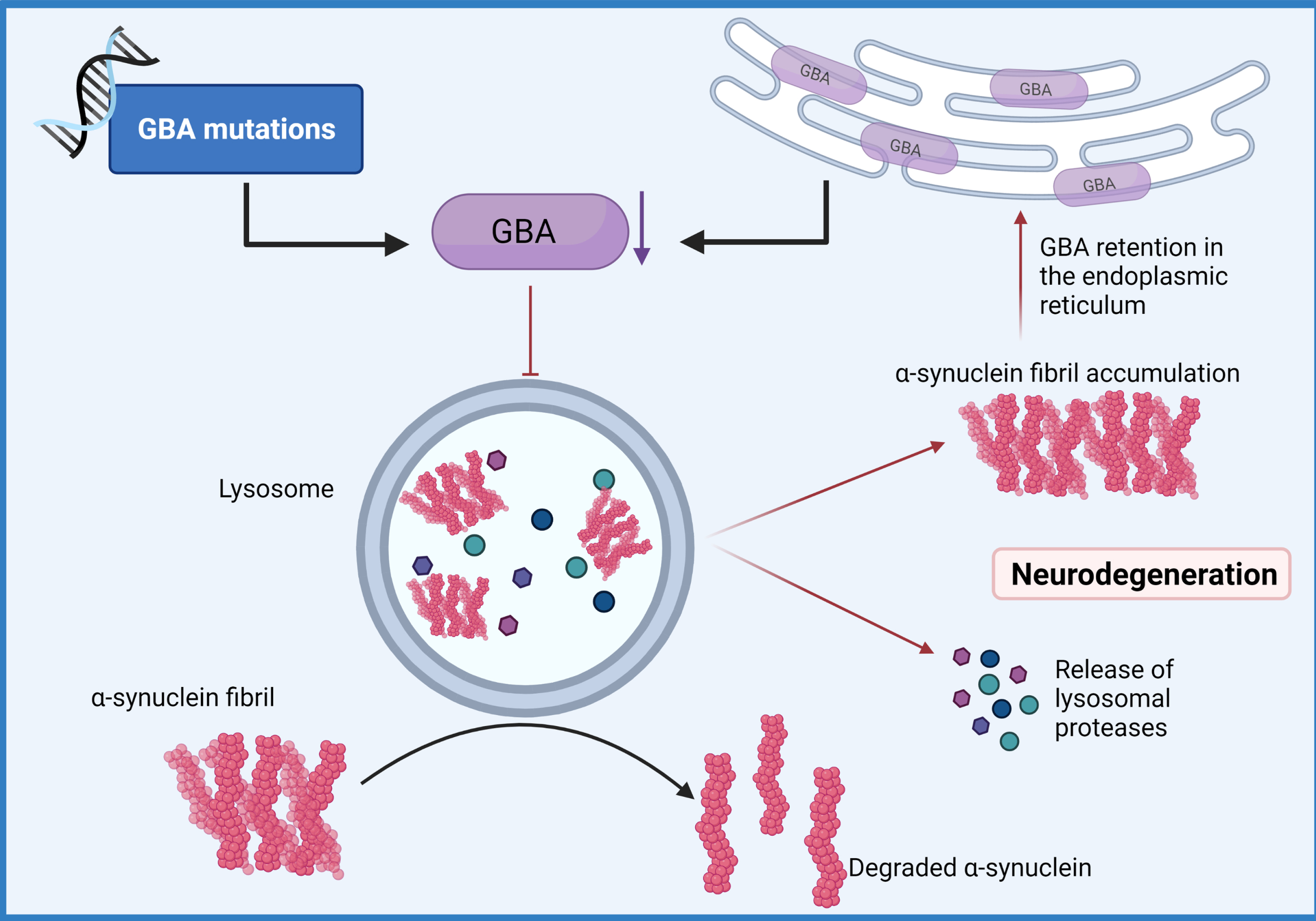
GBA: lysosomal pathway central in PD pathobiology
Rare coding variants in
CONCLUSIONS
Over the last two decades it has become clear that up to 10% of patients with EOPD harbor a major genetic determinant [126]. Nonetheless, in most patients, disease cannot be explained by a monogenetic form of inheritance, rather a genetic-environment interaction appears to play a pivotal role in the onset and progression of the disease [57]. GWAS have identified susceptible gene loci for many diseases, including neurodegenerative disorders, however, the variability in common alleles can’t explain heritability of common disorders by itself. Moreover, GWAS cannot detect those genes that influence disease onset/progression dependent upon an interaction with other genes or with the environment [127].
Functional analyses of lipid metabolism, mitochondrial health, neurometabolic cellular activity, and autophagic-lysosome capacity will be critical to place these proposed gene-environment interactions into context. Specific readouts such as phosphorylated ubiquitin, the joint PINK1-PARKIN product and so-called mitophagy tag, may not only be helpful to reveal cross-talk between and potential convergence of PD pathways in cells, tissues, and biofluids, but may also aid in better stratification of patients [128–130]. Likewise, further approaches to identify and compare specific molecular signatures, could be critical to re-categorize forms of PD based on biological pathways, in addition to clinical and pathological definitions, rather than simply age at onset [131].
After the initial discovery of
A major question that remains unanswered by current genetic-environment studies is whether EOPD and LOPD are distinct disease entities or different staging on the same spectrum of disease. As discussed herein, there are clinical and neuropathologic differences in the phenotypic manifestation of disease signs, but it is not clear whether these are a simply a reflection of overall health (biological age) in the younger patients. Age is recognized as the major risk factor for LOPD, but this is absent in patients with EOPD and may reflect a stronger genetic/environmental component in the latter. However, given the presented genetic and environmental data it may be the scenario whereby there are distinct forms of PD, e.g., α-synuclein-centric versus mitophagy-driven, that may produce/influence both EOPD and LOPD. If this is the case, then whether it is a benefit to split patients by an arbitrary age cut-off is up for debate. What we really need as a field is an unbiased biologic readout that categorizes patients into subtypes and allows a more accurate definition of disease for clinical trial participation. A good example of this is the application of seed assays, such as RT-QuIC (real-time quaking induced conversion), which will facilitate the identification of patients with toxic α-synuclein protein driven forms of disease [138, 139]. Also, the recent work on phospho-ubiquitin (Serine-65) as a marker of mitochondrial health and readout for the mitophagy/mitochondrial quality control pathway, is another example of a potential biologic marker of a specific disease process [128]. Finally, the exposure to toxins may also in part define these subtypes, creating an environment on which the genetic determinants will act to drive phenotypic heterogeneity. Whether we can use these combinations to define specific patients’ subtypes, beyond the simple dichotomy of EOPD or LOPD, will be critical for drug development and successful clinical trials. As we start using machine-learning approaches to integrate large scale multimodal datasets, we can perhaps identify these key interactions, tease out cause versus effect questions, and redefine early-onset forms of PD.
Footnotes
ACKNOWLEDGMENTS
We acknowledge the support of the many people with Parkinson’s disease, both early and late-onset forms, and their families and caregivers who have driven many of the studies described in this review. In addition, we note the support of many clinical neurologists, clinical study coordinators, research technicians, fellows and students who have contributed to the studies on early-onset PD. Mayo Clinic is an American Parkinson Disease Association (APDA) Mayo Clinic Information and Referral Center, an APDA Center for Advanced Research, and the Mayo Clinic Lewy Body Dementia Association (LBDA) Research Center of Excellence. Mayo Clinic is also an LBD Center without Walls (U54-NS110435). OAR is supported by National Institute of Health/National Institute of Neurological Disorders and Stroke (NIH/NINDS) [R01 NS085070 and U54 NS110435], the Department of Defense Congressionally Directed Medical Research Programs (CDMRP) [W81XWH-17-1-0249], the Michael J. Fox Foundation for Parkinson’s Research (MJFF), the American Parkinson Disease Association (APDA), and American Brain Foundation. PJM is supported by NIH/NINDS R01 NS085070, R01 NS110085 and U54 NS110435, the APDA Center for Advanced Research, the Mayo Clinic Foundation, and the American Brain Foundation. WS is supported by National Institute on Aging (NIH/NIA) [R56 AG062556], National Institute of Neurological Disorders and Stroke (NIH/NINDS) [R01 NS085070, R01 NS110085 and U54 NS110435], the Department of Defense Congressionally Directed Medical Research Programs (CDMRP) [W81XWH-17-1-0248], the Florida Department of Health - Ed and Ethel Moore Alzheimer’s Disease Research Program [![]() ], the Michael J. Fox Foundation for Parkinson’s Research (MJFF), the American Parkinson Disease Association (APDA), Mayo Clinic Foundation, the Center for Biomedical Discovery (CBD) and the Robert and Arlene Kogod Center on Aging, and a sponsored research agreement with Amazentis SA.
], the Michael J. Fox Foundation for Parkinson’s Research (MJFF), the American Parkinson Disease Association (APDA), Mayo Clinic Foundation, the Center for Biomedical Discovery (CBD) and the Robert and Arlene Kogod Center on Aging, and a sponsored research agreement with Amazentis SA.
CONFLICT OF INTEREST
PJM: Support from NIH/NINDS R01 NS085070, R01 NS110085 and U54 NS110435, the APDA Center for Advanced Research, the Mayo Clinic Foundation, and the American Brain Foundation.
WS: Support from National Institute on Aging (NIH/NIA) [R56 AG062556], National Institute of Neurological Disorders and Stroke (NIH/NINDS) [R01 NS085070, R01 NS110085 and U54 NS110435], the Department of Defense Congressionally Directed Medical Research Programs (CDMRP) [W81XWH-17-1-0248], the Michael J. Fox Foundation for Parkinson’s Research (MJFF), the American Parkinson Disease Association (APDA),
RS: Support from the National Institute on Aging, the National Institute of Neurological Disorders and Stroke, the Parkinson’s Disease Foundation, and Acadia Pharmaceuticals.
OAR: Support from National Institute of Health/National Institute of Neurological Disorders and Stroke (NIH/NINDS) [R01 NS085070 and U54 NS110435], the Department of Defense Congressionally Directed Medical Research Programs (CDMRP) [W81XWH-17-1-0249], the Michael J. Fox Foundation for Parkinson’s Research (MJFF), the American Parkinson Disease Association (APDA), and American Brain Foundation.
The other authors have no relevant disclosures to report.
